Treatment of Men with Central Hypogonadism: Alternatives for Testosterone Replacement Therapy
Abstract
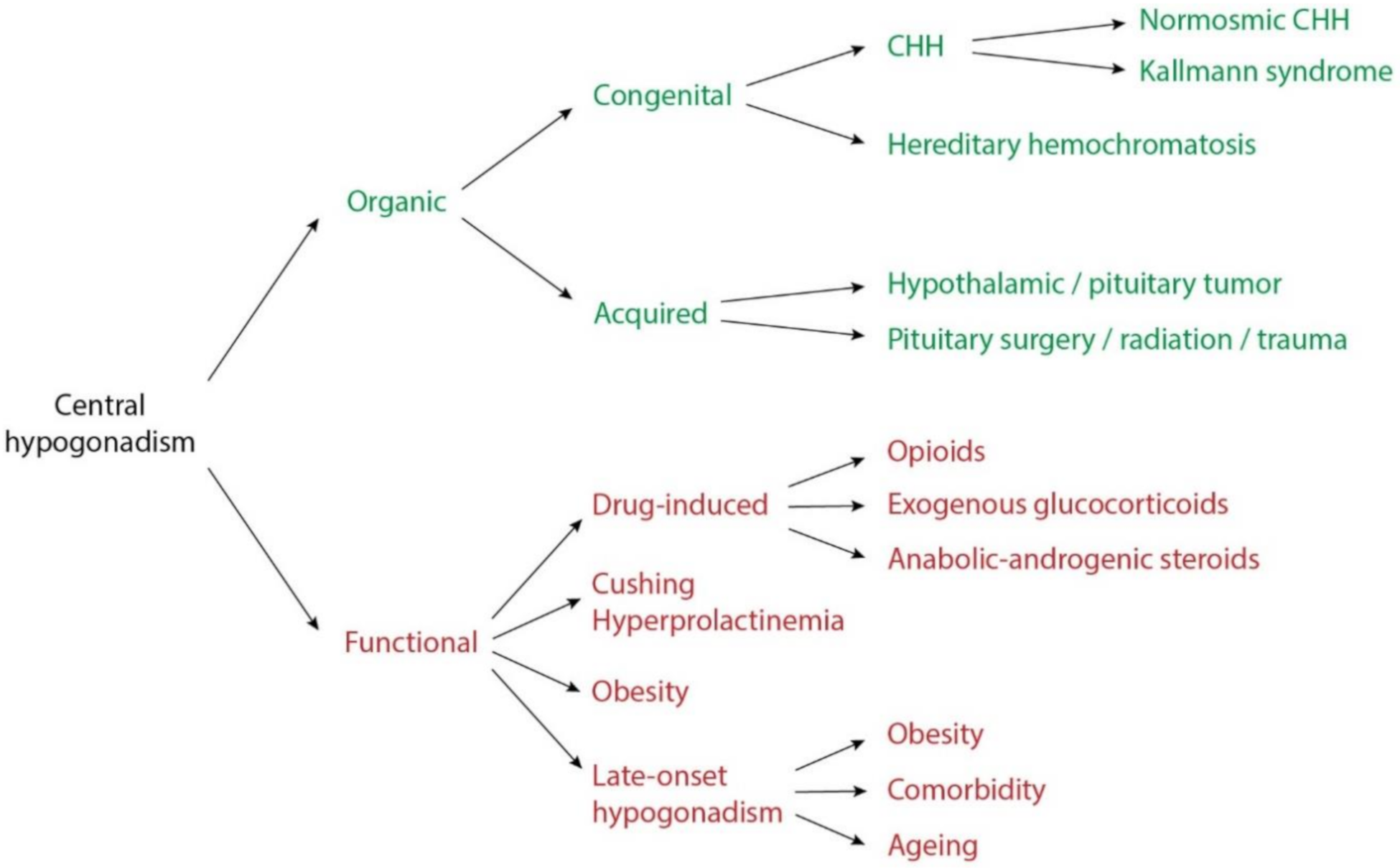
1. Introduction
2. Non-Pharmacological Treatment
3. Gonadotropins
3.1. Mechanism of Action
3.2. Indications
3.3. Treatment Regimens
3.4. Results
3.5. Adverse Effects and Monitoring of Therapy
4. Pulsatile GnRH Therapy
5. Clomiphene Citrate and Tamoxifen
5.1. Mechanism of Action
5.2. Off-Label Use and Treatment Regimens
5.3. Results
5.4. Adverse Effects
6. Aromatase Inhibitors
6.1. Mechanism of Action and Off-Label Use
6.2. Results
6.3. Adverse Effects
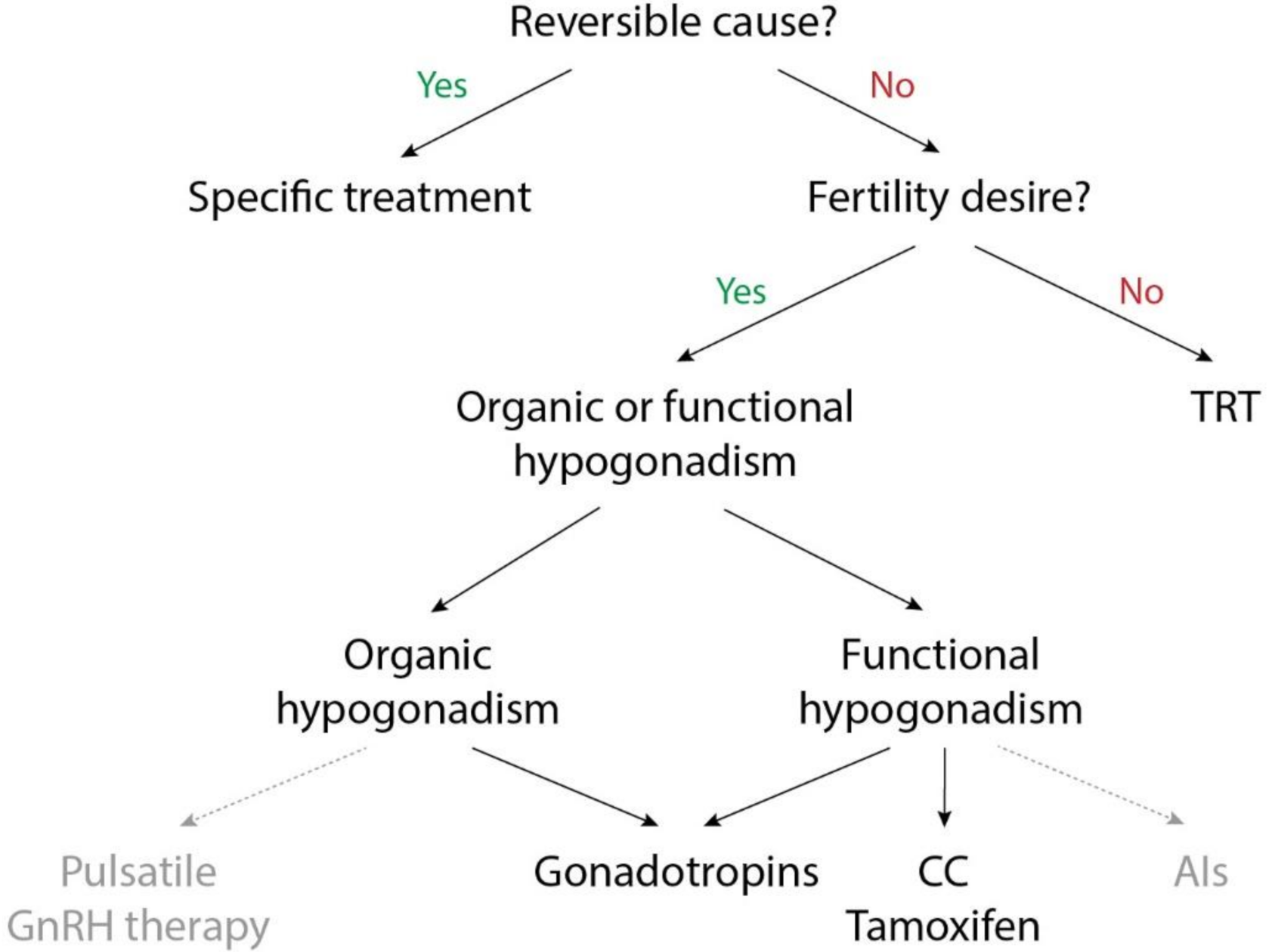
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| TRT | Testosterone replacement therapy |
| HPG | Hypothalamic-pituitary-gonadal |
| GnRH | Gonadotropin releasing hormone |
| LH | Luteinizing hormone |
| FSH | Follicle-stimulating hormone |
| CHH | Congenital hypogonadotropic hypogonadism |
| EMAS | European Male Aging Study |
| hCG | Human chorionic gonadotropin |
| hMG | Human menopausal gonadotropin |
| SERMs | Selective estrogen receptor modulators |
| ADAM | Androgen deficiency in males |
| RCT | Randomized controlled trial |
| VTE | Venous thromboembolism |
| AIs | Aromatase inhibitors |
References
- Salonia, A.; Rastrelli, G.; Hackett, G.; Seminara, S.B.; Huhtaniemi, I.T.; Rey, R.A.; Hellstrom, W.J.G.; Palmert, M.R.; Corona, G.; Dohle, G.R.; et al. Paediatric and adult-onset male hypogonadism. Nat. Rev. Dis. Prim. 2019, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Goulis, D.G.; Huhtaniemi, I.; Zitzmann, M.; Toppari, J.; Forti, G.; Vanderschueren, D.; Wu, F.C.; Corona, G.; Goulis, D.G.; et al. European Academy of Andrology (EAA) guidelines on investigation, treatment and monitoring of functional hypogonadism in males. Andrology 2020, 12770. [Google Scholar] [CrossRef] [PubMed]
- Bhasin, S.; Brito, J.P.; Cunningham, G.R.; Hayes, F.J.; Hodis, H.N.; Matsumoto, A.M.; Snyder, P.J.; Swerdloff, R.S.; Wu, F.C.; Yialamas, M.A. Testosterone Therapy in Men with Hypogonadism: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2018, 103, 1715–1744. [Google Scholar] [CrossRef] [PubMed]
- Awouters, M.; Vanderschueren, D.; Antonio, L. Aromatase inhibitors and selective estrogen receptor modulators: Unconventional therapies for functional hypogonadism? Andrology 2019, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Boehm, U.; Bouloux, P.M.; Dattani, M.T.; de Roux, N.; Dodé, C.; Dunkel, L.; Dwyer, A.A.; Giacobini, P.; Hardelin, J.P.; Juul, A.; et al. Expert consensus document: European Consensus Statement on congenital hypogonadotropic hypogonadism-pathogenesis, diagnosis and treatment. Nat. Rev. Endocrinol. 2015, 11, 547–564. [Google Scholar] [CrossRef]
