Design of Novel Oligomeric Mixed Ligand Complexes: Preparation, Biological Applications and the First Example of Their Nanosized Scale
Abstract
:1. Introduction
2. Results and Discussion
2.1. Infrared Spectra
2.2. Mass Spectra
2.3. UV-Vis Spectra
2.4. Magnetic Measurements
2.5. Thermal Analysis
2.6. In Silico Predictions
2.7. Biological Activity
2.8. Anticancer Activity
2.9. Synthesis of Nanosized Coordinated Complexes
3. Materials and Methods
3.1. Materials
3.2. Instrumentation
3.3. Synthesis of Ternary Metal Complexes
3.3.1. Synthesis of Divalent Metal Complexes of Co, Ni, Cu or Zn
3.3.2. Synthesis of Trivalent Metal Complexes of Cr or Fe
3.3.3. Synthesis of Nanosized Coordination Polymers
3.4. In Silico Predictions
3.4.1. Bioactivity Predictions Using Molinspiration
3.4.2. Physicochemical and Pharmacokinetic Predictions Using Swiss ADME
3.5. Biological Tests
3.5.1. Cell Culture
3.5.2. Cytotoxicity Evaluation Using Viability Assay
3.6. Antimicrobial Assay
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kan, W.Q.; Ma, J.F.; Liu, Y.Y.; Yang, J. A series of coordination polymers based on 5-(2-carboxybenzyloxy) isophthalic acid and bis(imidazole) ligands: Syntheses, topological structures and photoluminescent properties. CrystEngComm 2012, 14, 2316–2326. [Google Scholar] [CrossRef]
- Chen, H.; Ruckenstein, E. Nanoparticle aggregation in the presence of a block copolymer. J. Chem. Phys. 2009, 131, 244904. [Google Scholar] [CrossRef]
- Li, H.; Shin, K.; Henkelman, G. Effects of ensembles, ligand, and strain on adsorbate binding to alloy surfaces. J. Chem. Phys. 2018, 149, 174705. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Ruckenstein, E. Structure and particle aggregation in block copolymer-binary nanoparticle composites. Polymer 2010, 51, 5869–5882. [Google Scholar] [CrossRef]
- Li, H.; Luo, L.; Kunal, P.; Bonifacio, C.S.; Duan, Z.; Yang, J.; Humphrey, S.M.; Crooks, R.M.; Henkelman, G. Oxygen Reduction Reaction on Classically Immiscible Bimetallics: A Case Study of RhAu. J. Phys. Chem. C 2018, 122, 2712–2716. [Google Scholar] [CrossRef]
- Chen, H.; Ruckenstein, E. Micellar Structures in Nanoparticle-Multiblock Copolymer Complexes. Langmuir 2014, 30, 3723–3728. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Ruckenstein, E. Aggregation of nanoparticles in a block copolymer bilayer. J. Coll. Interf. Sci. 2011, 363, 573–578. [Google Scholar] [CrossRef]
- Naushad, M.; Sharma, G.; Kumar, A.; Sharma, S.; Ghfar, A.A.; Bhatnagar, A.; Stadler, F.J.; Khan, M.K. Efficient removal of toxic phosphate anions from aqueous environment using pectin based quaternary amino anion exchanger. Int. J. Biol. Macromol. 2018, 106, 1–10. [Google Scholar] [CrossRef]
- Sumida, K.; Rogow, D.L.; Mason, J.A.; McDonald, T.M.; Bloch, E.D.; Herm, Z.R.; Bae, T.-H.; Long, J.R. Carbon dioxide capture in metal-organic frameworks. Chem. Rev. 2012, 112, 724–781. [Google Scholar] [CrossRef]
- Qiu, S.; Xuea, M.; Zhua, G. Metal–organic framework membranes: From synthesis to separation application. Chem. Soc. Rev. 2014, 43, 6116–6140. [Google Scholar] [CrossRef]
- Son, H.J.; Jin, S.; Patwardhan, S.; Wezenberg, S.J.; Jeong, N.C.; So, M.; Wilmer, C.E.; Sarjeant, A.A.; Schatz, G.C.; Snurr, R.Q.; et al. Light-harvesting and ultrafast energy migration in porphyrin-based metal-organic frameworks. J. Am. Chem. Soc. 2013, 135, 862–869. [Google Scholar] [CrossRef]
