Therapeutic Targeting Steroid Resistant Pro-Inflammatory NK and NKT-Like Cells in Chronic Inflammatory Lung Disease
Abstract
:1. Introduction
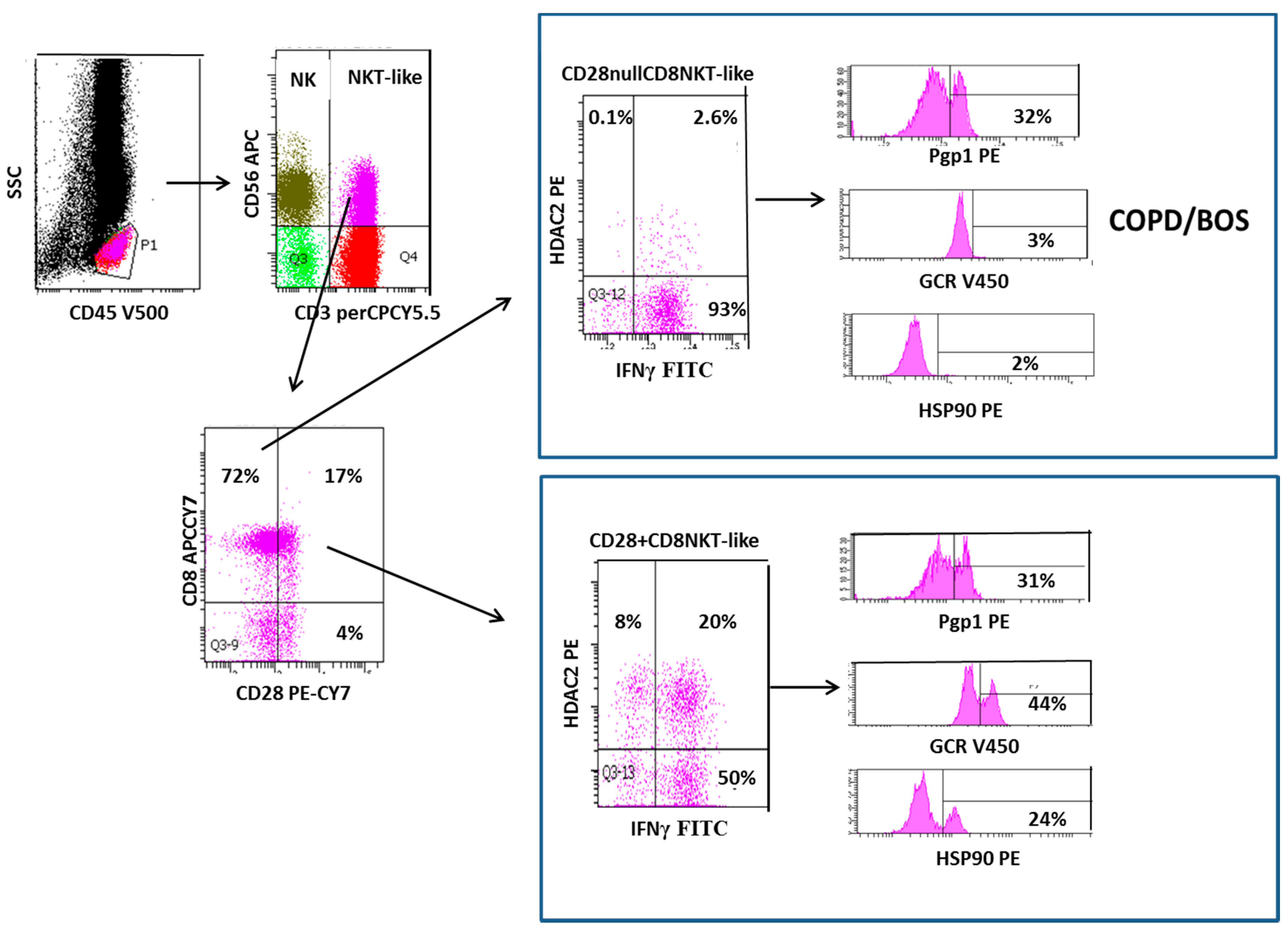
2. Loss of CD28 on Steroid Resistant Senescent NKT-Like Lymphocytes in COPD and BOS
3. Increased Transmembrane Pump, P-Glycoprotein 1, In NK and NKT-Like Cells
