Effects of Prolonged Dietary Curcumin Exposure on Skeletal Muscle Biochemical and Functional Responses of Aged Male Rats
Abstract
:1. Introduction
2. Results
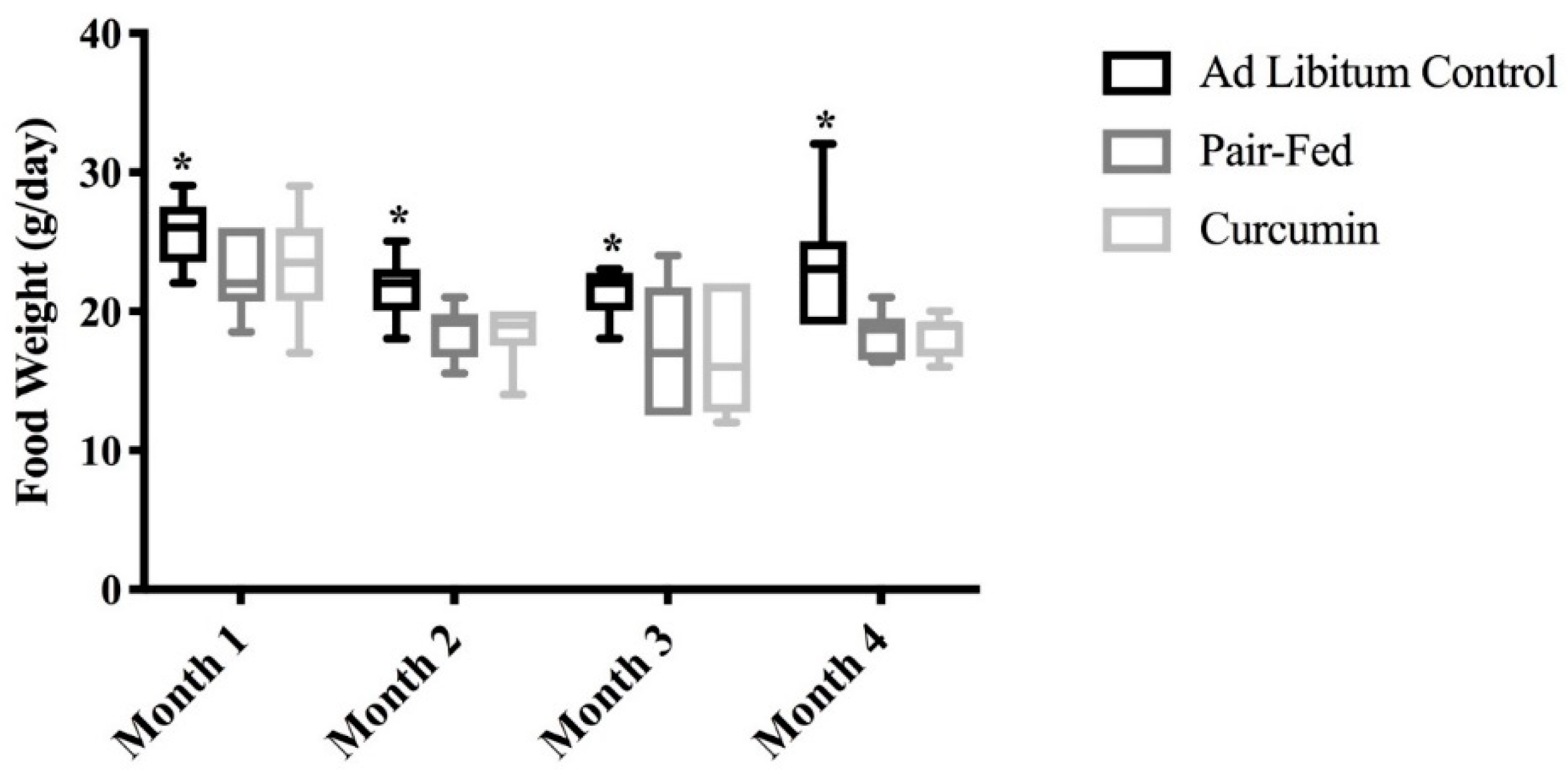
2.1. Food Intake, Body Mass and Muscle Mass
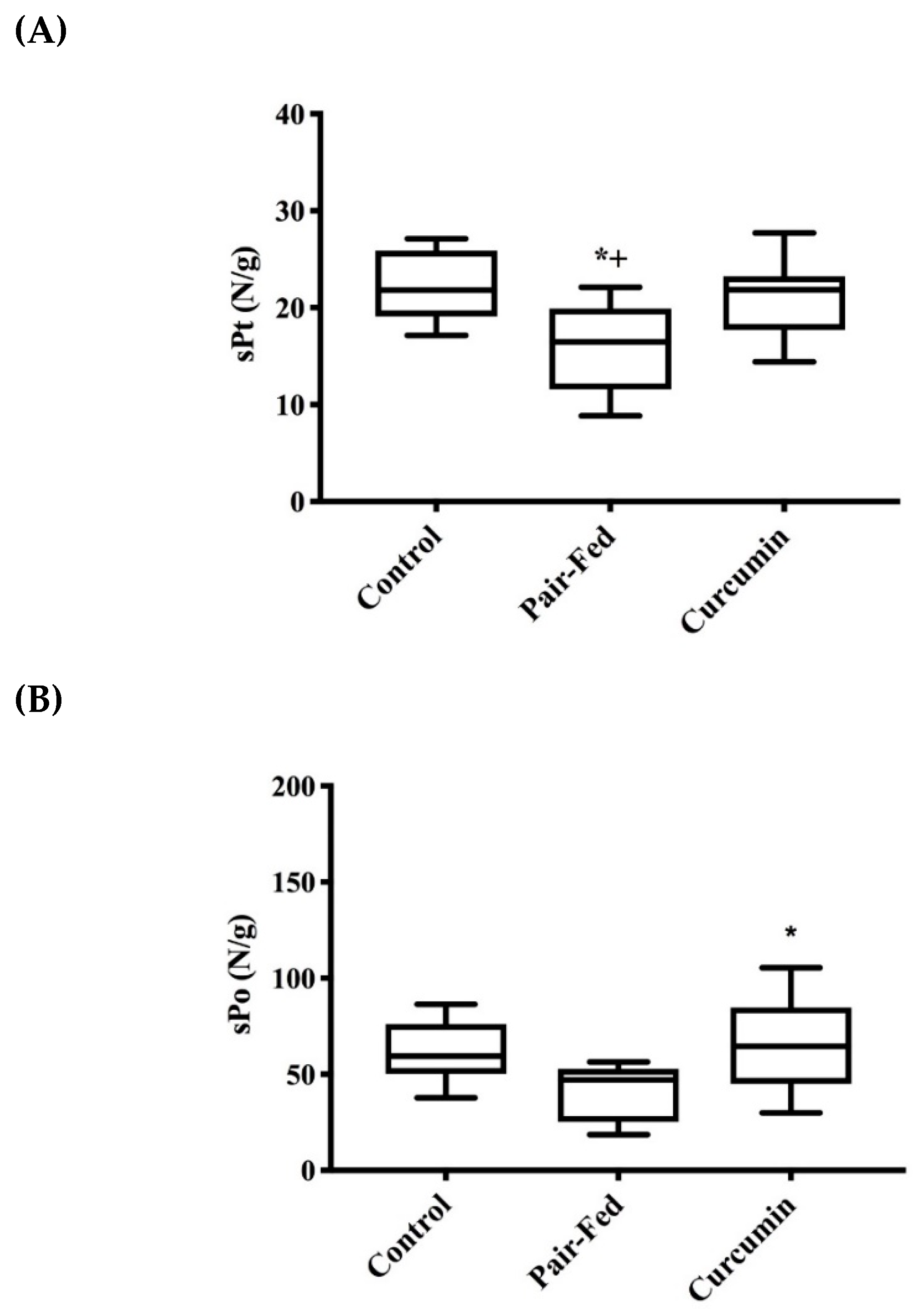
2.2. Muscle Contractile Function
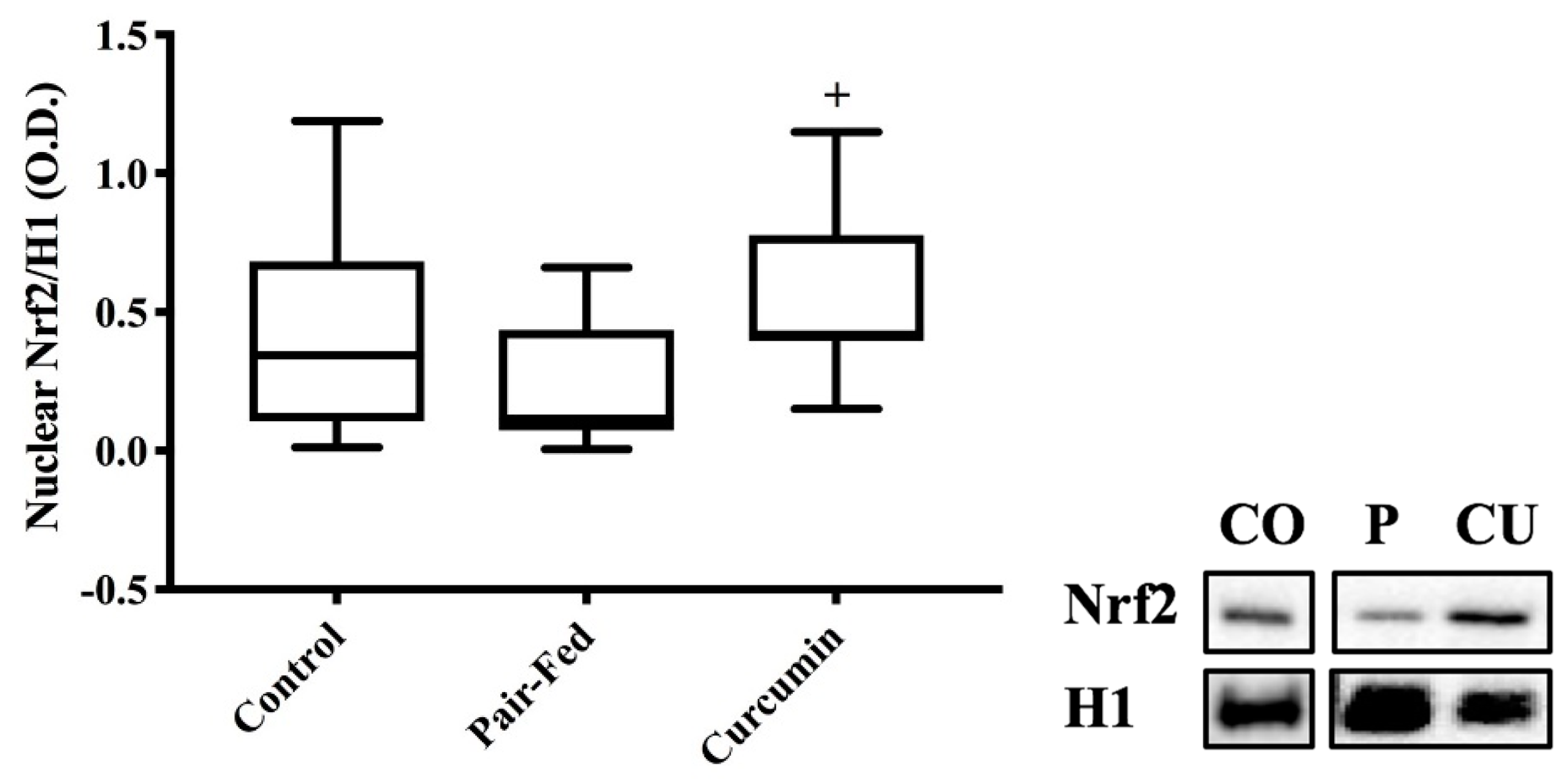
2.3. Skeletal Muscle Nuclear Nrf2 Expression & Antioxidant Measures
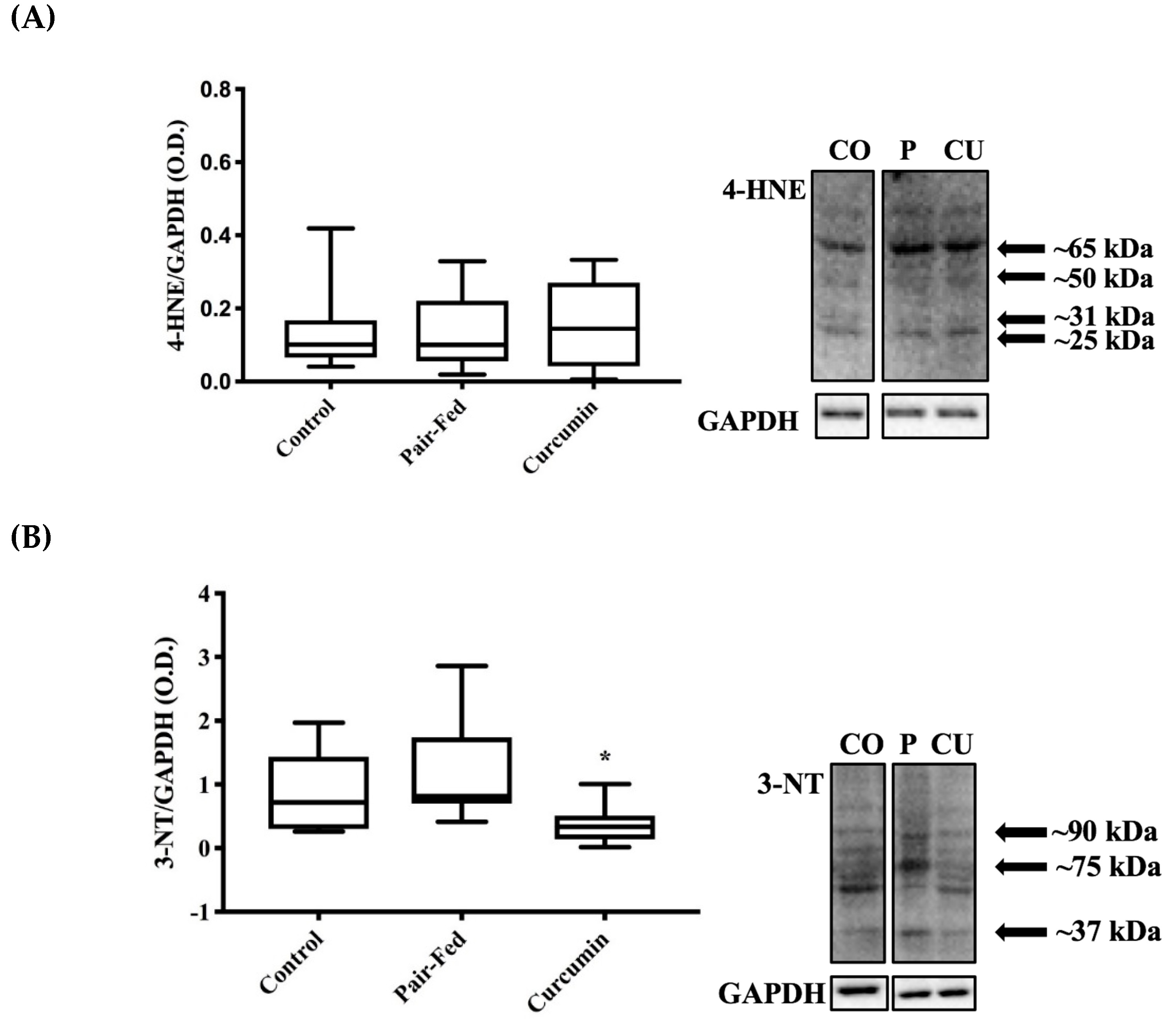
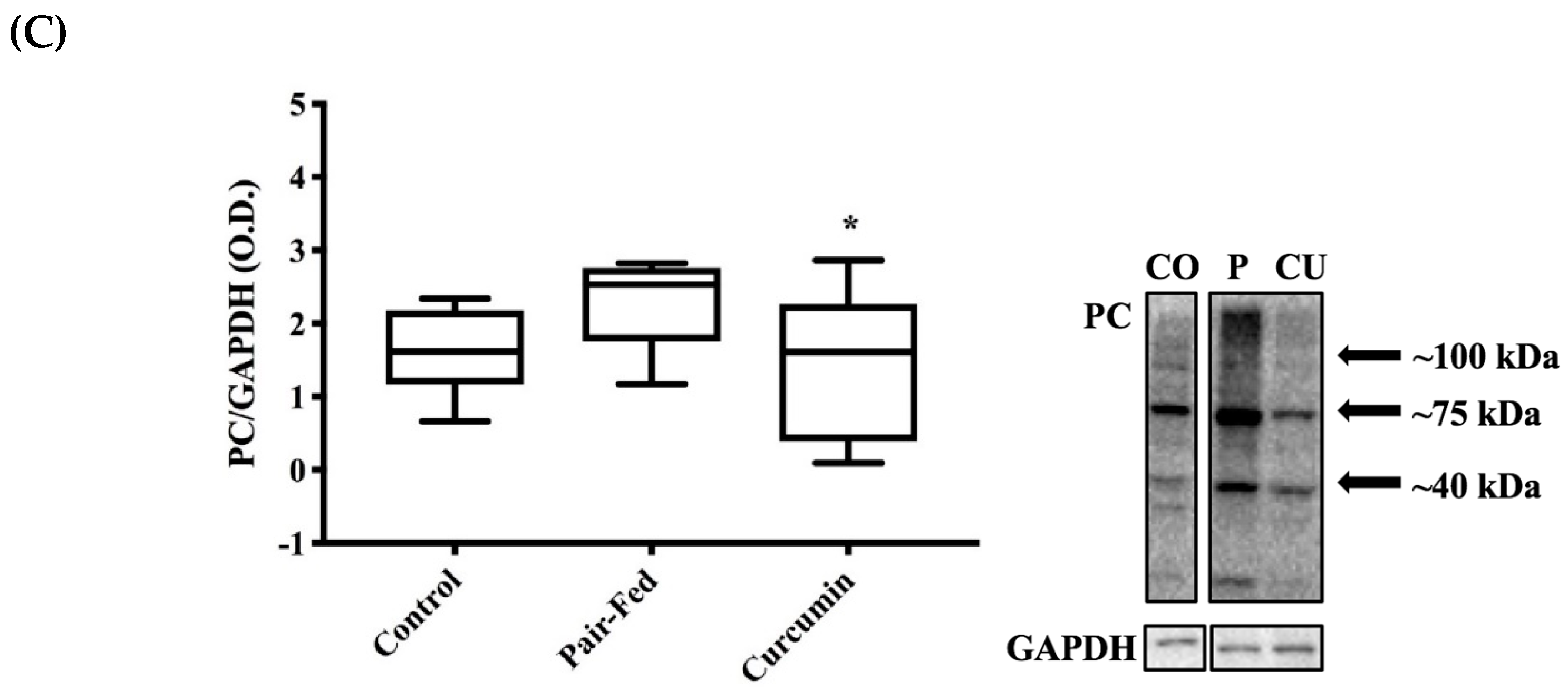
2.4. Skeletal Muscle Oxidative Stress
2.5. Curcumin Administration via Osmotic Pumps: Complementary Study
3. Discussion
3.1. Rats Consuming Curcumin Showed Reduced Food Intake
3.2. Exposure to Dietary Curcumin Altered Muscle Mass, but not Body Mass
3.3. Curcumin Exposure Effects on Nuclear Nrf2 Protein Expression and Antioxidant Responses
3.4. Curcumin Exposure and Muscle Oxidative Damage
3.5. Curcumin Administration to Aged Rats via Osmotic Pump Results in Greater Skeletal Muscle Mass
3.6. Limitations
4. Materials and Methods
4.1. Ethical Approval
4.2. Animals
4.3. Experimental Design
4.4. Plantaris Muscle Contractility
4.5. Tissue Collection
4.6. Western Blot
4.7. Protein Carbonyls (PC)
4.8. Nuclear Protein Fractions for Nrf2
4.9. Muscle Antioxidant Capacity
4.10. Catalase Activity
4.11. Manganese Superoxide Dismutase (SOD) Activity
4.12. Complimentary Study: Administration of Curcumin via Subcutaneous Osmotic Pumps
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Von Haehling, S.; Morley, J.E.; Anker, S.D. An overview of sarcopenia: Facts and numbers on prevalence and clinical impact. J. Caxhexia Sarcopenia Muscle 2010, 1, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Bowen, T.S.; Schuler, G.; Adams, V. Skeletal muscle wasting in cachexia and sarcopenia: Molecular pathophysiology and impact of exercise training. J. Caxhexia Sarcopenia Muscle 2015, 6, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Moreland, J.D.; Richardson, J.A.; Goldsmith, C.H.; Clase, C.M. Muscle weakness and falls in older adults: A systematic review and meta-analysis. J. Am. Geriatr. Soc. 2004, 52, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.P.; Duthie, E.H.; Gambert, S.R.; Sepic, S.B.; Mollinger, L.A. AGE-RELATED DIFFERENCES IN KNEE MUSCLE STRENGTH IN NORMAL WOMEN. J. Gerontol. 1985, 40, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.P.; Gardner, G.M.; Mollinger, L.A.; Sepic, S.B. STRENGTH OF ISOMETRIC AND ISOKINETIC CONTRACTIONS—KNEE MUSCLES OF MEN AGED 20 TO 86. Phys. Ther. 1980, 60, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.; Visser, M.; DeMeersman, R.E.; Sepulveda, D.; Baumgartner, R.N.; Pierson, R.N.; Harris, T.; Heymsfield, S.B. Appendicular skeletal muscle mass: Effects of age, gender, and ethnicity. J. Appl. Physiol. 1997, 83, 229–239. [Google Scholar] [CrossRef]
- Kregel, K.C.; Zhang, H.J. An integrated view of oxidative stress in aging: Basic mechanisms, functional effects, and pathological considerations. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2007, 292, R18–R36. [Google Scholar] [CrossRef]
- Fano, G.; Mecocci, P.; Vecchiet, J.; Belia, S.; Fulle, S.; Polidori, M.C.; Felzani, G.; Senin, U.; Vecchiet, L.; Beal, M.F. Age and sex influence on oxidative damage and functional status in human skeletal muscle. J. Muscle Res. Cell Motil. 2001, 22, 345–351. [Google Scholar] [CrossRef]
- Leeuwenburgh, C.; Fiebig, R.; Chandwaney, R.; Ji, L.L. Aging and exercise training in skeletal-muscle—responses of glutathione and antioxidant enzyme-systems. Am. J. Physiol. 1994, 267, R439–R445. [Google Scholar] [CrossRef]
