Is There a Place for Immune Checkpoint Inhibitors in Vulvar Neoplasms? A State of the Art Review
Abstract
1. Background
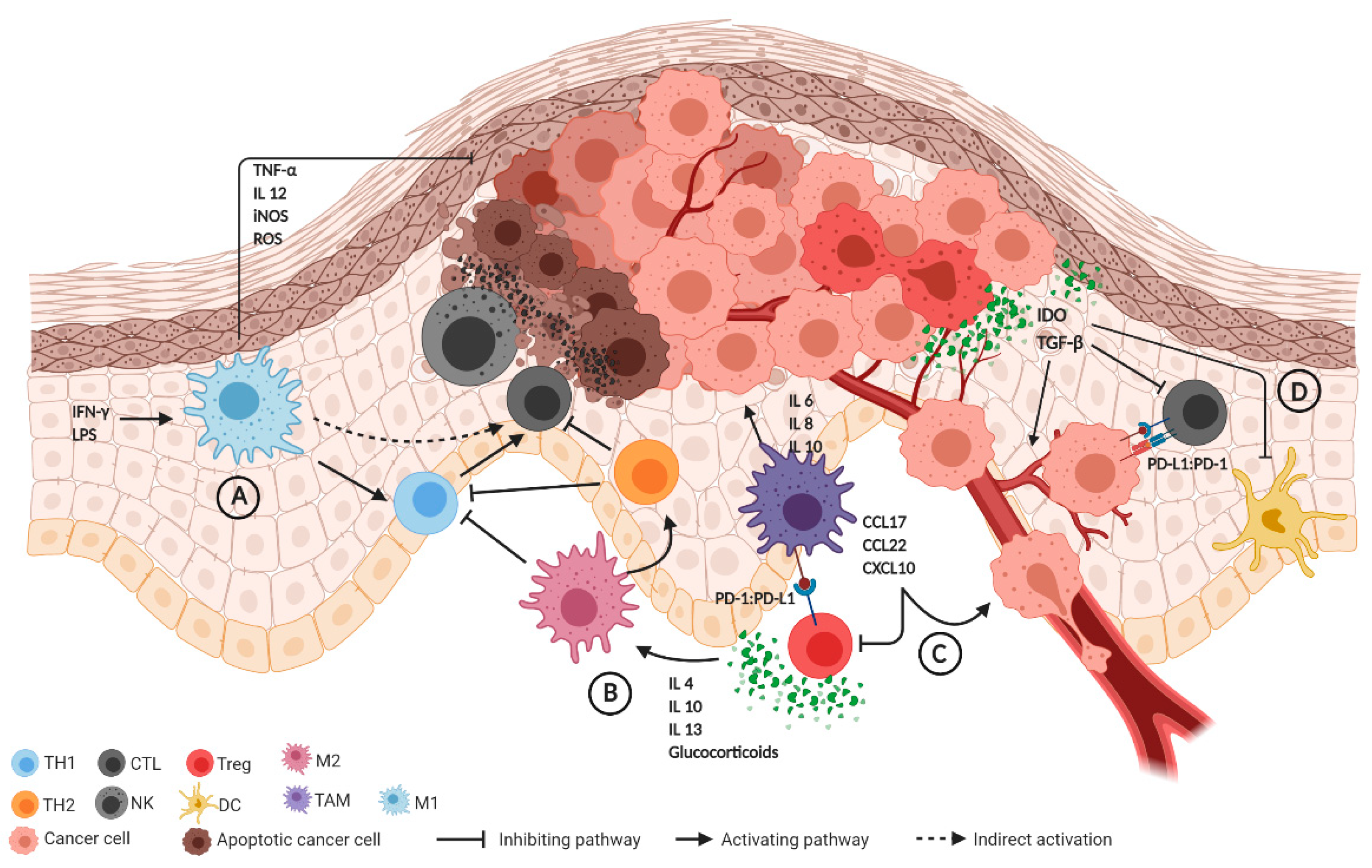
2. Immunopathology of VC
2.1. Innate Immune System
2.2. Adaptive Immune System
2.3. The Role of Immune Checkpoint Regulators
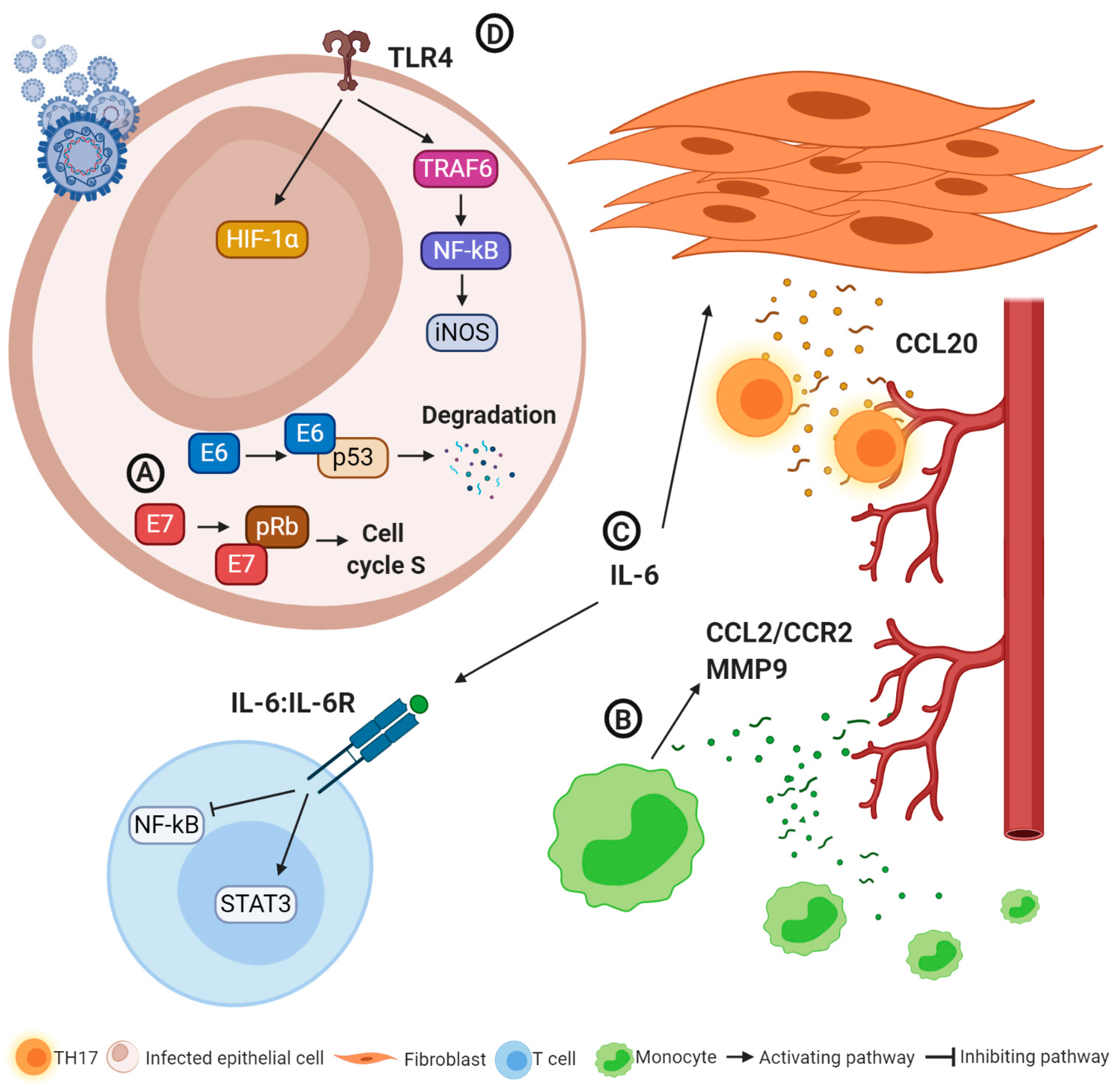
2.4. Immunological Changes Induced by HPV Infections
3. Clinical Role of ICIs in VSCC
4. Potential Role of ICIs in Rare Vulvar Tumor Types
4.1. Vulvar Melanoma
4.2. Vulvar Paget’s Disease
4.3. Neuroendocrine Tumors
5. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AEs | adverse effects |
| APC | antigen-presenting cell |
| CCL | C-C motif chemokine ligand |
| CCR-2 | C-C chemokine receptor type 2 |
| CPS | combined positivity score |
| CR | complete response |
| CTL | cytotoxic T cells |
| CTLA-4 | cytotoxic T-lymphocyte-associated protein 4 |
| CXCL | C-X-C motif chemokine ligand |
| DCs | dendritic cells |
| dMMR | deficient mismatch repair |
| DOI | depth of invasion |
| DSS | disease-specific survival |
| dVIN | differentiated vulvar intraepithelial neoplasia |
| EMPD | extramammary Paget’s disease |
| FDA | Food and Drug Administration |
| FIGO | International Federation of Gynecology and Obstetrics |
| FOXP3 | forkhead box P3 |
| HIF-1α | hypoxia-inducible factor-1α |
| HLA | human leukocyte antigen |
| HPF | high-power field |
| HPV | human papilloma virus |
| ICI | immune checkpoint inhibitor |
| IDO | indoleamine 2,3-dioxygenase |
| IL | interleukin |
| ILR | interleukin receptor |
| INF-γ | interferon-γ |
| iNOS | inducible nitric oxygen synthase |
| LCs | Langerhans cells |
| Lm-LLO | Listeria monocytogenes-listeriolysin lipopolysaccharide |
| LPS | lipopolysaccharide |
| MAGE-A | melanoma-associated antigen |
| MAPK | mitogen-activated protein kinase |
| MHC | major histocompatibility complex |
| MMR | mismatch repair |
| MMP-9 | matrix-metalloproteinase 9 |
| mPFS | median progression-free survival |
| MSCs | myeloid-derived suppressor cells |
| NET | neuroendocrine tumor |
| MSI-h | microsatellite instability-high |
| NF-kB | nuclear aactor kappa-light-chain-enhancer of activated B cells |
| NK | natural killer |
| NKT | natural killer T cells |
| NY-ESO-1 | New York esophageal squamous cell carcinoma 1 |
| ORR | objective response rate |
| OS | overall survival |
| PAMPs | pathogen-associated molecular patterns |
| PD | progression disease |
| PD-1 | programmed death-1 |
| PD-L1 | programmed death-ligand 1 |
| PD-L2 | programmed death-ligand 2 |
| PFS | progression-free survival |
| PR | partial response |
| PRRs | pattern-recognition receptors |
| RANK | receptor activator of nuclear factor kappa-Β |
| Rb | retinoblastoma |
| RECIST | Response Evaluation Criteria in Solid Tumours |
| ROS | reactive oxygen species |
| RFS | recurrence-free survival |
| RT | radiotherapy |
| SD | stable disease |
| SEER | Surveillance, Epidemiology and End Results |
| SLP | synthetic long peptide |
| STAT3 | signal transducer and activator of transcription 3 |
| TAMs | tumor-associated macrophages |
| TGF-β | transforming growth factor-β |
| TH | T helper |
| TH1 | T helper 1 |
| TH2 | T helper 2 |
| TILs | tumor infiltrating lymphocytes |
| TLR | toll-like receptors |
| TMB | tumor mutational burden |
| TNF-α | tumor necrosis factor α |
| TRAF6 | tumor necrosis factor receptor-associated factor 6 |
| T reg | regulatory T cells |
| VC | vulvar cancer |
| VHSIL | vulvar high-grade squamous intraepithelial neoplasia |
| VPD | vulvar Paget’s disease |
| VSCC | vulvar squamous cell carcinoma |
References
- Cancer Stat Facts: Vulvar Cancer. Available online: https://seer.cancer.gov/statfacts/html/vulva.html (accessed on 10 November 2020).
