Focal Neuroendocrine Differentiation of Conventional Prostate Adenocarcinoma as a Prognostic Factor after Radical Prostatectomy: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Results
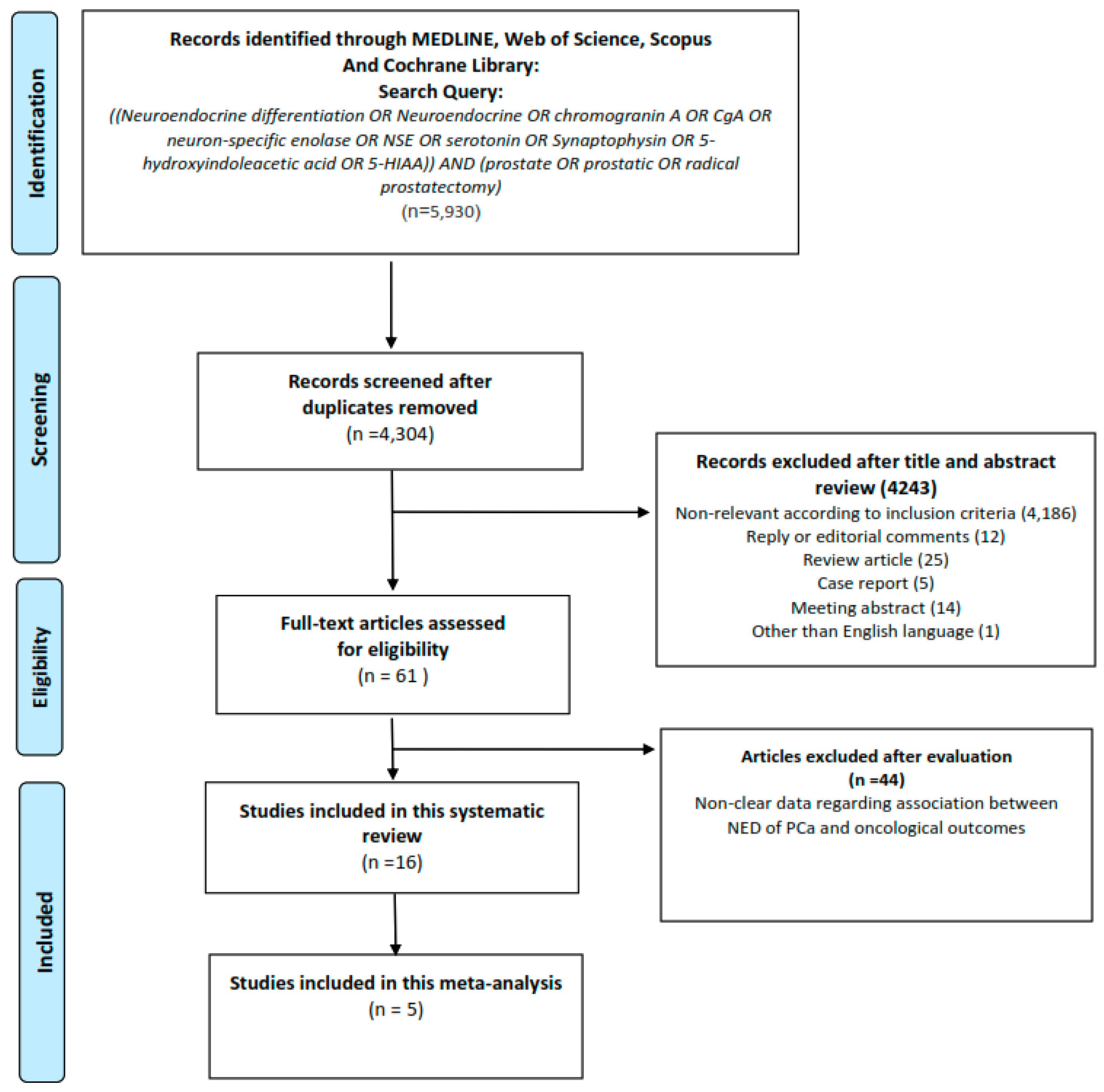
2.1. Results of Search
2.2. Characteristics of the Included Studies
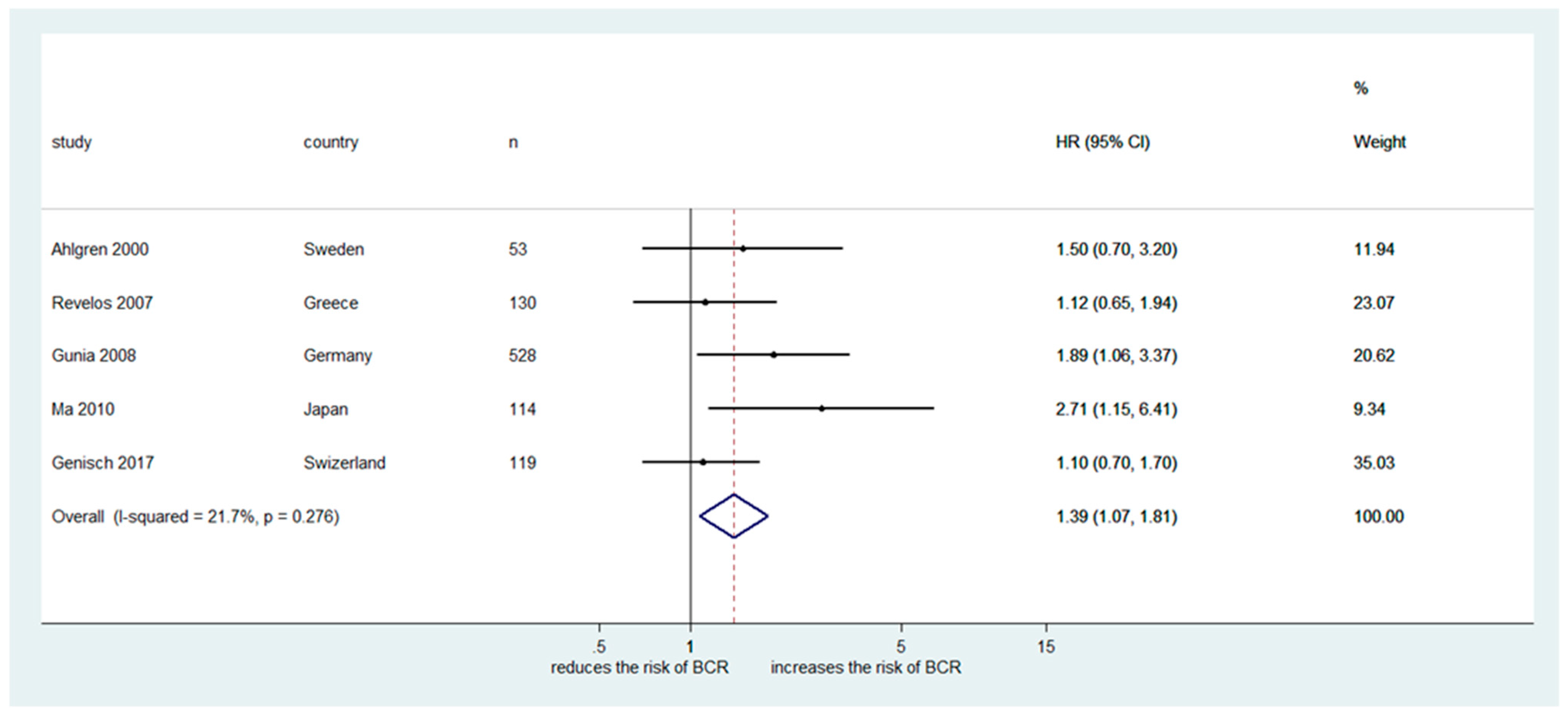
2.3. Meta-Analysis
3. Discussion
4. Materials and Methods
4.1. Searching Strategy
4.2. Inclusion Criteria
4.3. Data Extraction
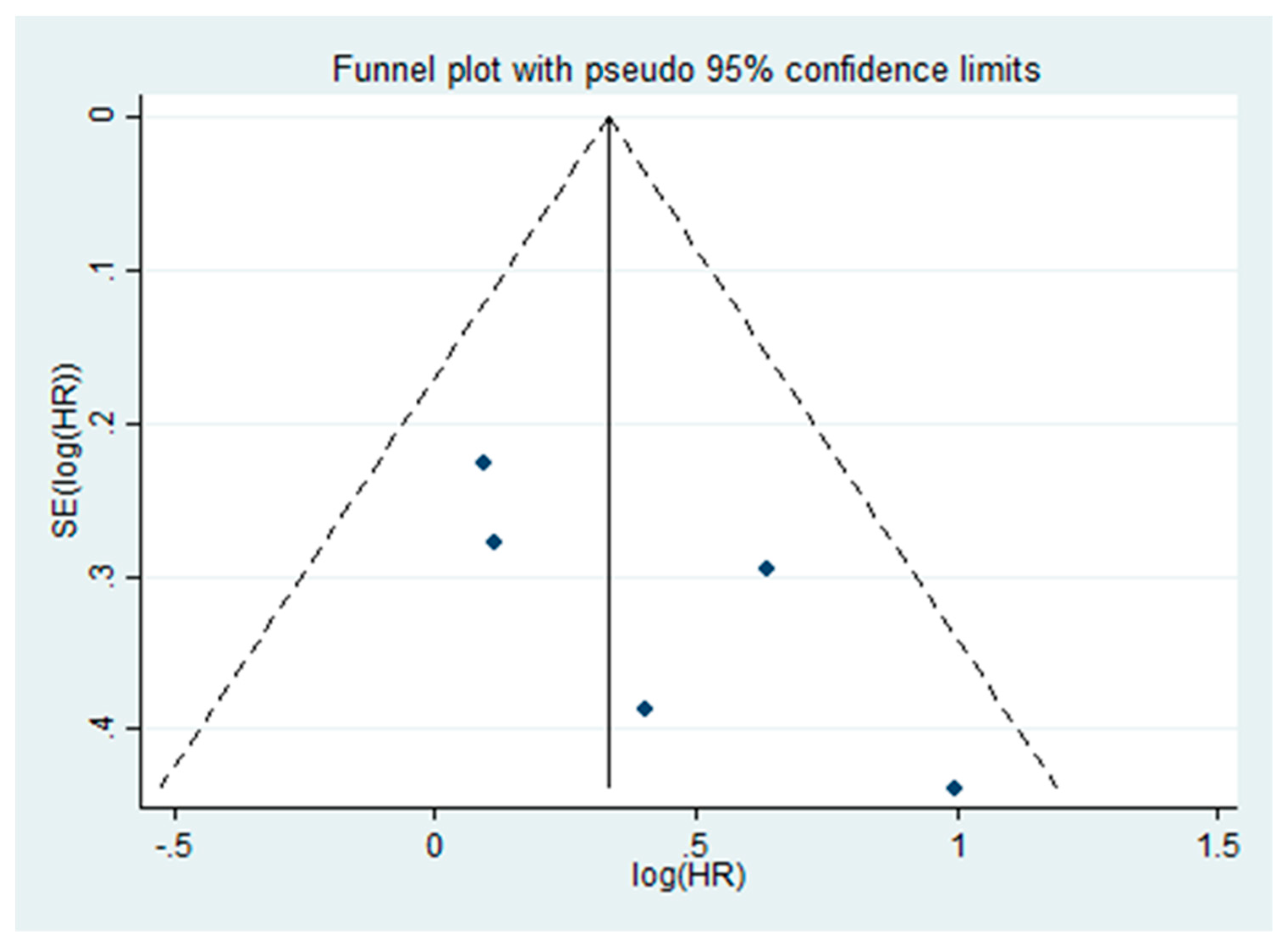
4.4. Statistical Analyses and Bias Risk Assessment
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| NED | neuroendocrine differentiation |
| PC | prostate adenocarcinoma |
| RP | radical prostatectomy |
| BCR | biochemical recurrence |
| PRISMA | Preferred Reporting Items for Systematic Review and Meta-Analysis |
| CgA | chromogranin A |
| NSE | neuron specific enolase |
| PSA | prostate specific antigen |
| ADT | androgen deprivation therapy |
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Mottet, N.; Bellmunt, J.; Bolla, M.; Briers, E.; Cumberbatch, M.G.; De Santis, M.; Fossati, N.; Gross, T.; Henry, A.M.; Joniau, S.; et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2017, 71, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Canto, E.I.; Shariat, S.F.; Kadmon, D.; Miles, B.J.; Wheeler, T.M.; Slawin, K.M. Improved detection of clinically significant, curable prostate cancer with systematic 12-core biopsy. J. Urol. 2004, 171, 1089–1092. [Google Scholar] [CrossRef] [PubMed]
- Freedland, S.J.; Humphreys, E.B.; Mangold, L.A.; Eisenberger, M.; Dorey, F.J.; Walsh, P.C.; Partin, A.W. Risk of prostate cancer-specific mortality following biochemical recurrence after radical prostatectomy. JAMA 2005, 294, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Trinh, Q.D.; Bjartell, A.; Freedland, S.J.; Hollenbeck, B.K.; Hu, J.C.; Shariat, S.F.; Sun, M.; Vickers, A.J. A systematic review of the volume-outcome relationship for radical prostatectomy. Eur. Urol. 2013, 64, 786–798. [Google Scholar] [CrossRef] [PubMed]
- Lughezzani, G.; Briganti, A.; Karakiewicz, P.I.; Kattan, M.W.; Montorsi, F.; Shariat, S.F.; Vickers, A.J. Predictive and prognostic models in radical prostatectomy candidates: A critical analysis of the literature. Eur. Urol. 2010, 58, 687–700. [Google Scholar] [CrossRef]
