Chitin Nanofibrils in Poly(Lactic Acid) (PLA) Nanocomposites: Dispersion and Thermo-Mechanical Properties
Abstract
1. Introduction
2. Results
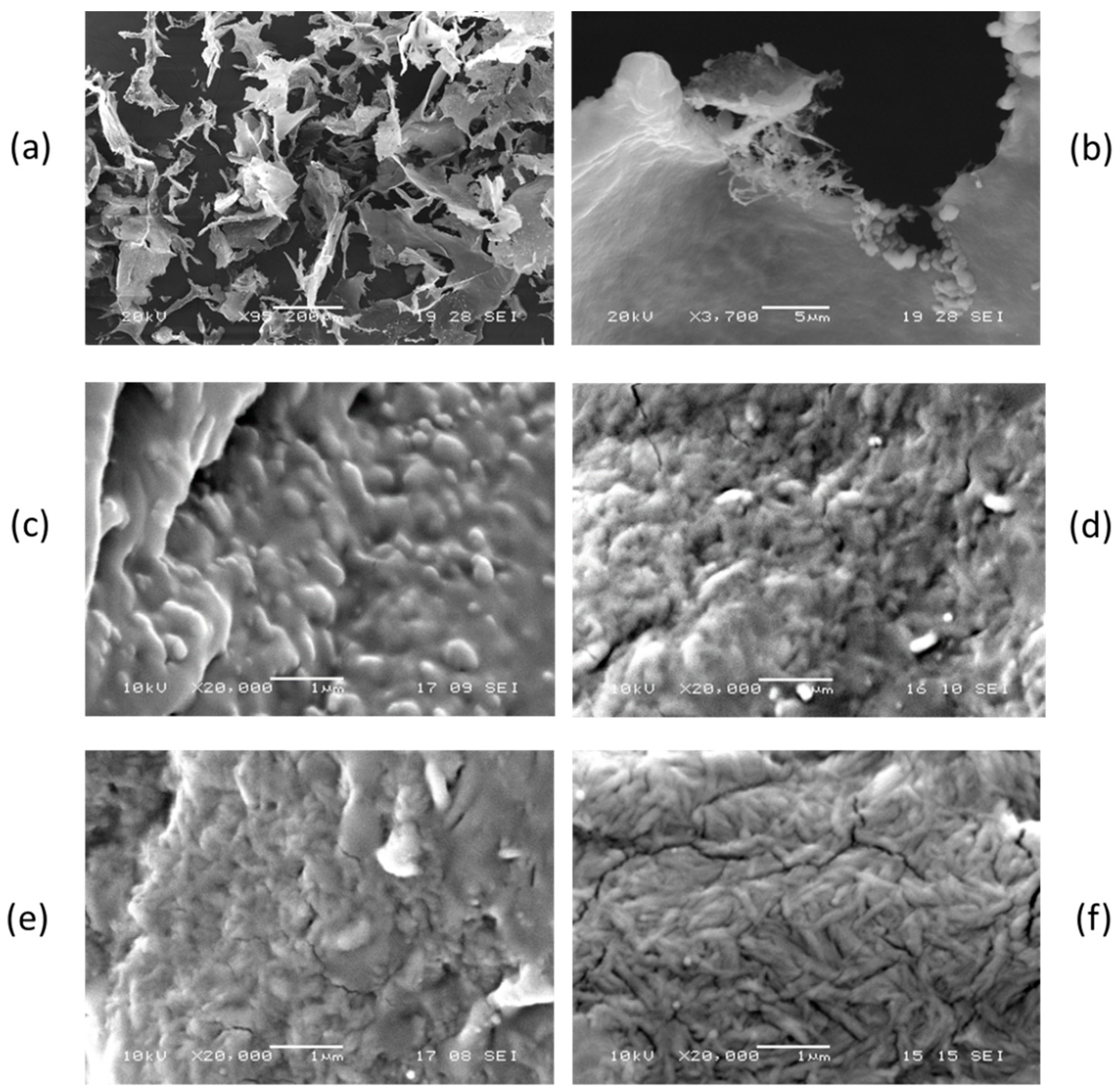
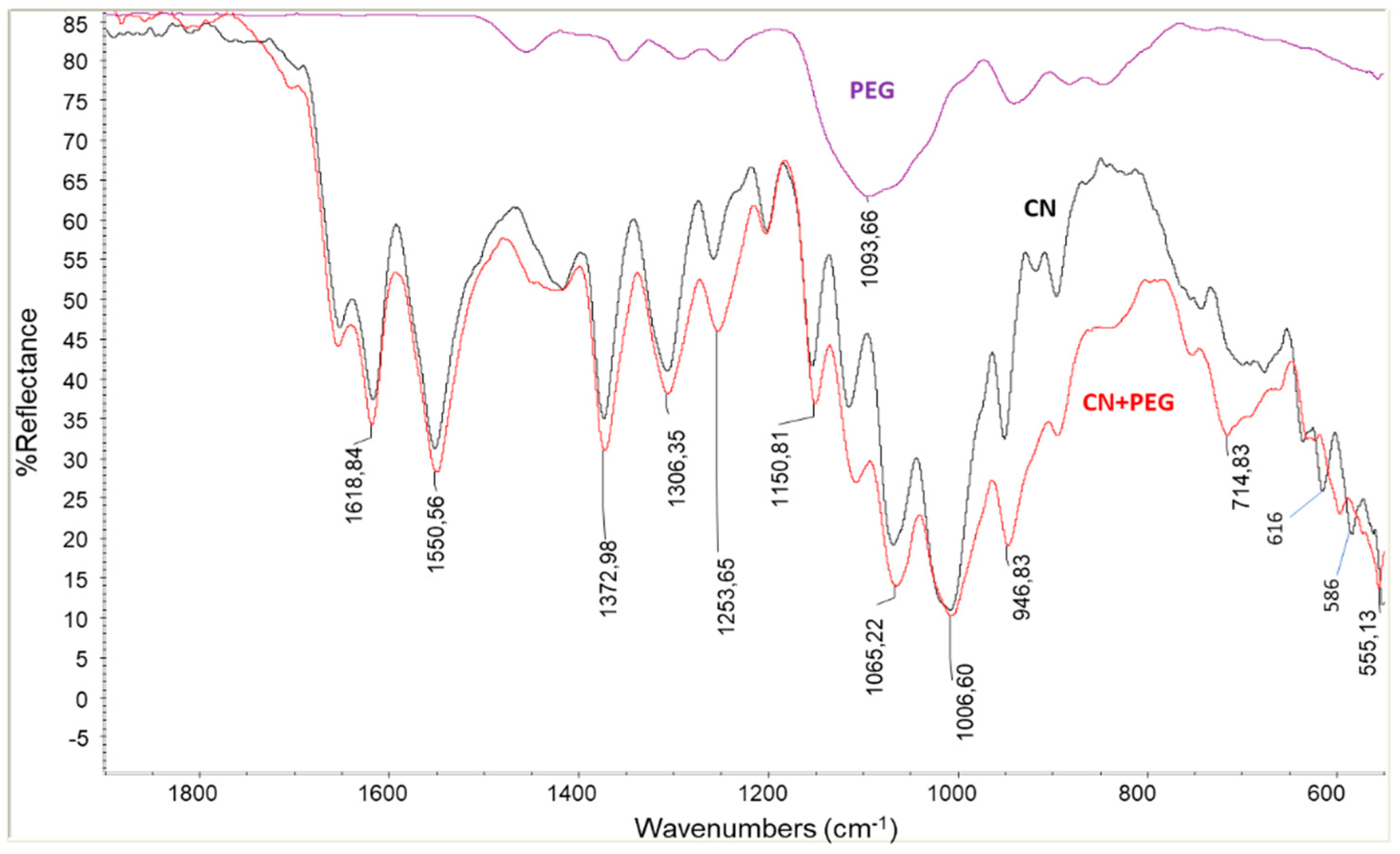
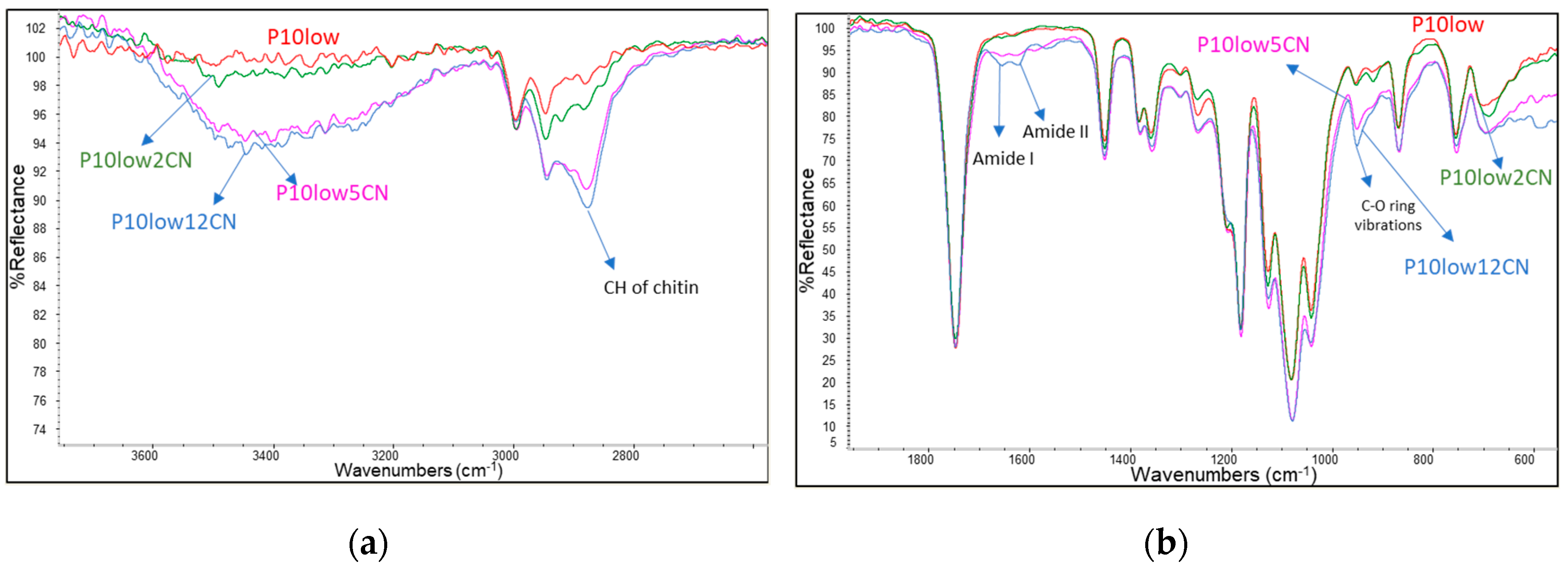
2.1. Dispersion of Chitin Nanofibrils in Plasticized Pla
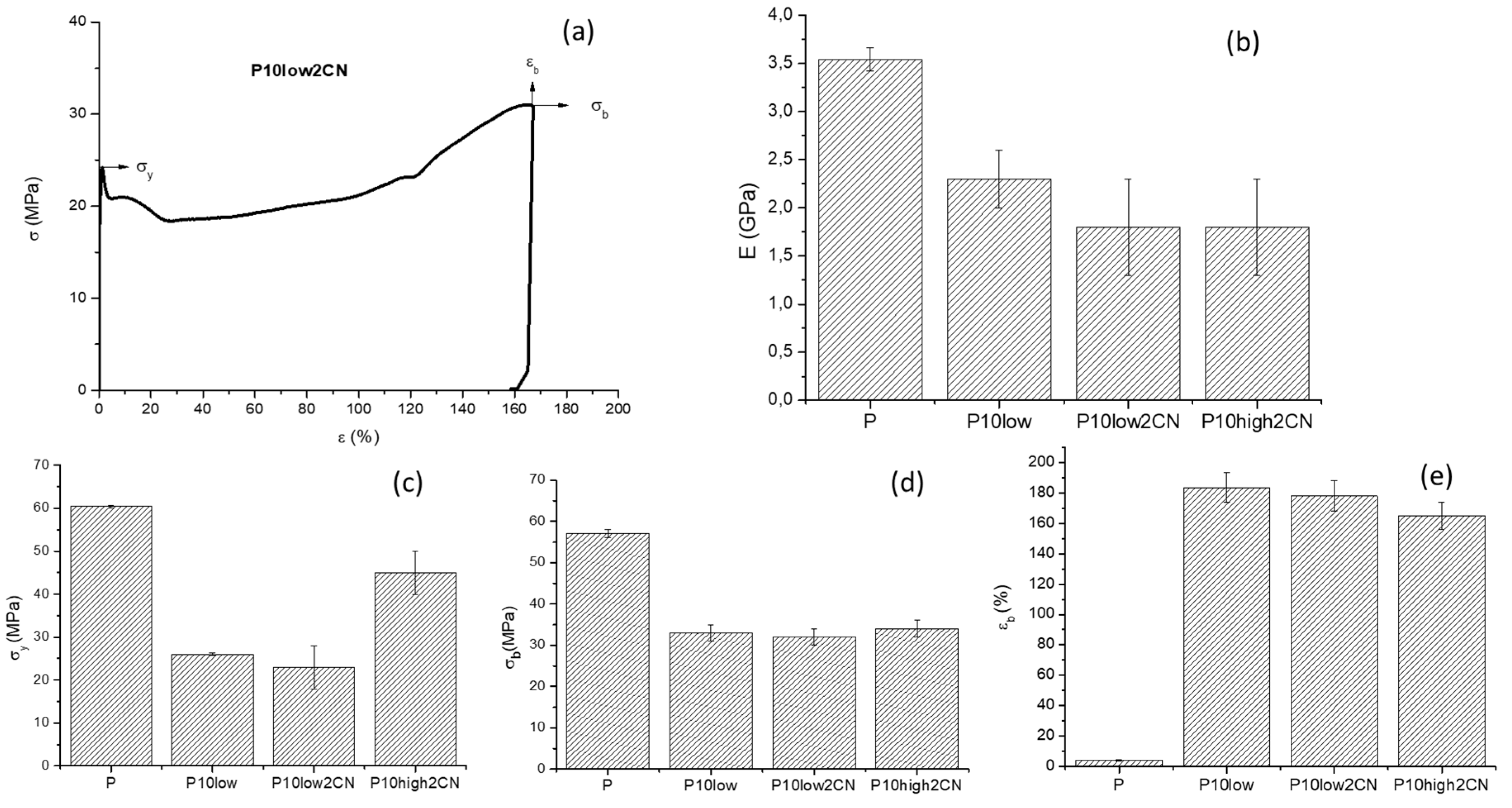
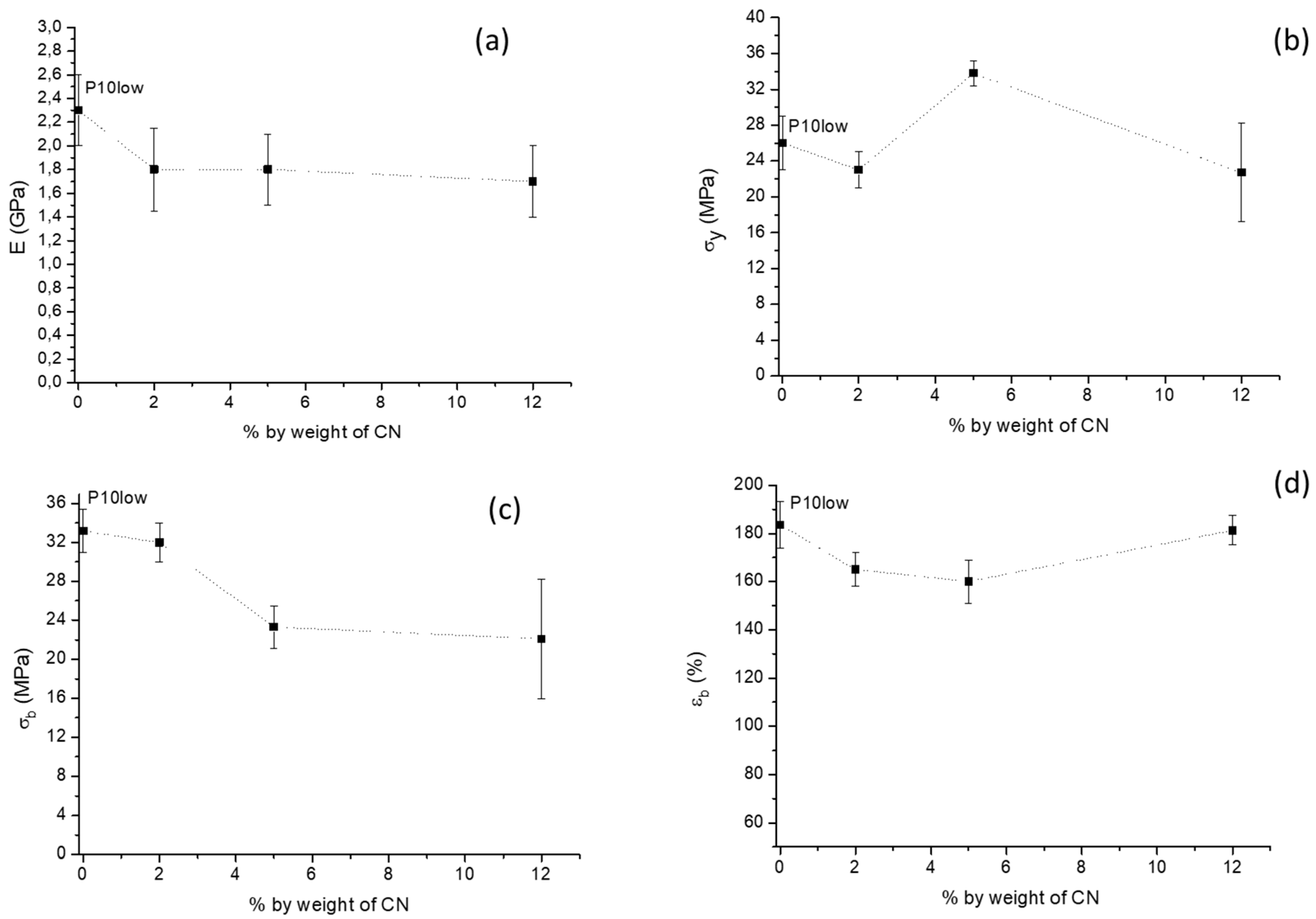
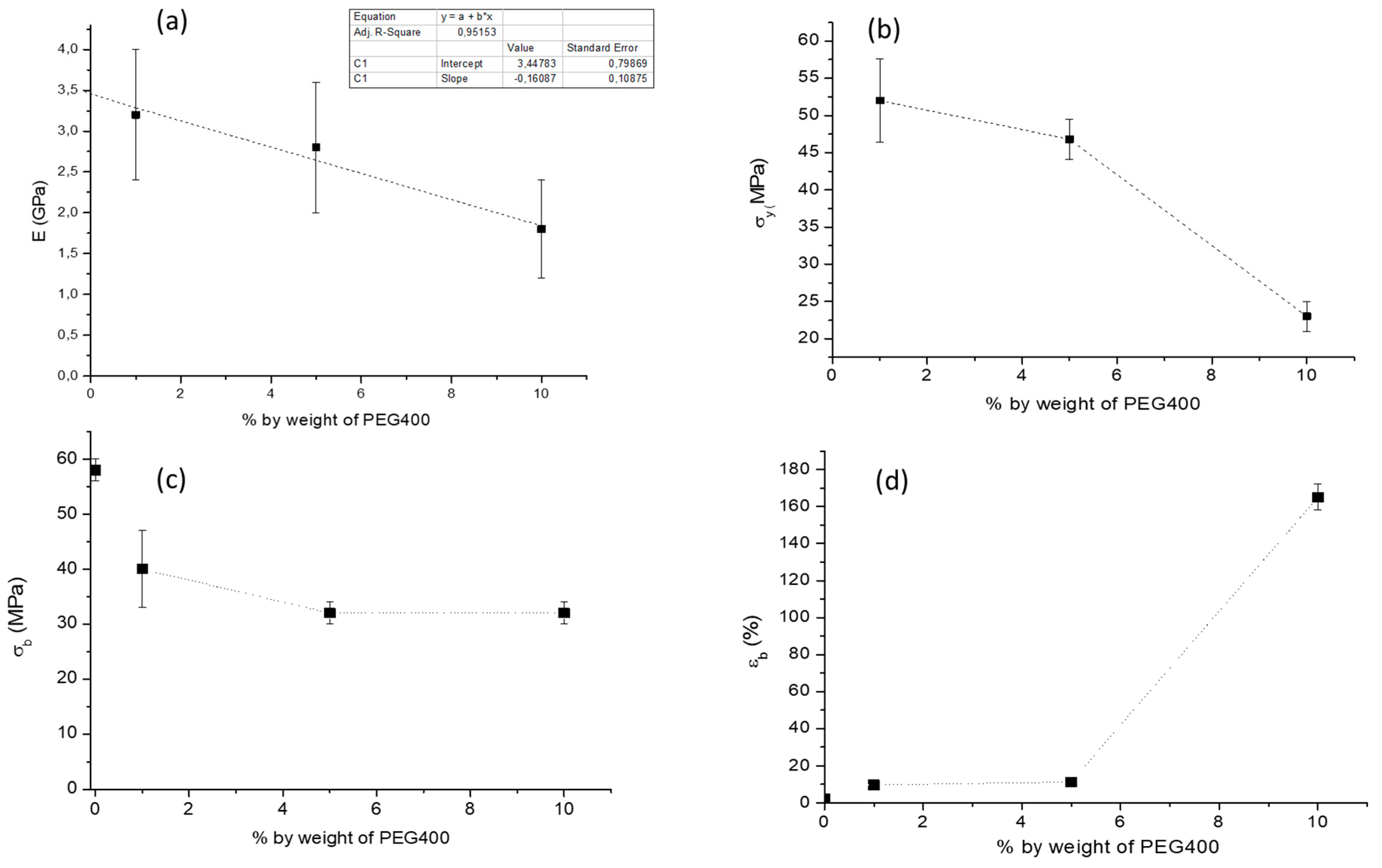
2.2. Tensile Properties
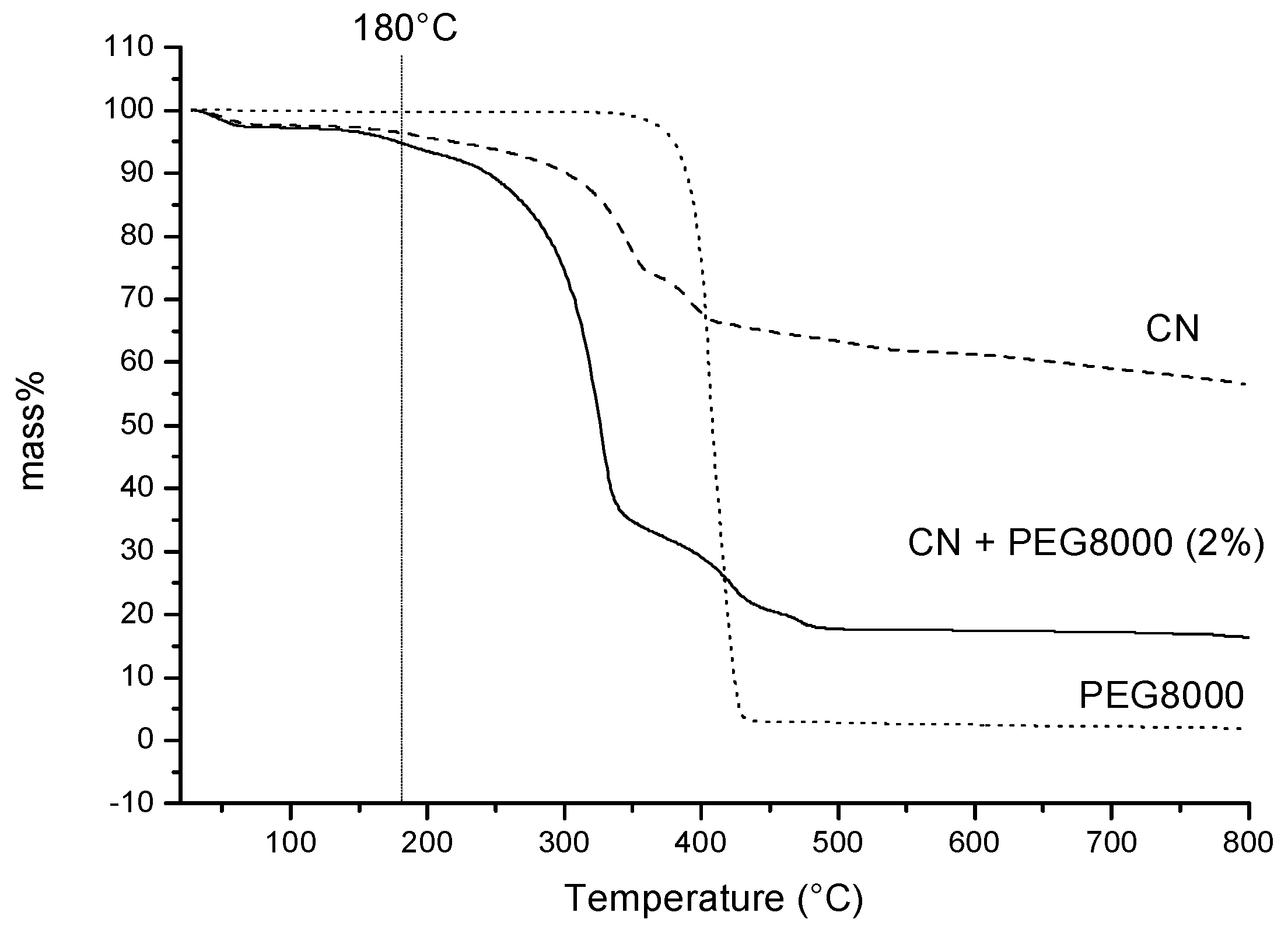
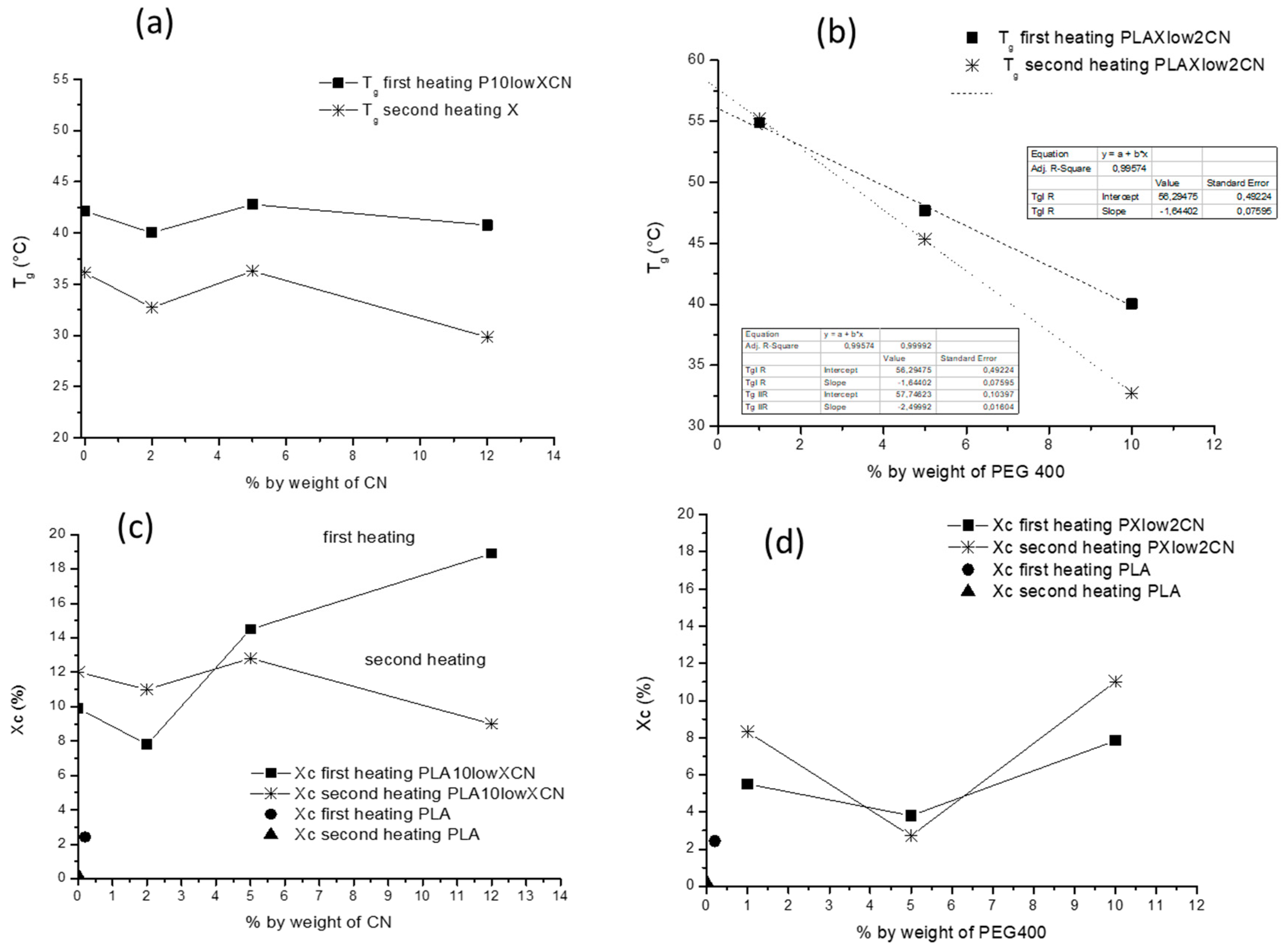
2.3. Thermal Properties
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Materials Preparation
4.3. Characterizations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, S.K. (Ed.) Chitin, Chitosan, Oligosaccharides and Their Derivatives, 1st ed.; CRC Press: Boca Raton, FL, USA, 2011; p. 241. [Google Scholar]
