Artificial Light at Night Alleviates the Negative Effect of Pb on Freshwater Ecosystems
Abstract
1. Introduction
2. Results
2.1. Water Chemistry
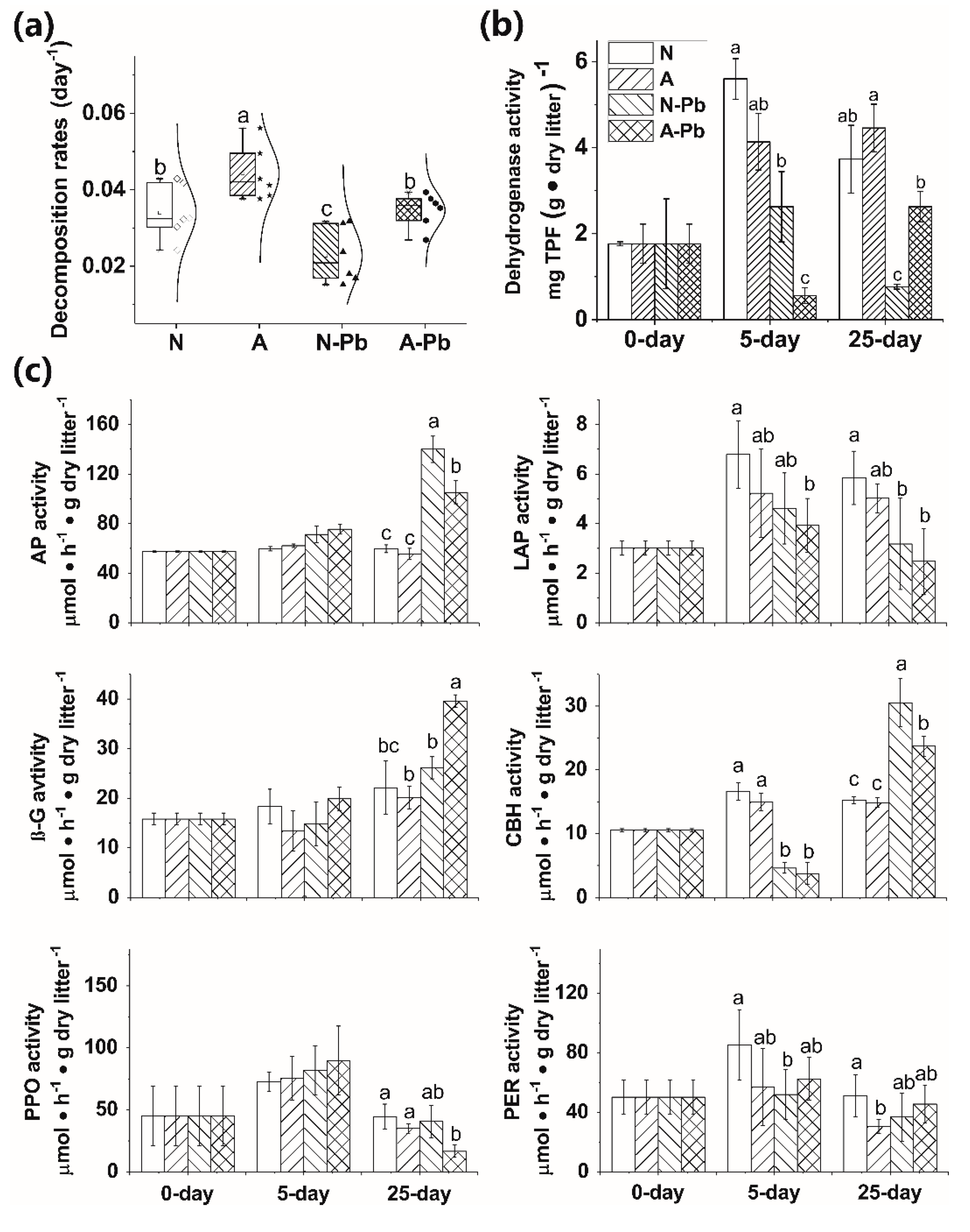
2.2. Leaf Litter Chemical Characteristics and Decomposition Rates
2.3. Microbial Biomass
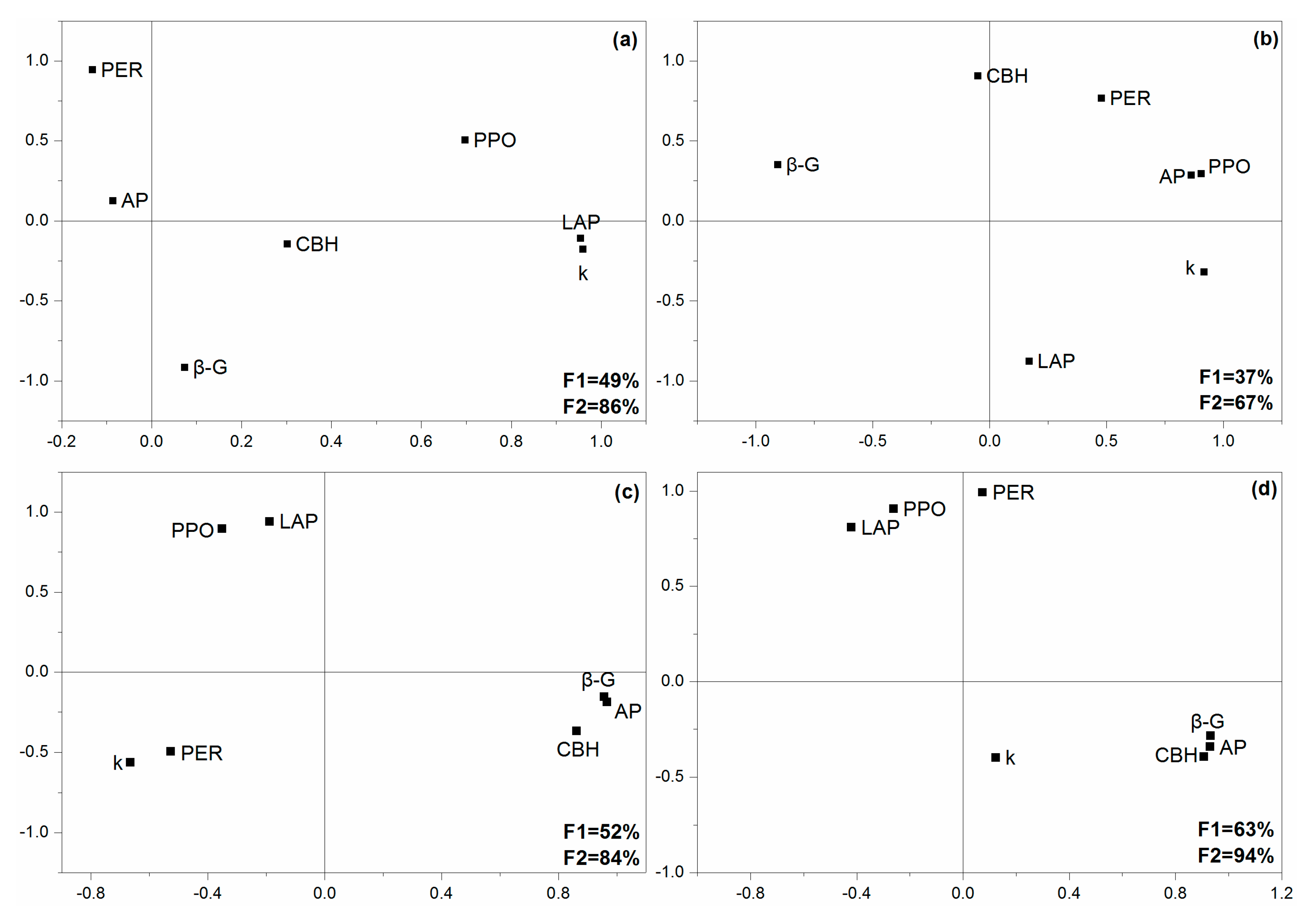
2.4. Changes in Extracellular Enzyme Activities
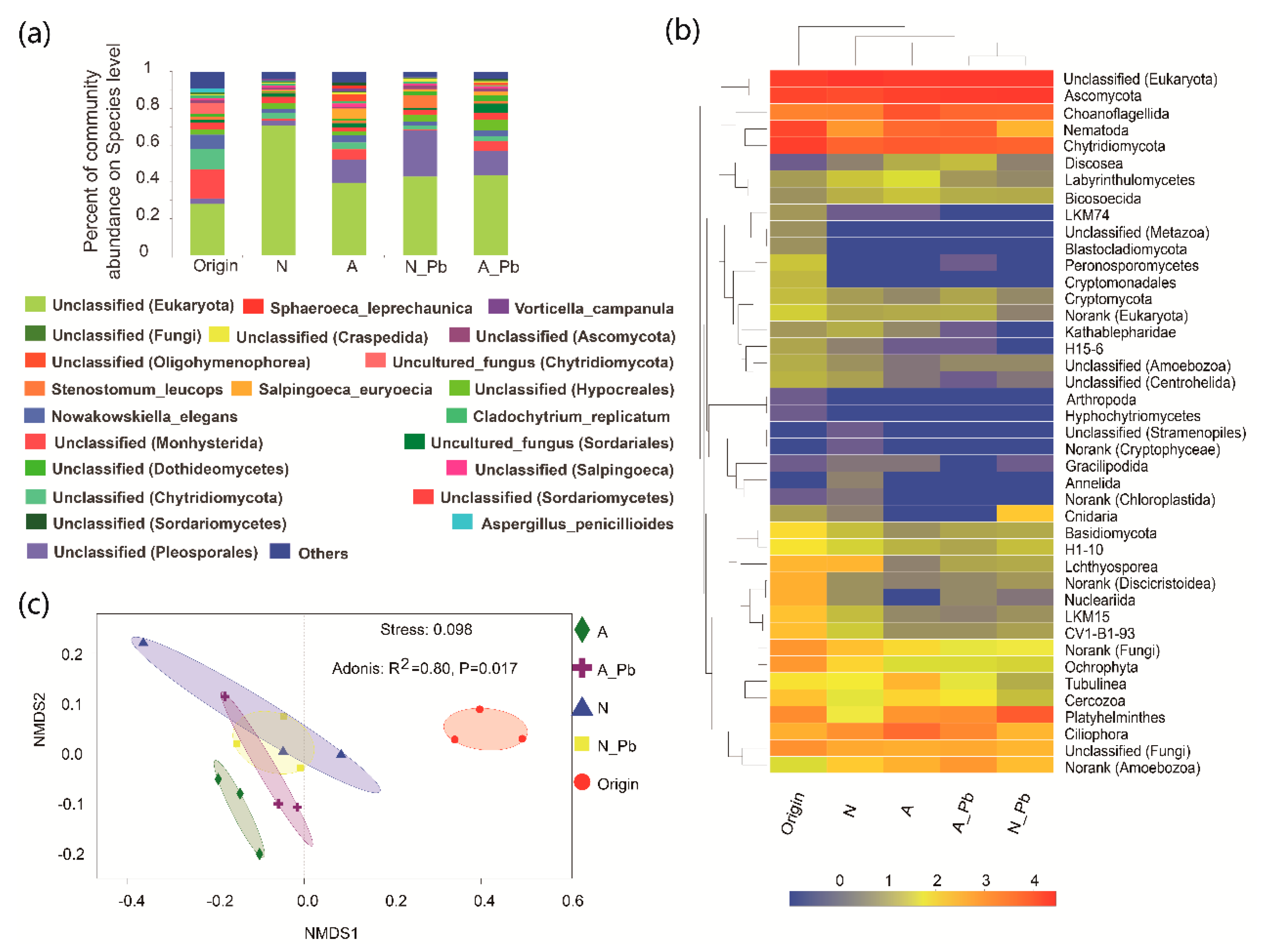
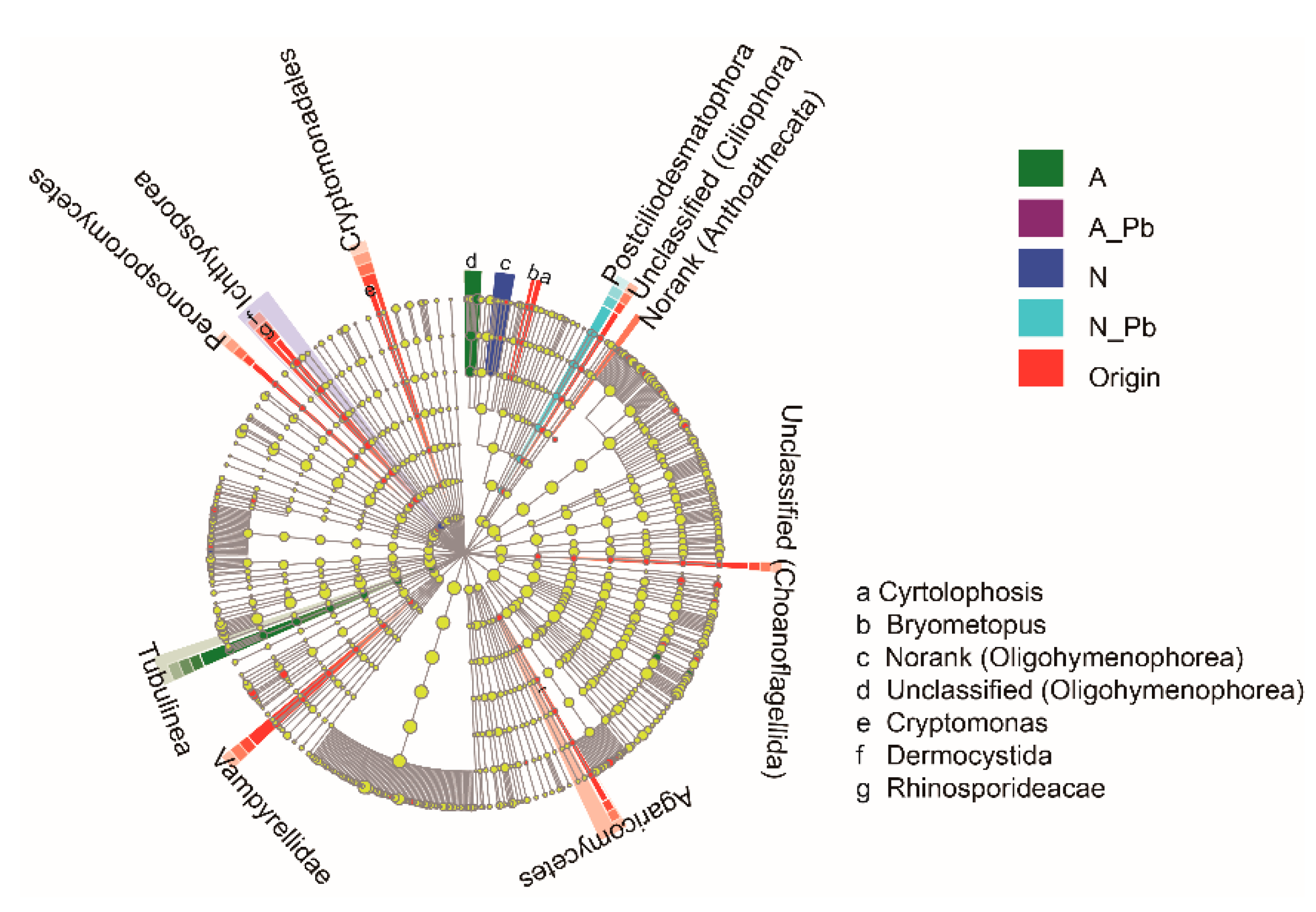
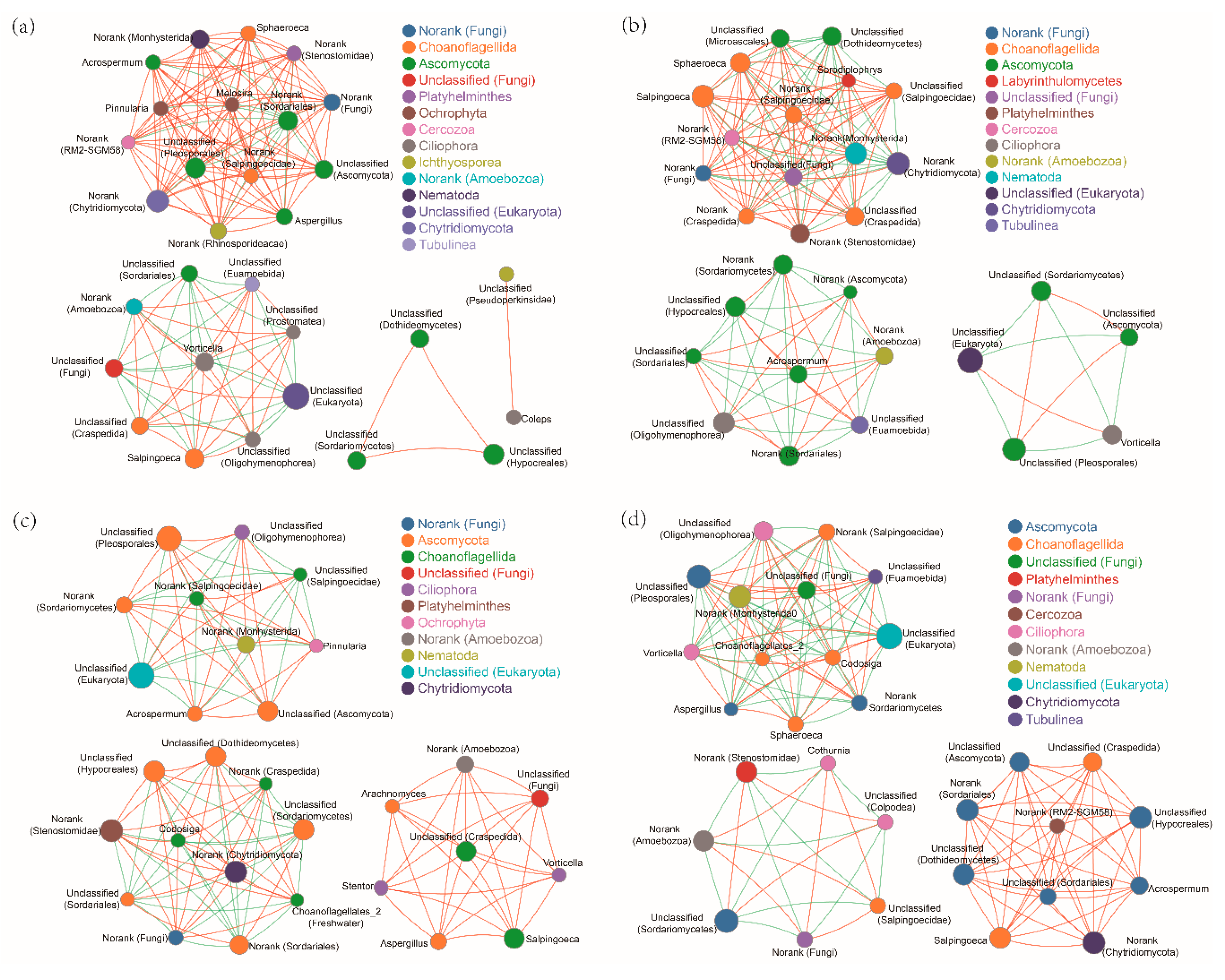
2.5. Fungal Communities
3. Discussion
4. Materials and Methods
4.1. Preparation of P. stenoptera Leaf Litter
4.2. Experimental Design
4.3. Leaf Mass Loss and Chemical Characteristics
4.4. Microbial Biomass
4.5. Extracellular Enzyme Activities
4.6. Fungal Diversity and Community Structure
4.7. Data Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| AP | β-G | LAP | CBH | PPO | PER | |
|---|---|---|---|---|---|---|
| Treatments | ||||||
| F | 87.54 | 14.83 | 5.95 | 4.96 | 0.04 | 0.53 |
| P | <0.001 | <0.001 | 0.003 | 0.008 | 0.989 | 0.667 |
| Sampling time | ||||||
| F | 137.22 | 56.00 | 10.65 | 221.92 | 5.36 | 1.64 |
| P | <0.001 | <0.001 | <0.001 | <0.001 | 0.012 | 0.215 |
| Treatments × Sampling time | ||||||
| F | 57.99 | 8.03 | 1.69 | 69.64 | 0.223 | 0.19 |
| P | <0.001 | <0.001 | 0.167 | <0.001 | 0.955 | 0.976 |
| Par. | pH | DO (mg·L−1) | Cond. (μs·cm−1) | Sal | NTU | OPR (mv) | Chla (μg·L−1) | Ca2+ (mg·L−1) |
| water | 6.21 | 3.88 | 0.033 | 0.013 | 0.47 | 154 | 0.65 | 75 |
| Par. | TSS (mg·L−1) | DOC (mg·L−1) | TP | NH4+ (ppm) | Temp. (°C) | TN | Mg2+ (mg·L−1) | Pb3+ (mg·L−1) |
| water | 3.55 | 1.25 | 0.05 | 0.73 | 20.01 | 1.44 | 4.12 | < 0.01 |
References
- Tlili, A.; Jabiol, J.; Behra, R.; Gil-Allué, C.; Gessner, M.O. Chronic exposure effects of silver nanoparticles on stream microbial decomposer communities and ecosystem functions. Environ. Sci. Technol. 2017, 51, 2447–2455. [Google Scholar] [CrossRef]