- Morris, W.; Volosskiy, B.; Demir, S.; Gándara, F.; McGrier, P.L.; Furukawa, H.; Cascio, D.; Stoddart, J.F.; Yaghi, O.M. Synthesis, Structure, and Metalation of Two New Highly Porous Zirconium Metal–Organic Frameworks. Inorg. Chem. 2012, 51, 6443–6445. [Google Scholar] [CrossRef]
- Liu, J.; Chen, L.; Cui, H.; Zhang, J.; Zhang, L.; Su, CY. Applications of metal-organic frameworks in heterogeneous supramolecular catalysis. Chem. Soc. Rev. 2014, 43, 6011–6061. [Google Scholar] [CrossRef] [PubMed]
- Phuengphai, P.; Youngme, S.; Gamezb, P.; Reedijkb, J. Catalytic properties of a series of coordination networks: Cyanosilylation of aldehydes catalyzed by Zn(II)-4,4′-bpy-carboxylato complexes. Dalt. Trans. 2010, 39, 7936–7942. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, Y.; Zhao, Y.; Liu, C. Recent progresses in the size and structure control of MOF supported noble metal catalysts. Catal. Today 2016, 263, 61–68. [Google Scholar] [CrossRef]
- Bhunia, A.; Dey, S.; Moreno, J.M.; Diaz, U.; Concepcion, P.; Hecke, K.V.; Janiak, C.; Voort, P.V.D. A homochiral vanadium–salen based cadmium bpdc MOF with permanent porosity as an asymmetric catalyst in solvent-free cyanosilylation. Chem. Commun. Commun. 2016, 52, 1401–1404. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Yue, Y.; Qian, G.; Chen, B. Luminescent Functional Metal–Organic Frameworks. Chem. Rev. 2012, 112, 1126–1162. [Google Scholar] [CrossRef]
- Heine, J.; Muller-Buschbaum, K. Engineering metal-based luminescence in coordination polymers and metal-organic frameworks. Chem. Soc. Rev. 2013, 42, 9232–9242. [Google Scholar] [CrossRef]
- Lu, K.; He, C.; Lin, W. A Chlorin-Based Nanoscale Metal–Organic Framework for Photodynamic Therapy of Colon Cancers. J. Am. Chem. Soc. 2015, 137, 7600–7603. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Liu, D.; Lin, W. Nanomedicine Applications of Hybrid Nanomaterials Built from Metal–Ligand Coordination Bonds: Nanoscale Metal–Organic Frameworks and Nanoscale Coordination Polymers. Chem. Rev. 2015, 115, 11079–11108. [Google Scholar] [CrossRef]
- Hu, Z.; Deibert, B.J.; Li, J. Luminescent metal-organic frameworks for chemical sensing and explosive detection. Chem. Soc. Rev. 2014, 43, 5815–5840. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Liu, C.P.; Jiang, F.L.; Wu, M.Y.; Chen, L.; Qian, J.J.; Su, K.Z.; Wan, X.Y.; Hong, M.C. Diverse architectures and luminescence properties of two novel copper(I) coordination polymers assembled from 2,6-bis[3-(pyrid-4-yl)-1,2,4-triazolyl]pyridine ligands. CrystEngComm 2015, 17, 1541–1548. [Google Scholar] [CrossRef]
- Kreno, L.E.; Leong, K.; Farha, O.K.; Allendorf, M.; Van Duyne, R.P.; Hupp, J.T. Metal–Organic Framework Materials as Chemical Sensors. Chem. Rev. 2012, 112, 1105–1125. [Google Scholar] [CrossRef] [PubMed]
- Charlot, M.; Kahn, O.; Jeannin, S.; Jeannin, Y. Exchange interaction in roof-shaped hydroxo-bridged copper (II) dimers. Inorg. Chem. 1980, 19, 1410–1411. [Google Scholar] [CrossRef]
- Sigel, H.; Operschall, B.P.; Massoud, S.S.; Song, B.; Griesser, R. Evidence for intramolecular aromatic-ring stacking in the physiological pH range of the monodeprotonated xanthine residue in mixed-ligand complexes containing xanthosinate 5′-monophosphate (XMP). Dalt. Trans. 2006, 5521–5529. [Google Scholar] [CrossRef] [PubMed]