- Bonomi, M.; Vezzoli, V.; Krausz, C.; Guizzardi, F.; Vezzani, S.; Simoni, M.; Bassi, I.; Duminuco, P.; di Iorgi, N.; Giavoli, C.; et al. Characteristics of a nationwide cohort of patients presenting with isolated hypogonadotropic hypogonadism (IHH). Eur. J. Endocrinol. 2018, 178, 23–32. [Google Scholar] [CrossRef]
- Cangiano, B.; Swee, D.S.; Quinton, R.; Bonomi, M. Genetics of congenital hypogonadotropic hypogonadism: Peculiarities and phenotype of an oligogenic disease. Qual. Life Res. 2020, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Crownover, B.K.; Covey, C.J. Hereditary Hemochromatosis. Am. Fam. Physician 2013, 87, 183–190. [Google Scholar]
- Grossmann, M.; Matsumoto, A.M. A Perspective on Middle-Aged and Older Men with Functional Hypogonadism: Focus on Holistic Management. J. Clin. Endocrinol. Metab. 2017, 102, 1067–1075. [Google Scholar] [CrossRef]
- Huhtaniemi, I. Late-onset hypogonadism: Current concepts and controversies of pathogenesis, diagnosis and treatment. Asian J. Androl. 2014, 16, 192–202. [Google Scholar] [CrossRef]
- Wu, F.C.W.; Tajar, A.; Beynon, J.M.; Pye, S.R.; Phil, M.; Silman, A.J.; Finn, J.D.; O’neill, T.W.; Bartfai, G.; Casanueva, F.F.; et al. Identification of Late-Onset Hypogonadism in Middle-Aged and Elderly Men. N. Engl. J. Med. 2010, 363, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.C.W.; Tajar, A.; Pye, S.R.; Silman, A.J.; Finn, J.D.; O’Neill, T.W.; Bartfai, G.; Casanueva, F.; Forti, G.; Giwercman, A.; et al. Hypothalamic-pituitary-testicular axis disruptions in older men are differentially linked to age and modifiable risk factors: The European male aging study. J. Clin. Endocrinol. Metab. 2008, 93, 2737–2745. [Google Scholar] [CrossRef] [PubMed]
- Rastrelli, G.; Carter, E.L.; Ahern, T.; Finn, J.D.; Antonio, L.; O’Neill, T.W.; Bartfai, G.; Casanueva, F.F.; Forti, G.; Keevil, B.; et al. Development of and Recovery from Secondary Hypogonadism in Aging Men: Prospective Results from the EMAS. J. Clin. Endocrinol. Metab. 2015, 100, 3172–3182. [Google Scholar] [CrossRef] [PubMed]
- Cangiano, B.; Duminuco, P.; Vezzoli, V.; Guizzardi, F.; Chiodini, I.; Corona, G.; Maggi, M.; Persani, L.; Bonomi, M. Evidence for a Common Genetic Origin of Classic and Milder Adult-Onset Forms of Isolated Hypogonadotropic Hypogonadism. J. Clin. Med. 2019, 8, 126. [Google Scholar] [CrossRef] [PubMed]
- Indirli, R.; Cangiano, B.; Profka, E.; Mantovani, G.; Persani, L.; Arosio, M.; Bonomi, M.; Ferrante, E. A Rare SPRY4 Gene Mutation Is Associated with Anosmia and Adult-Onset Isolated Hypogonadotropic Hypogonadism. Front. Endocrinol. 2019, 10, 781. [Google Scholar] [CrossRef]
- Snyder, P.J.; Bhasin, S.; Cunningham, G.R.; Matsumoto, A.M.; Stephens-Shields, A.J.; Cauley, J.A.; Gill, T.M.; Barrett-Connor, E.; Swerdloff, R.S.; Wang, C.; et al. Lessons from the Testosterone Trials. Endocr. Rev. 2018, 39, 369–386. [Google Scholar] [CrossRef] [PubMed]
