Possible Receptor Mechanisms Underlying Cannabidiol Effects on Addictive-like Behaviors in Experimental Animals
Abstract
1. The History of Medicinal Cannabidiol
2. CBD Attenuates Opioid Addictive-Like Behaviors, Possibly through CB1 and 5-HT1A Receptor Mechanisms
3. CBD Attenuates Cocaine Addictive-Like Behavior by the CB1, CB2, 5-HT1A and TRPV1 Receptor Mechanisms
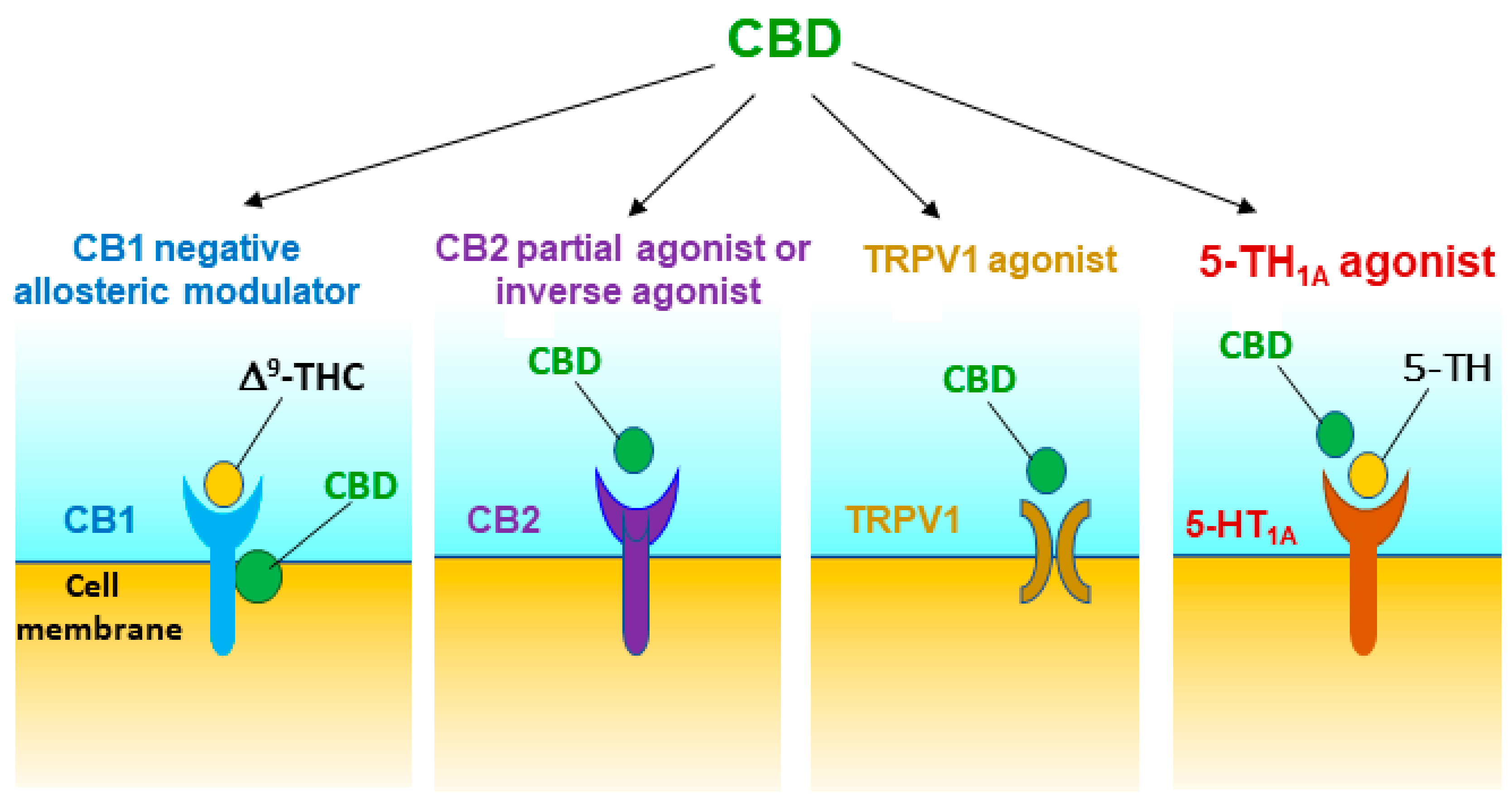
3.1. CB1R Mechanism
3.2. CB2R Mechanism
| Receptor Mechanism | Major Findings | References |
|---|---|---|
| CB1 | CBD blocked CB1R up-regulation in rats after extinction from heroin self-administration | [29] |
| Repeated treatment with CBD increased CB1R expression in the striatum | [36] | |
| Deletion of CB1R potentiated CBD-induced reduction in sucrose self-administration in CB1-KO mice | [52] | |
| AM251 failed to alter CBD-induced reduction in cocaine self-administration in rats | [37] | |
| SR141716A failed to alter CBD-potentiated extinction of cocaine- or amphetamine-induced CPP in rats | [38] | |
| AM251 failed to alter CBD-induced reduction in 75 mg/kg of cocaine-induced seizure | [65] | |
| AM251 blocked CBD-induced reduction in aggressive behavior caused by social isolation in mice | [66] | |
| CB2 | Deletion of CB2R blocked CBD-induced reduction in sucrose self-administration | [52] |
| AM630 attenuated CBD-induced reduction in cocaine self-administration | [37] | |
| AM630 attenuated CBD-induced reduction in food intake, body weight and obesity | [67,68] | |
| AM630 failed to alter high doses (75 mg/kg) of cocaine-induced seizure in mice | [65] | |
| TRPV1 | TRPV1 antagonist blocks CBD-induced reduction in cocaine self-administration in rats | [37] |
| TRPV1 deletion attenuates CBD-induced reduction in electrical shock-induced seizure in mice | [69] | |
| 5-HT1A | The 5-HT1A antagonist, WAY100635, blocked CBD’s action in cocaine self-administration in rats | [37] |
| WAY100635 attenuated CBD’s action on morphine-enhanced brain stimulation reward in rats | [32] | |
| Deletion of 5-HT1A in the DRN abolished cocaine-induced CPP. 5-HT1A antagonism in the DRN inhibited cocaine self-administration in rats | [70] | |
| Intra-NAc CBD inhibited spontaneous DA neuronal firing, which can be reversed by WAY100635 | [71] | |
| WAY100635 attenuated CBD’s action in alcohol consumption in rats | [72] | |