- Kang, Y.J.; Smith, M.; Barlow, E.; Coffey, K.; Hacker, N.; Canfell, K. Vulvar cancer in high-income countries: Increasing burden of disease. Int. J. Cancer 2017, 141, 2174–2186. [Google Scholar] [CrossRef]
- Mancini, S.; Bucchi, L.; Baldacchini, F.; Giuliani, O.; Ravaioli, A.; Vattiato, R.; Preti, M.; Tumino, R.; Ferretti, S.; Biggeri, A.; et al. Incidence trends of vulvar squamous cell carcinoma in Italy from 1990 to 2015. Gynecol. Oncol. 2020, 157, 656–663. [Google Scholar] [CrossRef]
- Panatto, D.; Amicizia, D.; Trucchi, C.; Casabona, F.; Lai, P.L.; Bonanni, P.; Boccalini, S.; Bechini, A.; Tiscione, E.; Zotti, C.M.; et al. Sexual behaviour and risk factors for the acquisition of human papillomavirus infections in young people in Italy: Suggestions for future vaccination policies. BMC Public Health 2012, 12, 623. [Google Scholar] [CrossRef] [PubMed]
- Joura, E.A.; Lösch, A.; Haider-Angeler, M.G.; Breitenecker, G.; Leodolter, S. Trends in vulvar neoplasia. Increasing incidence of vulvar intraepithelial neoplasia and squamous cell carcinoma of the vulva in young women. J. Reprod. Med. 2000, 45, 613–615. [Google Scholar] [PubMed]
- Jones, R.W.; Baranyai, J.; Stables, S. Trends in squamous cell carcinoma of the vulva: The influence of vulvar intraepithelial neoplasia. Obstet. Gynecol. 1997, 90, 448–452. [Google Scholar] [CrossRef]
- Baandrup, L.; Varbo, A.; Munk, C.; Johansen, C.; Frisch, M.; Kjaer, S.K. In situ and invasive squamous cell carcinoma of the vulva in Denmark 1978–2007—A nationwide population-based study. Gynecol. Oncol. 2011, 122, 45–49. [Google Scholar] [CrossRef]
- Schuurman, M.S.; van den Einden, L.C.G.; Massuger, L.F.A.G.; Kiemeney, L.A.; van der Aa, M.A.; de Hullu, J.A. Trends in incidence and survival of Dutch women with vulvar squamous cell carcinoma. Eur. J. Cancer 2013, 49, 3872–3880. [Google Scholar] [CrossRef]
- De Sanjosé, S.; Alemany, L.; Ordi, J.; Tous, S.; Alejo, M.; Bigby, S.M.; Joura, E.A.; Maldonado, P.; Laco, J.; Bravo, I.G.; et al. Worldwide human papillomavirus genotype attribution in over 2000 cases of intraepithelial and invasive lesions of the vulva. Eur. J. Cancer 2013, 49, 3450–3461. [Google Scholar] [CrossRef]
- Hoang, L.N.; Park, K.J.; Soslow, R.A.; Murali, R. Squamous precursor lesions of the vulva: Current classification and diagnostic challenges. Pathology 2016, 48, 291–302. [Google Scholar] [CrossRef]
- Preti, M.; Rotondo, J.C.; Holzinger, D.; Micheletti, L.; Gallio, N.; McKay-Chopin, S.; Carreira, C.; Privitera, S.S.; Watanabe, R.; Ridder, R.; et al. Role of human papillomavirus infection in the etiology of vulvar cancer in Italian women. Infect. Agent. Cancer 2020, 15, 20. [Google Scholar] [CrossRef]
- Xing, D.; Fadare, O. Molecular events in the pathogenesis of vulvar squamous cell carcinoma. Semin. Diagn. Pathol. 2020, 38, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Kortekaas, K.E.; Bastiaannet, E.; van Doorn, H.C.; de Vos van Steenwijk, P.J.; Ewing-Graham, P.C.; Creutzberg, C.L.; Akdeniz, K.; Nooij, L.S.; van der Burg, S.H.; Bosse, T.; et al. Vulvar cancer subclassification by HPV and p53 status results in three clinically distinct subtypes. Gynecol. Oncol. 2020, 159, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Proctor, L.; Hoang, L.; Moore, J.; Thompson, E.; Leung, S.; Natesan, D.; Chino, J.; Gilks, B.; McAlpine, J.N. Association of human papilloma virus status and response to radiotherapy in vulvar squamous cell carcinoma. Int. J. Gynecol. Cancer 2020, 30, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Bruyere, D.; Monnien, F.; Colpart, P.; Roncarati, P.; Vuitton, L.; Hendrick, E.; Lepinoy, A.; Luquain, A.; Pilard, C.; Lerho, T.; et al. Treatment algorithm and prognostic factors for patients with stage I–III carcinoma of the anal canal: A 20-year multicenter study. Mod. Pathol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Nooij, L.S.; ter Haar, N.T.; Ruano, D.; Rakislova, N.; van Wezel, T.; Smit, V.T.H.B.M.; Trimbos, B.J.B.M.Z.; Ordi, J.; van Poelgeest, M.I.E.; Bosse, T. Genomic Characterization of Vulvar (Pre)cancers Identifies Distinct Molecular Subtypes with Prognostic Significance. Clin. Cancer Res. 2017, 23, 6781–6789. [Google Scholar] [CrossRef] [PubMed]
- Pecorelli, S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int. J. Gynecol. Obstet. 2009, 105, 103–104. [Google Scholar] [CrossRef]
- Yoder, B.J.; Rufforny, I.; Massoll, N.A.; Wilkinson, E.J. Stage IA Vulvar Squamous Cell Carcinoma. Am. J. Surg. Pathol. 2008, 32, 765–772. [Google Scholar] [CrossRef]
- Ferrari, F.; Forte, S.; Ardighieri, L.; Bonetti, E.; Fernando, B.; Sartori, E.; Odicino, F. Multivariate analysis of prognostic factors in primary squamous cell vulvar cancer: The role of perineural invasion in recurrence and survival. Eur. J. Surg. Oncol. 2019, 45, 2115–2119. [Google Scholar] [CrossRef]
- Micheletti, L.; Preti, M. Surgery of the vulva in vulvar cancer. Best Pract. Res. Clin. Obstet. Gynaecol. 2014, 28, 1074–1087. [Google Scholar] [CrossRef]
- Kortekaas, K.E.; Santegoets, S.J.; Abdulrahman, Z.; van Ham, V.J.; van der Tol, M.; Ehsan, I.; van Doorn, H.C.; Bosse, T.; van Poelgeest, M.I.E.; van der Burg, S.H. High numbers of activated helper T cells are associated with better clinical outcome in early stage vulvar cancer, irrespective of HPV or p53 status. J. Immunother. Cancer 2019, 7, 236. [Google Scholar] [CrossRef]
- Wohlmuth, C.; Wohlmuth-Wieser, I.; Laframboise, S. Clinical Characteristics and Treatment Response with Checkpoint Inhibitors in Malignant Melanoma of the Vulva and Vagina. J. Low. Genit. Tract Dis. 2020, 4, 12. [Google Scholar] [CrossRef]
- Yoneda, K.; Imanishi, N.; Ichiki, Y.; Tanaka, F. Immune Checkpoint Inhibitors (ICIs) in Non-Small Cell Lung Cancer (NSCLC). J. UOEH 2018, 40, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Musacchio, L.; Boccia, S.M.; Caruso, G.; Santangelo, G.; Fischetti, M.; Tomao, F.; Perniola, G.; Palaia, I.; Muzii, L.; Pignata, S.; et al. Immune Checkpoint Inhibitors: A Promising Choice for Endometrial Cancer Patients? J. Clin. Med. 2020, 9, 1721. [Google Scholar] [CrossRef] [PubMed]
- Attademo, L.; Tuninetti, V.; Pisano, C.; Cecere, S.C.; Di Napoli, M.; Tambaro, R.; Valabrega, G.; Musacchio, L.; Setola, S.V.; Piccirillo, P.; et al. Immunotherapy in cervix cancer. Cancer Treat. Rev. 2020, 90, 102088. [Google Scholar] [CrossRef]
- Borella, F.; Ghisoni, E.; Giannone, G.; Cosma, S.; Benedetto, C.; Valabrega, G.; Katsaros, D. Immune Checkpoint Inhibitors in Epithelial Ovarian Cancer: An Overview on Efficacy and Future Perspectives. Diagnostics 2020, 10, 146. [Google Scholar] [CrossRef]
- Fridman, W.H.; Pagès, F.; Sautès-Fridman, C.; Galon, J. The immune contexture in human tumours: Impact on clinical outcome. Nat. Rev. Cancer 2012, 12, 298–306. [Google Scholar] [CrossRef]