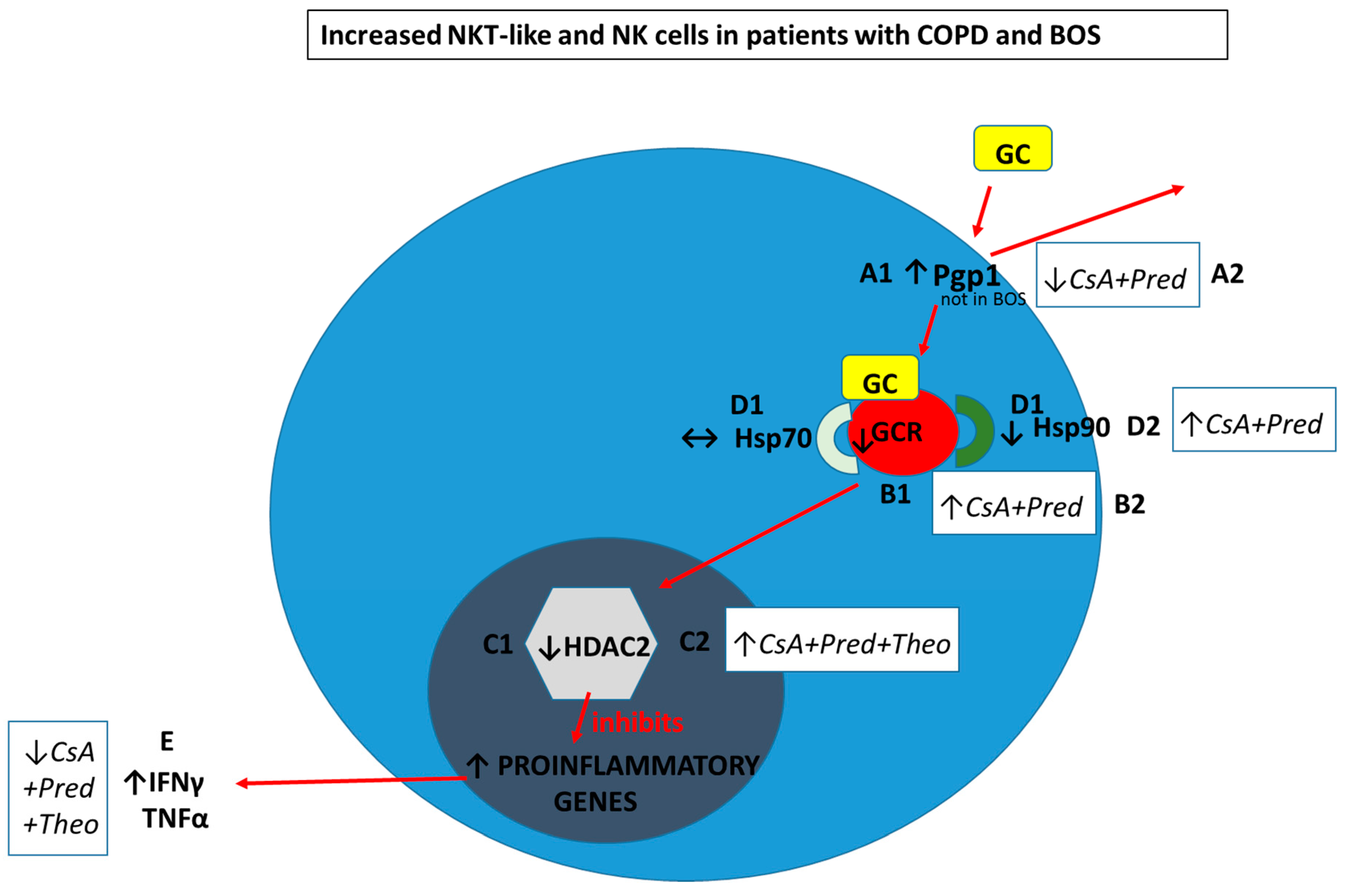
- A1. Pgp1+ NKT-like cells are increased in COPD (but not BOS), reducing intracellular levels of glucocorticoid (GC). Expression of GCR (B1), HDAC2 (C1) and Hsp90 (D1) are decreased in CD28nullCD8+NKT-like cells and NK cells (with no change in Hsp70) in COPD and BOS patients reducing steroid effectiveness.