- Wilkinson, J.; Hooda, P.S.; Barker, J.; Barton, S.; Swinden, J. Occurrence, fate and transformation of emerging contaminants in water: An overarching review of the field. Environ. Pollut. 2017, 231, 954–970. [Google Scholar] [CrossRef] [PubMed]
- Sanders, D.; Gaston, K.J. How ecological communities respond to artificial light at night. J. Exp. Zool. Part A Ecol. Integr. Physiol. 2018, 329, 8–9. [Google Scholar] [CrossRef] [PubMed]
- Holmstrup, M.; Sorensen, J.G.; Overgaard, J.; Bayley, M.; Bindesbøl, A.M.; Slotsbo, S.; Fisker, K.V.; Maraldo, K.; Waagner, D.; Labouriau, R.; et al. Body metal concentrations and glycogen reserves in earthworms (Dendrobaena octraedra) from contaminated and uncontaminated forest soil. Environ. Pollut. 2010, 159, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Rich, C.; Longcore, T. Ecological Consequences of Artificial Night Lighting; Island Press: Washington, DC, USA, 2006. [Google Scholar]
- Gaston, K.J.; Bennie, J.; Davies, T.W.; Hopkins, J. The ecological impacts of nighttime light pollution: A mechanistic appraisal. Biol. Rev. 2013, 88, 912–927. [Google Scholar] [CrossRef]
- Bennie, J.; Davies, T.W.; Cruse, D.; Bell, F.; Gaston, K.J. Artificial light at night alters grassland vegetation species composition and phenology. J. Appl. Ecol. 2017, 55, 442–450. [Google Scholar] [CrossRef]
- Cabrera-Cruz, S.A.; Smolinsky, J.A.; Buler, J.J. Light pollution is greatest within migration passage areas for nocturnally-migrating birds around the world. Sci. Rep. 2018, 8, 3261. [Google Scholar] [CrossRef] [PubMed]
- Singhal, R.K.; Kumar, M.; Bose, B. Ecophysiological responses of artificial night light pollution in plants. Russ. J. Plant Physiol. 2018. [Google Scholar] [CrossRef]
- Davies, T.W.; Smyth, T. Why artificial light at night should be a focus for global change research in the 21st century. Glob. Chang. Biol. 2018, 24, 872–882. [Google Scholar] [CrossRef]