- Chun, F.K.; Briganti, A.; Shariat, S.F.; Graefen, M.; Montorsi, F.; Erbersdobler, A.; Steuber, T.; Salonia, A.; Currlin, E.; Scattoni, V.; et al. Significant upgrading affects a third of men diagnosed with prostate cancer: Predictive nomogram and internal validation. BJU Int. 2006, 98, 329–334. [Google Scholar] [CrossRef]
- Surcel, C.I.; van Oort, I.M.; Sooriakumaran, P.; Briganti, A.; De Visschere, P.J.; Fütterer, J.J.; Ghadjar, P.; Isbarn, H.; Ost, P.; van den Bergh, R.C.; et al. Prognostic effect of neuroendocrine differentiation in prostate cancer: A critical review. Urol. Oncol. 2015, 33, 265.e1–265.e7. [Google Scholar] [CrossRef]
- Mazzucchelli, R.; Morichetti, D.; Lopez-Beltran, A.; Cheng, L.; Scarpelli, M.; Kirkali, Z.; Montironi, R. Neuroendocrine tumours of the urinary system and male genital organs: Clinical significance. BJU Int. 2009, 103, 1464–1470. [Google Scholar] [CrossRef]
- Abrahamsson, P.A.; Cockett, A.T.; di Sant’Agnese, P.A. Prognostic significance of neuroendocrine differentiation in clinically localized prostatic carcinoma. Prostate Suppl. 1998, 8, 37–42. [Google Scholar] [CrossRef]
- Bostwick, D.G.; Qian, J.; Pacelli, A.; Zincke, H.; Blute, M.; Bergstralh, E.J.; Slezak, J.M.; Cheng, L. Neuroendocrine expression in node positive prostate cancer: Correlation with systemic progression and patient survival. J. Urol. 2002, 168, 1204–1211. [Google Scholar] [CrossRef]
- Ishida, E.; Nakamura, M.; Shimada, K.; Tasaki, M.; Konishi, N. Immunohistochemical analysis of neuroendocrine differentiation in prostate cancer. Pathobiology 2009, 76, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, E.; Trojan, L.; Friedrich, D.; Voss, M.; Weiss, C.; Michel, M.S.; Grobholz, R. Neuroendocrine tumor cells in prostate cancer: Evaluation of the neurosecretory products serotonin, bombesin, and gastrin—Impact on angiogenesis and clinical follow-up. Prostate 2011, 71, 1752–1758. [Google Scholar] [CrossRef] [PubMed]
- Bubendorf, L.; Sauter, G.; Moch, H.; Schmid, H.P.; Gasser, T.C.; Jordan, P.; Mihatsch, M.J. Ki67 labelling index: An independent predictor of progression in prostate cancer treated by radical prostatectomy. J. Pathol. 1996, 178, 437–441. [Google Scholar] [CrossRef]