- Khera, M.; Adaikan, G.; Buvat, J.; Carrier, S.; El-Meliegy, A.; Hatzimouratidis, K.; Mccullough, A.; Morgentaler, A.; Torres, L.O.; Salonia, A. Diagnosis and Treatment of Testosterone Deficiency: Recommendations from the Fourth International Consultation for Sexual Medicine (ICSM 2015). J. Sex. Med. 2016, 13, 1787–1804. [Google Scholar] [CrossRef]
- Travison, T.G.; Araujo, A.B.; Kupelian, V.; O’Donnell, A.B.; McKinlay, J.B. The Relative Contributions of Aging, Health, and Lifestyle Factors to Serum Testosterone Decline in Men. J. Clin. Endocrinol. Metab. 2007, 92, 549–555. [Google Scholar] [CrossRef]
- Bacon, C.G.; Mittleman, M.A.; Kawachi, I.; Giovannucci, E.; Glasser, D.B.; Rimm, E.B. Sexual Function in Men Older Than 50 Years of Age: Results from the Health Professionals Follow-up Study. Ann. Intern. Med. 2003, 139, 161–168. [Google Scholar] [CrossRef]
- Camacho, E.M.; Huhtaniemi, I.T.; O’Neill, T.W.; Finn, J.D.; Pye, S.R.; Lee, D.M.; Tajar, A.; Bartfai, G.; Boonen, S.; Casanueva, F.F.; et al. Age-associated changes in hypothalamic-pituitary-testicular function in middle-aged and older men are modified by weight change and lifestyle factors: Longitudinal results from the European Male Ageing Study. Eur. J. Endocrinol. 2013, 168, 445–455. [Google Scholar] [CrossRef]
- Corona, G.; Rastrelli, G.; Monami, M.; Saad, F.; Luconi, M.; Lucchese, M.; Facchiano, E.; Sforza, A.; Forti, G.; Mannucci, E.; et al. Body weight loss reverts obesity-associated hypogonadotropic hypogonadism: A systematic review and meta-analysis. Eur. J. Endocrinol. 2013, 168, 829–843. [Google Scholar] [CrossRef] [PubMed]
- Heufelder, A.E.; Saad, F.; Bunck, M.C.; Gooren, L. Fifty-two-Week Treatment with Diet and Exercise Plus Transdermal Testosterone Reverses the Metabolic Syndrome and Improves Glycemic Control in Men with Newly Diagnosed Type 2 Diabetes and Subnormal Plasma Testosterone. J. Androl. 2009, 30, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Mongioì, L.M.; Cimino, L.; Condorelli, R.A.; Magagnini, M.C.; Barbagallo, F.; Cannarella, R.; la Vignera, S.; Calogero, A.E. Effectiveness of a Very Low Calorie Ketogenic Diet on Testicular Function in Overweight/Obese Men. Nutrients 2020, 12, 2967. [Google Scholar] [CrossRef]
- La Vignera, S.; Cannarella, R.; Galvano, F.; Grillo, A.; Aversa, A.; Cimino, L.; Magagnini, C.M.; Mongioì, L.M.; Condorelli, R.A.; Calogero, A.E. The ketogenic diet corrects metabolic hypogonadism and preserves pancreatic ß-cell function in overweight/obese men: A single-arm uncontrolled study. Endocrine 2020, 1–8. [Google Scholar] [CrossRef]