- Hölker, F.; Wurzbacher, C.; Weißenborn, C.; Monaghan, M.T.; Holzhauer, S.I.; Premke, K. Microbial diversity and community respiration in freshwater sediments influenced by artificial light at night. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2015, 370, 20140130. [Google Scholar] [CrossRef] [PubMed]
- Bolton, D.; Mayer-Pinto, M.; Clark, G.F.; Dafforn, K.A.; Brassil, W.A.; Becker, A.; Johnston, E.L. Coastal urban lighting has ecological consequences for multiple trophic levels under the sea. Sci. Total Environ. 2017, 576, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, A.; Holmes, N.D.; Ryan, P.G.; Wilson, K.J.; Faulquier, L.; Murillo, Y.; Raine, A.F.; Penniman, J.F.; Neves, V.; Rodríguez, B.; et al. Seabird mortality induced by land-based artificial lights. Conserv. Biol. 2017, 31, 986–1001. [Google Scholar] [CrossRef] [PubMed]
- Poulin, C.; Bruyant, F.; Laprise, M.H.; Cockshutt, A.M.; Vandenhecke, M.R.; Huot, Y. The impact of light pollution on diel changes in the photophysiology of Microcystis aeruginosa. J. Plankton Res. 2014, 36, 286–291. [Google Scholar] [CrossRef]
- Grubisic, M.; Singer, G.; Bruno, M.C.; van Grunsven, R.H.A.; Manfrin, A.; Monaghan, M.T.; Holker, F. Artificial light at night decreases biomass and alters community composition of benthic primary producers in a sub-alpine stream. Limnol. Oceanogr. 2017, 62, 2799–2810. [Google Scholar] [CrossRef]
- Wallace, J.B.; Eggert, S.L.; Meyer, J.L.; Webster, J.R. Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 1997, 277, 102–104. [Google Scholar] [CrossRef]
- Woodward, G.; Gessner, M.O.; Giller, P.S.; Gulis, V.; Hladyz, S.; Lecerf, A.; Malmqvist, B.; McKie, B.G.; Tiegs, S.D.; Cariss, H.; et al. Continental-scale effects of nutrient pollution on stream ecosystem functioning. Science 2012, 336, 1438–1440. [Google Scholar] [CrossRef]