| WAY100635 attenuated CBD-induced reduction in aggressive behavior caused by social isolation | [66] | |
| GPR55 | GPR55 antagonism did not alter CBD’s action in cocaine self-administration in rats | [37] |
| GPR55 deletion failed to alter CBD’s action in cocaine self-administration in mice | ||
| MOR & DOR | Naloxone had no effect on CBD action in cocaine self-administration | [37] |
3.3. 5-HT1A Mechanism
3.4. TRPV1 Mechanism
4. CBD Inhibits Methamphetamine Reward and Relapse by DA-Related Mechanisms
5. CBD Attenuates Alcohol Taking Possibly by the CB1, CB2 and 5-HT1A Receptor Mechanisms
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, H.-L. An archaeological and historical account of cannabis in China. Econ. Bot. 1973, 28, 437–448. [Google Scholar] [CrossRef]
- Zuardi, A.W. History of cannabis as a medicine: A review. Braz. J. Psychiatr. 2006, 28, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Mikuriya, T.H. Marijuana in medicine: Past, present and future. Calif. Med. 1969, 110, 34–40. [Google Scholar] [PubMed]
- Touw, M. The religious and medicinal uses of cannabis in China, India and Tibet. J. Psychoact. Drugs 1981, 13, 23–34. [Google Scholar] [CrossRef]
- Gabay, M. The federal controlled substances act: Schedules and pharmacy Registration. Hosp. Pharm. 2013, 48, 473–474. [Google Scholar] [CrossRef]
- Gaoni, Y.; Mechoulam, R. Isolation, structure, and partial synthesis of an active constituent of hashish. J. Am. Chem. Soc. 1964, 86, 1646–1647. [Google Scholar] [CrossRef]
- Pertwee, R.G. The pharmacology of cannabinoid receptors and their ligands: An overview. Int. J. Obes. 2006, 30, S13. [Google Scholar] [CrossRef]
- Mechoulam, R.; Shani, A.; Edery, H.; Grunfeld, Y. Chemical basis of hashish activity. Science 1970, 169, 611–612. [Google Scholar] [CrossRef]
- Cunha, J.M.; Carlini, E.A.; Pereira, A.E.; Ramos, O.L.; Pimentel, C.; Gagliardi, R.; Sanvito, W.L.; Lander, N.; Mechoulam, R. Chronic administration of cannabidiol to healthy volunteers and epileptic patients. Pharmacology 1980, 21, 175–185. [Google Scholar] [CrossRef]
- Maa, E.; Figi, P. The case for medical marijuana in epilepsy. Epilepsia 2014, 55, 783–786. [Google Scholar] [CrossRef]
- FDA EPIDIOLEX (Cannabidiol) Oral Solution. Highlights of Prescribing Information; June 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210365lbl.pdf (accessed on 24 December 2020).