- A2. Pgp1 is synergistically decreased in the presence of 2.5 ng/mL cyclosporine A (CsA) and 1 µM prednisolone in the blood of COPD patients ex vivo.
- B2. GCR expression is increased in the presence of 2.5 ng/mL cyclosporine A (CsA) and 1 µM prednisolone in the blood of COPD and BOS patients ex vivo.
- C2. HDAC2 expression is increased in the presence of 5 mg/mL theophylline, 2.5 ng/mL CsA and 1 µM prednisolone in the blood of COPD and BOS patients ex vivo.
- D2. Hsp90 expression is increased in the presence of 2.5 ng/mL CsA and 1 µM prednisolone in the blood of COPD and BOS patients ex vivo.
- E. Therapeutic targeting results in decreased IFNγ and TNFα pro-inflammatory cytokine expression in CD28nullCD8+NKT-like cells and NK cells in the blood of COPD and BOS patients ex vivo.
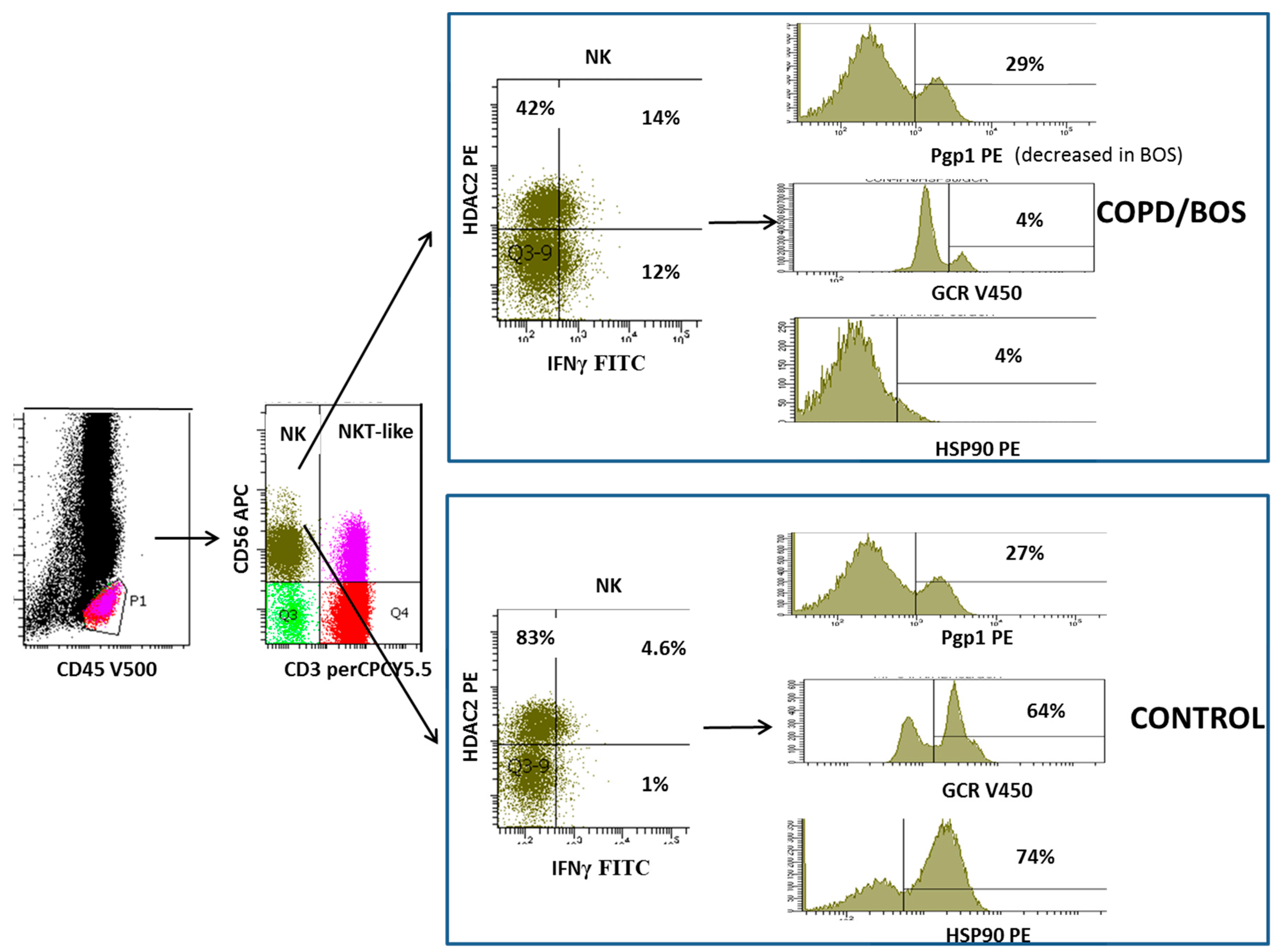
4. Loss of Glucocorticoid Receptor in NKT-like and NK Cells
5. Decreased Histone Deacetylase 2 in NKT-Like and NK Cells
6. Decreased Heat Shock Protein in NKT-Like and NK Cells
7. Future Therapy for Chronic Inflammatory Lung Disease
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| COPD | Chronic obstructive pulmonary disease |
| BOS | Bronchiolitis obliterans syndrome |
| NK | Natural killer cell |
| NKT-like | Natural killer T cell like |
| GC | Glucocorticoid |
| GCR | Glucocorticoid receptor |
| IFNγ | Interferon gamma |
| TNFα | Tumor necrosis factor alpha |
| Pgp1 | P-glycoprotein 1 |
| HDAC2 | Histone deacetylase 2 |
| Hsp90 | Heat shock protein 90 |
| CD | Cluster of differentiation |
| FEV1 | Forced expiratory volume in 1 s |
| BAL | Bronchoalveolar lavage |
References
- Barnes, P.J. Glucocorticosteroids: Current and future directions. Brit. J. Pharmacol. 2011, 163, 29–43. [Google Scholar] [CrossRef]
- Lama, R.; Santos, F.; Alvarez, A.; Algar, F.J.; Baamonde, C.; Quero, F.; Cerezo, F.; Salvatierra, A. Analysis of lung transplant recipients surviving beyond 5 years. Transpl. Proc. 2005, 37, 1523–1525. [Google Scholar] [CrossRef]
- Hodge, G.; Nairne, J.; Holmes, M.; Reynolds, P.N.; Hodge, S. Increased intracellular T helper 1 pro-inflammatory cytokine production in peripheral blood, bronchoalveolar lavage and intraepithelial T cells of COPD patients. Clin. Exp. Immunol. 2007, 150, 22–29. [Google Scholar] [CrossRef]
- Hodge, G.; Hodge, S.; Reynolds, P.; Holmes, M. Intracellular cytokines in blood T-cells in lung transplant patients- a more relevant indicator of immunosuppression than drug levels. Clin. Exp. Immunol. 2005, 139, 159–164. [Google Scholar] [CrossRef]