- Chew, V.; Toh, H.C.; Abastado, J.-P. Immune Microenvironment in Tumor Progression: Characteristics and Challenges for Therapy. J. Oncol. 2012, 2012, 608406. [Google Scholar] [CrossRef]
- Galon, J.; Bruni, D. Tumor Immunology and Tumor Evolution: Intertwined Histories. Immunity 2020, 52, 55–81. [Google Scholar] [CrossRef]
- Schenten, D.; Medzhitov, R. The Control of Adaptive Immune Responses by the Innate Immune System. In Advances in Immunology; Academic Press Inc.: Cambridge, AZ, USA, 2011; pp. 87–124. [Google Scholar]
- Rich, R.; Fleisher, T.; Shearer, W.; Schroeder, H.; Frew, A.; Weyand, C. Clinical Immunology: Principles and Practice, 5th ed.; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Hagerling, C.; Casbon, A.-J.; Werb, Z. Balancing the innate immune system in tumor development. Trends Cell Biol. 2015, 25, 214–220. [Google Scholar] [CrossRef]
- Berraondo, P.; Minute, L.; Ajona, D.; Corrales, L.; Melero, I.; Pio, R. Innate immune mediators in cancer: Between defense and resistance. Immunol. Rev. 2016, 274, 290–306. [Google Scholar] [CrossRef]
- Chávez-Galán, L.; Olleros, M.L.; Vesin, D.; Garcia, I. Much More than M1 and M2 Macrophages, There are also CD169+ and TCR+ Macrophages. Front. Immunol. 2015, 6, 263. [Google Scholar] [CrossRef] [PubMed]
- van der Sluis, T.C.; Sluijter, M.; van Duikeren, S.; West, B.L.; Melief, C.J.M.; Arens, R.; van der Burg, S.H.; van Hall, T. Therapeutic Peptide Vaccine-Induced CD8 T Cells Strongly Modulate Intratumoral Macrophages Required for Tumor Regression. Cancer Immunol. Res. 2015, 3, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Schmid, M.C.; Varner, J.A. Myeloid Cells in the Tumor Microenvironment: Modulation of Tumor Angiogenesis and Tumor Inflammation. J. Oncol. 2010, 2010, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.K.; Sica, A.; Lewis, C.E. Plasticity of Macrophage Function during Tumor Progression: Regulation by Distinct Molecular Mechanisms. J. Immunol. 2008, 180, 2011–2017. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Soto, A.; Sierra-Filardi, E.; Puig-Kröger, A.; Pérez-Maceda, B.; Gómez-Aguado, F.; Corcuera, M.T.; Sánchez-Mateos, P.; Corbí, A.L. Dendritic Cell-Specific ICAM-3–Grabbing Nonintegrin Expression on M2-Polarized and Tumor-Associated Macrophages Is Macrophage-CSF Dependent and Enhanced by Tumor-Derived IL-6 and IL-10. J. Immunol. 2011, 186, 2192–2200. [Google Scholar] [CrossRef] [PubMed]
- Wan, S.; Zhao, E.; Kryczek, I.; Vatan, L.; Sadovskaya, A.; Ludema, G.; Simeone, D.M.; Zou, W.; Welling, T.H. Tumor-Associated Macrophages Produce Interleukin 6 and Signal via STAT3 to Promote Expansion of Human Hepatocellular Carcinoma Stem Cells. Gastroenterology 2014, 147, 1393–1404. [Google Scholar] [CrossRef]
- Fu, X.-T.; Dai, Z.; Song, K.; Zhang, Z.-J.; Zhou, Z.-J.; Zhou, S.-L.; Zhao, Y.-M.; Xiao, Y.-S.; Sun, Q.-M.; Ding, Z.-B.; et al. Macrophage-secreted IL-8 induces epithelial-mesenchymal transition in hepatocellular carcinoma cells by activating the JAK2/STAT3/Snail pathway. Int. J. Oncol. 2015, 46, 587–596. [Google Scholar] [CrossRef]
- Noy, R.; Pollard, J.W. Tumor-Associated Macrophages: From Mechanisms to Therapy. Immunity 2014, 41, 49–61. [Google Scholar] [CrossRef]
- Fujimura, T.; Aiba, S. Significance of Immunosuppressive Cells as a Target for Immunotherapies in Melanoma and Non-Melanoma Skin Cancers. Biomolecules 2020, 10, 1087. [Google Scholar] [CrossRef]
- Ohue, Y.; Nishikawa, H. Regulatory T (Treg) cells in cancer: Can Treg cells be a new therapeutic target? Cancer Sci. 2019, 110, 2080–2089. [Google Scholar] [CrossRef]
- Welters, M.J.; van der Sluis, T.C.; van Meir, H.; Loof, N.M.; van Ham, V.J.; van Duikeren, S.; Santegoets, S.J.; Arens, R.; de Kam, M.L.; Cohen, A.F.; et al. Vaccination during myeloid cell depletion by cancer chemotherapy fosters robust T cell responses. Sci. Transl. Med. 2016, 8, 334ra52. [Google Scholar] [CrossRef] [PubMed]
- van Esch, E.M.G.; van Poelgeest, M.I.E.; Trimbos, J.B.M.Z.; Fleuren, G.J.; Jordanova, E.S.; van der Burg, S.H. Intraepithelial macrophage infiltration is related to a high number of regulatory T cells and promotes a progressive course of HPV-induced vulvar neoplasia. Int. J. Cancer 2015, 136, E85–E94. [Google Scholar] [CrossRef] [PubMed]
- Hecking, T.; Thiesler, T.; Schiller, C.; Lunkenheimer, J.-M.; Ayub, T.H.; Rohr, A.; Condic, M.; Keyver-Paik, M.-D.; Fimmers, R.; Kirfel, J.; et al. Tumoral PD-L1 expression defines a subgroup of poor-prognosis vulvar carcinomas with non-viral etiology. Oncotarget 2017, 8, 92890–92903. [Google Scholar] [CrossRef] [PubMed]
- Sznurkowski, J.J.; Żawrocki, A.; Biernat, W. Local immune response depends on p16INK4a status of primary tumor in vulvar squamous cell carcinoma. Oncotarget 2017, 8, 46204–46210. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Veglia, F.; Gabrilovich, D.I. Dendritic cells in cancer: The role revisited. Curr. Opin. Immunol. 2017, 45, 43–51. [Google Scholar] [CrossRef]
- Wculek, S.K.; Cueto, F.J.; Mujal, A.M.; Melero, I.; Krummel, M.F.; Sancho, D. Dendritic cells in cancer immunology and immunotherapy. Nat. Rev. Immunol. 2020, 20, 7–24. [Google Scholar] [CrossRef]
- Deckers, J.; Hammad, H.; Hoste, E. Langerhans Cells: Sensing the Environment in Health and Disease. Front. Immunol. 2018, 9, 93. [Google Scholar] [CrossRef]
- Rotsztejn, H.; Trznadel-Budźko, E.; Jesionek-Kupnicka, D. Langerhans cells in vulvar lichen sclerosus and vulvar squamous cell carcinoma. Arch. Immunol. Ther. Exp. (Warsz) 2006, 54, 363–366. [Google Scholar] [CrossRef]
- Sznurkowski, J.J.; Żawrocki, A.; Biernat, W. Subtypes of cytotoxic lymphocytes and natural killer cells infiltrating cancer nests correlate with prognosis in patients with vulvar squamous cell carcinoma. Cancer Immunol. Immunother. 2014, 63, 297–303. [Google Scholar] [CrossRef][Green Version]
- Vivier, E.; Ugolini, S.; Blaise, D.; Chabannon, C.; Brossay, L. Targeting natural killer cells and natural killer T cells in cancer. Nat. Rev. Immunol. 2012, 12, 239–252. [Google Scholar] [CrossRef]
- Terabe, M.; Berzofsky, J.A. NKT cells in immunoregulation of tumor immunity: A new immunoregulatory axis. Trends Immunol. 2007, 28, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Sznurkowski, J.J.; Żawrocki, A.; Emerich, J.; Sznurkowska, K.; Biernat, W. Expression of indoleamine 2,3-dioxygenase predicts shorter survival in patients with vulvar squamous cell carcinoma (vSCC) not influencing on the recruitment of FOXP3-expressing regulatory T cells in cancer nests. Gynecol. Oncol. 2011, 122, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.-S.; Lee, S.-M.; Kim, M.-K.; Park, S.-G.; Choi, I.-W.; Choi, I.; Joo, Y.-D.; Park, S.-J.; Kang, S.-W.; Seo, S.-K. The tryptophan metabolite 3-hydroxyanthranilic acid suppresses T cell responses by inhibiting dendritic cell activation. Int. Immunopharmacol. 2013, 17, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Chinn, Z.; Stoler, M.H.; Mills, A.M. PD-L1 and IDO expression in cervical and vulvar invasive and intraepithelial squamous neoplasias: Implications for combination immunotherapy. Histopathology 2019, 74, 256–268. [Google Scholar] [CrossRef]