- Burstein, S. Cannabidiol (CBD) and its analogs: A review of their effects on inflammation. Bioorg. Med. Chem. 2015, 23, 1377–1385. [Google Scholar] [CrossRef] [PubMed]
- Zlebnik, N.E.; Cheer, J.F. Beyond the CB1 receptor: Is cannabidiol the answer for disorders of motivation? Annu. Rev. Neurosci. 2016, 39, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Maroon, J.; Bost, J. Review of the neurological benefits of phytocannabinoids. Surg. Neurol. Int. 2018, 9, 91. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.B. Cannabis therapeutics and the future of neurology. Front. Integr. Neurosci. 2018, 12. [Google Scholar] [CrossRef]
- Mandolini, G.M.; Lazzaretti, M.; Pigoni, A.; Oldani, L.; Delvecchio, G.; Brambilla, P. Pharmacological properties of cannabidiol in the treatment of psychiatric disorders: A critical overview. Epidemiol. Psychiatr. Sci. 2018, 27, 327–335. [Google Scholar] [CrossRef]
- Calpe-López, C.; García-Pardo, M.P.; Aguilar, M.A. Cannabidiol treatment might promote resilience to cocaine and methamphetamine use disorders: A review of possible mechanisms. Molecules 2019, 24, 2583. [Google Scholar] [CrossRef]
- Chye, Y.; Christensen, E.; Solowij, N.; Yücel, M. The endocannabinoid system and cannabidiol’s promise for the treatment of substance use disorder. Front. Psychiatr. 2019, 10. [Google Scholar] [CrossRef]
- Elsaid, S.; Kloiber, S.; Le Foll, B. Effects of cannabidiol (CBD) in neuropsychiatric disorders: A review of pre-clinical and clinical findings. Prog. Mol. Biol. Transl. Sci 2019, 167, 25–75. [Google Scholar] [CrossRef]
- Rodrigues, L.A.; Caroba, M.E.S.; Taba, F.K.; Filev, R.; Gallassi, A.D. Evaluation of the potential use of cannabidiol in the treatment of cocaine use disorder: A systematic review. Pharmacol. Biochem. Behav. 2020, 196. [Google Scholar] [CrossRef]
- Hurd, Y.L.; Yoon, M.; Manini, A.F.; Hernandez, S.; Olmedo, R.; Ostman, M.; Jutras-Aswad, D. Early phase in the development of cannabidiol as a treatment for addiction: Opioid relapse takes initial center stage. Neurotherapeutics 2015, 12, 807–815. [Google Scholar] [CrossRef]
- Prud’homme, M.; Cata, R.; Jutras-Aswad, D. Cannabidiol as an Intervention for Addictive Behaviors: A systematic review of the evidence. Subst. Abus. 2015, 9, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Freeman, A.M.; Petrilli, K.; Lees, R.; Hindocha, C.; Mokrysz, C.; Curran, H.V.; Saunders, R.; Freeman, T.P. How does cannabidiol (CBD) influence the acute effects of delta-9-tetrahydrocannabinol (THC) in humans? A systematic review. Neurosci. Biobehav. Rev. 2019, 107, 696–712. [Google Scholar] [CrossRef]
- Turna, J.; Syan, S.K.; Frey, B.N.; Rush, B.; Costello, M.J.; Weiss, M.; MacKillop, J. Cannabidiol as a novel candidate alcohol use disorder pharmacotherapy: A systematic review. Alcohol. Clin. Exp. Res. 2019, 43, 550–563. [Google Scholar] [CrossRef]
- Nona, C.N.; Hendershot, C.S.; Le Foll, B. Effects of cannabidiol on alcohol-related outcomes: A review of preclinical and human research. Exp. Clin. Psychopharmacol. 2019, 27, 359–369. [Google Scholar] [CrossRef]
- Hine, B.; Torrelio, M.; Gershon, S. Differential effect of cannabinol and cannabidiol on THC-induced responses during abstinence in morphine-dependent rats. Res. Commun. Chem. Pathol. Pharmacol. 1975, 12, 185–188. [Google Scholar] [PubMed]
- Hine, B.; Torrelio, M.; Gershon, S. Interactions between cannabidiol and Δ9-THC during abstinence in morphine-dependent rats. Life Sci. 1975, 17, 851–857. [Google Scholar] [CrossRef]
- Bhargava, H.N. Effect of some cannabinoids on naloxone-precipitated abstinence in morphine-dependent mice. Psychopharmacology 1976, 49, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Whittard, J.; Higuera-Matas, A.; Morris, C.V.; Hurd, Y.L. Cannabidiol, a nonpsychotropic component of cannabis, inhibits cue-induced heroin seeking and normalizes discrete mesolimbic neuronal disturbances. J. Neurosci. 2009, 29, 14764–14769. [Google Scholar] [CrossRef] [PubMed]
- Markos, J.R.; Harris, H.M.; Gul, W.; ElSohly, M.A.; Sufka, K.J. Effects of cannabidiol on morphine conditioned place preference in mice. Planta Med. 2018, 84, 221–224. [Google Scholar] [CrossRef]