- Hodge, G.; Hodge, S.; Li-Liew, C.; Chambers, D.; Hopkins, P.; Reynolds, P.N.; Holmes, M. Time post-transplant correlates with increasing peripheral blood T-cell granzyme B and pro-inflammatory cytokines. Clin. Exp. Immunol. 2010, 161, 584–590. [Google Scholar] [CrossRef]
- Hodge, G.; Hodge, S.; Reynolds, P.N.; Holmes, M. Compartmentalisation of intracellular cytokines in bronchial intra-epithelial T-cells of stable lung transplant patients. Clin. Exp. Immunol. 2006, 145, 413–419. [Google Scholar] [CrossRef]
- Hodge, G.; Reynolds, P.N.; Holmes, M.; Hodge, S. Differential expression of pro-inflammatory cytokines in intra-epithelial T cells between trachea and bronchi distinguishes severity of COPD. Cytokine 2012, 60, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Maeno, T.; Houghton, A.M.; Quintero, P.A.; Grumelli, S.; Owen, C.A.; Shapiro, S.D. CD8+ T cells are required for inflammation and destruction in cigarette smoke-induced emphysema in mice. J. Immunol. 2007, 178, 8090–8096. [Google Scholar] [CrossRef]
- Saetta, M.; Baraldo, S.; Corbino, L.; Turato, G.; Braccioni, F.; Rea, F.; Cavallesco, G.; Tropeano, G.; Mapp, C.E.; Maestrelli, P. CD8+ve cells in the lungs of smokers with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 1999, 160, 711–717. [Google Scholar] [CrossRef]
- Hodge, G.; Hodge, S. Steroid resistant CD8+CD28nullNKT-like pro-inflammatory cytotoxic cells in chronic obstructive pulmonary disease. Front. Immunol. 2016, 7, 617. [Google Scholar] [CrossRef]
- Rajavec, M.; Volarevic, S.; Osolnic, K.; Kosnok, M.; Korosec, P. Natural Niller T cells in pulmonary disorders. Respir. Med. 2011, 105 (Suppl. 1), S20–S25. [Google Scholar] [CrossRef]
- Urbanowicz, R.A.; Lamb, J.R.; Todd, I.; Corne, J.M.; Fiarclough, L.C. Altered effector function of peripheral cytotoxic cells in COPD. Respir. Res. 2009, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Hodge, G.; Mukaro, V.; Holmes, M.; Reynolds, P.N.; Hodge, S. Enhanced cytotoxic function of natural killer T-like cells associated with decreased CD94 (Kp43) in the chronic obstructive pulmonary disease pathway. Respirology 2013, 18, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Li, X.; Wang, M.; Zou, Q.; Zhao, S.; Sun, B.; Xu, L.; Jiang, Y. Increased numbers of NK cells, NKT-like cells and NK inhibitory receptors in peripheral blood of patients with chronic obstructive pulmonary disease. Clin. Dev. Immunol. 2013, 2013, 721782. [Google Scholar] [CrossRef]
- Freeman, C.M.; Stolberg, V.R.; Crudgington, S.; Martinez, F.J.; Han, M.K.; Chensue, S.W.; Arenberg, D.A.; Meldrum, C.A.; McCloskey, L.; Curtis, J.L. Human CD56+ cytotoxic lung lymphocytes kill autologous lung cells in chronic obstructive pulmonary disease. PLoS ONE 2014, 9, e103840. [Google Scholar] [CrossRef]