- Cohen, M.K.; Arber, D.A.; Coffield, K.S.; Keegan, G.T.; McClintock, J.; Speights, V.O., Jr. Neuroendocrine differentiation in prostatic adenocarcinoma and its relationship to tumor progression. Cancer 1994, 74, 1899–1903. [Google Scholar] [CrossRef]
- Ahlegren, G.; Pedersen, K.; Lundberg, S.; Aus, G.; Hugosson, J.; Abrahamsson, P. Neuroendocrine differentiation is not prognostic of failure after radical prostatectomy but correlates with tumor volume. Urology 2000, 56, 1011–1015. [Google Scholar] [CrossRef]
- Gunia, S.; Albrecht, K.; Koch, S.; Herrmann, T.; Ecke, T.; Loy, V.; Linke, J.; Siegsmund, M.; May, M. Ki67 staining index and neuroendocrine differentiation aggravate adverse prognostic parameters in prostate cancer and are characterized by negligible inter-observer variability. World J. Urol. 2008, 26, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Krupski, T.; Petroni, G.R.; Frierson, H.F., Jr.; Theodorescu, J.U. Microvessel density, p53, retinoblastoma, and chromogranin A immunohistochemistry as predictors of disease-specific survival following radical prostatectomy for carcinoma of the prostate. Urology 2000, 55, 743–749. [Google Scholar] [CrossRef]
- Ma, Z.; Tsuchiya, N.; Yuasa, T.; Huang, M.; Obara, T.; Narita, S.; Horikawa, Y.; Tsuruta, H.; Saito, M.; Satoh, S.; et al. Clinical significance of polymorphism and expression of chromogranin a and endothelin-1 in prostate cancer. J. Urol. 2010, 184, 1182–1188. [Google Scholar] [CrossRef]
- Revelos, K.; Petraki, C.; Scorilas, A.; Stefanakis, S.; Malovrouvas, D.; Alevizopoulos, N.; Kanellis, G.; Halapas, A.; Koutsilieris, M. Correlation of androgen receptor status, neuroendocrine differentiation and angiogenesis with time-to-biochemical failure after radical prostatectomy in clinically localized prostate cancer. Anticancer Res. 2007, 27, 3651–3660. [Google Scholar]
- Veltri, R.W.; Isharwal, S.; Miller, M.C.; Epstein, J.I.; Mangold, L.A.; Humphreys, E.; Partin, A.W. Long-term assessment of prostate cancer progression free survival: Evaluation of pathological parameters, nuclear shape and molecular biomarkers of pathogenesis. Prostate 2008, 68, 1806–1815. [Google Scholar] [CrossRef]
- Weinstein, M.H.; Partin, A.W.; Veltri, R.W.; Epstein, J.I. Neuroendocrine differentiation in prostate cancer: Enhanced prediction of progression after radical prostatectomy. Hum. Pathol. 1996, 27, 683–687. [Google Scholar] [CrossRef]