- De Carvalho, C.R.; Takahashi, R.N. Cannabidiol disrupts the reconsolidation of contextual drug-associated memories in Wistar rats. Addict. Biol. 2017, 22, 742–751. [Google Scholar] [CrossRef]
- Katsidoni, V.; Apazoglou, K.; Panagis, G. Role of serotonin 5-HT2A and 5-HT2C receptors on brain stimulation reward and the reward-facilitating effect of cocaine. Psychopharmacology 2011, 213, 337–354. [Google Scholar] [CrossRef] [PubMed]
- Pellinen, P.; Honkakoski, P.; Stenbäck, F.; Niemitz, M.; Alhava, E.; Pelkonen, O.; Lang, M.A.; Pasanen, M. Cocaine N-demethylation and the metabolism-related hepatotoxicity can be prevented by cytochrome P450 3A inhibitors. Eur. J. Pharmacol. 1994, 270, 35–43. [Google Scholar] [CrossRef]
- Bornheim, L.M. Effect of cytochrome P450 inducers on cocaine-mediated hepatotoxicity. Toxicol. Appl. Pharmacol. 1998, 150, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, A.; Gallant, S.; Sedki, F.; D’Cunha, T.; Shalev, U. Effects of an acute cannabidiol treatment on cocaine self-administration and cue-induced cocaine seeking in male rats. J. Psychopharmacol. 2017, 31, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Luján, M.Á.; Castro-Zavala, A.; Alegre-Zurano, L.; Valverde, O. Repeated Cannabidiol treatment reduces cocaine intake and modulates neural proliferation and CB1R expression in the mouse hippocampus. Neuropharmacology 2018, 143, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Galaj, E.; Bi, G.-H.; Yang, H.-J.; Xi, Z.-X. Cannabidiol attenuates the rewarding effects of cocaine in rats by CB2, 5-HT1A and TRPV1 receptor mechanisms. Neuropharmacology 2020, 167. [Google Scholar] [CrossRef]
- Parker, L.A.; Burton, P.; Sorge, R.E.; Yakiwchuk, C.; Mechoulam, R. Effect of low doses of delta9-tetrahydrocannabinol and cannabidiol on the extinction of cocaine-induced and amphetamine-induced conditioned place preference learning in rats. Psychopharmacology 2004, 175, 360–366. [Google Scholar] [CrossRef]
- Gerdeman, G.L.; Schechter, J.B.; French, E.D. Context-specific reversal of cocaine sensitization by the CB1 cannabinoid receptor antagonist rimonabant. Neuropsychopharmacology 2008, 33, 2747–2759. [Google Scholar] [CrossRef]
- Gonzalez-Cuevas, G.; Martin-Fardon, R.; Kerr, T.M.; Stouffer, D.G.; Parsons, L.H.; Hammell, D.C.; Banks, S.L.; Stinchcomb, A.L.; Weiss, F. Unique treatment potential of cannabidiol for the prevention of relapse to drug use: Preclinical proof of principle. Neuropsychopharmacology 2018, 43, 2036–2045. [Google Scholar] [CrossRef]
- Thomas, A.; Baillie, G.L.; Phillips, A.M.; Razdan, R.K.; Ross, R.A.; Pertwee, R.G. Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br. J. Pharmacol. 2007, 150, 613–623. [Google Scholar] [CrossRef]
- Tham, M.; Yilmaz, O.; Alaverdashvili, M.; Kelly, M.E.M.; Denovan-Wright, E.M.; Laprairie, R.B. Allosteric and orthosteric pharmacology of cannabidiol and cannabidiol-dimethylheptyl at the type 1 and type 2 cannabinoid receptors. Br. J. Pharmacol. 2019, 176, 1455–1469. [Google Scholar] [CrossRef] [PubMed]
- Laprairie, R.B.; Bagher, A.M.; Kelly, M.E.M.; Denovan-Wright, E.M. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br. J. Pharmacol. 2015, 172, 4790–4805. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Pinilla, E.; Varani, K.; Reyes-Resina, I.; Angelats, E.; Vincenzi, F.; Ferreiro-Vera, C.; Oyarzabal, J.; Canela, E.I.; Lanciego, J.L.; Nadal, X.; et al. Binding and Signaling Studies Disclose a Potential Allosteric Site for Cannabidiol in Cannabinoid CB2 Receptors. Front. Pharmacol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Seeman, P. Cannabidiol is a partial agonist at dopamine D2High receptors, predicting its antipsychotic clinical dose. Transl. Psychiatry 2016, 6, e920. [Google Scholar] [CrossRef] [PubMed]
- Bisogno, T.; Hanuš, L.; Petrocellis, L.D.; Tchilibon, S.; Ponde, D.E.; Brandi, I.; Moriello, A.S.; Davis, J.B.; Mechoulam, R.; Marzo, V.D. Molecular targets for cannabidiol and its synthetic analogues: Effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br. J. Pharmacology 2001, 134, 845–852. [Google Scholar] [CrossRef]