- Farver, O.; Pecht, I. Structure—reactivity studies of blue copper preteins. Affinity labeling of electron transfer proteins by transition metal coordination. Coord. Chem. Rev. 1989, 94, 17–45. [Google Scholar] [CrossRef]
- Burns, C.; Field, L.; Hambley, T.; Lin, T.; Ridley, D.; Turner, P.; Wilkinson, M. X-Ray crystal structural determination of copper ( II ) -nitrilotriacetic acid-bis ( N -methylimidazol-2-yl ) ketone ternary complex. ARKIVOCvii 2001, 7, 157–165. [Google Scholar]
- Nowack, B. Environmental chemistry of aminopolycarboxylate chelating agents. Environ. Sci. Technol. 2002, 36, 4009–4016. [Google Scholar] [CrossRef]
- Arnold, F.H. Metal-affinity separations: A new dimension in protein processing. Biotechnology 1991, 9, 151–156. [Google Scholar] [CrossRef]
- Hochuli, E.; Dobeli, H.; Schacher, A. New metal chelate adsorbent selective for proteins and peptides containing neighbouring histidine residues. J. Chromatogr. 1987, 411, 177–184. [Google Scholar] [CrossRef]
- Nancharaiah, Y.V.; Schwarzenbeck, N.; Mohan, T.V.K.; Narasimhan, S.V.; Wilderer, P.A.; Venugopalan, V.P. Biodegradation of nitrilotriacetic acid (NTA) and ferric-NTA complex by aerobic microbial granules. Water Res. 2006, 40, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Mendola, M.E.; Paul, T.; Strathmann, T.J.; Carbonaro, R.F. Investigation of the kinetics of aquation of the 1:2 complex between CrIII and nitrilotriacetic acid. Polyhedron 2009, 28, 269–278. [Google Scholar] [CrossRef]
- Kornev, V.I.; Semenova, M.G.; Merkulov, D.A. Homo- and heteroleptic cobalt(II) and nickel(II) complexes with nitrilotriacetic and dibasic carboxylic acids. Russ. J. Coord. Chem. 2009, 35, 519–525. [Google Scholar] [CrossRef]
- Andrianirinaharivelo, S.L.; Pilichowski, J.; Bolte, M. Nitrilotriacetic acid transformation photo-induced by complexation with iron (III) in aqueous solution. Transit. Met. Chem. 1993, 18, 37–41. [Google Scholar] [CrossRef]
- Dutta, S.; Ray, S.; Nagarajan, K. Glutamic acid as anticancer agent: An overview. Saudi Pharm. J. 2013, 21, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Latha, M.P.; Rao, V.M.; Rao, T.S.; Rao, G.N. Speciation of ternary complexes of some essential metal ions with L-glutamic acid and L-methionine in 1, 2-propanediol - water mixtures. Acta Chim. Slov. 2008, 55, 248–256. [Google Scholar]
- Bartaria, D.; Chandra, P.; Krishna, V. A Study on speciation and coordination tendency of glutamic acid and uracil for ternary complexation towards some toxic metal ions. Chem. Sci. Rev. Lett. 2013, 1, 201–208. [Google Scholar]
- Wenlock, M.C.; Barton, P. In silico physicochemical parameter predictions. Mol. Pharm. 2013, 10, 1224–1235. [Google Scholar] [CrossRef]
- Tian, S.; Wand, J.; Li, Y.; Li, D.; Xu, L.; Hou, T. The application of in silico drug-likeness predictions in pharmaceutical research. Adv. Drug Deliv. Rev. 2015, 86, 2–10. [Google Scholar] [CrossRef]
- Tian, F.; Tian, F.; Prina-Mello, A.; Estrada, G.; Beyerle, A.; Möller, W.; Schulz, H.; Kreyling, W.; Stoeger, T. A novel assay for the quantification of internalized nanoparticles in macrophages. Nanotoxicology 2008, 2, 1–11. [Google Scholar] [CrossRef]