- Esposito, K.; Giugliano, F.; di Palo, C.; Giugliano, G.; Marfella, R.; D’Andrea, F.; D’Armiento, M.; Giugliano, D. Effect of lifestyle changes on erectile dysfunction in obese men: A randomized controlled trial. J. Am. Med. Assoc. 2004, 291, 2978–2984. [Google Scholar] [CrossRef]
- Xu, J.; Wu, Q.; Zhang, Y.; Pei, C. Effect of Bariatric Surgery on Male Sexual Function: A Meta-Analysis and Systematic Review. Sex. Med. 2019, 7, 270–281. [Google Scholar] [CrossRef]
- Bawor, M.; Bami, H.; Dennis, B.B.; Plater, C.; Worster, A.; Varenbut, M.; Daiter, J.; Marsh, D.C.; Steiner, M.; Anglin, R.; et al. Testosterone suppression in opioid users: A systematic review and meta-analysis. Drug Alcohol Depend. 2015, 149, 1–9. [Google Scholar] [CrossRef]
- Rosa, M.; Ciccarelli, A.; Zarrilli, S.; Guerra, E.; Gaccione, M.; Sarno, A.; Lombardi, G.; Colao, A. The treatment with cabergoline for 24 month normalizes the quality of seminal fluid in hyperprolactinaemic males. Clin. Endocrinol. 2006, 64, 307–313. [Google Scholar] [CrossRef]
- Lenzi, A.; Balercia, G.; Bellastella, A.; Colao, A.; Fabbri, A.; Foresta, C.; Galdiero, M.; Gandini, L.; Krausz, C.; Lombardi, G.; et al. Epidemiology; diagnosis, and treatment of male hypogonadotropic hypogonadism. J. Endocrinol. Investig. 2009, 32, 934–938. [Google Scholar] [CrossRef]
- Colao, A.; Vitale, G.; Cappabianca, P.; Briganti, F.; Ciccarelli, A.; De Rosa, M.; Zarrilli, S.; Lombardi, G. Outcome of Cabergoline Treatment in Men with Prolactinoma: Effects of a 24-Month Treatment on Prolactin Levels, Tumor Mass, Recovery of Pituitary Function, and Semen Analysis. J. Clin. Endocrinol. Metab. 2004, 89, 1704–1711. [Google Scholar] [CrossRef]
- Rastrelli, G.; Corona, G.; Mannucci, E.; Maggi, M. Factors affecting spermatogenesis upon gonadotropin-replacement therapy: A meta-analytic study. Andrology 2014, 2, 794–808. [Google Scholar] [CrossRef] [PubMed]
- Finkel, D.M.; Phillips, J.L.; Snyder, P.J. Stimulation of Spermatogenesis by Gonadotropins in Men with Hypogonadotropic Hypogonadism. N. Engl. J. Med. 1985, 313, 651–655. [Google Scholar] [CrossRef] [PubMed]
- Nieschlag, E.; Bouloux, P.M.G.; Stegmann, B.J.; Shankar, R.R.; Guan, Y.; Tzontcheva, A.; McCrary Sisk, C.; Behre, H.M. An open-label clinical trial to investigate the efficacy and safety of corifollitropin alfa combined with hCG in adult men with hypogonadotropic hypogonadism. Reprod. Biol. Endocrinol. 2017, 15, 17. [Google Scholar] [CrossRef] [PubMed]
- Sinisi, A.A.; Esposito, D.; Bellastella, G.; Maione, L.; Palumbo, V.; Gandini, L.; Lombardo, F.; De Bellis, A.; Lenzi, A.; Bellastella, A. Efficacy of recombinant human follicle stimulating hormone at low doses in inducing spermatogenesis and fertility in hypogonadotropic hypogonadism. J. Endocrinol. Investig. 2010, 33, 618–623. [Google Scholar] [CrossRef]