- Genitsch, V.; Zlobec, I.; Seiler, R.; Thalmann, G.N.; Fleischmann, A. Neuroendocrine Differentiation in Metastatic Conventional Prostate Cancer Is Significantly Increased in Lymph Node Metastases Compared to the Primary Tumors. Int. J. Mol. Sci. 2017, 18, 1640. [Google Scholar] [CrossRef]
- Theodorescu, D.; Broder, S.R.; Boyd, J.C.; Mills, S.E.; Frierson, H.F., Jr. Cathepsin D and chromogranin A as predictors of long term disease specific survival after radical prostatectomy for localized carcinoma of the prostate. Cancer 1997, 80, 2109–2119. [Google Scholar] [CrossRef]
- Noordzij, M.A.; van der Kwast, T.H.; van Steenbrugge, G.J.; Hop, W.J.; Schroder, F.H. The prognostic influence of neuroendocrine cells in prostate cancer: Results of a long-term follow-up study with patients treated by radical prostatectomy. Int. J. Cancer 1995, 62, 252–258. [Google Scholar] [CrossRef]
- Parimi, V.; Goyal, R.; Poropatich, K.; Yang, X.J. Neuroendocrine differentiation of prostate cancer: A review. Am. J. Clin. Exp. Urol. 2014, 2, 273–285. [Google Scholar]
- Xing, N.; Qian, J.; Bostwick, D.; Bergstralh, E.; Young, C.Y. Neuroendocrine cells in human prostate over-express the anti-apoptosis protein survivin. Prostate 2001, 48, 7–15. [Google Scholar] [CrossRef]
- Mathieu, R.; Lucca, I.; Vartolomei, M.D.; Mbeutcha, A.; Klatte, T.; Seitz, C.; Karakiewicz, P.I.; Fajkovic, H.; Sun, M.; Lotan, Y.; et al. Role of survivin expression in predicting biochemical recurrence after radical prostatectomy: A multi-institutional study. BJU Int. 2017, 119, 234–238. [Google Scholar] [CrossRef]
- Shariat, S.F.; Lotan, Y.; Saboorian, H.; Khoddami, S.M.; Roehrborn, C.G.; Slawin, K.M.; Ashfaq, R. Survivin expression is associated with features of biologically aggressive prostate carcinoma. Cancer 2004, 100, 751–757. [Google Scholar] [CrossRef]
- Puca, L.; Vlachostergios, P.J.; Beltran, H. Neuroendocrine Differentiation in Prostate Cancer: Emerging Biology, Models, and Therapies. Cold Spring Harb. Perspect. Med. 2018, 29, a030593. [Google Scholar] [CrossRef]
- Terry, S.; Beltran, H. The many faces of neuroendocrine differentiation in prostate cancer progression. Front. Oncol. 2014, 25, 60. [Google Scholar] [CrossRef]