- Cui, D.; Tian, F.; Coyer, S.R.; Wang, J.; Pan, B.; Gao, F.; He, R.; Zhang, Y. Effects of antisense-myc-conjugated single-walled carbon nanotubes on HL-60 cells. J. Nanosci. Nanotechnol. 2007, 7, 1–8. [Google Scholar] [CrossRef]
- Edelstein, R.L.; Tamanaha, C.R.; Sheehan, P.E.; Miller, M.M.; Baselt, D.R.; Whitman, L.J.; Colton, R.J. The BARC biosensor applied to the detection of biological warfare agents. Biosens. Bioelectron. 2000, 14, 805–813. [Google Scholar] [CrossRef]
- Isla, A.d.l.; Brostow, W.; Bujard, B.; Estevez, M.; Rodriguez, J.R.; Vargas, S.; Castaño, V.M. Nanohybrid scratch resistant coatings for teeth and bone viscoelasticity manifested in tribology. Mater. Res. Innov. 2003, 7, 110–114. [Google Scholar] [CrossRef]
- Williams, M.; Okasha, R.M.; Nairn, J.; Twamley, B.; Afifi, T.H.; Shapiro, P.J. A photochemical route to discrete, ternary metal chalcogenide clusters. Chem. Commun. 2007, 3177–3179. [Google Scholar] [CrossRef] [PubMed]
- Baranwal, B.P.; Fatma, T.; Singh, A.K.; Varma, A. Nano-sized titanium(IV) ternary and quaternary complexes with electron-rich oxygen-based bidentate ligands. Inorganica Chim. Acta 2009, 362, 3461–3464. [Google Scholar] [CrossRef]
- Baranwal, B.P.; Fatma, T.; Varma, A.; Singh, A.K. Substitution reactions of thorium(IV) acetate to synthesize nano-sized carboxylate complexes. Spectrochim. Acta. A Mol. Biomol. Spectrosc. 2010, 75, 1177–1180. [Google Scholar] [CrossRef] [PubMed]
- Baranwal, B.P.; Singh, A.K. Nano-sized, quaternary titanium(IV) metal-organic frameworks with multidentate ligands. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 77, 938–941. [Google Scholar] [CrossRef]
- Souaya, E.R.; Ismail, E.H.; Mohamed, A.A.; Milad, N.E. Preparation, characterization and thermal studies of some transition metal ternary complexes. J. Therm. Anal. Calorim. 2009, 95, 253–258. [Google Scholar] [CrossRef]
- Ismail, E.H.; Souaya, E.R.; Mohamed, A.A. Synthesis, characterization, and thermal analysis of two polymeric chromium ternary complexes with nitrilotriacetic acid and alanine or glycine. J. Appl. Polym. Sci. 2011, 124, 1976–1980. [Google Scholar] [CrossRef]
- Khalil, M.M.H.; Ismail, E.H.; Azim, S.A.; Souaya, E.R. Synthesis, characterization, and thermal analysis of ternary complexes of nitrilotriacetic acid and alanine or phenylalanine with some transition metals. J. Therm. Anal. Calorim. 2010, 101, 129–135. [Google Scholar] [CrossRef]
- Brain, G.C. Acids and Bases: Solvent Effects on Acid-Base Strength; Oxford University Press: Oxford, UK, 2013. [Google Scholar] [CrossRef]
- Chow, S.T.; McAuliffe, C.A.; Sayle, B.J. Metal complexes of amino acids and derivatives—VIII [1]: The reaction of D, L-penicillamine with some transition and non-transition metal salts. J. Inorg. Nucl. Chem. 1973, 35, 4349–4354. [Google Scholar] [CrossRef]
- Chen, Y.J.; Nuevo, M.; Hsieh, J.M.; Yih, T.S.; Sun, W.H.; Ip, W.H.; Fung, H.S.; Chiang, S.Y.; Lee, Y.Y.; Chen, J.M.; et al. Carbamic acid produced by the UV / EUV irradiation of interstellar ice analogs. Astron. Astrophys. 2007, 464, 253–257. [Google Scholar] [CrossRef]
- Rosado, M.T.; Duarte, M.L.T.S.; Fausto, R. Vibrational spectra of acid and alkaline glycine salts. Vib. Spectrosc. 1998, 16, 35–54. [Google Scholar] [CrossRef]