- Elmes, M.W.; Kaczocha, M.; Berger, W.T.; Leung, K.; Ralph, B.P.; Wang, L.; Sweeney, J.M.; Miyauchi, J.T.; Tsirka, S.E.; Ojima, I.; et al. Fatty acid-binding proteins (FABPs) are intracellular carriers for Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD). J. Biol. Chem. 2015, 290, 8711–8721. [Google Scholar] [CrossRef]
- Russo, E.B.; Burnett, A.; Hall, B.; Parker, K.K. Agonistic Properties of cannabidiol at 5-HT1a receptors. Neurochem. Res. 2005, 30, 1037–1043. [Google Scholar] [CrossRef]
- Pertwee, R.G. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: Δ9-tetrahydrocannabinol, cannabidiol and Δ9-tetrahydrocannabivarin. Br. J. Pharmacol. 2008, 153, 199–215. [Google Scholar] [CrossRef]
- Kathmann, M.; Flau, K.; Redmer, A.; Tränkle, C.; Schlicker, E. Cannabidiol is an allosteric modulator at mu- and delta-opioid receptors. Naunyn. Schmied. Arch. Pharmacol. 2006, 372, 354–361. [Google Scholar] [CrossRef]
- Straiker, A.; Mitjavila, J.; Yin, D.; Gibson, A.; Mackie, K. Aiming for allosterism: Evaluation of allosteric modulators of CB1 in a neuronal model. Pharmacol. Res. 2015, 99, 370–376. [Google Scholar] [CrossRef]
- Bi, G.-H.; Galaj, E.; He, Y.; Xi, Z.-X. Cannabidiol inhibits sucrose self-administration by CB1 and CB2 receptor mechanisms in rodents. Addict. Biol. 2019, e12783. [Google Scholar] [CrossRef]
- Xi, Z.-X.; Spiller, K.; Pak, A.C.; Gilbert, J.; Dillon, C.; Li, X.; Peng, X.-Q.; Gardner, E.L. Cannabinoid CB1 receptor antagonists attenuate cocaine’s rewarding effects: Experiments with self-administration and brain-stimulation reward in rats. Neuropsychopharmacology 2008, 33, 1735–1745. [Google Scholar] [CrossRef] [PubMed]
- Casarotto, P.C.; Gomes, F.V.; Resstel, L.B.M.; Guimarães, F.S. Cannabidiol inhibitory effect on marble-burying behaviour: Involvement of CB1 receptors. Behav. Pharmacol. 2010, 21, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Lesscher, H.M.B.; Hoogveld, E.; Burbach, J.P.H.; van Ree, J.M.; Gerrits, M.A.F.M. Endogenous cannabinoids are not involved in cocaine reinforcement and development of cocaine-induced behavioural sensitization. Eur. Neuropsychopharmacol. 2005, 15, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Filip, M.; Gołda, A.; Zaniewska, M.; McCreary, A.C.; Nowak, E.; Kolasiewicz, W.; Przegaliński, E. Involvement of cannabinoid CB1 receptors in drug addiction: Effects of rimonabant on behavioral responses induced by cocaine. Pharmacol. Rep. 2006, 58, 806–819. [Google Scholar] [PubMed]
- Adamczyk, P.; Miszkiel, J.; McCreary, A.C.; Filip, M.; Papp, M.; Przegaliński, E. The effects of cannabinoid CB1, CB2 and vanilloid TRPV1 receptor antagonists on cocaine addictive behavior in rats. Brain Res. 2012, 1444, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Thapa, D.; Toguri, J.T.; Szczesniak, A.M.; Kelly, M.E. The Non-psychoactive phytocannabinoid, cannabidiol (CBD), and the synthetic derivatives, HU308 and CBD-DMH, reduces hyperalgesia and inflammation in a mouse model of corneal injury. FASEB J. 2017, 31. [Google Scholar] [CrossRef]
- De Gregorio, D.; McLaughlin, R.J.; Posa, L.; Ochoa-Sanchez, R.; Enns, J.; Lopez-Canul, M.; Aboud, M.; Maione, S.; Comai, S.; Gobbi, G. Cannabidiol modulates serotonergic transmission and reverses both allodynia and anxiety-like behavior in a model of neuropathic pain. Pain 2019, 160, 136–150. [Google Scholar] [CrossRef]
- Navarro, G.; Reyes-Resina, I.; Rivas-Santisteban, R.; Sánchez de Medina, V.; Morales, P.; Casano, S.; Ferreiro-Vera, C.; Lillo, A.; Aguinaga, D.; Jagerovic, N.; et al. Cannabidiol skews biased agonism at cannabinoid CB1 and CB2 receptors with smaller effect in CB1-CB2 heteroreceptor complexes. Biochem. Pharmacol. 2018, 157, 148–158. [Google Scholar] [CrossRef]
- Ishiguro, H.; Carpio, O.; Horiuchi, Y.; Shu, A.; Higuchi, S.; Schanz, N.; Benno, R.; Arinami, T.; Onaivi, E.S. A nonsynonymous polymorphism in cannabinoid CB2 receptor gene is associated with eating disorders in humans and food intake is modified in mice by its ligands. Synapse 2010, 64, 92–96. [Google Scholar] [CrossRef]
- Ignatowska-Jankowska, B.; Jankowski, M.M.; Swiergiel, A.H. Cannabidiol decreases body weight gain in rats: Involvement of CB2 receptors. Neurosci. Lett. 2011, 490, 82–84. [Google Scholar] [CrossRef] [PubMed]