- Ulloa-Aguirre, A.; Lira-Albarrán, S. Clinical Applications of Gonadotropins in the Male. Prog. Mol. Biol. Sci. 2016, 143, 121–174. [Google Scholar] [CrossRef]
- Rohayem, J.; Hauffa, B.P.; Zacharin, M.; Kliesch, S.; Zitzmann, M. Testicular growth and spermatogenesis: New goals for pubertal hormone replacement in boys with hypogonadotropic hypogonadism? A multicentre prospective study of hCG/rFSH treatment outcomes during adolescence. Clin. Endocrinol. 2017, 86, 75–87. [Google Scholar] [CrossRef]
- Thirumalai, A.; Berkseth, K.E.; Amory, J.K. Treatment of hypogonadism: Current and future therapies. F1000Research 2017, 6, 68. [Google Scholar] [CrossRef]
- George, B.; Bantwal, G. Endocrine management of male subfertility. Indian J. Endocrinol. Metab. 2013, 17, 32–34. [Google Scholar] [CrossRef]
- Dwyer, A.A.; Sykiotis, G.P.; Hayes, F.J.; Boepple, P.A.; Lee, H.; Loughlin, K.R.; Dym, M.; Sluss, P.M.; Crowley, W.F.; Pitteloud, N. Trial of Recombinant Follicle-Stimulating Hormone Pretreatment for GnRH-Induced Fertility in Patients with Congenital Hypogonadotropic Hypogonadism. J. Clin. Endocrinol. Metab. 2013, 98, E1790–E1795. [Google Scholar] [CrossRef]
- Dwyer, A.A.; Raivio, T.; Pitteloud, N. Gonadotrophin replacement for induction of fertility in hypogonadal men. Best Pract. Res. Clin. Endocrinol. Metab. 2015, 29, 91–103. [Google Scholar] [CrossRef]
- Burris, A.S.; Rodbard, H.W.; Winters, S.J.; Sherins, R.J. Gonadotropin therapy in men with isolated hypogonadotropic hypogonadism: The response to human chorionic gonadotropin is predicted by initial testicular size. J. Clin. Endocrinol. Metab. 1988, 66, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Vicari, E.; Mongioì, A.; Calogero, A.E.; Moncada, M.L.; Sidoti, G.; Polosa, P.; D’agata, R. Therapy with human chorionic gonadotrophin alone induces spermatogenesis in men with isolated hypogonadotrophic hypogonadism-long-term follow-up. Int. J. Androl. 1992, 15, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhang, S.X.; Dong, Q.; Xiong, Z.B.; Li, X. Application of hormonal treatment in hypogonadotropic hypogonadism: More than ten years experience. Int. Urol. Nephrol. 2012, 44, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Warne, D.W.; Decosterd, G.; Okada, H.; Yano, Y.; Koide, N.; Howles, C.M. A combined analysis of data to identify predictive factors for spermatogenesis in men with hypogonadotropic hypogonadism treated with recombinant human follicle-stimulating hormone and human chorionic gonadotropin. Fertil. Steril. 2009, 92, 594–604. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.Y.; Wishart, S.M.; Handelsman, D.J. A Double-Blind, Placebo-Controlled, Randomized Clinical Trial of Recombinant Human Chorionic Gonadotropin on Muscle Strength and Physical Function and Activity in Older Men with Partial Age-Related Androgen Deficiency. J. Clin. Endocrinol. Metab. 2002, 87, 3125–3135. [Google Scholar] [CrossRef] [PubMed]