- Shariat, S.F.; Anwuri, V.A.; Lamb, D.J.; Shah, N.V.; Wheeler, T.M.; Slawin, K.M. Association of preoperative plasma levels of vascular endothelial growth factor and soluble vascular cell adhesion molecule-1 with lymph node status and biochemical progression after radical prostatectomy. J. Clin. Oncol. 2004, 22, 1655–1663. [Google Scholar] [CrossRef]
- Shariat, S.F.; Roehrborn, C.G.; McConnell, J.D.; Park, S.; Alam, N.; Wheeler, T.M.; Slawin, K.M. Association of the circulating levels of the urokinase system of plasminogen activation with the presence of prostate cancer and invasion, progression, and metastasis. J. Clin. Oncol. 2007, 25, 349–355. [Google Scholar] [CrossRef]
- Wang, H.T.; Yao, Y.H.; Li, B.G.; Tang, Y.; Chang, J.W.; Zhang, J. Neuroendocrine Prostate Cancer (NEPC) progressing from conventional prostatic adenocarcinoma: Factors associated with time to development of NEPC and survival from NEPC diagnosis-a systematic review and pooled analysis. J. Clin. Oncol. 2014, 32, 3383–3390. [Google Scholar] [CrossRef]
- Epstein, J.I.; Amin, M.B.; Beltran, H.; Lotan, T.L.; Mosquera, J.M.; Reuter, V.E.; Robinson, B.D.; Troncoso, P.; Rubin, M.A. Proposed morphologic classification of prostate cancer with neuroendocrine differentiation. Am. J. Surg. Pathol. 2014, 38, 756–767. [Google Scholar] [CrossRef]
- Shariat, S.F.; Semjonow, A.; Lilja, H.; Savage, C.; Vickers, A.J.; Bjartell, A. Tumor markers in prostate cancer I: Blood-based markers. Acta Oncol. 2011, 50, 61–75. [Google Scholar] [CrossRef]
- Niedworok, C.; Tschirdewahn, S.; Reis, H.; Lehmann, N.; Szücs, M.; Nyirády, P.; Romics, I.; Rübben, H.; Szarvas, T. Serum Chromogranin A as a Complementary Marker for the Prediction of Prostate Cancer-Specific Survival. Pathol. Oncol. Res. 2017, 23, 643–650. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernán, M.A.; Reeves, B.C.; Savović, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
| Author | Year | Region | Design | Recruitment Period | No. pts | Markers | Oncological End Point |
|---|---|---|---|---|---|---|---|
| Cohen [15] | 1994 | USA | Retrospective | 1986–1989 | 38 | CgA, NSE | Disease progression (LR, BCR, DM) |
| Noordzij [25] | 1995 | Netherlands | Retrospective | 1977–1987 | 90 | CgA | Disease progression (LR, DM), CSS |
| Bubendorf [14] | 1996 | Switzerland | Retrospective | 1978–1993 | 137 | CgA, NSE | Disease progression (LR, BCR, DM) |
| Weinstein [22] | 1996 | USA | Retrospective | N/A | 104 | CgA | PFS (BCR) |