- Boujelbene, N.; Ben Yahia, H.; Babay, W.; Gadria, S.; Zemni, I.; Azaiez, H.; Dhouioui, S.; Zidi, N.; Mchiri, R.; Mrad, K.; et al. HLA-G, HLA-E, and IDO overexpression predicts a worse survival of Tunisian patients with vulvar squamous cell carcinoma. HLA 2019, 94, 11–24. [Google Scholar] [CrossRef]
- Chaplin, D.D. Overview of the immune response. J. Allergy Clin. Immunol. 2010, 125, S3–S23. [Google Scholar] [CrossRef]
- Spurrell, E.L.; Lockley, M. Adaptive immunity in cancer immunology and therapeutics. Ecancermedicalscience 2014, 8, 441. [Google Scholar] [CrossRef]
- Ostrand-Rosenberg, S. Immune surveillance: A balance between protumor and antitumor immunity. Curr. Opin. Genet. Dev. 2008, 18, 11–18. [Google Scholar] [CrossRef]
- Karoń, P.; Olejek, A.; Olszak-Wąsik, K. TGF-β expression in vulvar cancer. Pol. Gynaecol. 2014, 85, 847–851. [Google Scholar] [CrossRef]
- Liu, S.; Chen, S.; Zeng, J. TGF-β signaling: A complex role in tumorigenesis (Review). Mol. Med. Rep. 2017, 17, 699–704. [Google Scholar] [CrossRef]
- Batlle, E.; Massagué, J. Transforming Growth Factor-β Signaling in Immunity and Cancer. Immunity 2019, 50, 924–940. [Google Scholar] [CrossRef] [PubMed]
- Sznurkowski, J.J.; Żawrocki, A.; Emerich, J.; Biernat, W. Prognostic Significance of CD4 + and CD8 + T Cell Infiltration Within Cancer Cell Nests in Vulvar Squamous Cell Carcinoma. Int. J. Gynecol. Cancer 2011, 21, 717–721. [Google Scholar] [CrossRef] [PubMed]
- de Jong, R.A.; Toppen, N.L.; ten Hoor, K.A.; Boezen, H.M.; Kema, I.P.; Hollema, H.; Nijman, H.W. Status of cellular immunity lacks prognostic significance in vulvar squamous carcinoma. Gynecol. Oncol. 2012, 125, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Menon, S.; Shin, S.; Dy, G. Advances in Cancer Immunotherapy in Solid Tumors. Cancers 2016, 8, 106. [Google Scholar] [CrossRef]
- Kooshkaki, O.; Derakhshani, A.; Safarpour, H.; Najafi, S.; Vahedi, P.; Brunetti, O.; Torabi, M.; Lotfinejad, P.; Paradiso, A.V.; Racanelli, V.; et al. The Latest Findings of PD-1/PD-L1 Inhibitor Application in Gynecologic Cancers. Int. J. Mol. Sci. 2020, 21, 5034. [Google Scholar] [CrossRef]
- Giannone, G.; Ghisoni, E.; Genta, S.; Scotto, G.; Tuninetti, V.; Turinetto, M.; Valabrega, G. Immuno-Metabolism and Microenvironment in Cancer: Key Players for Immunotherapy. Int. J. Mol. Sci. 2020, 21, 4414. [Google Scholar] [CrossRef]
- Herbst, R.S.; Baas, P.; Kim, D.-W.; Felip, E.; Pérez-Gracia, J.L.; Han, J.-Y.; Molina, J.; Kim, J.-H.; Arvis, C.D.; Ahn, M.-J.; et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomised controlled trial. Lancet 2016, 387, 1540–1550. [Google Scholar] [CrossRef]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Pembrolizumab versus Chemotherapy for PD-L1–Positive Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2016, 375, 1823–1833. [Google Scholar] [CrossRef]
- Balar, A.V.; Castellano, D.; O’Donnell, P.H.; Grivas, P.; Vuky, J.; Powles, T.; Plimack, E.R.; Hahn, N.M.; de Wit, R.; Pang, L.; et al. First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (KEYNOTE-052): A multicentre, single-arm, phase 2 study. Lancet Oncol. 2017, 18, 1483–1492. [Google Scholar] [CrossRef]
- Apolo, A.B.; Infante, J.R.; Balmanoukian, A.; Patel, M.R.; Wang, D.; Kelly, K.; Mega, A.E.; Britten, C.D.; Ravaud, A.; Mita, A.C.; et al. Avelumab, an Anti–Programmed Death-Ligand 1 Antibody, In Patients With Refractory Metastatic Urothelial Carcinoma: Results From a Multicenter, Phase Ib Study. J. Clin. Oncol. 2017, 35, 2117–2124. [Google Scholar] [CrossRef]
- Mezache, L.; Paniccia, B.; Nyinawabera, A.; Nuovo, G.J. Enhanced expression of PD L1 in cervical intraepithelial neoplasia and cervical cancers. Mod. Pathol. 2015, 28, 1594–1602. [Google Scholar] [CrossRef] [PubMed]
- Badoual, C.; Hans, S.; Merillon, N.; Van Ryswick, C.; Ravel, P.; Benhamouda, N.; Levionnois, E.; Nizard, M.; Si-Mohamed, A.; Besnier, N.; et al. PD-1–Expressing Tumor-Infiltrating T Cells Are a Favorable Prognostic Biomarker in HPV-Associated Head and Neck Cancer. Cancer Res. 2013, 73, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Lyford-Pike, S.; Peng, S.; Young, G.D.; Taube, J.M.; Westra, W.H.; Akpeng, B.; Bruno, T.C.; Richmon, J.D.; Wang, H.; Bishop, J.A.; et al. Evidence for a Role of the PD-1:PD-L1 Pathway in Immune Resistance of HPV-Associated Head and Neck Squamous Cell Carcinoma. Cancer Res. 2013, 73, 1733–1741. [Google Scholar] [CrossRef] [PubMed]
- Cocks, M.; Chaux, A.; Jenson, E.G.; Miller, J.A.; Rodriguez Pena, M.D.C.; Tregnago, A.C.; Taheri, D.; Eich, M.-L.; Sharma, R.; Vang, R.; et al. Immune checkpoint status and tumor microenvironment in vulvar squamous cell carcinoma. Virchows Arch. 2020, 477, 93–102. [Google Scholar] [CrossRef]
- Lérias, S.; Esteves, S.; Silva, F.; Cunha, M.; Cochicho, D.; Martins, L.; Félix, A. CD274 (PD-L1), CDKN2A (p16), TP53, and EGFR immunohistochemical profile in primary, recurrent and metastatic vulvar cancer. Mod. Pathol. 2020, 33, 893–904. [Google Scholar] [CrossRef]
- Sznurkowski, J.J.; Żawrocki, A.; Sznurkowska, K.; Pęksa, R.; Biernat, W. PD-L1 expression on immune cells is a favorable prognostic factor for vulvar squamous cell carcinoma patients. Oncotarget 2017, 8, 89903–89912. [Google Scholar] [CrossRef]
- Howitt, B.E.; Sun, H.H.; Roemer, M.G.M.; Kelley, A.; Chapuy, B.; Aviki, E.; Pak, C.; Connelly, C.; Gjini, E.; Shi, Y.; et al. Genetic Basis for PD-L1 Expression in Squamous Cell Carcinomas of the Cervix and Vulva. JAMA Oncol. 2016, 2, 518. [Google Scholar] [CrossRef]
- Choschzick, M.; Gut, A.; Fink, D. PD-L1 receptor expression in vulvar carcinomas is HPV-independent. Virchows Arch. 2018, 473, 513–516. [Google Scholar] [CrossRef]
- Thangarajah, F.; Morgenstern, B.; Pahmeyer, C.; Schiffmann, L.M.; Puppe, J.; Mallmann, P.; Hamacher, S.; Buettner, R.; Alidousty, C.; Holz, B.; et al. Clinical impact of PD-L1 and PD-1 expression in squamous cell cancer of the vulva. J. Cancer Res. Clin. Oncol. 2019, 145, 1651–1660. [Google Scholar] [CrossRef]
- Egawa, N.; Egawa, K.; Griffin, H.; Doorbar, J. Human Papillomaviruses; Epithelial Tropisms, and the Development of Neoplasia. Viruses 2015, 7, 3863–3890. [Google Scholar] [CrossRef]