- Deveaux, V.; Cadoudal, T.; Ichigotani, Y.; Teixeira-Clerc, F.; Louvet, A.; Manin, S.; Nhieu, J.T.-V.; Belot, M.P.; Zimmer, A.; Even, P.; et al. Cannabinoid CB2 receptor potentiates obesity-associated inflammation, insulin resistance and hepatic steatosis. PLoS ONE 2009, 4, e5844. [Google Scholar] [CrossRef] [PubMed]
- Castillo, A.; Tolón, M.R.; Fernández-Ruiz, J.; Romero, J.; Martinez-Orgado, J. The neuroprotective effect of cannabidiol in an in vitro model of newborn hypoxic-ischemic brain damage in mice is mediated by CB(2) and adenosine receptors. Neurobiol. Dis. 2010, 37, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Gobira, P.H.; Vilela, L.R.; Gonçalves, B.D.; Santos, R.P.; de Oliveira, A.C.; Vieira, L.B.; Aguiar, D.C.; Crippa, J.A.; Moreira, F.A. Cannabidiol, a Cannabis sativa constituent, inhibits cocaine-induced seizures in mice: Possible role of the mTOR pathway and reduction in glutamate release. Neurotoxicology 2015, 50, 116–121. [Google Scholar] [CrossRef]
- Hartmann, A.; Lisboa, S.F.; Sonego, A.B.; Coutinho, D.; Gomes, F.V.; Guimarães, F.S. Cannabidiol attenuates aggressive behavior induced by social isolation in mice: Involvement of 5-HT1A and CB1 receptors. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 94, 109637. [Google Scholar] [CrossRef]
- Zhang, H.-Y.; Gao, M.; Liu, Q.-R.; Bi, G.-H.; Li, X.; Yang, H.-J.; Gardner, E.L.; Wu, J.; Xi, Z.-X. Cannabinoid CB2 receptors modulate midbrain dopamine neuronal activity and dopamine-related behavior in mice. Proc. Natl. Acad. Sci. USA 2014, 111, E5007–E5015. [Google Scholar] [CrossRef]
- Zhang, H.-Y.; Gao, M.; Shen, H.; Bi, G.-H.; Yang, H.-J.; Liu, Q.-R.; Wu, J.; Gardner, E.L.; Bonci, A.; Xi, Z.-X. Expression of functional cannabinoid CB2 receptor in VTA dopamine neurons in rats. Addict. Biol. 2017, 22, 752–765. [Google Scholar] [CrossRef]
- Gray, R.A.; Stott, C.G.; Jones, N.A.; Di Marzo, V.; Whalley, B.J. Anticonvulsive properties of cannabidiol in a model of generalized seizure are transient receptor potential vanilloid 1 dependent. Cannabis Cannabinoid Res. 2020, 5, 145–149. [Google Scholar] [CrossRef]
- You, I.-J.; Wright, S.R.; Garcia-Garcia, A.L.; Tapper, A.R.; Gardner, P.D.; Koob, G.F.; David Leonardo, E.; Bohn, L.M.; Wee, S. 5-HT1A autoreceptors in the dorsal raphe nucleus convey vulnerability to compulsive cocaine seeking. Neuropsychopharmacology 2016, 41, 1210–1222. [Google Scholar] [CrossRef]
- Norris, C.; Loureiro, M.; Kramar, C.; Zunder, J.; Renard, J.; Rushlow, W.; Laviolette, S.R. Cannabidiol modulates fear memory formation through interactions with serotonergic transmission in the mesolimbic system. Neuropsychopharmacology 2016, 41, 2839–2850. [Google Scholar] [CrossRef]
- Viudez-Martínez, A.; García-Gutiérrez, M.S.; Fraguas-Sánchez, A.I.; Torres-Suárez, A.I.; Manzanares, J. Effects of cannabidiol plus naltrexone on motivation and ethanol consumption. Br. J. Pharmacol. 2018, 175, 3369–3378. [Google Scholar] [CrossRef] [PubMed]
- Jordan, C.; Xi, Z.X. Progress in brain cannabinoid CB2 receptors: From gene to behavior. Neurosci. Biobehav. Rev. 2019, 98, 208–220. [Google Scholar] [CrossRef] [PubMed]
- Manzanares, J.; Cabañero, D.; Puente, N.; García-Gutiérrez, M.S.; Grandes, P.; Maldonado, R. Role of the endocannabinoid system in drug addiction. Biochem. Pharmacol. 2018, 157, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Aracil-Fernández, A.; Trigo, J.M.; García-Gutiérrez, M.S.; Ortega-Álvaro, A.; Ternianov, A.; Navarro, D.; Robledo, P.; Berbel, P.; Maldonado, R.; Manzanares, J. Decreased cocaine motor sensitization and self-administration in mice overexpressing cannabinoid CB2 receptors. Neuropsychopharmacology 2012, 37, 1749–1763. [Google Scholar] [CrossRef] [PubMed]
- Foster, D.J.; Wilson, J.M.; Remke, D.H.; Mahmood, M.S.; Uddin, M.J.; Wess, J.; Patel, S.; Marnett, L.J.; Niswender, C.M.; Jones, C.K.; et al. Antipsychotic-like effects of M4 positive allosteric modulators are mediated by CB2 receptor-dependent inhibition of dopamine release. Neuron 2016, 91, 1244–1252. [Google Scholar] [CrossRef]