- Strelcova, Z.; Kulhanek, P.; Friak, M.; Fabritius, H.O.; Petrov, M.; Neugebauer, J.; Koca, J. The structure and dynamics of chitin nanofibrils in an aqueous environment revealed by molecular dynamics simulations. RSC Adv. 2016, 6, 30710–30721. [Google Scholar] [CrossRef]
- Fazeni, K.; Lindorfer, J. The Energy Institute at the Johannes Kepler University, 2011. Available online: http://www.energyefficiency.at/web/projekte/chibio-1-53-1-53-1-53-1-53.html (accessed on 28 February 2015).
- Nisticò, R. Aquatic-Derived Biomaterials for a Sustainable Future: A European Opportunity. Resources 2017, 6, 65. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [PubMed]
- Morganti, P.; Muzzarelli, C. Spray Dried Chitin Nanofibrils, Method for Production and Uses Thereof. U.S. Patent 2009/0203642, 13 August 2009. [Google Scholar]
- Gadgey, K.K.; Bahekar, A. Studies on extraction methods of chitin from crab shell and investigation of its mechanical properties. Int. J. Mech. Eng. Technol. 2017, 8, 220–231. [Google Scholar]
- Wu, J.; Zhang, K.; Girouard, N.; Carson Meredith, J. Facile Route to Produce Chitin Nanofibers as Precursors for Flexible and Transparent Gas Barrier Materials. Biomacromolecules 2014, 15, 4614–4620. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Chen, X.; Zhang, J.; Guo, W.; Jin, F.; Yan, N. Transformation of Chitin and Waste Shrimp Shells into Acetic Acid and Pyrrole. ACS Sustain. Chem. Eng. 2016, 4, 3912–3920. [Google Scholar] [CrossRef]
- De Azeredo, H.M.C. Antimicrobial nanostructures in food packaging. Trends Food Sci. Technol. 2013, 30, 56–69. [Google Scholar] [CrossRef]