| Theodorescu [24] | 1997 | USA | Retrospective | 1970–1984 | 71 | CgA | DSS, Long-term Survival |
| Abrahamsson [10] | 1998 | USA | Retrospective | 1973–1989 | 87 | S, CgA, NSE | Disease progression (LR, BCR, DM) |
| Krupski [18] | 2000 | USA | Retrospective | 1970–1984 | 42 | CgA | DSS (LR, BCR, DM) |
| Ahlgren [16] | 2000 | Sweden | Retrospective | N/A | 53 | CgA | PFS (BCR) |
| Bostwick [11] | 2002 | USA | Retrospective | 1987-1992 | 196 | S, CgA | DM, CSD, All cause death |
| Revelos [20] | 2007 | Greece | Retrospective | N/A | 130 | CgA | PFS (BCR) |
| Gunia [17] | 2008 | Germany | Retrospective | 1996–2003 | 528 | CgA | BFS (BCR) |
| Veltri [21] | 2008 | USA | Retrospective | 1975–1991 | 105 | CgA | PFS (LR, BCR, DM) |
| Ishida [12] | 2008 | Japan | Retrospective | N/A | RP (50) Vs NADT +RP (46) | S, CgA | BCR |
| Ma [19] | 2010 | Japan | Retrospective | N/A | RP (114) of PCa cases (435) | CgA | BFS (BCR) |
| Heinrich [13] | 2011 | Germany | Retrospective | N/A | 175 | S, CgA | BCR |
| Genitsch [23] | 2017 | Switzerland | Retrospective | 1989–2006 | 119 | CgA | PFS (BCR), CSS, OS |
| Author | Age, Year (mean/median) | Pre-Operative PSA, mg/dl (n) | Surgical GS (n) | Pathological Stage (n) | Follow-up Duration | Independent Correlation with Oncologic Outcomes |
|---|---|---|---|---|---|---|
| Cohen [15] | N/A | N/A | ≤6 (20), 7 (14), ≥8 (4) | II† (22), III (16) | Mean: 50.5 months (range, 2–77) | NS |
| Noordzij [25] | 62 (range, 47–74) | N/A | ≤6 (26)/7 (36)/≥8 (28) | T2 (22), T3 (66), T4 (2), N+ (7) | Mean: 86 months (range, 1–203) | NS |
| Bubendorf [14] | 65.3 (range, 45–82) | N/A | <7(68), ≥7 (69) | PT1 (4), PT2 (43), PT3 (90), N+(34) | Mean: 5.4 years (range, 1–15) | NS |
| Weinstein [22] | N/A | N/A | ≤6 (59), >6 (45) | Organ confined (21%) SVI (0), LNI (0) | Mean: 8 years (range,7–10) | S |
| Theodorescu [24] | 60.5 (range, 42–72) | N/A | ≤7 (48), ≥8 (23) | Capsular penetration: −(37), +(31), N/A(3) SVI: −(50), +(13), N/A(8) LNI: −(13), +(1), N/A(57) | N/A | S |
| Abrahamsson [10] | 66 (range, 50–77) | N/A | Mean GS: 6–7 | A (1), B (27), C (50), D (9) | Mean: 4.2 years (range, 1.8–10.1) | NS |
| Krupski [18] | 62 (range, 42–72) | N/A | ≤6 (22), 7 (3), ≥8 (17) | N/A | Median: 10 years (range, 3.5–20) | NS |