- Gowri, S.; Sathiyabama, J.; Rajendran, S. The inhibitive effect of glutamic acid on the corrosion of carbon steel in sea water. Int. J. Chem. Tech. Res. 2013, 5, 347–352. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1986. [Google Scholar] [CrossRef]
- Cotton, F.A.; Wilkinson, G.; Murillo, C.A.; Bochmann, M. Advanced Inorganic Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1999. [Google Scholar]
- Mahmoud, W.H.; Mohamed, G.G.; El-Dessouky, M.M.I. Synthesis, structural characterization, in vitro antimicrobial and anticancer activity studies of ternary metal complexes containing glycine amino acid and the anti-inflammatory drug lornoxicam. J. Mol. Struct. 2015, 1082, 12–22. [Google Scholar] [CrossRef]
- Khalill, M.M.H.; Souaya, E.R.; Ismail, E.H.; Rabie, E. Ternary transition metal complexes of nitrilotriacetic acid and valine or leucine: Synthesis and biological appplication. Chinese J. Inorg. Chem. 2013, 29, 1969–1978. [Google Scholar] [CrossRef]
- Gudasi, K.B.; Patil, S.A.; Vadavi, R.S.; Shenoy, R.V.; Patil, M.S. Synthesis and spectral studies of Cu(II), Ni(II), Co(II), Mn(II), Zn(II) and Cd(II) complexes of a new macroacyclic ligand N,N’-bis(2-benzothiazolyl)-2,6-pyridinedicarboxamide. J. Serbian Chem. Soc. 2006, 71, 529–542. [Google Scholar] [CrossRef]
- Reddy, S.L.; Endo, T.; Reddy, G.S. Advanced Aspects of Spectroscopy; InTech.: London, UK, 2012; pp. 4–48. [Google Scholar]
- Orabi, A.S.; Abbas, A.M.; Sallam, S.A. Spectral, magnetic, thermal, and DNA interaction of Ni(II) complexes of glutamic acid schiff bases. Synth. React. Inorganic, Met. Nano-Metal Chem. 2013, 43, 63–75. [Google Scholar] [CrossRef]
- Hatakeyama, T.; Liu, Z. Handbook of Thermal Analysis; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1998. [Google Scholar]
- European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Dieases (ESCMID). Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by agar dilution. Clin. Microbiol. Infect. 2000, 6, 509–515. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
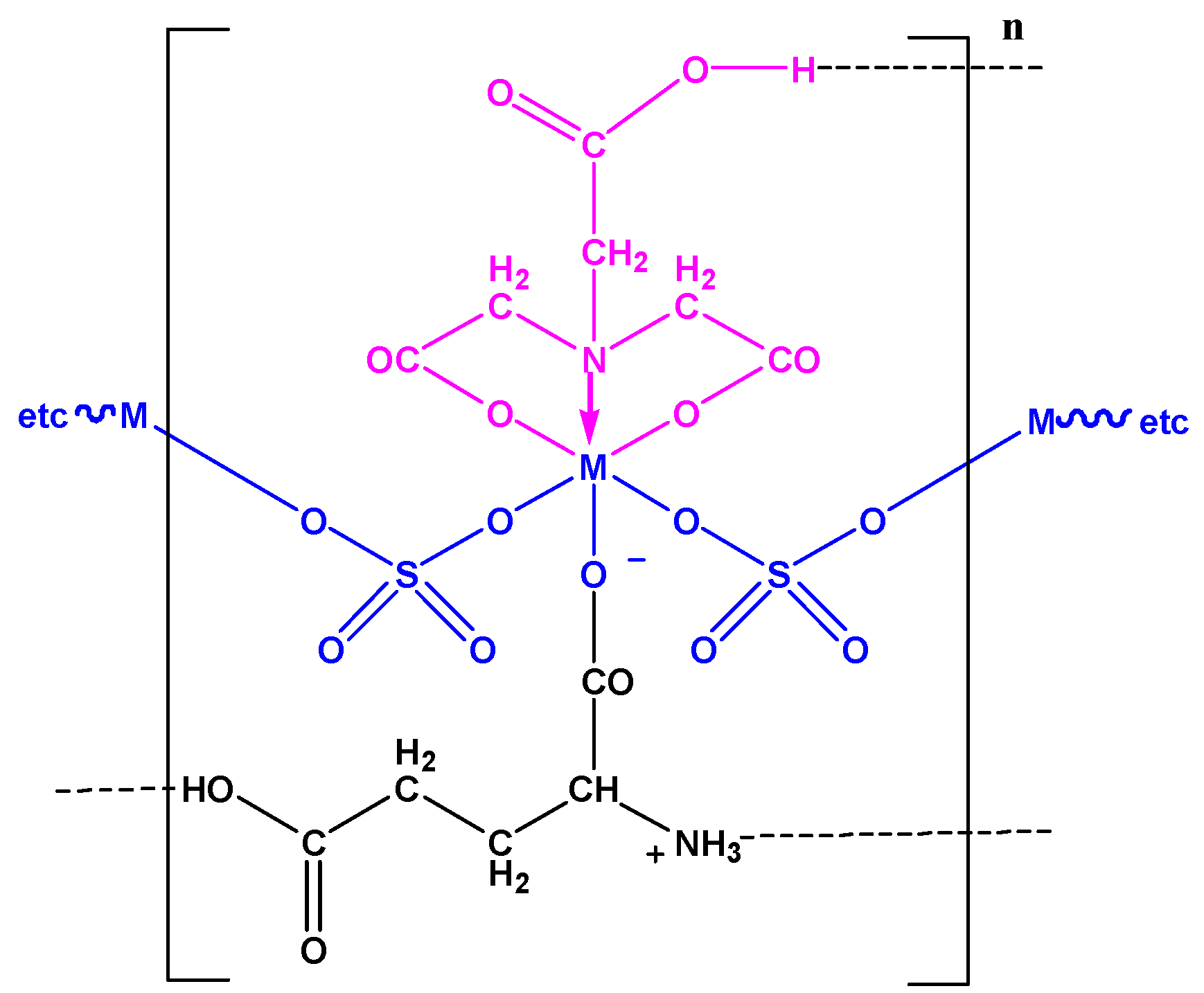
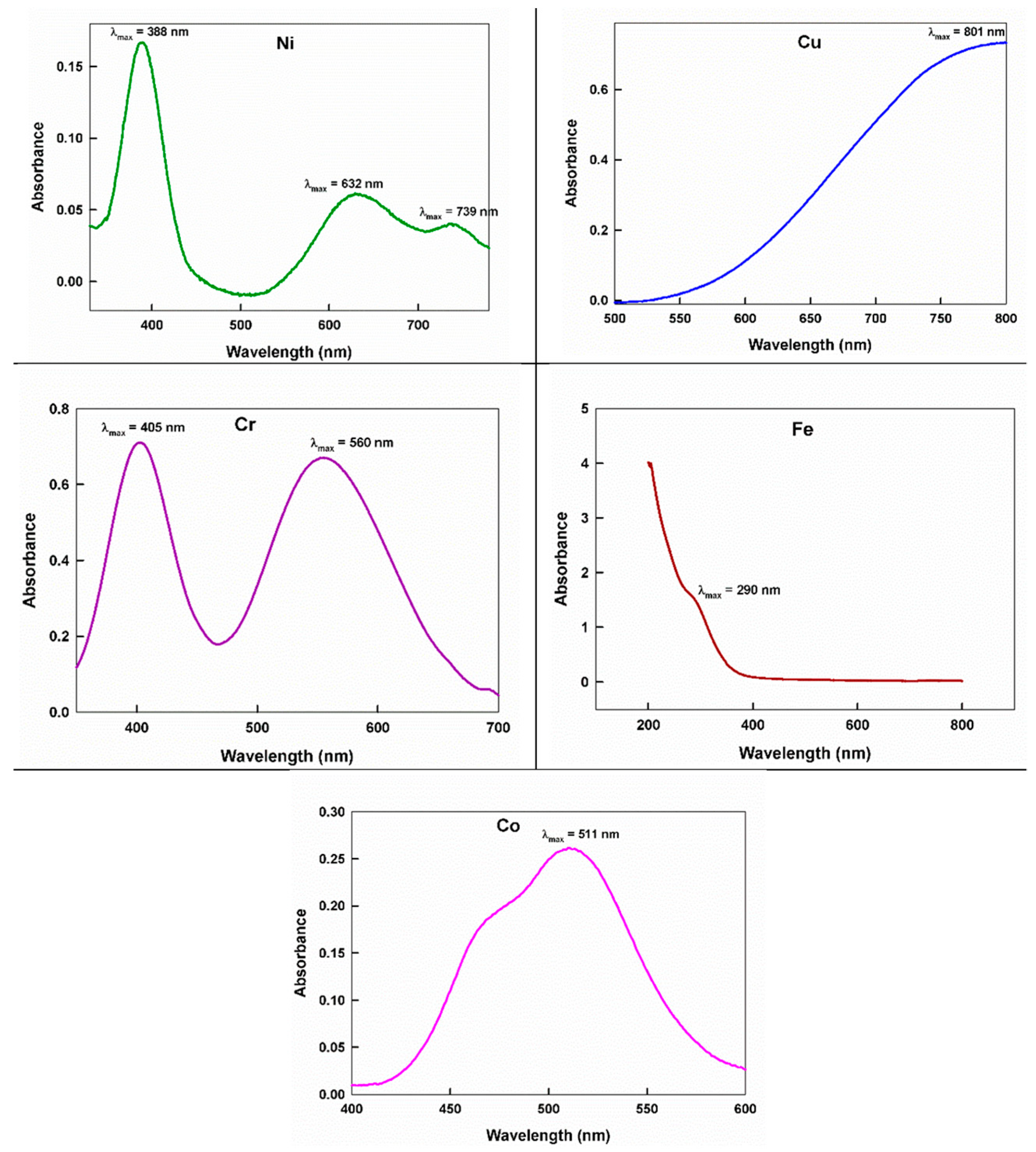
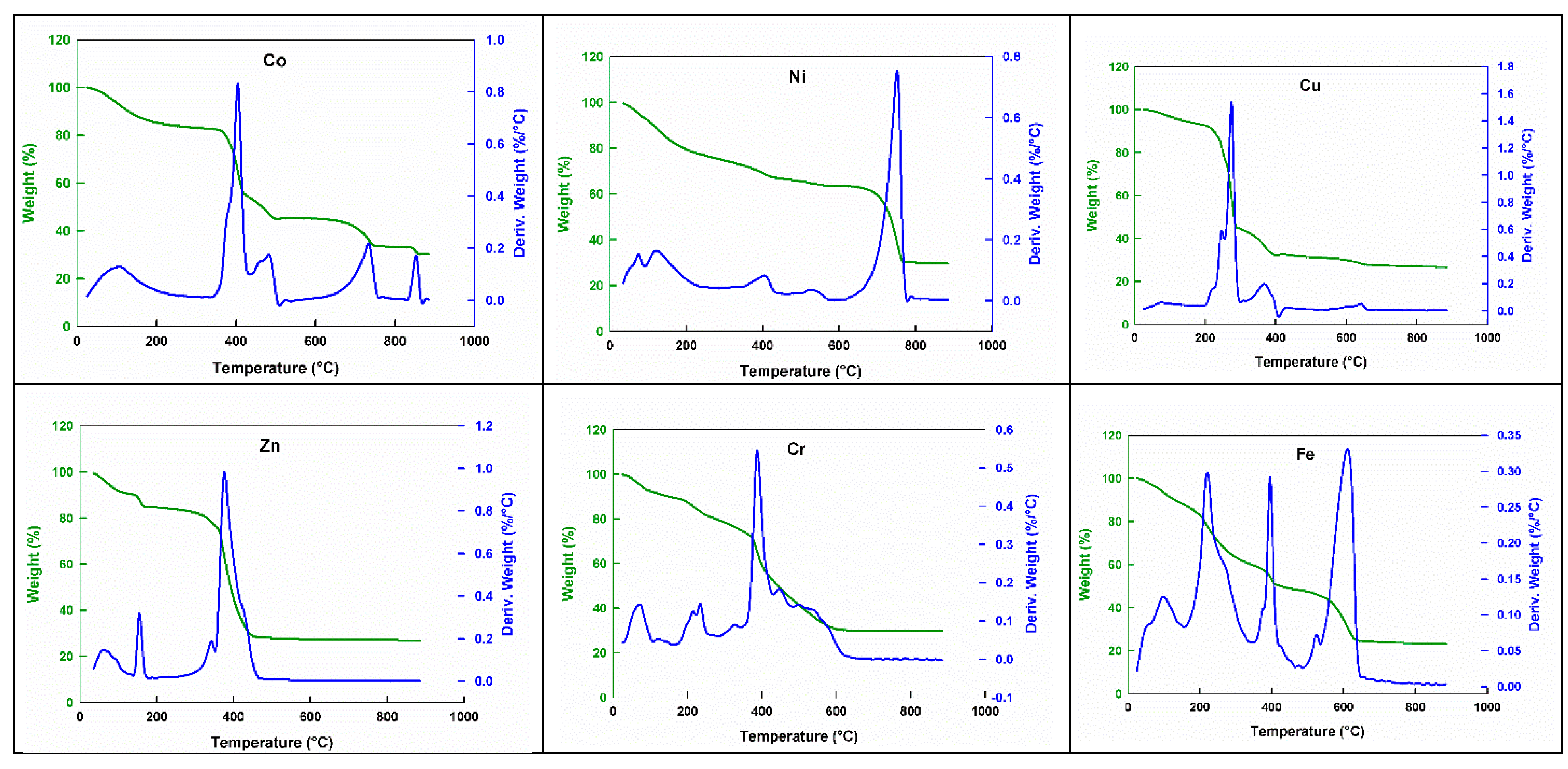

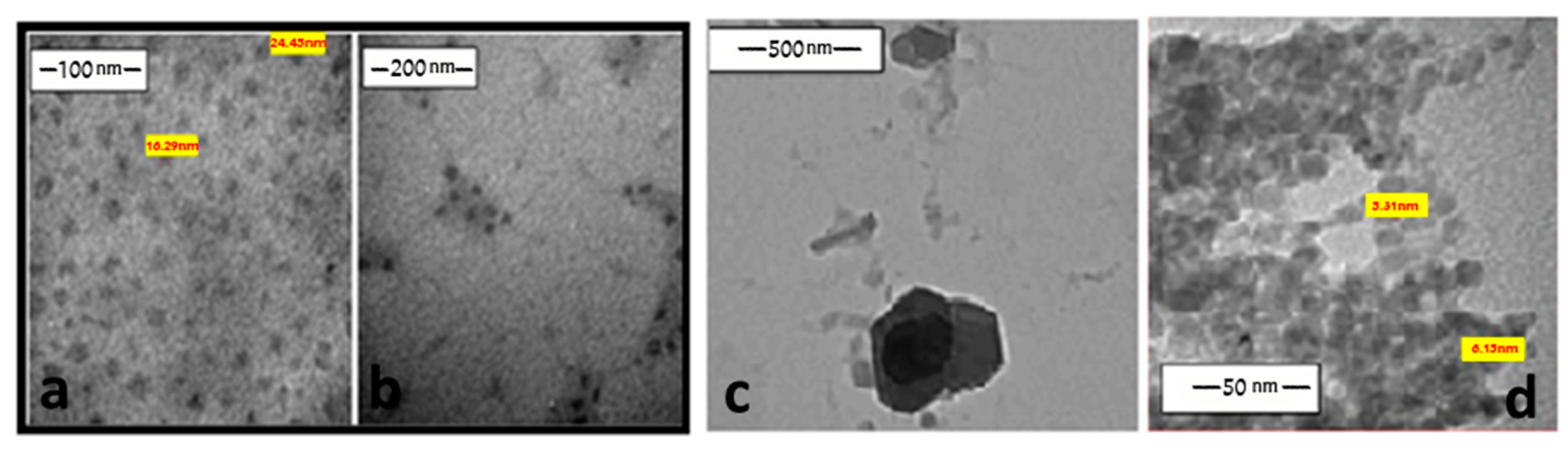



| No. | Monomeric Unit | Color | Mol. Wt. | pH | Magnetic Moment | C% cal/found | H% cal./found | N% cal./found | S% cal./found | M% cal./found |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | H2(Co(HNTA)(GluH)(SO4)) | Pink | 493.268 | 3.3 | 6.49 | 26.76/25.98 | 3.65/3.57 | 5.68/4.99 | 6.49/7.50 | 11.95/12.00 |
| 2 | H2(Ni(HNTA)(GluH)(SO4)) | Green | 493.028 | 4 | 2.99 | 26.77/25.98 | 3.65/3.85 | 5.68/4.86 | 6.49/7.55 | 11.90/11.75 |
| 3 | H2(Cu(HNTA)(GluH)(SO4)) | Blue | 497.881 | 4.4 | 1.16 | 26.51/26.96 | 3.62/2.95 | 5.62/5.38 | 6.43/5.74 | 12.76/12.87 |
| 4 | H2(Zn(HNTA)(GluH)(SO4)) | White | 499.744 | 2.3 | diamagnetic | 26.41/26.50 | 3.60/3.32 | 5.60/5.31 | 6.40/6.70 | 14.09/14.56 |
| 5 | H(Cr(HNTA)(GluH)(SO4)) | Blue violet | 486.331 | 3 | 4.96 | 27.14/27.49 | 3.70/3.70 | 5.75/5.26 | 6.58/8.21 | 10.69/10.49 |
| 6 | H(Fe(HNTA)(GluH)(SO4)) | Paige | 490.182 | 3 | 4.16 | 26.93/26.86 | 3.67/3.65 | 5.71/5.40 | 6.53/7.50 | 13.39/13.62 |