- Luhtala, T.A.; Roecker, E.B.; Pugh, T.; Feuers, R.J.; Weindruch, R. Dietary restriction attenuates age-related increases in rat skeletal-muscle antioxidant enzyme-activities. J. Gerontol. 1994, 49, B231–B238. [Google Scholar] [CrossRef]
- Vertechy, M.; Cooper, M.B.; Ghirardi, O.; Ramacci, M.T. Antioxidant enzyme-activities in heart and skeletal-muscle of rats of different ages. Exp. Gerontol. 1989, 24, 211–218. [Google Scholar] [CrossRef]
- Gianni, P.; Kaczor, J.J.; Douglas, M.J.; Stuart, P.M.; Tarnopolsky, M.A. Oxidative stress and the mitochondrial theory of aging in human skeletal muscle. Exp. Gerontol. 2004, 39, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.L.; Dillon, D.; Wu, E. Alteration of antioxidant enzymes with aging in rat skeletal-muscle and liver. Am. J. Physiol. 1990, 258, R918–R923. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K.N.; Wason, E.; Edrey, Y.H.; Kristan, D.M.; Nevo, E.; Buffenstein, R. Regulation of Nrf2 signaling and longevity in naturally long-lived rodents. Proc. Natl. Acad. Sci. USA 2015, 112, 3722–3727. [Google Scholar] [CrossRef] [PubMed]
- Itoh, K.; Wakabayashi, N.; Katoh, Y.; Ishii, T.; Igarashi, K.; Engel, J.D.; Yamamoto, M. Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev. 1999, 13, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Muthusamy, V.R.; Kannan, S.; Sadhaasivam, K.; Gounder, S.S.; Davidson, C.J.; Boeheme, C.; Hoidal, J.R.; Wang, L.; Rajasekaran, N.S. Acute exercise stress activates Nrf2/ARE signaling and promotes antioxidant mechanisms in the myocardium. Free Radic. Biol. Med. 2012, 52, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Kensler, T.W.; Wakabayash, N.; Biswal, S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Safdar, A.; DeBeer, J.; Tarnopolsky, M.A. Dysfunctional Nrf2-Keap1 redox signaling in skeletal muscle of the sedentary old. Free Radic. Biol. Med. 2010, 49, 1487–1493. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.H.; Shenvi, S.V.; Dixon, B.M.; Liu, H.L.; Jaiswal, A.K.; Liu, R.M.; Hagen, T.M. Decline in transcriptional activity of Nrf2 causes age-related loss of glutathione synthesis, which is reversible with lipoic acid. Proc. Natl. Acad. Sci. USA 2004, 101, 3381–3386. [Google Scholar] [CrossRef]
- Alamdari, N.; O’Neal, P.; Hasselgren, P.O. Curcumin and muscle wasting-A new role for an old drug? Nutrition 2009, 25, 125–129. [Google Scholar] [CrossRef]
- Anto, R.J.; Kuttan, G.; Babu, K.V.D.; Rajasekharan, K.N.; Kuttan, R. Anti-tumour and free radical scavenging activity of synthetic curcuminoids. Int. J. Pharm. 1996, 131, 1–7. [Google Scholar] [CrossRef]
- Jeong, S.O.; Oh, G.S.; Ha, H.Y.; Koo, B.S.; Kim, H.S.; Kim, Y.C.; Kim, E.C.; Lee, K.M.; Chung, H.T.; Pae, H.O. Dimethoxycurcumin, a synthetic curcumin analogue, induces heme oxygenase-1 expression through Nrf2 activation in RAW264.7 macrophages. J. Clin. Biochem. Nutr. 2009, 44, 79–84. [Google Scholar] [CrossRef]
- Shen, G.X.; Xu, C.J.; Hu, R.; Jain, M.R.; Gopalkrishnan, A.; Nair, S.; Huang, M.T.; Chan, J.Y.; Kong, A.N.T. Modulation of nuclear factor E2-related factor 2-mediated gene expression in mice liver and small intestine by cancer chemopreventive agent curcumin. Mol. Cancer Ther. 2006, 5, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Sahin, K.; Pala, R.; Tuzcu, M.; Ozdemir, O.; Orhan, C.; Sahin, N.; Juturu, V. Curcumin prevents muscle damage by regulating NF-kappa B and Nrf2 pathways and improves performance: An in vivo model. J. Inflamm. Res. 2016, 9, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Ono, T.; Takada, S.; Kinugawa, S.; Tsutsui, H. Curcumin ameliorates skeletal muscle atrophy in type 1 diabetic mice by inhibiting protein ubiquitination. Exp. Physiol. 2015, 100, 1052–1063. [Google Scholar] [CrossRef] [PubMed]