- Smola, S. Immunopathogenesis of HPV-Associated Cancers and Prospects for Immunotherapy. Viruses 2017, 9, 254. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, N.; Bosch, F.X.; de Sanjosé, S.; Herrero, R.; Castellsagué, X.; Shah, K.V.; Snijders, P.J.F.; Meijer, C.J.L.M. Epidemiologic Classification of Human Papillomavirus Types Associated with Cervical Cancer. N. Engl. J. Med. 2003, 348, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Bosch, F.X.; Broker, T.R.; Forman, D.; Moscicki, A.-B.; Gillison, M.L.; Doorbar, J.; Stern, P.L.; Stanley, M.; Arbyn, M.; Poljak, M.; et al. Comprehensive Control of Human Papillomavirus Infections and Related Diseases. Vaccine 2013, 31, H1–H31. [Google Scholar] [CrossRef]
- van der Burg, S.H.; de Jong, A.; Welters, M.J.; Offringa, R.; Melief, C.J. The status of HPV16-specific T-cell reactivity in health and disease as a guide to HPV vaccine development. Virus Res. 2002, 89, 275–284. [Google Scholar] [CrossRef]
- Bergers, G.; Brekken, R.; McMahon, G.; Vu, T.H.; Itoh, T.; Tamaki, K.; Tanzawa, K.; Thorpe, P.; Itohara, S.; Werb, Z.; et al. Matrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesis. Nat. Cell Biol. 2000, 2, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Schroer, N.; Pahne, J.; Walch, B.; Wickenhauser, C.; Smola, S. Molecular Pathobiology of Human Cervical High-Grade Lesions: Paracrine STAT3 Activation in Tumor-Instructed Myeloid Cells Drives Local MMP-9 Expression. Cancer Res. 2011, 71, 87–97. [Google Scholar] [CrossRef]
- Sansone, P.; Bromberg, J. Targeting the Interleukin-6/Jak/Stat Pathway in Human Malignancies. J. Clin. Oncol. 2012, 30, 1005–1014. [Google Scholar] [CrossRef]
- Walch-Ruckheim, B.; Mavrova, R.; Henning, M.; Vicinus, B.; Kim, Y.-J.; Bohle, R.M.; Juhasz-Boss, I.; Solomayer, E.-F.; Smola, S. Stromal Fibroblasts Induce CCL20 through IL6/C/EBP to Support the Recruitment of Th17 Cells during Cervical Cancer Progression. Cancer Res. 2015, 75, 5248–5259. [Google Scholar] [CrossRef]
- DeCarlo, C.A.; Rosa, B.; Jackson, R.; Niccoli, S.; Escott, N.G.; Zehbe, I. Toll-Like Receptor Transcriptome in the HPV-Positive Cervical Cancer Microenvironment. Clin. Dev. Immunol. 2012, 2012, 785825. [Google Scholar] [CrossRef]
- Yang, X.; Cheng, Y.; Li, C. The role of TLRs in cervical cancer with HPV infection: A review. Signal Transduct. Target. Ther. 2017, 2, 17055. [Google Scholar] [CrossRef]
- Cheng, Y.; Chen, G.; Wang, X.; Huang, Y.; Ding, J.; Huang, J.; Hong, L. TLR4 May Accelerate Hypoxia Reaction to Promote the Occurrence and Progress of Cervical Lesions by Infected Pathogenic Microorganisms Other than HPV. J. Cancer Ther. 2013, 4, 549–553. [Google Scholar] [CrossRef][Green Version]
- Rahkola, P.; Mikkola, T.S.; Ylikorkala, O.; Vaisanen-Tommiska, M. Association between high risk papillomavirus DNA and nitric oxide release in the human uterine cervix. Gynecol. Oncol. 2009, 114, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.-J.; Han, S.-C.; Yoon, W.-J.; Koh, Y.-S.; Hyun, J.-W.; Kang, H.-K.; Youl Cho, J.; Yoo, E.-S. Sargaquinoic acid isolated from Sargassum siliquastrum inhibits lipopolysaccharide-induced nitric oxide production in macrophages via modulation of nuclear factor-κB and c-Jun N -terminal kinase pathways. Immunopharmacol. Immunotoxicol. 2013, 35, 80–87. [Google Scholar] [CrossRef] [PubMed]
- DONG, J.; CHENG, M.; SUN, H. Function of inducible nitric oxide synthase in the regulation of cervical cancer cell proliferation and the expression of vascular endothelial growth factor. Mol. Med. Rep. 2014, 9, 583–589. [Google Scholar] [CrossRef]
- Garland, S.M.; Paavonen, J.; Jaisamrarn, U.; Naud, P.; Salmerón, J.; Chow, S.; Apter, D.; Castellsagué, X.; Teixeira, J.C.; Skinner, S.R.; et al. Prior human papillomavirus-16/18 AS04-adjuvanted vaccination prevents recurrent high grade cervical intraepithelial neoplasia after definitive surgical therapy: Post-hoc analysis from a randomized controlled trial. Int. J. Cancer 2016, 139, 2812–2826. [Google Scholar] [CrossRef]
- Hansen, B.T.; Campbell, S.; Nygård, M. Long-term incidence trends of HPV-related cancers, and cases preventable by HPV vaccination: A registry-based study in Norway. BMJ Open 2018, 8, e019005. [Google Scholar] [CrossRef]
- Garbuglia, A.R.; Lapa, D.; Sias, C.; Capobianchi, M.R.; Del Porto, P. The Use of Both Therapeutic and Prophylactic Vaccines in the Therapy of Papillomavirus Disease. Front. Immunol. 2020, 11, 188. [Google Scholar] [CrossRef]
- Miles, B.; Safran, H.P.; Monk, B.J. Therapeutic options for treatment of human papillomavirus-associated cancers—novel immunologic vaccines: ADXS11–001. Gynecol. Oncol. Res. Pract. 2017, 4, 10. [Google Scholar] [CrossRef]
- Basu, P.; Mehta, A.; Jain, M.; Gupta, S.; Nagarkar, R.V.; John, S.; Petit, R. A Randomized Phase 2 Study of ADXS11-001 Listeria monocytogenes–Listeriolysin O Immunotherapy With or Without Cisplatin in Treatment of Advanced Cervical Cancer. Int. J. Gynecol. Cancer 2018, 28, 764–772. [Google Scholar] [CrossRef]
- Huh, W.K.; Brady, W.E.; Fracasso, P.M.; Dizon, D.S.; Powell, M.A.; Monk, B.J.; Leath, C.A.; Landrum, L.M.; Tanner, E.J.; Crane, E.K.; et al. Phase II study of axalimogene filolisbac (ADXS-HPV) for platinum-refractory cervical carcinoma: An NRG oncology/gynecologic oncology group study. Gynecol. Oncol. 2020, 158, 562–569. [Google Scholar] [CrossRef]
- Ott, P.A.; Bang, Y.-J.; Piha-Paul, S.A.; Razak, A.R.A.; Bennouna, J.; Soria, J.-C.; Rugo, H.S.; Cohen, R.B.; O’Neil, B.H.; Mehnert, J.M.; et al. T-Cell–Inflamed Gene-Expression Profile, Programmed Death Ligand 1 Expression, and Tumor Mutational Burden Predict Efficacy in Patients Treated With Pembrolizumab Across 20 Cancers: KEYNOTE-028. J. Clin. Oncol. 2019, 37, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Naumann, R.W.; Hollebecque, A.; Meyer, T.; Devlin, M.-J.; Oaknin, A.; Kerger, J.; López-Picazo, J.M.; Machiels, J.-P.; Delord, J.-P.; Evans, T.R.J.; et al. Safety and Efficacy of Nivolumab Monotherapy in Recurrent or Metastatic Cervical, Vaginal, or Vulvar Carcinoma: Results From the Phase I/II CheckMate 358 Trial. J. Clin. Oncol. 2019, 37, 2825–2834. [Google Scholar] [CrossRef] [PubMed]
- Shields, L.B.E.; Gordinier, M.E. Pembrolizumab in Recurrent Squamous Cell Carcinoma of the Vulva: Case Report and Review of the Literature. Gynecol. Obstet. Investig. 2019, 84, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Yeku, O.; Russo, A.L.; Lee, H.; Spriggs, D. A phase 2 study of combined chemo-immunotherapy with cisplatin-pembrolizumab and radiation for unresectable vulvar squamous cell carcinoma. J. Transl. Med. 2020, 18, 350. [Google Scholar] [CrossRef]
- Gong, J.; Le, T.Q.; Massarelli, E.; Hendifar, A.E.; Tuli, R. Radiation therapy and PD-1/PD-L1 blockade: The clinical development of an evolving anticancer combination. J. Immunother. Cancer 2018, 6, 46. [Google Scholar] [CrossRef]