- Carrasquillo, R.; Chu, K.; Ramasamy, R. Novel Therapy for Male Hypogonadism. Curr. Urol. Rep. 2018, 19, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Giagulli, V.A.; Silvestrini, A.; Bruno, C.; Triggiani, V.; Mordente, A.; Mancini, A. Is There Room for SERMs or SARMs as Alternative Therapies for Adult Male Hypogonadism? Int. J. Endocrinol. 2020, 2020. [Google Scholar] [CrossRef]
- Mazzola, C.R.; Katz, D.J.; Loghmanieh, N.; Nelson, C.J.; Mulhall, J.P. Predicting Biochemical Response to Clomiphene Citrate in Men with Hypogonadism. J. Sex. Med. 2014, 11, 2302–2307. [Google Scholar] [CrossRef]
- Cangiano, B.; Cacciatore, C.; Persani, L.; Bonomi, M. Switch to restoration therapy in a testosterone treated central hypogonadism with erythrocytosis. Endocrinol. Diabetes Metab. Case Rep. 2017, 2017. [Google Scholar] [CrossRef]
- Wheeler, K.M.; Smith, R.P.; Kumar, R.A.; Setia, S.; Costabile, R.A.; Kavoussi, P.K. A Comparison of Secondary Polycythemia in Hypogonadal Men Treated with Clomiphene Citrate versus Testosterone Replacement: A Multi-Institutional Study. J. Urol. 2017, 197, 1127–1131. [Google Scholar] [CrossRef]
- Chua, M.E.; Escusa, K.G.; Luna, S.; Tapia, L.C.; Dofitas, B.; Morales, M. Revisiting oestrogen antagonists (clomiphene or tamoxifen) as medical empiric therapy for idiopathic male infertility: A meta-analysis. Andrology 2013, 1, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Shabsigh, A.; Kang, Y.; Shabsign, R.; Gonzalez, M.; Liberson, G.; Fisch, H.; Goluboff, E. Clomiphene citrate effects on testosterone/estrogen ratio in male hypogonadism. J. Sex. Med. 2005, 2, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Taylor, F.; Levine, L. Clomiphene Citrate and Testosterone Gel Replacement Therapy for Male Hypogonadism: Efficacy and Treatment Cost. J. Sex. Med. 2010, 7, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.J.; Nabulsi, O.; Tal, R.; Mulhall, J.P. Outcomes of clomiphene citrate treatment in young hypogonadal men. BJU Int. 2012, 110, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.D.; Mccullough, A.; Kaminetsky, J. Oral enclomiphene citrate raises testosterone and preserves sperm counts in obese hypogonadal men, unlike topical testosterone: Restoration instead of replacement. BJU Int. 2016, 117, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Pelusi, C.; Giagulli, V.A.; Baccini, M.; Fanelli, F.; Mezzullo, M.; Fazzini, A.; Bianchi, N.; Carbone, M.D.; de Pergola, G.; Mastroroberto, M.; et al. Clomiphene citrate effect in obese men with low serum testosterone treated with metformin due to dysmetabolic disorders: A randomized, double-blind, placebo-controlled study. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Wheeler, K.M.; Sharma, D.; Kavoussi, P.K.; Smith, R.P.; Costabile, R. Clomiphene Citrate for the Treatment of Hypogonadism. Sex. Med. Rev. 2019, 7, 272–276. [Google Scholar] [CrossRef]