- Poylin, V.; Fareed, M.U.; O’Neal, P.; Alamdari, N.; Reilly, N.; Menconi, M.; Hasselgren, P.O. The NF-kappa B inhibitor curcumin blocks sepsis-induced muscle proteolysis. Mediat. Inflamm. 2008, 2008. [Google Scholar] [CrossRef] [PubMed]
- Vazeille, E.; Slimani, L.; Claustre, A.; Magne, H.; Labas, R.; Bechet, D.; Taillandier, D.; Dardevet, D.; Astruc, T.; Attaix, D.; et al. Curcumin treatment prevents increased proteasome and apoptosome activities in rat skeletal muscle during reloading and improves subsequent recovery. J. Nutr. Biochem. 2012, 23, 245–251. [Google Scholar] [CrossRef]
- Franceschi, F.; Feregalli, B.; Togni, S.; Cornelli, U.; Giacomelli, L.; Eggenhoffner, R.; Belcaro, G. A novel phospholipid delivery system of curcumin (Meriva (R)) preserves muscular mass in healthy aging subjects. Eur. Rev. Med Pharmacol. Sci. 2016, 20, 762–766. [Google Scholar]
- He, J.; Xie, H.M.; Wu, S. Dietary supplementation of curcumin alleviates NF-kappa B-dependent skeletal muscle wasting in rat. Endocr. Metab. Immune Disord. Drug Targets 2016, 16, 140–147. [Google Scholar] [CrossRef]
- Kawamori, T.; Lubet, R.; Steele, V.E.; Kelloff, G.J.; Kaskey, R.B.; Rao, C.V.; Reddy, B.S. Chemopreventive effect of curcumin, a naturally occurring anti-inflammatory agent, during the promotion/progression stages of colon cancer. Cancer Res. 1999, 59, 597–601. [Google Scholar]
- Kempaiah, R.K.; Srinivasan, K. Influence of dietary curcumin, capsaicin and garlic on the antioxidant status of red blood cells and the liver in high-fat-fed rats. Ann. Nutr. Metab. 2004, 48, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Manjunatha, H.; Srinivasan, K. Hypolipidemic and antioxidant effects of curcumin and capsaicin in high-fat-fed rats. Can. J. Physiol. Pharmacol. 2007, 85, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Asai, A.; Miyazawa, T. Dietary curcuminoids prevent high-fat diet-induced lipid accumulation in rat liver and epididymal adipose tissue. J. Nutr. 2001, 131, 2932–2935. [Google Scholar] [CrossRef] [PubMed]
- Chin, D.; Huebbe, P.; Frank, J.; Rimbach, G.; Pallauf, K. Curcumin may impair iron status when fed to mice for six months. Redox Biol. 2014, 2, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Aggarwal, B.B. “Spicing up” of the immune system by curcumin. J. Clin. Immunol. 2007, 27, 19–35. [Google Scholar] [CrossRef] [PubMed]
- Kamal-Eldin, A.; Frank, J.; Razdan, A.; Tengblad, S.; Basu, S.; Vessby, B. Effects of dietary phenolic compounds on tocopherol, cholesterol, and fatty acids in rats. Lipids 2000, 35, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Turturro, A.; Witt, W.W.; Lewis, S.; Hass, B.S.; Lipman, R.D.; Hart, R.W. Growth curves and survival characteristics of the animals used in the biomarkers of aging program. J. Gerontol. Ser. A-Biol. Sci. Med Sci. 1999, 54, B492–B501. [Google Scholar] [CrossRef]
- Asai, A.; Miyazawa, T. Occurrence of orally administered curcuminoid as glucuronide and glucuronide/sulfate conjugates in rat plasma. Life Sci. 2000, 67, 2785–2793. [Google Scholar] [CrossRef]