- Hodge, G.; Hodge, S.; Yeo, A.; Nguyen, P.; Hopkins, E.; Holmes-Liew, C.L.; Reynolds, P.N.; Holmes, M. BOS is associated with increased cytotoxic proinflammatory CD8+T, NKT-like and NK cells in the small airways. Transplantation 2017, 101, 2469–2476. [Google Scholar] [CrossRef]
- Kaur, M.; Smyth, L.J.; Cadden, P.; Grundy, S.; Ray, D.; Plumb, J.; Singh, D. T lymphocyte insensitivity to corticosteroids in chronic obstructive pulmonary disease. Respir. Res. 2012, 13, 20. [Google Scholar] [CrossRef]
- Hodge, G.; Hodge, S.; Nguyen, P.; Yeo, A.; Sarkar, P.; Badiei, A.; Holmes-Liew, C.L.; Reynolds, P.N.; Holmes, M. Bronchiolitis obliterans syndrome is associated with increased p-glyycoprotein expression and loss of glucocorticoid receptor from steroid resistant proinflammatory CD8+ T cells. Clin. Exp. Immunol. 2018, 192, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Hodge, G.; Jersmann, H.; Tran, H.B.; Holmes, M.; Reynolds, P.N.; Hodge, S. Lymphocyte senescence in COPD is associated with loss of glucocorticoid receptor expression by pro-inflammatory/cytotoxic lymphocytes. Respir. Res. 2015, 16, 2. [Google Scholar] [CrossRef] [PubMed]
- Gadgil, A.; Duncan, S. Role of T-lymphocytes and proinflammatory mediators in the pathogenesis of chronic obstructive pulmonary disease. Int. J. Chron. Obstruct. Pulm. Dis. 2001, 3, 531–541. [Google Scholar]
- Hodge, G.; Hodge, S.; Ahern, J.; Holmes-Liew, C.L.; Reynolds, P.N.; Holmes, M. Upregulation of alternate costimulatory molecules on proinflammatory CD28null T cells in bronchiolitis obliterans syndrome. Clin. Exp. Immunol. 2013, 173, 150–160. [Google Scholar] [CrossRef]
- Fojo, A.T.; Ueda, K.; Slamon, D.J.; Poplack, D.G.; Gottesman, M.M. Expression of a multidrug resistant gene in human tumors and tissues. Proc. Natl. Acad. Sci. USA 1987, 84, 265–269. [Google Scholar] [CrossRef]
- Hodge, G.; Holmes, M.; Jersmann, H.; Reynolds, P.N.; Hodge, S. The drug efflux pump Pgp1 in pro-inflammatory lymphocytes is a target for novel treatment strategies in COPD. Respir. Res. 2013, 14, 63. [Google Scholar] [CrossRef]
- Pujols, L.; Xaubet, A.; Ramirez, J.; MUllol, J.; Roca-Ferrer, J.; Torrego, A.; Cidlowski, J.A.; Picado, C. Expression of glucocorticoid receptors alpha and beta in steroid sensitive and steroid insensitive interstitial lung diseases. Thorax 2004, 59, 687–693. [Google Scholar] [CrossRef]