- Quéreux, G.; Wylomanski, S.; Bouquin, R.; Saint-Jean, M.; Peuvrel, L.; Knol, A.C.; Hanf, M.; Dréno, B. Are checkpoint inhibitors a valuable option for metastatic or unresectable vulvar and vaginal melanomas? J. Eur. Acad. Dermatol. Venereol. 2018, 32, e39–e40. [Google Scholar] [CrossRef]
- Strosberg, J.; Mizuno, N.; Doi, T.; Grande, E.; Delord, J.-P.; Shapira-Frommer, R.; Bergsland, E.; Shah, M.; Fakih, M.; Takahashi, S.; et al. Efficacy and Safety of Pembrolizumab in Previously Treated Advanced Neuroendocrine Tumors: Results From the Phase II KEYNOTE-158 Study. Clin. Cancer Res. 2020, 26, 2124–2130. [Google Scholar] [CrossRef]
- Hou, J.Y.; Baptiste, C.; Hombalegowda, R.B.; Tergas, A.I.; Feldman, R.; Jones, N.L.; Chatterjee-Paer, S.; Bus-Kwolfski, A.; Wright, J.D.; Burke, W.M. Vulvar and vaginal melanoma: A unique subclass of mucosal melanoma based on a comprehensive molecular analysis of 51 cases compared with 2253 cases of nongynecologic melanoma. Cancer 2017, 123, 1333–1344. [Google Scholar] [CrossRef]
- Saglam, O.; Naqvi, S.M.H.; Zhang, Y.; Mesa, T.; Teer, J.K.; Yoder, S.; Lee, J.; Messina, J. Female genitourinary tract melanoma. Melanoma Res. 2018, 28, 586–591. [Google Scholar] [CrossRef]
- Pleunis, N.; Schuurman, M.S.; Van Rossum, M.M.; Bulten, J.; Massuger, L.F.; De Hullu, J.A.; Van der Aa, M.A. Rare vulvar malignancies; incidence, treatment and survival in the Netherlands. Gynecol. Oncol. 2016, 142, 440–445. [Google Scholar] [CrossRef]
- Sinasac, S.E.; Petrella, T.M.; Rouzbahman, M.; Sade, S.; Ghazarian, D.; Vicus, D. Melanoma of the Vulva and Vagina: Surgical Management and Outcomes Based on a Clinicopathologic Reviewof 68 Cases. J. Obstet. Gynaecol. Can. 2019, 41, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Wohlmuth, C.; Wohlmuth-Wieser, I.; May, T.; Vicus, D.; Gien, L.T.; Laframboise, S. Malignant Melanoma of the Vulva and Vagina: A US Population-Based Study of 1863 Patients. Am. J. Clin. Dermatol. 2020, 21, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.A.; Wolchok, J.D.; Sznol, M. Immunotherapy of Melanoma: Facts and Hopes. Clin. Cancer Res. 2019, 25, 5191–5201. [Google Scholar] [CrossRef] [PubMed]
- Petrova, V.; Arkhypov, I.; Weber, R.; Groth, C.; Altevogt, P.; Utikal, J.; Umansky, V. Modern Aspects of Immunotherapy with Checkpoint Inhibitors in Melanoma. Int. J. Mol. Sci. 2020, 21, 2367. [Google Scholar] [CrossRef]
- Testori, A.A.E.; Chiellino, S.; van Akkooi, A.C.J. Adjuvant Therapy for Melanoma: Past, Current, and Future Developments. Cancers 2020, 12, 1994. [Google Scholar] [CrossRef]
- Tumeh, P.C.; Harview, C.L.; Yearley, J.H.; Shintaku, I.P.; Taylor, E.J.M.; Robert, L.; Chmielowski, B.; Spasic, M.; Henry, G.; Ciobanu, V.; et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 2014, 515, 568–571. [Google Scholar] [CrossRef]
- Marzagalli, M.; Ebelt, N.D.; Manuel, E.R. Unraveling the crosstalk between melanoma and immune cells in the tumor microenvironment. Semin. Cancer Biol. 2019, 59, 236–250. [Google Scholar] [CrossRef]
- Gerber, A.L.; Münst, A.; Schlapbach, C.; Shafighi, M.; Kiermeir, D.; Hüsler, R.; Hunger, R.E. High expression of FOXP3 in primary melanoma is associated with tumour progression. Br. J. Dermatol. 2014, 170, 103–109. [Google Scholar] [CrossRef]
- Hino, R.; Kabashima, K.; Kato, Y.; Yagi, H.; Nakamura, M.; Honjo, T.; Okazaki, T.; Tokura, Y. Tumor cell expression of programmed cell death-1 ligand 1 is a prognostic factor for malignant melanoma. Cancer 2010, 116, 1757–1766. [Google Scholar] [CrossRef]
- Chłopik, A.; Selim, M.A.; Peng, Y.; Wu, C.-L.; Tell-Marti, G.; Paral, K.M.; Shalin, S.C.; Kraft, S.; Hsu, C.-K.; Shea, C.R.; et al. Prognostic role of tumoral PDL1 expression and peritumoral FoxP3+ lymphocytes in vulvar melanomas. Hum. Pathol. 2018, 73, 176–183. [Google Scholar] [CrossRef]
- Kakavand, H.; Vilain, R.E.; Wilmott, J.S.; Burke, H.; Yearley, J.H.; Thompson, J.F.; Hersey, P.; Long, G.V.; Scolyer, R.A. Tumor PD-L1 expression, immune cell correlates and PD-1+ lymphocytes in sentinel lymph node melanoma metastases. Mod. Pathol. 2015, 28, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Shang, B.; Liu, Y.; Jiang, S.; Liu, Y. Prognostic value of tumor-infiltrating FoxP3+ regulatory T cells in cancers: A systematic review and meta-analysis. Sci. Rep. 2015, 5, 15179. [Google Scholar] [CrossRef] [PubMed]
- Saleh, B.; Kriegsmann, J.; Falk, S.; Aulmann, S. Frequent PD-L1 Expression in Malignant Melanomas of the Vulva. Int. J. Gynecol. Pathol. 2018, 37, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Tse, K.-Y.; Lee, H.H.Y.; Chow, K.-L.; Tsang, H.-W.; Wong, R.W.C.; Cheung, E.T.Y.; Cheuk, W.; Lee, V.W.K.; Chan, W.-K.; et al. Predictive biomarkers and tumor microenvironment in female genital melanomas: A multi-institutional study of 55 cases. Mod. Pathol. 2020, 33, 138–152. [Google Scholar] [CrossRef]
- Thierauf, J.; Veit, J.A.; Hess, J.; Treiber, N.; Lisson, C.; Weissinger, S.E.; Bommer, M.; Hoffmann, T.K. Checkpoint inhibition for advanced mucosal melanoma. Eur. J. Dermatol. 2017, 27, 160–165. [Google Scholar] [CrossRef]
- D’Angelo, S.P.; Larkin, J.; Sosman, J.A.; Lebbé, C.; Brady, B.; Neyns, B.; Schmidt, H.; Hassel, J.C.; Hodi, F.S.; Lorigan, P.; et al. Efficacy and Safety of Nivolumab Alone or in Combination With Ipilimumab in Patients With Mucosal Melanoma: A Pooled Analysis. J. Clin. Oncol. 2017, 35, 226–235. [Google Scholar] [CrossRef]
- Li, J.; Kan, H.; Zhao, L.; Sun, Z.; Bai, C. Immune checkpoint inhibitors in advanced or metastatic mucosal melanoma: A systematic review. Ther. Adv. Med. Oncol. 2020, 12, 175883592092202. [Google Scholar] [CrossRef]
- Anko, M.; Nakamura, M.; Kobayashi, Y.; Tsuji, K.; Nakada, S.; Nakamura, Y.; Funakoshi, T.; Banno, K.; Aoki, D. Primary malignant melanoma of the uterine cervix or vagina which were successfully treated with nivolumab. J. Obstet. Gynaecol. Res. 2020, 46, 190–195. [Google Scholar] [CrossRef]
- Kim, M.S.; Choi, C.-H.; Kim, T.-J.; Lee, J.-W.; Lee, J.; Bae, D.-S.; Kim, B.-G. Primary malignant melanoma of the uterine cervix treated with pembrolizumab after radical surgery: A case report and literature review. Obstet. Gynecol. Sci. 2018, 61, 524. [Google Scholar] [CrossRef]
- Schiavone, M.B.; Broach, V.; Shoushtari, A.N.; Carvajal, R.D.; Alektiar, K.; Kollmeier, M.A.; Abu-Rustum, N.R.; Leitao, M.M. Combined immunotherapy and radiation for treatment of mucosal melanomas of the lower genital tract. Gynecol. Oncol. Rep. 2016, 16, 42–46. [Google Scholar] [CrossRef]
- Carcangiu, M.L.; Kurman, R.J.; Carcangiu, M.L.; Herrington, C.S. Tumours of the Vulva. In WHO Classification of Tumours of Female Reproductive Organs; International Agency for Research on Cancer: Lyon, France, 2014; pp. 236–237. [Google Scholar]