- A double-blind trial of clomiphene citrate for the treatment of idiopathic male infertility. Int. J. Androl. 1992, 15, 299–307. [CrossRef]
- Kaminetsky, J.; Werner, M.; Fontenot, G.; Wiehle, R.D. Oral enclomiphene citrate stimulates the endogenous production of testosterone and sperm counts in men with low testosterone: Comparison with testosterone gel. J. Sex. Med. 2013, 10, 1628–1635. [Google Scholar] [CrossRef]
- Cannarella, R.; Condorelli, R.A.; Mongioì, L.M.; Barbagallo, F.; Calogero, A.E.; La Vignera, S. Effects of the selective estrogen receptor modulators for the treatment of male infertility: A systematic review and meta-analysis. Expert Opin. Pharmacother. 2019, 20, 1517–1525. [Google Scholar] [CrossRef]
- Ramasamy, R.; Scovell, J.M.; Kovac, J.R.; Lipshultz, L.I. Testosterone Supplementation Versus Clomiphene Citrate for Hypogonadism: An Age Matched Comparison of Satisfaction and Efficacy. J. Urol. 2014, 192, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Soares, A.H.; Horie, N.C.; Chiang, L.A.P.; Caramelli, B.; Matheus, M.G.; Campos, A.H.; Marti, L.C.; Rocha, F.A.; Mancini, M.C.; Costa, E.M.F.; et al. Effects of clomiphene citrate on male obesity-associated hypogonadism: A randomized, double-blind, placebo-controlled study. Int. J. Obes. 2018, 42, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Krzastek, S.C.; Sharma, D.; Abdullah, N.; Sultan, M.; Machen, G.L.; Wenzel, J.L.; Ells, A.; Chen, X.; Kavoussi, M.; Costabile, R.A.; et al. Long-Term Safety and Efficacy of Clomiphene Citrate for the Treatment of Hypogonadism. J. Urol. 2019, 202, 1029–1035. [Google Scholar] [CrossRef] [PubMed]
- Moskovic, D.J.; Katz, D.J.; Akhavan, A.; Park, K.; Mulhall, J.P. Clomiphene citrate is safe and effective for long-term management of hypogonadism. BJU Int. 2012, 110, 1524–1528. [Google Scholar] [CrossRef] [PubMed]
- Wibowo, E.; Pollock, P.A.; Hollis, N.; Wassersug, R.J. Tamoxifen in men: A review of adverse events. Andrology 2016, 4, 776–788. [Google Scholar] [CrossRef] [PubMed]
- Helo, S.; Ellen, J.; Mechlin, C.; Feustel, P.; Grossman, M.; Ditkoff, E.; Mccullough, A. A Randomized Prospective Double-Blind Comparison Trial of Clomiphene Citrate and Anastrozole in Raising Testosterone in Hypogonadal Infertile Men. J. Sex. Med. 2015, 12, 1761–1769. [Google Scholar] [CrossRef]
- Burnett-Bowie, S.-A.M.; Roupenian, K.C.; Dere, M.E.; Lee, H.; Leder, B.Z. Effects of aromatase inhibition in hypogonadal older men: A randomized, double-blind, placebo-controlled trial. Clin. Endocrinol. 2009, 70, 116–123. [Google Scholar] [CrossRef]