- Renard, J.; Loureiro, M.; Rosen, L.G.; Zunder, J.; de Oliveira, C.; Schmid, S.; Rushlow, W.J.; Laviolette, S.R. Cannabidiol counteracts amphetamine-induced neuronal and behavioral sensitization of the mesolimbic dopamine pathway through a novel mTOR/p70S6 kinase signaling pathway. J. Neurosci. 2016, 36, 5160–5169. [Google Scholar] [CrossRef]
- Iannotti, F.A.; Hill, C.L.; Leo, A.; Alhusaini, A.; Soubrane, C.; Mazzarella, E.; Russo, E.; Whalley, B.J.; Di Marzo, V.; Stephens, G.J. Nonpsychotropic plant cannabinoids, cannabidivarin (CBDV) and cannabidiol (CBD), activate and desensitize transient receptor potential vanilloid 1 (TRPV1) channels in vitro: Potential for the treatment of neuronal hyperexcitability. ACS Chem. Neurosci. 2014, 5, 1131–1141. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Ligresti, A.; Moriello, A.S.; Allarà, M.; Bisogno, T.; Petrosino, S.; Stott, C.G.; Di Marzo, V. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. Br. J. Pharmacol. 2011, 163, 1479–1494. [Google Scholar] [CrossRef]
- Edwards, J.G. TRPV1 in the central nervous system: Synaptic plasticity, function, and pharmacological implications. Prog. Drug Res. 2014, 68, 77–104. [Google Scholar]
- Bisogno, T.; Maccarrone, M. Latest advances in the discovery of fatty acid amide hydrolase inhibitors. Exp. Opin. Drug Discov. 2013, 8, 509–522. [Google Scholar] [CrossRef]
- Arnold, J.C. The role of endocannabinoid transmission in cocaine addiction. Pharmacol. Biochem. Behav. 2005, 81, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Onaivi, E.S. An endocannabinoid hypothesis of drug reward and drug addiction. Ann. N. Y. Acad. Sci. 2008, 1139, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Hay, G.L.; Baracz, S.J.; Everett, N.A.; Roberts, J.; Costa, P.A.; Arnold, J.C.; McGregor, I.S.; Cornish, J.L. Cannabidiol treatment reduces the motivation to self-administer methamphetamine and methamphetamine-primed relapse in rats. J. Psychopharmacol. 2018, 32, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Haghighi, S.; Haghparast, A. Cannabidiol inhibits priming-induced reinstatement of methamphetamine in REM sleep deprived rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 82, 307–313. [Google Scholar] [CrossRef]
- Yang, G.; Liu, L.; Zhang, R.; Li, J.; Leung, C.-K.; Huang, J.; Li, Y.; Shen, B.; Zeng, X.; Zhang, D. Cannabidiol attenuates methamphetamine-induced conditioned place preference via the Sigma1R/AKT/GSK-3β/CREB signaling pathway in rats. Toxicol. Res. 2020, 9, 202–211. [Google Scholar] [CrossRef]
- Karimi-Haghighi, S.; Dargahi, L.; Haghparast, A. Cannabidiol modulates the expression of neuroinflammatory factors in stress- and drug-induced reinstatement of methamphetamine in extinguished rats. Addict. Biol. 2020, 25, e12740. [Google Scholar] [CrossRef]
- Palazzolo, D.L.; Quadri, S.K. Interleukin-1 stimulates catecholamine release from the hypothalamus. Life Sci. 1990, 47, 2105–2109. [Google Scholar] [CrossRef]
- Shintani, F.; Kanba, S.; Nakaki, T.; Nibuya, M.; Kinoshita, N.; Suzuki, E.; Yagi, G.; Kato, R.; Asai, M. Interleukin-1 beta augments release of norepinephrine, dopamine, and serotonin in the rat anterior hypothalamus. J. Neurosci. 1993, 13, 3574–3581. [Google Scholar] [CrossRef]
- Benjamins, J.A. Direct effects of secretory products of immune cells on neurons and glia. J. Neurolog. Sci. 2013, 333, 30–36. [Google Scholar] [CrossRef]
- Wang, X.; Northcutt, A.L.; Cochran, T.A.; Zhang, X.; Fabisiak, T.J.; Haas, M.E.; Amat, J.; Li, H.; Rice, K.C.; Maier, S.F.; et al. Methamphetamine activates toll-like receptor 4 to induce central immune signaling within the ventral tegmental area and contributes to extracellular dopamine increase in the nucleus accumbens shell. ACS Chem. Neurosci. 2019, 10, 3622–3634. [Google Scholar] [CrossRef]
- Viudez-Martínez, A.; García-Gutiérrez, M.S.; Navarrón, C.M.; Morales-Calero, M.I.; Navarrete, F.; Torres-Suárez, A.I.; Manzanares, J. Cannabidiol reduces ethanol consumption, motivation and relapse in mice. Addict. Biol. 2018, 23, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Viudez-Martínez, A.; García-Gutiérrez, M.S.; Manzanares, J. Gender differences in the effects of cannabidiol on ethanol binge drinking in mice. Addict. Biol. 2020, 25, e12765. [Google Scholar] [CrossRef] [PubMed]