- Wilkinson, E.J.; Brown, H.M. Vulvar Paget disease of urothelial origin: A report of three cases and a proposed classification of vulvar Paget disease. Hum. Pathol. 2002, 33, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Hatta, N.; Yamada, M.; Hirano, T.; Fujimoto, A.; Morita, R. Extramammary Paget’s disease: Treatment, prognostic factors and outcome in 76 patients. Br. J. Dermatol. 2008, 158, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Preti, M.; Micheletti, L.; Massobrio, M.; Ansai, S.; Wilkinson, E.J. Vulvar Paget Disease: One Century After First Reported. J. Low. Genit. Tract Dis. 2003, 7, 122–135. [Google Scholar] [CrossRef] [PubMed]
- van der Linden, M.; Meeuwis, K.A.P.; Bulten, J.; Bosse, T.; van Poelgeest, M.I.E.; de Hullu, J.A. Paget disease of the vulva. Crit. Rev. Oncol. Hematol. 2016, 101, 60–74. [Google Scholar] [CrossRef] [PubMed]
- van der Linden, M.; Oonk, M.H.M.; van Doorn, H.C.; Bulten, J.; van Dorst, E.B.L.; Fons, G.; Lok, C.A.R.; van Poelgeest, M.I.E.; Slangen, B.M.F.; Massuger, L.F.A.G.; et al. Vulvar Paget disease: A national retrospective cohort study. J. Am. Acad. Dermatol. 2019, 81, 956–962. [Google Scholar] [CrossRef] [PubMed]
- Carton, I.; Lebreton, M.; Tesson, C.; Henno, S.; Lavoué, V.; Levêque, J.; Nyangoh-Timoh, K. Paget’s disease of the vulva: A challenge for the gynaecologist. J. Gynecol. Obstet. Hum. Reprod. 2020, 101896. [Google Scholar] [CrossRef]
- Sopracordevole, F.; Di Giuseppe, J.; Giorda, G.; Alessandrini, L.; Canzonieri, V.; Ciavattini, A. Extramammary Paget disease of the vulva (VEMPD) with perianal involvement: A 30-year experience. G. Ital. Dermatol. Venereol. 2018. [Google Scholar] [CrossRef]
- Press, J.Z.; Allison, K.H.; Garcia, R.; Everett, E.N.; Pizer, E.; Swensen, R.E.; Tamimi, H.K.; Gray, H.J.; Peters, W.A.; Goff, B.A. FOXP3+ regulatory T-cells are abundant in vulvar Paget’s disease and are associated with recurrence. Gynecol. Oncol. 2011, 120, 296–299. [Google Scholar] [CrossRef]
- Fujimura, T.; Kambayashi, Y.; Hidaka, T.; Hashimoto, A.; Haga, T.; Aiba, S. Comparison of Foxp3+ Regulatory T cells and CD163+ Macrophages in Invasive and Non-invasive Extramammary Paget’s Disease. Acta Derm. Venereol. 2012, 92, 625–628. [Google Scholar] [CrossRef]
- Fujimura, T.; Kambayashi, Y.; Furudate, S.; Kakizaki, A.; Hidaka, T.; Aiba, S. Possible mechanisms of the crosstalk between Langerhans cells and regulatory T cells in extramammary Paget disease by receptor activator of nuclear factor kappa B (RANK)ligand/RANK pathways. Br. J. Dermatol. 2017, 176, 387–394. [Google Scholar] [CrossRef]
- van der Linden, M.; van Esch, E.; Bulten, J.; Dreef, E.; Massuger, L.; van der Steen, S.; Bosse, T.; de Hullu, J.; van Poelgeest, M. The immune cell infiltrate in the microenvironment of vulvar Paget disease. Gynecol. Oncol. 2018, 151, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Dogan, A.; Hilal, Z.; Krentel, H.; Cetin, C.; Hefler, L.A.; Grimm, C.; Tempfer, C.B. Paget’s Disease of the Vulva Treated with Imiquimod: Case Report and Systematic Review of the Literature. Gynecol. Obstet. Investig. 2017, 82, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mauzo, S.H.; Tetzlaff, M.T.; Milton, D.R.; Siroy, A.E.; Nagarajan, P.; Torres-Cabala, C.A.; Ivan, D.; Curry, J.L.; Hudgens, C.W.; Wargo, J.A.; et al. Expression of PD-1 and PD-L1 in Extramammary Paget Disease: Implications for Immune-Targeted Therapy. Cancers 2019, 11, 754. [Google Scholar] [CrossRef] [PubMed]
- Pourmaleki, M.; Young, J.H.; Socci, N.D.; Chiang, S.; Edelweiss, M.; Li, Y.; Zhang, M.; Roshal, L.; Chi, D.S.; Busam, K.J.; et al. Extramammary Paget disease shows differential expression of B7 family members B7-H3, B7-H4, PD-L1, PD-L2 and cancer/testis antigens NY-ESO-1 and MAGE-A. Oncotarget 2019, 10, 6152–6167. [Google Scholar] [CrossRef]
- Gatalica, Z.; Vranic, S.; Krušlin, B.; Poorman, K.; Stafford, P.; Kacerovska, D.; Senarathne, W.; Florento, E.; Contreras, E.; Leary, A.; et al. Comparison of the biomarkers for targeted therapies in primary extra-mammary and mammary Paget’s disease. Cancer Med. 2020, 9, 1441–1450. [Google Scholar] [CrossRef]
- Karpathiou, G.; Chauleur, C.; Hathroubi, S.; Habougit, C.; Peoc’h, M. Expression of CD3, PD-L1 and CTLA-4 in mammary and extra-mammary Paget disease. Cancer Immunol. Immunother. 2018, 67, 1297–1303. [Google Scholar] [CrossRef]
- Garganese, G.; Inzani, F.; Mantovani, G.; Santoro, A.; Valente, M.; Babini, G.; Petruzzellis, G.; Fragomeni, S.M.; Gentileschi, S.; Bove, S.; et al. The vulvar immunohistochemical panel (VIP) project: Molecular profiles of vulvar Paget’s disease. J. Cancer Res. Clin. Oncol. 2019, 145, 2211–2225. [Google Scholar] [CrossRef]
- Stasenko, M.; Jayakumaran, G.; Cowan, R.; Broach, V.; Chi, D.S.; Rossi, A.; Hollman, T.J.; Zehir, A.; Abu-Rustum, N.R.; Leitao, M.M. Genomic Alterations as Potential Therapeutic Targets in Extramammary Paget’s Disease of the Vulva. JCO Precis. Oncol. 2020, 4, 1054–1060. [Google Scholar] [CrossRef]
- Kang, Z.; Xu, F.; Zhu, Y.; Fu, P.; Zhang, Q.; Hu, T.; Li, X.; Zhang, Q.; Wu, Z.; Zhang, X.; et al. Genetic Analysis of Mismatch Repair Genes Alterations in Extramammary Paget Disease. Am. J. Surg. Pathol. 2016, 40, 1517–1525. [Google Scholar] [CrossRef]
- Oronsky, B.; Ma, P.C.; Morgensztern, D.; Carter, C.A. Nothing But NET: A Review of Neuroendocrine Tumors and Carcinomas. Neoplasia 2017, 19, 991–1002. [Google Scholar] [CrossRef]
- Maggio, I.; Manuzzi, L.; Lamberti, G.; Ricci, A.D.; Tober, N.; Campana, D. Landscape and Future Perspectives of Immunotherapy in Neuroendocrine Neoplasia. Cancers 2020, 12, 832. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, J.; Mei, S.; Economos, K.; Lee, Y.-C.; Kanis, M.J. Clinicopathologic features, incidence, and survival trends of gynecologic neuroendocrine tumors: A SEER database analysis. Am. J. Obstet. Gynecol. 2019, 221, 53.e1–53.e6. [Google Scholar] [CrossRef] [PubMed]
- Howitt, B.E.; Kelly, P.; McCluggage, W.G. Pathology of Neuroendocrine Tumours of the Female Genital Tract. Curr. Oncol. Rep. 2017, 19, 59. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.P.; Othus, M.; Chae, Y.K.; Giles, F.J.; Hansel, D.E.; Singh, P.P.; Fontaine, A.; Shah, M.H.; Kasi, A.; Baghdadi, T.A.; et al. A Phase II Basket Trial of Dual Anti-CTLA-4 and Anti-PD-1 Blockade in Rare Tumors (DART SWOG 1609) in Patients with Nonpancreatic Neuroendocrine Tumors. Clin. Cancer Res. 2020, 26, 2290–2296. [Google Scholar] [CrossRef] [PubMed]