- Oner-Iyidogan, Y.; Tanrikulu-Kucuk, S.; Seyithanoglu, M.; Kocak, H.; Dogru-Abbasoglu, S.; Aydin, A.F.; Beyhan-Ozdas, S.; Yapislar, H.; Kocak-Toker, N. Effect of curcumin on hepatic heme oxygenase 1 expression in high fat diet fed rats: Is there a triangular relationship? Can. J. Physiol. Pharmacol. 2014, 92, 805–812. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, L.Y.; Shen, Y.; Tan, T.; Xie, N.Z.; Luo, M.; Li, Z.H.; Xie, X.Y. Curcumin ameliorates ischemia-induced limb injury through immunomodulation. Med Sci. Monit. 2016, 22, 2035–2042. [Google Scholar] [CrossRef]
- Villareal, D.T.; Fontana, L.; Weiss, E.P.; Racette, S.B.; Steger-May, K.; Schechtman, K.B.; Klein, S.; Holloszy, J.O. Bone mineral density response to caloric restriction-induced weight loss or exercise-induced weight loss—A randomized controlled trial. Arch. Intern. Med. 2006, 166, 2502–2510. [Google Scholar] [CrossRef] [PubMed]
- Stein, T.P.; Rumpler, W.V.; Leskiw, M.J.; Staples, R.; Bodwell, C.E. Effect of reduced dietary-intake on energy-expenditure, protein-turnover, and glucose cycling in man. Metab.-Clin. Exp. 1991, 40, 478–483. [Google Scholar] [CrossRef]
- Weinheimer, E.; Sands, L.P.; Campbell, W.W. A systematic review of the separate and combined effects of energy restriction and exercise on fat-free mass in middle-aged and older adults: Implications for sarcopenic obesity. Faseb J. 2010, 24, 1. [Google Scholar] [CrossRef] [PubMed]
- Duclos, M.; Gatti, C.; Bessiere, B.; Mormede, P. Tonic and phasic effects of corticosterone on food restriction-induced hyperactivity in rats. Psychoneuroendocrinology 2009, 34, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Morse, A.D.; Russell, J.C.; Hunt, T.W.M.; Wood, G.O.; Epling, W.F.; Pierce, W.D. Diurnal variation of intensive running in food deprived rats. Can. J. Physiol. Pharmacol. 1995, 73, 1519–1523. [Google Scholar] [CrossRef] [PubMed]
- Done, A.J.; Gage, M.J.; Nieto, N.C.; Traustadottir, T. Exercise-induced Nrf2-signaling is impaired in aging. Free Radic. Biol. Med. 2016, 96, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.J.; Gounder, S.S.; Kannan, S.; Goutam, K.; Muthusamy, V.R.; Firpo, M.A.; Symons, J.D.; Paine, R.; Hoidal, J.R.; Rajasekaran, N.S. Disruption of Nrf2/ARE signaling impairs antioxidant mechanisms and promotes cell degradation pathways in aged skeletal muscle. Biochim. Et Biophys. Acta-Mol. Basis Dis. 2012, 1822, 1038–1050. [Google Scholar] [CrossRef]
- Baldelli, S.; Aquilano, K.; Ciriolo, M.R. Punctum on two different transcription factors regulated by PGC-1 alpha: Nuclear factor erythroid-derived 2-like 2 and nuclear respiratory factor 2. Biochim. Et Biophys. Acta-Gen. Subj. 2013, 1830, 4137–4146. [Google Scholar] [CrossRef]
- He, H.-J.; Wang, G.-Y.; Gao, Y.; Ling, W.-H.; Yu, Z.-W.; Jin, T.-R. Curcumin attenuates Nrf2 signaling defect, oxidative stress in muscle and glucose intolerance in high fat diet-fed mice. World J. Diabetes 2012, 3, 94–104. [Google Scholar] [CrossRef]
- Cui, Q.L.; Li, X.; Zhu, H.C. Curcumin ameliorates dopaminergic neuronal oxidative damage via activation of the Akt/Nrf2 pathway. Mol. Med. Rep. 2016, 13, 1381–1388. [Google Scholar] [CrossRef]
- Gao, S.; Duan, X.X.; Wang, X.; Dong, D.D.; Liu, D.; Li, X.; Sun, G.F.; Li, B. Curcumin attenuates arsenic-induced hepatic injuries and oxidative stress in experimental mice through activation of Nrf2 pathway, promotion of arsenic methylation and urinary excretion. Food Chem. Toxicol. 2013, 59, 739–747. [Google Scholar] [CrossRef]