| Ahlgren [16] | N/A | <10 (24), 11–20 (22), >20 (7) | ≤6 (19), 7 (16), ≥8 (18) | T1b-T1c (22), T2-3 (31) | Mean: 39 ± 1 months | NS |
| Bostwick [11] | 65.7 (range, 47–79) | Median: 21.4 (range, 0.9–616) | N/A | N+ (196) | Mean: 6.8 years (range, 0.3–11) | NS* |
| Revelos [20] | 66 (range, 47–76) | Median: 9.23 (2.5–45.0) | ≤6 (29), 7 (75), ≥8 (26) | ECE: +(70) −(60)/SVI: +(34) −(96), LNI: +(10) −(120) | Median: 28 months (1–97) | NS |
| Gunia [17] | 63.8 (range, 44–79) | ≤20 (472), >20 (56) | ≤6 (316), 7 (157), ≥8 (55) | T2 (367), T3 (149), T4 (12), N0 (412), N1 (38), Nx (78) | Median: 46.4 months (range, 10–116) | S |
| Veltri [21] | 59.62 | N/A | < 7 (64), ≥ 7 (41) | T2: (75), >T2 (30) SVI: +(1), −(104) LNI (0) | Mean: 17.3 years (range:2–26) | NS |
| Ishida [12] | 69 (range, 54–78) | Mean 7.5 (range 0.0–50.3) | RP: ≤6 (25), 7 (21), ≥8 (4)/NADT +RP: ≤6 (13), 7 (8), ≥8 (12) | I (24), II (29), III (25), IV(12) | N/A | NS |
| Ma [19] | 70.28±7.43 | N/A | ≤6 (14), 7 (202), ≥8 (164), N/A (55) | T1a-bN0M0 (10), T1c-2N0M0 (191), T3-4N0M0 (83), T1-4N1M0-1 (25), T1-4N0-1M1 (126) | N/A | S |
| Heinrich [13] | 63.3 ± 5.9 years | N/A | ≤6 (86), 7 (63), ≥8 (24) | T2 (85), T3 (86), T4 (3) | Medium:17.1 months (range, 2–44) | NS |
| Genitsch [23] | 65 (range, 45–75) | N/A | ≤6 (12), 7 (63), ≥8 (44) | T2: 14, T3:105, N+ (119) | Median: 5.9 years (0.1–15.2) | NS |
| Author | Confounding | Participant Selection | Classification of Interventions | Deviations from Intended Intervention | Missing Data | Measurement of Outcomes | Selection of the Reported Result | Overall |
|---|---|---|---|---|---|---|---|---|
| Abrahamsson [10] | Serious | Serious | Low | Serious | Low | Moderate | Low | Serious |
| Ahlgren [16] | Serious | Serious | Low | Moderate | Low | Low | Low | Serious |
| Bostwick [11] | Serious | Serious | Low | Serious | Low | Moderate | Moderate | Serious |
| Bubendorf [14] | Serious | Serious | Low | Moderate | Low | Low | Moderate | Serious |
| Cohen [15] | Serious | Serious | Low | Serious | Low | Moderate | Low | Serious |
| Gunia [17] | Serious | Serious | Low | Serious | Low | Moderate | Low | Serious |
| Heinrich [13] | Serious | Serious | Low | Serious | Low | Low | Moderate | Serious |
| Ishida [12] | Serious | Serious | Low | Serious | Low | Moderate | Moderate | Serious |