- Du, J.J.; Zhang, Y.; Liu, L.; Qv, M.; Lv, Y.; Yin, Y.; Zhou, Y.; Cui, M.; Zhu, Y.; Zhang, H. Can visible light impact litter decomposition under pollution of ZnO nanoparticles? Chemosphere 2017, 187, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Du, J.J.; Qv, M.X.; Zhang, Y.Y.; Yin, X.; Wan, N.; Zhang, B.; Zhang, H. The potential phototoxicity of nano-scale ZnO induced by visible light on freshwater ecosystems. Chemosphere 2018, 208, 698–706. [Google Scholar] [CrossRef]
- Pourrut, B.; Shahid, M.; Camille, D.; Winterton, P.; Pinelli, E. Lead uptake, toxicity, and detoxification in plants. Rev. Environ. Contam. Toxicol. 2011, 213, 113–136. [Google Scholar]
- Ashraf, U.; Kanu, A.S.; Deng, Q.; Mo, Z.; Pan, S.; Tian, H.; Tang, X. Lead (Pb) toxicity; physio-biochemical mechanisms, grain yield, quality, and Pb distribution proportions in scented rice. Front. Plant Sci. 2017, 8, 259. [Google Scholar] [CrossRef]
- Xia, J.; Jin, C.; Pan, Z.; Sun, L.; Fu, Z.; Jin, Y. Chronic exposure to low concentrations of lead induces metabolic disorder and dysbiosis of the gut microbiota in mice. Sci. Total Environ. 2018, 631–632, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Oguma, A.Y.; Klerks, P.L. Evidence for mild sediment Pb contamination affecting leaf-litter decomposition in a lake. Ecotoxicology 2015, 24, 1322–1329. [Google Scholar] [CrossRef] [PubMed]
- Fechner, L.C.; Dufour, M.; Gourlay-Francé, C. Pollution-induced community tolerance of freshwater biofilms: Measuring heterotrophic tolerance to Pb using an enzymatic toxicity test. Ecotoxicology 2012, 21, 2123–2131. [Google Scholar] [CrossRef] [PubMed]
- Zeng, G.; Wan, J.; Huang, D.; Hu, L.; Huang, C.; Cheng, M.; Xue, W.; Gong, X.; Wang, R.; Jiang, D. Precipitation, adsorption and rhizosphere effect: The mechanisms for phosphate-induced Pb immobilization in soils—A review. J. Hazard. Mater. 2017, 339, 354–367. [Google Scholar] [CrossRef]
- Babich, H.; Stotzky, G. Abiotic factors affecting the toxicity of lead to fungi. Appl. Environ. Microb. 1979, 38, 506–513. [Google Scholar]