- Dias, J.P.; Melvin, D.; Simonsick, E.M.; Carlson, O.; Shardell, M.D.; Ferrucci, L.; Chia, C.W.; Basaria, S.; Egan, J.M. Effects of aromatase inhibition vs. testosterone in older men with low testosterone: Randomized-controlled trial. Andrology 2016, 4, 33–40. [Google Scholar] [CrossRef]
| Therapy | Indication | Modality | Results | Possible Adverse Effects |
|---|---|---|---|---|
| TRT | Organic and functional HG without fertility desire | IM SC Transdermal Nasal Oral | T-levels ++ Sexual function ++ Body composition ++ BMD + | Sperm production ↓ Erythrocytosis ++ PSA ↑ Gynecomastia |
| Non-pharmacological | Reversible causes | Lifestyle modification Weight loss Improved glycemic control in T2DM | T-levels + Sexual function + Body composition + BMD? | / |
| Stop opioids/glucocorticoids | T-levels + Sexual function + Body composition + BMD + | Withdrawal | ||
| Dopamine agonists | HyperPRL | Oral | T-levels + Sexual function + Body composition / BMD + | Headache Orthostatic hypotension Nausea |
| Gonadotropins | Organic and functional HG Fertility | hCG IM/SC +/− FSH/hMG SC | T-levels ++ Spermatogenesis ++ Sexual function? Body composition? BMD? | Gynecomastia Erythrocytosis + |
| Pulsatile GnRH therapy | HG due to hypothalamic disorders | SC/IV pulsatile | T-levels ++ Spermatogenesis ++ Sexual function? Body composition? BMD? | Erythrocytosis + Expensive Requires experience and material |
| Clomiphene citrate Tamoxifen | Functional HG (off-label) | Oral | T-levels + Spermatogenesis + Sexual function? Body composition? BMD? | (Mood changes, blurred vision, breast tenderness, weight gain, VTE) |
| Aromatase inhibitors | Functional HG (off-label) | Oral (Anastrozole or Letrozole) | T-levels + Sexual function / Body composition / BMD− | Osteopenia Hot flashes Weight gain Insomnia (VTE) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ide, V.; Vanderschueren, D.; Antonio, L. Treatment of Men with Central Hypogonadism: Alternatives for Testosterone Replacement Therapy. Int. J. Mol. Sci. 2021, 22, 21. https://doi.org/10.3390/ijms22010021
Ide V, Vanderschueren D, Antonio L. Treatment of Men with Central Hypogonadism: Alternatives for Testosterone Replacement Therapy. International Journal of Molecular Sciences. 2021; 22(1):21. https://doi.org/10.3390/ijms22010021
Chicago/Turabian StyleIde, Veerle, Dirk Vanderschueren, and Leen Antonio. 2021. "Treatment of Men with Central Hypogonadism: Alternatives for Testosterone Replacement Therapy" International Journal of Molecular Sciences 22, no. 1: 21. https://doi.org/10.3390/ijms22010021
APA StyleIde, V., Vanderschueren, D., & Antonio, L. (2021). Treatment of Men with Central Hypogonadism: Alternatives for Testosterone Replacement Therapy. International Journal of Molecular Sciences, 22(1), 21. https://doi.org/10.3390/ijms22010021