| No. | Ligands and Oligomers | υ (OH) | υ (NH3+) | υ (COOH) | υ (COO−) | υ (C–N) | υ (M–N) | υ (M–O) | ν1 (SO4) | ν2 (SO4) | ν3 (SO4) | ν4 (SO4) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NTA | 3434 | - | 1725 | 1575 (as) 1530 (s) | 1206 | - | - | - | - | - | - | |
| Glu | 3434 | 3062 | 1728 | 1641 (as) 1418 (s) | 1242 | - | - | - | - | - | - | |
| 1 | (H2(Co(HNTA)(GluH)(SO4)))n | 3417 | 3048 | - | 1587 (as) 1455–1421 (s) | 1026 | 551 | 395 | 989 | 446 | 1026 1177 1102 | 551 633 |
| 2 | (H2(Ni(HNTA)(GluH)(SO4)))n | 3395 | 3056 | - | 1589 (as) 1462–1414 (s) | 1088 | 556 | 343 | 981 | 443 | 1064–1088 1185 1122 | 564 626 669 |
| 3 | H2(Cu(HNTA)(GluH)(SO4)))n | 3424 | 3193 | - | 1597–1567 (as) 1435–1418 (s) | 1048 | 552 | 332 | 983 | 467 | 1048 1193 1102 | 596 604 641 |
| 4 | (H2(Zn(HNTA)(GluH)(SO4)))n | 3452 | 3048 | 1724 | 1622–1584 (as) 1461–1406 (s) | 1064 | 557 | 318 | 988 | 477 | 1064 1169 1153 | 570 627 677 |
| 5 | (H(Cr(HNTA)(GluH)(SO4)))n | 3450 | 3056 | - | 1644 (as) 1435–1387 (s) | 1051 | 550 | 330 | 995 | 479 | 1051 1185 1104 | 598 620 653 |
| 6 | (H(Fe(HNTA)(GluH)(SO4)))n | 3445 | 3080 | - | 1628–1548 (as) 1445 (s) | 1044 | 556 | 320 | 997 | 482 | 1072 1169 1104 | 588 629 661 |
| No. | Oligomers | Monomer Mol. Wt. | Monomer m/z | m/z |
|---|---|---|---|---|
| 1 | (H2(Co(HNTA)(GluH)(SO4)))n | 493.268 | 497 | 1678 |
| 2 | (H2(Ni(HNTA)(GluH)(SO4)))n | 493.028 | 499 | 1321 |
| 3 | (H2(Cu(HNTA)(GluH)(SO4)))n | 497.881 | 495 | 1217 |
| 4 | (H2(Zn(HNTA)(GluH)(SO4)))n | 499.744 | 509 | 1307 |
| 5 | (H(Cr(HNTA)(GluH)(SO4)))n | 486.331 | 499 | 1627 |
| 6 | (H(Fe(HNTA)(GluH)(SO4)))n | 490.182 | 425 | 2402 |
| No. | Oligomers | Mol. Wt. | TGA Range (°C) | Mass Loss (%) | Total Mass Loss (%) | Assignment | ||
|---|---|---|---|---|---|---|---|---|
| Fou. | Calc. | Fou. | Calc. | |||||
| 1 | (H2(Co(HNTA) (GluH)(SO4)))n | 493.268 | 25–150 | 10.50 | 10.34 | 74.20 | 75.41 | H2S+NH3 |
| 150–441 | 37.50 | 38.72 | Maleic acid+glycine | |||||
| 441–779 | 23.00 | 23.11 | 2CO2+C2H2 | |||||
| 779–868 | 3.20 | 3.24 | CH4 | |||||
| Above 868 | 25.80 | 24.92 | Residue | CoSO2 | ||||
| 2 | (H2(Ni(HNTA) (GluH)(SO4)))n | 493.028 | 25–127 | 10.00 | 10.34 | 70.50 | 72.20 | H2S+NH3 |
| 127–429 | 22.00 | 23.53 | Maleic acid | |||||
| 429–779 | 38.50 | 37.53 | Glycine+3CO+C2H2 | |||||
| Above 779 | 29.50 | 28.13 | Residue | NiSO3 | ||||
| 3 | (H2(Cu(HNTA) (GluH)(SO4)))n | 497.881 | 25–200 | 7.00 | 6.8 | 73.50 | 74.69 | H2S |
| 200–664 | 66.50 | 67.89 | Maleic acid+ glycine+ CH4+2CO2+C2H2 +NH3 | |||||
| Above 664 | 26.50 | 25.62 | Residue | CuSO2 | ||||
| 4 | (H2(Zn(HNTA) (GluH)(SO4)))n | 499.744 | 25–135 | 10.00 | 10.21 | 73.00 | 74.44 | H2S + NH3 |
| 135–500 | 63.00 | 64.23 | Maleic acid + glyciene + 2CO2 + C2H2+ CH4 | |||||
| Above 500 | 26.90 | 25.89 | Residue | ZnSO4 | ||||
| 5 | (H(Cr(HNTA) (GluH)(SO4)))n | 486.331 | 25–100 | 7.00 | 6.99 | 70.50 | 71.26 | H2S |
| 100–212 | 6.50 | 6.79 | NH3 +CH4 | |||||
| 212–650 | 57.00 | 56.55 | Maleic acid + Glycine + 3CO | |||||
| Above 650 | 29.50 | 30.43 | Residue | CrSO4 | ||||
| 6 | H(Fe(HNTA) (GluH)(SO4)) | 490.182 | 25–122 | 7.00 | 6.94 | 77.00 | 75.9 | H2S |
| 122–400 | 41.00 | 39.99 | Glycine + 2CO2 + NH3 + CH4 | |||||
| 400–650 | 29.00 | 28.97 | Maleic acid + C2H2 | |||||
| Above 650 | 23.00 | 24.45 | Residue | FeSO4 | ||||