- Farombi, E.O.; Shrotriya, S.; Na, H.K.; Kim, S.H.; Surh, Y.J. Curcumin attenuates dimethylnitrosamine-induced liver injury in rats through Nrf2-mediated induction of heme oxygenase-1. Food Chem. Toxicol. 2008, 46, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Nakhostin-Roohi, B.; Moradlou, A.N.; Hamidabad, S.M.; Ghanivand, B. The effect of curcumin supplementation on selected markers of delayed onset muscle soreness (DOMS). Ann. Appl. Sport Sci. 2016, 4, 25–31. [Google Scholar] [CrossRef]
- Takahashi, M.; Suzuki, K.; Kim, H.K.; Otsuka, Y.; Imaizumi, A.; Miyashita, M.; Sakamoto, S. Effects of curcumin supplementation on exercise-induced oxidative stress in humans. Int. J. Sports Med. 2014, 35, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Libera, L.; Ravara, B.; Gobbo, V.; Tarricone, E.; Vitadello, M.; Biolo, G.; Vescovo, G.; Gorza, L. A transient antioxidant stress response accompanies the onset of disuse atrophy in human skeletal muscle. J. Appl. Physiol. 2009, 107, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.C.; Li, S.J.; Yang, C.L.; Xue, R.L.; Xi, Y.Y.; Wang, L.; Zhao, Q.L.; Li, D.J. Sulforaphane attenuates muscle inflammation in dystrophin-deficient mdx mice via NF-E2-related factor 2 (Nrf2)-mediated inhibition of NF-kappa B signaling pathway. J. Biol. Chem. 2015, 290, 17784–17795. [Google Scholar] [CrossRef]
- Buford, T.W.; Cooke, M.B.; Manini, T.M.; Leeuwenburgh, C.; Willoughby, D.S. Effects of Age and Sedentary Lifestyle on Skeletal Muscle NF-kappa B Signaling in Men. J. Gerontol. Ser. A-Biol. Sci. Med Sci. 2010, 65, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Cuthbertson, D.; Smith, K.; Babraj, J.; Leese, G.; Waddell, T.; Atherton, P.; Wackerhage, H.; Taylor, P.M.; Rennie, M.J. Anabolic signaling deficits underlie amino acid resistance of wasting, aging muscle. Faseb J. 2004, 18, 422–424. [Google Scholar] [CrossRef]
- Zhang, N.; Valentine, J.M.; Zhou, Y.; Li, M.E.; Zhang, Y.; Bhattacharya, A.; Walsh, M.E.; Fischer, K.E.; Austad, S.N.; Osmulski, P.; et al. Sustained NF kappa B inhibition improves insulin sensitivity but is detrimental to muscle health. Aging Cell 2017, 16, 847–858. [Google Scholar] [CrossRef]
- Guttridge, D.C.; Mayo, M.W.; Madrid, L.V.; Wang, C.Y.; Baldwin, A.S. NF-kappa B-induced loss of MyoD messenger RNA: Possible role in muscle decay and cachexia. Science 2000, 289, 2363–2366. [Google Scholar] [CrossRef]
- Vitadello, M.; Germinario, E.; Ravara, B.; Dalla Libera, L.; Danieli-Betto, D.; Gorza, L. Curcumin counteracts loss of force and atrophy of hindlimb unloaded rat soleus by hampering neuronal nitric oxide synthase untethering from sarcolemma. J. Physiol. Lond. 2014, 592, 2637–2652. [Google Scholar] [CrossRef] [PubMed]
- Nageshwar, M.; Sudhakar, K.; Reddy, N.C.C.; Reddy, K.P. Neuroprotective effects of curcumin on sodium fluoride induced behavioural and enzymatic changes in brain and muscles of rat. J. Environ. Biol. 2017, 38, 675–681. [Google Scholar] [CrossRef]
- Smith, M.A.; Reid, M.B. Redox modulation of contractile function in respiratory and limb skeletal muscle. Respir. Physiol. Neurobiol. 2006, 151, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Harman, D. FREE-RADICAL THEORY OF AGING. Mutat. Res. 1992, 275, 257–266. [Google Scholar] [CrossRef]
- Dairam, A.; Fogel, R.; Daya, S.; Limson, J.L. Antioxidant and iron-binding properties of curcumin, capsaicin, and S-Allylcysteine reduce oxidative stress in rat brain homogenate. J. Agric. Food Chem. 2008, 56, 3350–3356. [Google Scholar] [CrossRef] [PubMed]
- Kunchandy, E.; Rao, M.N.A. Oxygen radical scavenging activity of curcumin. Int. J. Pharm. 1990, 58, 237–240. [Google Scholar] [CrossRef]