- Khan, S.; Cao, Q.; Chen, B.D.; Zhu, Y.G. Humic acids increase the phytoavailability of Cd and Pb to wheat plants cultivated in freshly spiked, contaminated soil. J. Soil Sediment. 2006, 6, 236–242. [Google Scholar]
- Kurtyka, R.; MałKowski, E.; Burdach, Z.; Kita, A.; Karcz, W. Interactive effects of temperature and heavy metals (Cd, Pb) on the elongation growth in maize coleoptiles. C. R. Biol. 2012, 335, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Li, R.Y.; Zhou, Z.G.; Xie, X.J.; Li, Y.X.; Zhang, Y.H.; Xu, X.H. Effects of dissolved organic matter on uptake and translocation of lead in Brassica chinensis and potential health risk of Pb. Int. J. Environ. Res. Public Health. 2016, 13, 687. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, Y.F.; Zhang, X.Y.; Li, R.Z.; Chen, Y.K.; Meng, Q.J. Investigating the behavior of binding properties between dissolved organic matter (DOM) and Pb(II) during the soil sorption process using parallel factor analysis (PARAFAC) and two-dimensional correlation spectroscopy (2D-COS). Environ. Sci. Pollut. Res. 2017, 24, 25156–25165. [Google Scholar] [CrossRef]
- Sánchez-Marín, P.; Liu, F.J.; Chen, Z.Z.; Fortin, C.; Campbell, P.G.C. Microalgal-driven pH changes in the boundary layer lead to apparent increases in Pb internalization by a unicellular alga in the presence of citrate. Limnol. Oceanogr. 2018, 63, 1328–1339. [Google Scholar] [CrossRef]
- Reddy, Y.S.; Srivalliputturu, S.B.; Bharatraj, D.K. The effect of lead (Pb) exposure and iron (Fe) deficiency on intestinal lactobacilli, E. coli and yeast: A study in experimental rats. J. Occup. Health. 2018, 60, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Svoboda, K.R.; Lenz, K.A.; Pattison, C.; Ma, H. Toxicity interactions between manganese (Mn) and lead (Pb) or cadmium (Cd) in a model organism the nematode C. elegans. Environ. Sci. Pollut. Res. 2018, 25, 15378–15389. [Google Scholar] [CrossRef]