- Hodge, G.; Jersmann, H.; Tran, H.B.; Roscioli, E.; Holmes, M.; Reynolds, P.N.; Hodge, S. Lymphocyte senescence in COPD is associated with decreased histone deacetylase 2 expression by pro-inflammatory lymphocytes. Respir. Res. 2015, 16, 130. [Google Scholar] [CrossRef] [PubMed]
- Hodge, G.; Hodge, S.; Holmes-Liew, C.L.; Reynolds, P.N.; Holmes, M. Histone deacetylase 2 is decreased in peripheral blood pro-inflammatory CD8+T and NKT-like lymphocytes following lung transplant. Respirology 2017, 22, 394–400. [Google Scholar] [CrossRef]
- Hodge, G.; Hodge, S.; Yeo, A.; Nguyen, P.; Hopkins, E.; Holmes-Liew, C.L.; Reynolds, P.N.; Holmes, M. BOS is associated with decreased HDAC2 from steroid resistant lymphocytes in the small airways. Clin. Exp. Immunol. 2019, 195, 277–285. [Google Scholar] [CrossRef]
- Arcasoy, S.M.; Berry, G.; Marboe, C.C.; Tazelaar, H.D.; Zamora, M.R.; Wolters, H.J.; Fang, K.C.; Keshavjee, S. Pathologic interpretation of transbronchial biopsy for acute rejection of lung allograft is highly variable. Am. J. Transplant. 2011, 11, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Massaque, J. Nuclear-cytoplasmic shuttling of signal transducers. Nat. Rev. Mol. Cell Biol. 2004, 5, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Hodge, G.; Roscioli, E.; Jersmann, H.; Tran, H.B.; Holmes, M.; Reynolds, P.N.; Hodge, S. Steroid resistance in COPD is associated with impaired molecular chaperone Hsp90 expression by pro-inflammatory lymphocytes. Respir. Res. 2016, 17, 1–12. [Google Scholar] [CrossRef]
- Barnes, P.J. Corticosteroid resistance in patients with asthma and chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 2013, 131, 636–645. [Google Scholar] [CrossRef]
- Téo, F.H.; de Oliveira, R.T.; Mamoni, R.L.; Ferreira, M.C.; Nadruz Jr, W.; Coelho, O.R.; de Lara Fernandes, J.; Blotta, M.H. Characterisation of CD4+CD28null T cells in patients with coronary artery disease and individuals with risk factors for atherosclerosis. Cell Immunol. 2013, 281, 11–19. [Google Scholar] [CrossRef]
- Thewissen, M.; Somers, V.; Hellings, N.; Fraussen, J.; Damoiseaux, J.; Stinissen, P. CD4+CD28null T cells in autoimmune disease: Pathologenic features and decreased susceptibility to immunoregulation. J. Immunol. 2007, 179, 6514–6523. [Google Scholar] [CrossRef]
- Fasth, A.E.; Snir, O.; Johansson, A.A.; Nordmark, B.; Rahbar, A.; af Klint, E.; Björkström, N.K.; Ulfgren, A.K.; van Vollenhoven, R.F.; Malmström, V.; et al. Skewed distribution of pro-inflammatory CD4+CD28null T cells in rheumatoid arthritis. Arthritis Res. Ther. 2007, 9, R87. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, Y.; Fukunaga, K.; Ikeuchi, H.; Kamikozuru, K.; Hida, N.; Ohda, Y.; Iimuro, M.; Yoshida, K.; Kikuyama, R.; Kato, K.; et al. The CD4CD28null and the regulatory CD4+CD25High T-cell phenotypes in patients with ulcerative colitis during active and quiescent disease, following colectomy. Cytokine 2011, 56, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.; Rahman, I. Role of histone deacetylase 2 in epigenetics and cellular senescence: Implications in lung inflammaging and COPD. Am. Physiol. Lung Cell Mol. Physiol. 2012, 303, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Burgel, P.R.; Bergeron, A.; De Blic, J.; Bonniaud, P.; Bourdin, A.; Chanez, P.; Chinet, T.; Dalphin, J.C.; Devillier, P.; Deschildre, A.; et al. Small airways diseases, excluding asthma and COPD: An overview. Eur. Respir. Rev. 2013, 22, 131–147. [Google Scholar] [CrossRef]
- Hodge, G.; Hodge, S.; Chambers, D.; Reynolds, P.N.; Holmes, M. Bronchiolitis obliterans syndrome is associated with absence of suppression of peripheral blood Th1 pro-inflammatory cytokines. Transplantation 2009, 88, 211–218. [Google Scholar] [CrossRef]
- Hodge, S.; Hodge, G.; Ahern, J.; Liew, C.L.; Hopkins, E.; Chambers, D.C.; Reynolds, P.N.; Holmes, M. Increased levels of granzyme b in bronchiolitis obliterans syndrome are not suppressed adequately by current immunosuppressive regimes. Clin. Exp. Immunol. 2009, 158, 230–236. [Google Scholar] [CrossRef]
- Hodge, S.; Holmes, M.; Banerjee, B.; Musk, M.; Kicic, A.; Wtaerer, G.; Reynolds, P.N.; Hodge, G.; Chambers, D.C. Posttransplant bronchiolitis obliterans syndrome is associated with bronchial to epithelial to mesenchymal transition. Am. J. Transplant. 2009, 9, 727–733. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hodge, G.; Hodge, S. Therapeutic Targeting Steroid Resistant Pro-Inflammatory NK and NKT-Like Cells in Chronic Inflammatory Lung Disease. Int. J. Mol. Sci. 2019, 20, 1511. https://doi.org/10.3390/ijms20061511
Hodge G, Hodge S. Therapeutic Targeting Steroid Resistant Pro-Inflammatory NK and NKT-Like Cells in Chronic Inflammatory Lung Disease. International Journal of Molecular Sciences. 2019; 20(6):1511. https://doi.org/10.3390/ijms20061511
Chicago/Turabian StyleHodge, Greg, and Sandra Hodge. 2019. "Therapeutic Targeting Steroid Resistant Pro-Inflammatory NK and NKT-Like Cells in Chronic Inflammatory Lung Disease" International Journal of Molecular Sciences 20, no. 6: 1511. https://doi.org/10.3390/ijms20061511
APA StyleHodge, G., & Hodge, S. (2019). Therapeutic Targeting Steroid Resistant Pro-Inflammatory NK and NKT-Like Cells in Chronic Inflammatory Lung Disease. International Journal of Molecular Sciences, 20(6), 1511. https://doi.org/10.3390/ijms20061511