- Kowluru, R.A.; Kanwar, M. Effects of curcumin on retinal oxidative stress and inflammation in diabetes. Nutr. Metab. 2007, 4. [Google Scholar] [CrossRef]
- Strasser, E.M.; Wessner, B.; Manhart, N.; Roth, E. The relationship between the anti-inflammatory effects of curcumin and cellular glutathione content in myelomonocytic cells. Biochem. Pharmacol. 2005, 70, 552–559. [Google Scholar] [CrossRef]
- Andreatta, R.D.; Stemple, J.C.; Seward, T.S.; McMullen, C.A. Subcutaneous neurotrophin 4 infusion using osmotic pumps or direct muscular injection enhances aging rat laryngeal muscles. JoVE J. Vis. Exp. 2017. [Google Scholar] [CrossRef]
- Chang, C.Z.; Wu, S.C.; Lin, C.L.; Kwan, A.L. Curcumin, encapsulated in nano-sized PLGA, down-regulates nuclear factor kappa B (p65) and subarachnoid hemorrhage induced early brain injury in a rat model. Brain Res. 2015, 1608, 215–224. [Google Scholar] [CrossRef]
- Moreau, M.L.; Sauvant, J.; Moos, F.; Palin, K. Brain insulin growth factor-I induces diuresis increase through the inhibition of arginin-vasopressin release in aged rats. Neurobiol. Aging 2010, 31, 532–536. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.M.; Cui, X.X.; Wu, R.Q.; Dong, W.F.; Zhou, M.; Hu, M.W.; Simms, H.H.; Wang, P. The anti-inflammatory effect of curcumin in an experimental model of sepsis is mediated by up-regulation of peroxisome proliferator-activated receptor-gamma. Crit. Care Med. 2006, 34, 1874–1882. [Google Scholar] [CrossRef] [PubMed]
- Rao, C.V.; Rivenson, A.; Simi, B.; Reddy, B.S. Chemoprevention of colon carcinogenesis by dietary curcumin, a naturally-occurring plant phenolic compound. Cancer Res. 1995, 55, 259–266. [Google Scholar] [PubMed]
- Mitchell, W.K.; Williams, J.; Atherton, P.; Larvin, M.; Lund, J.; Narici, M. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Front. Physiol. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Research, I.F.L.A. Guide for the Care and Use of Laboratory Animals, 8th ed.; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Patel, V.B.; Spencer, C.H.; Young, T.A.; Lively, M.O.; Cunningham, C.C. Effects of 4-hydroxynonenal on mitochondrial 3-hydroxy-3-methylglutaryl (HMG-CoA) synthase. Free Radic. Biol. Med. 2007, 43, 1499–1507. [Google Scholar] [CrossRef]
- Penkowa, M.; Carrasco, J.; Giralt, M.; Molinero, A.; Hernandez, J.; Campbell, I.L.; Hidalgo, J. Altered central nervous system cytokine-growth factor expression profiles and angiogenesis in metallothionein-I plus II deficient mice. J. Cereb. Blood Flow Metab. 2000, 20, 1174–1189. [Google Scholar] [CrossRef] [PubMed]
- Penkowa, M.; Giralt, M.; Lago, N.; Camats, J.; Caffasco, J.; Hernandez, J.; Molinero, A.; Campbell, I.L.; Hidalgo, J. Astrocyte-targeted expression of IL-6 protects the CNS against a focal brain injury. Exp. Neurol. 2003, 181, 130–148. [Google Scholar] [CrossRef]
- Muscoli, C.; Lauro, F.; Dagostino, C.; Ilari, S.; Giancotti, L.A.; Gliozzi, M.; Costa, N.; Carresi, C.; Musolino, V.; Casale, F.; et al. Olea europea derived phenolic products attenuate antinociceptive morphine tolerance: An innovative strategic approach to treat cancer pain. J. Biol. Regul. Homeost. Agents 2014, 28, 105–116. [Google Scholar]
- Wang, S.S.; Yang, S.C.; Vlantis, A.C.; Liu, S.Y.W.; Ng, E.K.W.; Chan, A.B.W.; Wu, J.; Du, J.; Wei, W.; Liu, X.L.; et al. Expression of Antioxidant Molecules and Heat Shock Protein 27 in Thyroid Tumors. J. Cell. Biochem. 2016, 117, 2473–2481. [Google Scholar] [CrossRef]