- Frumovitz, M.; Westin, S.N.; Salvo, G.; Zarifa, A.; Xu, M.; Yap, T.A.; Rodon, A.J.; Karp, D.D.; Abonofal, A.; Jazaeri, A.A.; et al. Phase II study of pembrolizumab efficacy and safety in women with recurrent small cell neuroendocrine carcinoma of the lower genital tract. Gynecol. Oncol. 2020, 158, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Sharabi, A.; Kim, S.S.; Kato, S.; Sanders, P.D.; Patel, S.P.; Sanghvi, P.; Weihe, E.; Kurzrock, R. Exceptional Response to Nivolumab and Stereotactic Body Radiation Therapy (SBRT) in Neuroendocrine Cervical Carcinoma with High Tumor Mutational Burden: Management Considerations from the Center For Personalized Cancer Therapy at UC San Diego Moores Cance. Oncologist 2017, 22, 631–637. [Google Scholar] [CrossRef]
- Nghiem, P.T.; Bhatia, S.; Lipson, E.J.; Kudchadkar, R.R.; Miller, N.J.; Annamalai, L.; Berry, S.; Chartash, E.K.; Daud, A.; Fling, S.P.; et al. PD-1 Blockade with Pembrolizumab in Advanced Merkel-Cell Carcinoma. N. Engl. J. Med. 2016, 374, 2542–2552. [Google Scholar] [CrossRef]
- Nghiem, P.; Bhatia, S.; Lipson, E.J.; Sharfman, W.H.; Kudchadkar, R.R.; Brohl, A.S.; Friedlander, P.A.; Daud, A.; Kluger, H.M.; Reddy, S.A.; et al. Durable Tumor Regression and Overall Survival in Patients With Advanced Merkel Cell Carcinoma Receiving Pembrolizumab as First-Line Therapy. J. Clin. Oncol. 2019, 37, 693–702. [Google Scholar] [CrossRef]
- Kaufman, H.L.; Russell, J.; Hamid, O.; Bhatia, S.; Terheyden, P.; D’Angelo, S.P.; Shih, K.C.; Lebbé, C.; Linette, G.P.; Milella, M.; et al. Avelumab in patients with chemotherapy-refractory metastatic Merkel cell carcinoma: A multicentre, single-group, open-label, phase 2 trial. Lancet Oncol. 2016, 17, 1374–1385. [Google Scholar] [CrossRef]
- D’Angelo, S.P.; Russell, J.; Lebbé, C.; Chmielowski, B.; Gambichler, T.; Grob, J.-J.; Kiecker, F.; Rabinowits, G.; Terheyden, P.; Zwiener, I.; et al. Efficacy and Safety of First-line Avelumab Treatment in Patients With Stage IV Metastatic Merkel Cell Carcinoma. JAMA Oncol. 2018, 4, e180077. [Google Scholar] [CrossRef]
- Lee, L.; Matulonis, U. Immunotherapy and radiation combinatorial trials in gynecologic cancer: A potential synergy? Gynecol. Oncol. 2019, 154, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.L.; Lakomy, D.S.; Ning, M.S.; Simpkins, F.; Jhingran, A. Combining novel agents with radiotherapy for gynecologic malignancies: Beyond the era of cisplatin. Int. J. Gynecol. Cancer 2020, 30, 409–423. [Google Scholar] [CrossRef] [PubMed]
- Massarelli, E.; William, W.; Johnson, F.; Kies, M.; Ferrarotto, R.; Guo, M.; Feng, L.; Lee, J.J.; Tran, H.; Kim, Y.U.; et al. Combining Immune Checkpoint Blockade and Tumor-Specific Vaccine for Patients With Incurable Human Papillomavirus 16–Related Cancer. JAMA Oncol. 2019, 5, 67. [Google Scholar] [CrossRef] [PubMed]
- Scarfì, F.; Patrizi, A.; Veronesi, G.; Lambertini, M.; Tartari, F.; Mussi, M.; Melotti, B.; Dika, E. The role of topical imiquimod in melanoma cutaneous metastases: A critical review of the literature. Dermatol. Ther. 2020, e14165. [Google Scholar] [CrossRef]
- Adams, S.; Kozhaya, L.; Martiniuk, F.; Meng, T.-C.; Chiriboga, L.; Liebes, L.; Hochman, T.; Shuman, N.; Axelrod, D.; Speyer, J.; et al. Topical TLR7 Agonist Imiquimod Can Induce Immune-Mediated Rejection of Skin Metastases in Patients with Breast Cancer. Clin. Cancer Res. 2012, 18, 6748–6757. [Google Scholar] [CrossRef]
- Salazar, L.G.; Lu, H.; Reichow, J.L.; Childs, J.S.; Coveler, A.L.; Higgins, D.M.; Waisman, J.; Allison, K.H.; Dang, Y.; Disis, M.L. Topical Imiquimod Plus Nab-paclitaxel for Breast Cancer Cutaneous Metastases. JAMA Oncol. 2017, 3, 969. [Google Scholar] [CrossRef]
- Joseph, R.W.; Cappel, M.; Tzou, K.; Bagaria, S.; Gilstrap, C.; Swaika, A.; Jambusaria-Pahlajani, A. Treatment of in-transit and metastatic melanoma in two patients treated with ipilimumab and topical imiquimod. Melanoma Res. 2016, 26, 409–412. [Google Scholar] [CrossRef]
- Fujimura, T.; Kambayashi, Y.; Sato, Y.; Tanita, K.; Furudate, S.; Tsukada, A.; Tono, H.; Hashimoto, A.; Aiba, S. Successful Treatment of Nivolumab-Resistant Multiple In-Transit Melanomas with Ipilimumab and Topical Imiquimod. Case Rep. Oncol. 2018, 11, 1–5. [Google Scholar] [CrossRef]
| Type of Study | Antibody | Clinical Setting | Number of Patients | Outcomes | Reference |
|---|---|---|---|---|---|
| KEYNOTE-028 Non-randomized, multicenter, multicohort phase Ib trial | Pembrolizumab (PD-1 inhibitor) | Avanced tumors | 18 VSCC out of 474 tumors | PFS was 20% and 7% at 6 and 12 months; OS rate was 42% and 28% at 6 and 12 months | Ott et al. [104] |
| CheckMate 358 trial Phase I-II study | Nivolumab (PD-1 inhibitor) | Metastatic/recurrent tumors | 19 cervical tumors, 5 vulvar/vaginal tumors | In vulvar/vaginal cohort: OS was 40% and 20% at 12-month and 18 month OS; PFS was 40% at 6 months | Naumann et al. [105] |
| Case report | Pembrolizumab (PD-1 inhibitor) | Recurrent VC with PD-L1 and PD-1 mutation | 1 | Near CR at 6 months of treatrment | Shields et al. [106] |
| Single-arm phase II clinical study | Pembrolizumab (PD-1 inhibitor) | Unoperable locally advanced or metastatic tumors | Recruiting. Target enrollement: 24 patients. | Primary endpoint: 95% CI for ORR. Secondary endopint: RFS-6 | Yeku et al. [107] |
| Retrospective series | Anti-CTLA-4 | Metastatic vulvar-vaginal melanomas | 6 | 33% survival rate at 1 year | Quéreux et al. [109] |
| Open-label, phase II basket trial | Pembrolizumab (PD-1 inhibitor) | Recurrent small cell NET | 6 cervical, 1 vulvar NETs | Median OS not achieved (18.8-not reached) OS 84.6% at 6 months | Strosberg et al. [110] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borella, F.; Preti, M.; Bertero, L.; Collemi, G.; Castellano, I.; Cassoni, P.; Cosma, S.; Carosso, A.R.; Bevilacqua, F.; Gallio, N.; et al. Is There a Place for Immune Checkpoint Inhibitors in Vulvar Neoplasms? A State of the Art Review. Int. J. Mol. Sci. 2021, 22, 190. https://doi.org/10.3390/ijms22010190
Borella F, Preti M, Bertero L, Collemi G, Castellano I, Cassoni P, Cosma S, Carosso AR, Bevilacqua F, Gallio N, et al. Is There a Place for Immune Checkpoint Inhibitors in Vulvar Neoplasms? A State of the Art Review. International Journal of Molecular Sciences. 2021; 22(1):190. https://doi.org/10.3390/ijms22010190
Chicago/Turabian StyleBorella, Fulvio, Mario Preti, Luca Bertero, Giammarco Collemi, Isabella Castellano, Paola Cassoni, Stefano Cosma, Andrea Roberto Carosso, Federica Bevilacqua, Niccolò Gallio, and et al. 2021. "Is There a Place for Immune Checkpoint Inhibitors in Vulvar Neoplasms? A State of the Art Review" International Journal of Molecular Sciences 22, no. 1: 190. https://doi.org/10.3390/ijms22010190
APA StyleBorella, F., Preti, M., Bertero, L., Collemi, G., Castellano, I., Cassoni, P., Cosma, S., Carosso, A. R., Bevilacqua, F., Gallio, N., Benedetto, C., & Micheletti, L. (2021). Is There a Place for Immune Checkpoint Inhibitors in Vulvar Neoplasms? A State of the Art Review. International Journal of Molecular Sciences, 22(1), 190. https://doi.org/10.3390/ijms22010190