| No. | GPCR Ligand | Ion Channel Modulator | Kinase Inhibitor | Nuclear Receptor Ligand | Protease Inhibitor | Enzyme Inhibitor |
|---|---|---|---|---|---|---|
| 1 | 0.04 | 0.21 | −0.00 | 0.01 | 0.47 | 0.50 |
| 2 | 0.04 | 0.21 | −0.00 | 0.01 | 0.47 | 0.50 |
| 3 | 0.04 | 0.21 | −0.00 | 0.01 | 0.47 | 0.50 |
| 4 | 0.40 | 0.21 | −0.00 | 0.01 | 0.47 | 0.54 |
| 5 | 0.04 | 0.21 | −0.00 | 0.01 | 0.47 | 0.50 |
| 6 | 0.45 | 0.21 | −0.00 | 0.01 | 0.47 | 0.53 |
| No. | Monomeric Unit | Mol. wt. | Fraction Csp3 a | HBA b | HBD c | Molar Refractivity | Water Solubility | TPSA d |
|---|---|---|---|---|---|---|---|---|
| 1 | H2(Co(HNTA)(GluH)(SO4)) | 493.268 | 0.58 | 19 | 7 | 100.09 | Yes | 364.14 |
| 2 | H2(Ni(HNTA)(GluH)(SO4)) | 493.028 | 0.57 | 19 | 7 | 100.15 | Yes | 364.16 |
| 3 | H2(Cu(HNTA)(GluH)(SO4)) | 497.881 | 0.55 | 19 | 7 | 100.15 | Yes | 364.17 |
| 4 | H2(Zn(HNTA)(GluH)(SO4)) | 499.744 | 0.56 | 19 | 7 | 100.11 | Yes | 364.13 |
| 5 | H(Cr(HNTA)(GluH)(SO4)) | 486.331 | 0.54 | 19 | 7 | 100.13 | Yes | 364.15 |
| 6 | H(Fe(HNTA)(GluH)(SO4)) | 490.182 | 0.55 | 19 | 7 | 100.15 | Yes | 364.17 |
| No. | Monomeric Unit | GI abs a | BBB Permeant b | P-Gpsubstrate c | CYP1A2 Inhibitor d | Log Kp e |
|---|---|---|---|---|---|---|
| 1 | H2(Co(HNTA)(GluH)(SO4)) | Low | No | Yes | No | −16.21 |
| 2 | H2(Ni(HNTA)(GluH)(SO4)) | Low | No | Yes | No | −16.21 |
| 3 | H2(Cu(HNTA)(GluH)(SO4)) | Low | No | Yes | No | −16.24 |
| 4 | H2(Zn(HNTA)(GluH)(SO4)) | Low | No | Yes | No | −16.25 |
| 5 | H(Cr(HNTA)(GluH)(SO4)) | Low | No | Yes | No | −16.17 |
| 6 | H(Fe(HNTA)(GluH)(SO4)) | Low | No | Yes | No | −16.19 |
| No. | Cpd. | S. pneum. (+ve) | B. Subtilis (+ve) | P. Aerug. (−ve) | E. coli (−ve) | A. fumig. | S. raceme. | G. candi. | C. albic. |
|---|---|---|---|---|---|---|---|---|---|
| Amphotericin B | NA | NA | NA | NA | 23.7 ± 0.1 (0.24) | 19.7 ± 0.2 (3.9) | 28.7 ± 0.2 (0.015) | 25.4 ± 0.1 (0.12) | |
| Ampicillin | 23.8 ± 0.2 (0.24 ) | 32.4 ± 0.3 (0.007 ) | NA | NA | NA | NA | NA | ||
| Gentamicin | NA | NA | 17.3 ± 0.1 (15.63 ) | 19.9 ± 0.3 (3.9 ) | NA | NA | NA | NA | |
| 1 | (H2(Co(HNTA) (GluH)(SO4)))n | NA | 12.3 ± 0.37 | NA | 13.6 ± 0.58 | 13.4 ± 0.63 | 11.6 ± 0.58 | 12.6 ± 0.25 | 10.6 ± 0.44 |
| 2 | (H2(Ni(HNTA) (GluH)(SO4)))n | 22.0 ± 0.29 (1.95) | 26.1 ± 0.44 (0.24) | 22.9 ± 0.58 (7.81) | 21.9 ± 0.72 (1.95) | 20.3 ± 0.58 (3.9) | 19.4 ± 0.44(31.25) | 22.3 ± 0.63 (3.9) | 18.4 ± 0.44 (125) |
| 3 | (H2(Cu(HNTA) (GluH)(SO4)))n | 20.9 ± 0.44 (1.95) | 23.8 ± 0.17 (0.24) | 22.3 ± 0.17 (0.98) | 25.2 ± 0.63 (0.12) | 24.8 ± 0.58 (0.12) | 23.8 ± 0.58 (0.24) | 24.2 ± 0.44 (0.24) | 20.9 ± 0.37 (1.95) |
| 4 | (H2(Zn(HNTA) (GluH)(SO4)))n | 18.2 ± 0.63 (15.63) | 19.3 ± 0.72 (7.81) | NA | 11.8 ± 0.58 (500) | 18.4 ± 0.58 (15.63) | 15.2 ± 0.58 (62.5) | 15.9 ± 0.08 (62.5) | 9.8 ± 0.44 (NA) |
| 5 | (H(Cr(HNTA) (GluH)(SO4)))n | 14.5 ± 0.44 (62.5) | 22.3 ± 0.37 (0.98) | NA | 11.6 ± 0.25 (125) | 17.2 ± 0.63 (31.25) | 16.8 ± 0.44 (31.25) | 18.3 ± 0.44 (15.63) | NA |
| 6 | (H(Fe(HNTA) (GluH)(SO4)))n | 24.7 ± 0.58 (0.12) | 28.2 ± 0.58 (0.015) | 22.8 ± 0.44 (0.49) | 24.4 ± 0.63 (0.24) | 24.4 ± 0.37 (0.24) | 17.2 ± 0.72 (31.25) | 25.3 ± 0.44 (0.12) | 19.9 ± 0.77 (3.9) |
| No. | Oligomers | Sample Conc. (µg/mL) | MCF-7 Viability% | IC50 (µg/mL) | HCT-116 Viability (µg/mL) | IC50 (µg/mL) | HepG-2 Viability% | IC50 (µg/mL) |