- Hyatt, H.W.; Smuder, A.J.; Sollanek, K.J.; Morton, A.B.; Roberts, M.D.; Kavazis, A.N. Comparative changes in antioxidant enzymes and oxidative stress in cardiac, fast twitch and slow twitch skeletal muscles following endurance exercise training. Int. J. Physiol. Pathophysiol. Pharmacol. 2016, 8, 160–168. [Google Scholar]
- Yang, C.C.; Hsu, S.P.; Chen, K.H.; Chien, C.T. Effect of Adenoviral Catalase Gene Transfer on Renal Ischemia/Reperfusion Injury in Rats. Chin. J. Physiol. 2015, 58, 420–430. [Google Scholar] [CrossRef]
- Jabs, A.; Oelze, M.; Mikhed, Y.; Stamm, P.; Kroeller-Schoen, S.; Welschof, P.; Jansen, T.; Hausding, M.; Kopp, M.; Steven, S.; et al. Effect of soluble guanylyl cyclase activator and stimulator therapy on nitroglycerin-induced nitrate tolerance in rats. Vasc. Pharmacol. 2015, 71, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Shinjo, T.; Tanaka, T.; Okuda, H.; Kawaguchi, A.T.; Oh-hashi, K.; Terada, Y.; Isonishi, A.; Morita-Takemura, S.; Tatsumi, K.; Kawaguchi, M.; et al. Propofol induces nuclear localization of Nrf2 under conditions of oxidative stress in cardiac H9c2 cells. PLoS ONE 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Lappalainen, Z.; Lappalainen, J.; Laaksonen, D.E.; Oksala, N.K.J.; Khanna, S.; Sen, C.K.; Atalay, M. Acute Exercise and Thioredoxin-1 in Rat Brain, and Alpha-Lipoic Acid and Thioredoxin-Interacting Protein Response, in Diabetes. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Zhong, L.; Lonn, M.E.; Burk, R.F.; Hill, K.E.; Holmgren, A. Penultimate selenocysteine residue replaced by cysteine in thioredoxin reductase from selenium-deficient rat liver. Faseb J. 2009, 23, 2394–2402. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Cha-Molstad, H.; Szabo, A.; Shalev, A. Diabetes induces and calcium channel blockers prevent cardiac expression of proapoptotic thioredoxin-interacting protein. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E1133–E1139. [Google Scholar] [CrossRef] [PubMed]
- Tyburski, A.L.; Cheng, L.; Assari, S.; Darvish, K.; Elliott, M.B. Frequent mild head injury promotes trigeminal sensitivity concomitant with microglial proliferation, astrocytosis, and increased neuropeptide levels in the trigeminal pain system. J. Headache Pain 2017, 18, 16. [Google Scholar] [CrossRef]
- Wu, Y.; Ma, S.; Xia, Y.; Lu, Y.; Xiao, S.; Cao, Y.; Zhuang, S.; Tan, X.; Fu, Q.; Xie, L.; et al. Loss of GCN5 leads to increased neuronal apoptosis by upregulating E2F1-and Egr-1-dependent BH3-only protein Bim. Cell Death Dis. 2017, 8, e2570. [Google Scholar] [CrossRef]
- Aslan, A.; Gok, O.; Erman, O.; Kuloglu, T. Ellagic acid impedes carbontetrachloride-induced liver damage in rats through suppression of NF-kB, Bcl-2 and regulating Nrf-2 and caspase pathway. Biomed. Pharmacother. 2018, 105, 662–669. [Google Scholar] [CrossRef]
- Huang, W.-Y.; Lin, S.; Chen, H.-Y.; Chen, Y.-P.; Chen, T.-Y.; Hsu, K.-S.; Wu, H.-M. NADPH oxidases as potential pharmacological targets against increased seizure susceptibility after systemic inflammation. J. Neuroinflamm. 2018, 15, 140. [Google Scholar] [CrossRef]
- Wang, P.; Powell, S.R. Decreased sensitivity associated with an altered formulation of a commercially available kit for detection of protein carbonyls. Free Radic. Biol. Med. 2010, 49, 119–121. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Rong, S.; Feng, Y.; Zhao, L.; Hong, J.; Wang, R.; Yuan, W. Simvastatin attenuates renal ischemia/reperfusion injury from oxidative stress via targeting Nrf2/HO-1 pathway. Exp. Ther. Med. 2017, 14, 4460–4466. [Google Scholar] [CrossRef]