| Krupski [18] | Serious | Serious | Low | Serious | Low | Moderate | Moderate | Moderate |
| Ma [19] | Serious | Serious | Low | Low | Low | Moderate | Low | Serious |
| Noordzij [25] | Serious | Serious | Low | Moderate | Low | Low | Moderate | Serious |
| Revelos [20] | Moderate | Serious | Low | Serious | Low | Low | Moderate | Serious |
| Theodorescu [24] | Serious | Moderate | Low | Serious | Low | Moderate | Low | Serious |
| Veltri [21] | Serious | Serious | Low | Serious | Low | Low | Moderate | Serious |
| Weinstein [22] | Moderate | Moderate | Low | Moderate | Low | Moderate | Low | Moderate |
| Genitsch [23] | Serious | Serious | Low | Serious | Low | Moderate | Moderate | Serious |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kardoust Parizi, M.; Iwata, T.; Kimura, S.; Janisch, F.; Abufaraj, M.; Karakiewicz, P.I.; Enikeev, D.; Rapoport, L.M.; Hutterer, G.; Shariat, S.F. Focal Neuroendocrine Differentiation of Conventional Prostate Adenocarcinoma as a Prognostic Factor after Radical Prostatectomy: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2019, 20, 1374. https://doi.org/10.3390/ijms20061374
Kardoust Parizi M, Iwata T, Kimura S, Janisch F, Abufaraj M, Karakiewicz PI, Enikeev D, Rapoport LM, Hutterer G, Shariat SF. Focal Neuroendocrine Differentiation of Conventional Prostate Adenocarcinoma as a Prognostic Factor after Radical Prostatectomy: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2019; 20(6):1374. https://doi.org/10.3390/ijms20061374
Chicago/Turabian StyleKardoust Parizi, Mehdi, Takehiro Iwata, Shoji Kimura, Florian Janisch, Mohammad Abufaraj, Pierre I. Karakiewicz, Dmitry Enikeev, Leonid M. Rapoport, Georg Hutterer, and Shahrokh F. Shariat. 2019. "Focal Neuroendocrine Differentiation of Conventional Prostate Adenocarcinoma as a Prognostic Factor after Radical Prostatectomy: A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 20, no. 6: 1374. https://doi.org/10.3390/ijms20061374
APA StyleKardoust Parizi, M., Iwata, T., Kimura, S., Janisch, F., Abufaraj, M., Karakiewicz, P. I., Enikeev, D., Rapoport, L. M., Hutterer, G., & Shariat, S. F. (2019). Focal Neuroendocrine Differentiation of Conventional Prostate Adenocarcinoma as a Prognostic Factor after Radical Prostatectomy: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 20(6), 1374. https://doi.org/10.3390/ijms20061374