- Yin, Y.W.; Zhang, P.J.; Yue, X.Y.; Du, X.Y.; Li, W.; Yin, Y.L.; Yi, C.; Li, Y.H. Effect of sub-chronic exposure to lead (Pb) and Bacillus subtilis on Carassius auratus gibelio: Bioaccumulation, antioxidant responses and immune responses. Ecotoxcol. Environ. Saf. 2018, 161, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Abdelkrim, S.; Jebara, S.H.; Saadani, O.; Chiboub, M.; Jebara, M. Effect of Pb-resistant plant growth-promoting rhizobacteria inoculation on growth and lead uptake by Lathyrus sativus. J. Basic. Microbiol. 2018, 58, 579–589. [Google Scholar] [CrossRef]
- Batista, D.; Pascoal, C.; Cássio, F. Temperature modulates AgNP impacts on microbial decomposer activity. Sci. Total Environ. 2017, 601–602, 1324–1332. [Google Scholar] [CrossRef]
- Austin, A.T.; Ballare, C.L. Dual role of lignin in plant litter decomposition in terrestrial ecosystems. Proc. Natl. Acad. Sci. USA 2010, 107, 4618–4622. [Google Scholar] [CrossRef]
- King, J.; Brandt, L.; Adair, E.C. Shedding light on plant litter decomposition: Advances, implications and new directions in understanding the role of photodegradation. Biogeochemistry 2012, 111, 57–81. [Google Scholar] [CrossRef]
- Wang, J.; Liu, L.L.; Wang, X.; Chen, Y.W. Interaction between abiotic photodegradation and microbial decomposition under ultraviolet radiation. Glob. Chang. Biol. 2015, 21, 2095–2104. [Google Scholar] [CrossRef]
- Krauss, G.J.; Solé, M.; Krauss, G.; Schlosser, D.; Wesenberg, D.; Bärlocher, F. Fungi in freshwaters: Ecology, physiology and biochemical potential. FEMS Microbiol. Rev. 2011, 35, 620–651. [Google Scholar] [CrossRef]
- Ferreira, V.; Castela, J.; Rosa, P.; Tonin, A.M.; Boyero, L.; Graça, M.A.S. Aquatic hyphomycetes, benthic macroinvertebrates and leaf litter decomposition in streams naturally differing in riparian vegetation. Aquat. Ecol. 2016, 50, 711–725. [Google Scholar] [CrossRef]