- Filev, R.; Engelke, D.S.; Da Silveira, D.X.; Mello, L.E.; Santos-Junior, J.G. THC inhibits the expression of ethanol-induced locomotor sensitization in mice. Alcohol 2017, 65, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Galaj, E.; Xi, Z.-X. Potential of cannabinoid receptor ligands as treatment for substance use disorders. CNS Drugs 2019, 33, 1001–1030. [Google Scholar] [CrossRef]
- De Almeida, D.L.; Devi, L.A. Diversity of molecular targets and signaling pathways for CBD. Pharmacol. Res. Perspect. 2020, 8, e00682. [Google Scholar] [CrossRef]
- Izzo, A.A.; Borrelli, F.; Capasso, R.; Di Marzo, V.; Mechoulam, R. Non-psychotropic plant cannabinoids: New therapeutic opportunities from an ancient herb. Trends Pharmacol. Sci. 2009, 30, 515–527. [Google Scholar] [CrossRef]
- Ryberg, E.; Larsson, N.; Sjögren, S.; Hjorth, S.; Hermansson, N.-O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The orphan receptor GPR55 is a novel cannabinoid receptor. Br. J. Pharmacol. 2007, 152, 1092–1101. [Google Scholar] [CrossRef]
| Target | Function | CBD Action | References |
|---|---|---|---|
| CB1 | Antagonist | Low affinity to CB1Rs in [3H]CP55940 binding assay (Ki = 3.3~4.9 μM); ↓ CP55940-enhanced GTPγS binding (KB = 79 nM); ↓ CP55940-induced inhibition of cAMP formation (KB = 545 nM); ↓ CP55940-induced β-arrestin recruitmrnt in CB1-expressing HEK-CRE cells (KB = 547 nM) | [41,42] |
| NAM | ↓ Δ9-THC- or 2-AG efficacy and potency on arrestin2, PLCβ3 and ERK1/2 signaling in CB1-expressing HEK 293A cells (IC50 = 0.27~0.96 μM) | [43] | |
| CB2 | Antagonist | Low affinity to CB2Rs in [3H]CP55940 or [3H]WIN55212-2 binding assays (CBD-Ki = 4.2 μM vs. ∆9-THC-Ki = 3.3 μM) ↓ CP55940-enahnced GTPγS binding in CB2-expressing CHO cells (KB = 65 nM) ↓ CP55940-induced inhibition of cAMP formation (KB = 641 nM); ↓ CP55940-induced β-arrestin recruitmrnt in CB2-expressing HEK-CRE cells (KB = 420 nM); | [41,42,44,45] |
| NAM | ↓ JWH133-induced reduction in forskolin-stimulated cAMP formation (IC50 = 3 nM) ↓ JWH133-induced ERK1/2 phosphorylation in HEK cells expressed hCB2 (IC50 = 29 nM) | [44] | |
| Inverse agonist | ↓ GTPγS binding in CB2-expressing CHO cells (EC50 = 503 nM) | [41] | |
| Partial agonist | Unexpectedly high affinity to CB2Rs in [3H]CP55940 binding assays (CBD-Ki = 34 nM vs. ∆9-THC-Ki = 15 nM) ↑ BRET response, ↑ intracellular CRE activity in CRE and BRET assays in CB2-expressing HEK cells | [42] | |
| GPR55 | Antagonist | ↓ CP55940-enhanced GTPγS binding in GPR55-expressing cells (IC50 = 445 nM) | [45] |
| TRPV1 | Agonist | ↑ [Ca++] levels in TRPV1-expressing HEK 293 cells (EC50 = 3.5 μM) | [46] |
| FAAH | Inhibitor | ↓ [14C] AEA update in N18TG2 cells (IC50 = 27.5 µM) ↓ [14C] AEA update in HeLa cells expressing rat, but not human, FAAH at 50–200 μM | [46,47] |
| 5-HT1A | Agonist | ↓ [3H]8-OH-DPAT binding; ↑ GTPγS binding in 5-HT1A-expressing CHO cells | [48] |
| D2 | Partial agonist | ↓ [3H]domperidone binding to D2 receptors (Ki = 11 nM at D2High; Ki = 2800 nm at D2Low) | [45] |
| MOR & DOR | Allosteric modulator | ↓ [3DNM] binding to MOR (IC50 = 7 μM) At 100 μM, ↑ [3H]DAMGO dissociation from MOR (pE50 = 4.38 μM) ↑ [3H]-naltrindole dissociation from DOR (pE50 = 4.1 μM) | [49,50] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galaj, E.; Xi, Z.-X. Possible Receptor Mechanisms Underlying Cannabidiol Effects on Addictive-like Behaviors in Experimental Animals. Int. J. Mol. Sci. 2021, 22, 134. https://doi.org/10.3390/ijms22010134
Galaj E, Xi Z-X. Possible Receptor Mechanisms Underlying Cannabidiol Effects on Addictive-like Behaviors in Experimental Animals. International Journal of Molecular Sciences. 2021; 22(1):134. https://doi.org/10.3390/ijms22010134
Chicago/Turabian StyleGalaj, Ewa, and Zheng-Xiong Xi. 2021. "Possible Receptor Mechanisms Underlying Cannabidiol Effects on Addictive-like Behaviors in Experimental Animals" International Journal of Molecular Sciences 22, no. 1: 134. https://doi.org/10.3390/ijms22010134
APA StyleGalaj, E., & Xi, Z.-X. (2021). Possible Receptor Mechanisms Underlying Cannabidiol Effects on Addictive-like Behaviors in Experimental Animals. International Journal of Molecular Sciences, 22(1), 134. https://doi.org/10.3390/ijms22010134