- Zhao, L.; Liu, Z.B.; Jia, H.Q.; Feng, Z.H.; Liu, J.K.; Li, X.S. Lipoamide Acts as an Indirect Antioxidant by Simultaneously Stimulating Mitochondrial Biogenesis and Phase II Antioxidant Enzyme Systems in ARPE-19 Cells. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Bi, Z.; Yang, T.; Wang, W.; Li, Z.; Huang, W.; Wang, L.; Zhang, S.; Zhou, Y.; Fan, N.; et al. Regulation of PKM2 and Nrf2-ARE Pathway during Benzoquinone Induced Oxidative Stress in Yolk Sac Hematopoietic Stem Cells. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Pekovic-Vaughan, V.; Gibbs, J.; Yoshitane, H.; Yang, N.; Pathiranage, D.; Guo, B.; Sagami, A.; Taguchi, K.; Bechtold, D.; Loudon, A.; et al. The circadian clock regulates rhythmic activation of the NRF2/glutathione-mediated antioxidant defense pathway to modulate pulmonary fibrosis. Genes Dev. 2014, 28, 548–560. [Google Scholar] [CrossRef]
- Aminzadeh, M.A.; Reisman, S.A.; Vaziri, N.D.; Shelkovnikov, S.; Farzaneh, S.H.; Khazaeli, M.; Meyer, C.J. The synthetic triterpenoid RTA dh404 (CDDO-dhTFEA) restores endothelial function impaired by reduced Nrf2 activity in chronic kidney disease. Redox Biol. 2013, 1, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; Lei, L.; Zhang, Z.; Cheng, Y. Neuroprotective effects of lycopene pretreatment on transient global cerebral ischemia-reperfusion in rats: The role of the Nrf2/HO-1 signaling pathway. Mol. Med. Rep. 2016, 13, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Aebi, H. Catalase invitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar]
| Body Mass (g) | Plantaris Muscle Mass (g) | |
|---|---|---|
| CON | 506.4 (51.1) | 0.285 (0.056) |
| PAIR | 503.7 (73.5) | 0.198 (0.046) * |
| CUR | 504.1 (95.6) | 0.326 (0.060) |
| CON | PAIR | CUR | p-Value | |
|---|---|---|---|---|
| Protein Expression | ||||
| Catalase | 1.08 (0.83) | 0.83 (0.53) | 1.03 (0.35) | 0.44 |
| MnSOD | 2.13 (1.21) | 1.96 (0.93) | 2.64 (1.52) | 0.23 |
| HO-1 | 2.38 (1.84) | 3.42 (1.33) | 3.28 (1.42) | 0.17 |
| TRX/TxNip | 0.52 (0.48) | 0.67 (0.60) | 1.11 (0.84) | 0.34 |
| Activity | ||||
| Catalase | 0.16 (0.07) | 0.18 (0.10) | 0.18 (0.07) | 0.79 |
| MnSOD | 3.96 (1.34) | 3.12 (1.65) | 4.56 (1.57) | 0.40 |
| TAC | 0.21 (0.10) | 0.23 (0.10) | 0.22 (0.07) | 0.88 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Receno, C.N.; Liang, C.; Korol, D.L.; Atalay, M.; Heffernan, K.S.; Brutsaert, T.D.; DeRuisseau, K.C. Effects of Prolonged Dietary Curcumin Exposure on Skeletal Muscle Biochemical and Functional Responses of Aged Male Rats. Int. J. Mol. Sci. 2019, 20, 1178. https://doi.org/10.3390/ijms20051178
Receno CN, Liang C, Korol DL, Atalay M, Heffernan KS, Brutsaert TD, DeRuisseau KC. Effects of Prolonged Dietary Curcumin Exposure on Skeletal Muscle Biochemical and Functional Responses of Aged Male Rats. International Journal of Molecular Sciences. 2019; 20(5):1178. https://doi.org/10.3390/ijms20051178
Chicago/Turabian StyleReceno, Candace N., Chen Liang, Donna L. Korol, Mustafa Atalay, Kevin S. Heffernan, Tom D. Brutsaert, and Keith C. DeRuisseau. 2019. "Effects of Prolonged Dietary Curcumin Exposure on Skeletal Muscle Biochemical and Functional Responses of Aged Male Rats" International Journal of Molecular Sciences 20, no. 5: 1178. https://doi.org/10.3390/ijms20051178
APA StyleReceno, C. N., Liang, C., Korol, D. L., Atalay, M., Heffernan, K. S., Brutsaert, T. D., & DeRuisseau, K. C. (2019). Effects of Prolonged Dietary Curcumin Exposure on Skeletal Muscle Biochemical and Functional Responses of Aged Male Rats. International Journal of Molecular Sciences, 20(5), 1178. https://doi.org/10.3390/ijms20051178