- Rozema, J.; Björn, L.O.; Bornman, J.F.; Gaberscik, A.; Häder, D.P.; Trost, T.; Germ, M.; Klisch, M.; Gröniger, A.; Sinha, R.P.; et al. The role of UV-B radiation in aquatic and terrestrial ecosystems-an experimental and functional analysis of the evolution of UV-absorbing compounds. J. Photochem. Photobiol. B 2002, 66, 2–12. [Google Scholar] [CrossRef]
- Manfrin, A.; Singer, G.; Larsen, S.; Manfrin, A.; Singer, G.; Larsen, S.; Weiß, N.; van Grunsven, R.H.A.; Weiß, N.S.; Wohlfahrt, S.; et al. Artificial light at night affects organism flux across ecosystem boundaries and drives community structure in the recipient ecosystem. Front. Environ. Sci. 2017, 5, 61. [Google Scholar] [CrossRef]
- Gessner, M.O. Proximate lignin and cellulose. In Methods to Study Litter Decomposition: A Practical Guide; Graça, M.A.S., Bärlocher, F., Gessner, M.O., Eds.; Springer: Berlin, Germany, 2005; pp. 115–120. [Google Scholar]
- Cochran, V.L.; Elliott, L.F.; Lewis, C.E. Soil microbial biomass and enzyme activity in subarctic agricultural and forest soils. Biol. Fertil. Soils 1989, 7, 283–288. [Google Scholar] [CrossRef]
- Hoostal, M.; Bidart-Bouzat, M.; Bouzat, J. Local adaptation of microbial communities to heavy metal stress in polluted sediments of Lake Erie. FEMS Microbiol. Ecol. 2008, 65, 156–168. [Google Scholar] [CrossRef]
- The Allison Lab. Enzyme Assays for Fresh Litter and Soil Adapted from Bob Sinsabaugh Lab. Available online: http://allison.bio.uci.edu/protocols/ (accessed on 25 January 2008).
- Rousk, J.; Bååth, E.; Brookes, P.C.; Lauber, C.L.; Lozupone, C.; Caporaso, J.G.; Knight, R.; Fierer, N. Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J. 2010, 4, 1340–1351. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Magoc, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
| Tr | pH | DO (mg·L−1) | NH4+ (ppm) | NTU | TDS (mg·L−1) | TSS (mg·L−1) | Cond. (μs·cm−1) |
|---|---|---|---|---|---|---|---|
| Origin | 6.77c | 5.32 | 41c | 434b | 24.400a | 2.033a | 0.036c |