|---|---|---|---|---|---|---|---|---|
| 1 | (H2(Co(HNTA) (GluH)(SO4)))n | 50 | 64.28 | >50 | 68.57 | >50 | 60.86 | >50 |
| 25 | 72.73 | 82.46 | 78.13 | |||||
| 12.5 | 89.42 | 93.12 | 91.48 | |||||
| 6.25 | 94.54 | 98.78 | 97.24 | |||||
| 3.125 | 98.16 | 100 | 100 | |||||
| 1.56 | 100 | 100 | 100 | |||||
| 0 | 100 | 100 | 100 | |||||
| 2 | (H2(Ni(HNTA) (GluH)(SO4)))n | 50 | 49.84 | 49.7 ± 0.23 | 57.42 | >50 | 46.18 | 44.8 ± 0.24 |
| 25 | 63.51 | 69.78 | 64.39 | |||||
| 12.5 | 71.62 | 83.14 | 80.61 | |||||
| 6.25 | 82.94 | 92.65 | 91.74 | |||||
| 3.125 | 91.05 | 98.78 | 96.52 | |||||
| 1.56 | 97.48 | 100 | 99.06 | |||||
| 0 | 100 | 100 | 100 | |||||
| 3 | (H2(Cu(HNTA) (GluH)(SO4)))n | 50 | 24.32 | 11.5 ± 0.09 | 26.98 | 18.00 ± 0.08 | 21.54 | 12.00 ± 0.05 |
| 25 | 33.57 | 39.44 | 36.93 | |||||
| 12.5 | 45.34 | 58.23 | 48.22 | |||||
| 6.25 | 74.69 | 75.16 | 71.36 | |||||
| 3.125 | 85.71 | 89.28 | 84.59 | |||||
| 1.56 | 93.56 | 95.34 | 92.64 | |||||
| 0 | 100 | 100 | 100 | |||||
| 4 | (H2(Zn(HNTA) (GluH)(SO4)))n | 50 | 20.43 | 10.7 ± 0.11 | 29.17 | 20.1 ± 0.10 | 23.67 | 16.1 ± 0.06 |
| 25 | 34.68 | 45.26 | 40.89 | |||||
| 12.5 | 42.97 | 57.41 | 53.72 | |||||
| 6.25 | 67.25 | 72.41 | 69.17 | |||||
| 3.125 | 80.13 | 81.04 | 86.28 | |||||
| 1.56 | 87.49 | 89.56 | 93.02 | |||||
| 0 | 100 | 100 | 100 | |||||
| 5 | (H(Cr(HNTA) (GluH)(SO4)))n | 50 | 77.18 | >50 | 84.62 | >50 | 76.28 | >50 |
| 25 | 86.27 | 93.13 | 91.36 | |||||
| 12.5 | 95.84 | 98.74 | 97.96 | |||||
| 6.25 | 99.08 | 100 | 100 | |||||
| 3.125 | 100 | 100 | 100 | |||||
| 1.56 | 100 | 100 | 100 | |||||
| 0 | 100 | 100 | 100 | |||||
| 6 | (H(Fe(HNTA) (GluH)(SO4)))n | 50 | 45.96 | 47.3 ± 0.19 | 61.84 | >50 | 42.69 | 44.1 ± 0.17 |
| 25 | 82.74 | 83.12 | 73.48 | |||||
| 12.5 | 90.36 | 91.46 | 82.97 | |||||
| 6.25 | 94.28 | 97.38 | 91.64 | |||||
| 3.125 | 98.93 | 99.17 | 98.26 | |||||
| 1.56 | 100 | 100 | 100 | |||||
| 0 | 100 | 100 | 100 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Okasha, R.M.; AL-Shaikh, N.E.; Aljohani, F.S.; Naqvi, A.; Ismail, E.H. Design of Novel Oligomeric Mixed Ligand Complexes: Preparation, Biological Applications and the First Example of Their Nanosized Scale. Int. J. Mol. Sci. 2019, 20, 743. https://doi.org/10.3390/ijms20030743
Okasha RM, AL-Shaikh NE, Aljohani FS, Naqvi A, Ismail EH. Design of Novel Oligomeric Mixed Ligand Complexes: Preparation, Biological Applications and the First Example of Their Nanosized Scale. International Journal of Molecular Sciences. 2019; 20(3):743. https://doi.org/10.3390/ijms20030743
Chicago/Turabian StyleOkasha, Rawda M., Najla E. AL-Shaikh, Faizah S. Aljohani, Arshi Naqvi, and Eman H. Ismail. 2019. "Design of Novel Oligomeric Mixed Ligand Complexes: Preparation, Biological Applications and the First Example of Their Nanosized Scale" International Journal of Molecular Sciences 20, no. 3: 743. https://doi.org/10.3390/ijms20030743
APA StyleOkasha, R. M., AL-Shaikh, N. E., Aljohani, F. S., Naqvi, A., & Ismail, E. H. (2019). Design of Novel Oligomeric Mixed Ligand Complexes: Preparation, Biological Applications and the First Example of Their Nanosized Scale. International Journal of Molecular Sciences, 20(3), 743. https://doi.org/10.3390/ijms20030743