| N | 7.34ab | 4.58 | 57b | 612a | 0.011b | 2.489a | 0.017c |
| A | 7.50a | 4.72 | 77a | 678a | 0.012b | 0.003c | 0.016c |
| N-Pb | 7.21b | 4.65 | 64b | 225b | 0.072c | 0.901b | 0.107b |
| A-Pb | 7.18b | 5.71 | 20c | 235b | 0.074c | 0.794b | 0.115a |
| Treatments | Carbon (mg·g−1) | Nitrogen (mg·g−1) | Phosphorus (mg·g−1) | Lignin (%) |
|---|---|---|---|---|
| N | 286.73b | 25.29 | 1.32 | 5.10a |
| A | 348.04ab | 26.48 | 1.28 | 2.96c |
| N-Pb | 419.64a | 27.84 | 1.38 | 6.76a |
| A-Pb | 333.61ab | 28.96 | 1.41 | 3.13b |
| Samples | Sequence | OTUs | Sobs | Shannon | Simpson | ACE | Chao1 |
|---|---|---|---|---|---|---|---|
| Origin | 58,462a | 351a | 274a | 3.08a | 0.15b | 293a | 293a |
| N | 49,342b | 319b | 219b | 1.98 b | 0.38a | 246b | 251b |
| A | 49,508b | 275c | 201b | 2.60a | 0.21b | 219b | 217b |
| N-Pb | 57,400a | 277c | 202b | 2.09b | 0.33a | 233b | 234b |
| A-Pb | 59,372a | 286c | 206b | 2.54a | 0.21b | 236b | 236b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pu, G.; Zeng, D.; Mo, L.; Liao, J.; Chen, X. Artificial Light at Night Alleviates the Negative Effect of Pb on Freshwater Ecosystems. Int. J. Mol. Sci. 2019, 20, 1343. https://doi.org/10.3390/ijms20061343
Pu G, Zeng D, Mo L, Liao J, Chen X. Artificial Light at Night Alleviates the Negative Effect of Pb on Freshwater Ecosystems. International Journal of Molecular Sciences. 2019; 20(6):1343. https://doi.org/10.3390/ijms20061343
Chicago/Turabian StylePu, Gaozhong, Danjuan Zeng, Ling Mo, Jianxiong Liao, and Xiaxia Chen. 2019. "Artificial Light at Night Alleviates the Negative Effect of Pb on Freshwater Ecosystems" International Journal of Molecular Sciences 20, no. 6: 1343. https://doi.org/10.3390/ijms20061343
APA StylePu, G., Zeng, D., Mo, L., Liao, J., & Chen, X. (2019). Artificial Light at Night Alleviates the Negative Effect of Pb on Freshwater Ecosystems. International Journal of Molecular Sciences, 20(6), 1343. https://doi.org/10.3390/ijms20061343