- Tsai, G.-J.; Su, W.-H.; Chen, H.-C.; Pan, C.-L. Antimicrobial activity of shrimp chitin and chitosan from different treatments and applications of fish preservation. Fish. Sci. 2002, 68, 170–177. [Google Scholar] [CrossRef]
- Jayakumara, R.; Prabaharan, M.; Sudheesh Kumar, P.T.; Naira, S.V.; Tamura, H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol. Adv. 2011, 29, 322–337. [Google Scholar] [CrossRef]
- Garrison, T.F.; Murawski, A.; Quirino, R.L. Bio-Based Polymers with Potential for Biodegradability. Polymers 2016, 8, 262. [Google Scholar] [CrossRef]
- Pandey, A.; Chauhan, N.P.S.; Shabafrooz, V.; Ameta, R.; Mozafari, M. Polylactic acid and polyethylene glycol as antimicrobial agents. In Biocidal Polymers, 1st ed.; Chauhan, N.P.S., Ed.; Smither Rapra: Shawbury, UK, 2016; pp. 131–144. [Google Scholar]
- Rabnawaz, M.; Wyman, I.; Auras, R.; Cheng, S. A roadmap towards green packaging: The current status and future outlook for polyesters in the packaging industry. Green Chem. 2017, 19, 4737–4753. [Google Scholar] [CrossRef]
- Chen, Y.; Geever, L.M.; Killion, J.A.; Lyons, J.G.; Higginbotham, C.L.; Devine, D.M. Review of Multifarious Applications of Poly (Lactic Acid). Polym.-Plast. Technol. Eng. 2016, 55, 1057–1075. [Google Scholar] [CrossRef]
- Saini, P.; Arora, M.; Kumar, M.N.V.R. Poly(lactic acid) blends in biomedical applications. Adv. Drug Deliv. Rev. 2016, 107, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Y.; Zhang, H.; Song, M.L.; Zhou, Y.; Yao, J.; Ni, Q.Q. From Cellulose Nanospheres, Nanorods to Nanofibers: Various Aspect Ratio Induced Nucleation/Reinforcing Effects on Polylactic Acid for Robust-Barrier Food Packaging. ACS Appl. Mater. Interfaces 2017, 9, 43920–43938. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Nazarkovsky, M.; Ghadi, R.; Khan, W.; Domb, A.J. Poly(lactic acid)-based nanocomposites. Polym. Adv. Technol. 2017, 28, 919–930. [Google Scholar] [CrossRef]
- Scaffaro, R.; Botta, L.; Lopresti, F.; Maio, A.; Sutera, F. Polysaccharide nanocrystals as fillers for PLA based nanocomposites. Cellulose 2017, 24, 447–478. [Google Scholar] [CrossRef]
- Jonoobi, M.; Harun, J.; Mathew, A.P.; Oksman, K. Mechanical properties of cellulose nanofiber (CNF) reinforced polylactic acid (PLA) prepared by twin screw extrusion. Compos. Sci. Technol. 2010, 70, 1742–1747. [Google Scholar] [CrossRef]
- Trifol Guzman, J.; Plackett, D.; Sillard, C.; Szabo, P.; Bras, J.; Daugaard, A.E. Hybrid poly(lactic acid)/nanocellulose/nanoclay composites with synergistically enhanced barrier properties and improved thermomechanical resistance. Polym. Int. 2016, 65, 988–995. [Google Scholar] [CrossRef]
- Nagarajan, V.; Mohanty, A.K.; Misra, M. Perspective on Polylactic Acid (PLA) based Sustainable Materials for Durable Applications: Focus on Toughness and Heat Resistance. ACS Sustain. Chem. Eng. 2016, 4, 2899–2916. [Google Scholar] [CrossRef]
- Fan, Y.M.; Saito, T.; Isogai, A. Preparation of chitin nanofibers from squid pen beta-chitin by simple mechanical treatment under acid conditions. Biomacromolecules 2008, 9, 1919–1923. [Google Scholar] [CrossRef]
- Nakagaito, A.N.; Kanzawa, S.; Takagi, H. Polylactic Acid Reinforced with Mixed Cellulose and Chitin Nanofibers—Effect of Mixture Ratio on the Mechanical Properties of Composites. J. Compos. Sci. 2018, 2, 36. [Google Scholar] [CrossRef]
- Araki, J. Preparation of Sterically Stabilized Chitin Nanowhisker Dispersions by Grafting of Poly(ethylene glycol) and Evaluation of Their Dispersion Stability. Biomacromolecules 2015, 16, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Araki, J.; Wada, M.; Kuga, S. Steric Stabilization of a Cellulose Microcrystal Suspension by Poly(ethylene glycol) Grafting. Langmuir 2001, 17, 21–27. [Google Scholar] [CrossRef]
- Araki, J.; Kuga, S.; Magoshi, J. Influence of reagent addition on carbodiimide-mediated amidation for poly(ethylene glycol) grafting. J. Appl. Polym. Sci. 2002, 85, 1349–1352. [Google Scholar] [CrossRef]
- Zhang, Q.; Wei, S.; Huang, J.; Feng, J.; Chang, P.R. Effect of Surface Acetylated-Chitin Nanocrystals on Structure and Mechanical Properties of Poly (lactic acid). J. Appl. Polym. Sci. 2014, 131, 39809. [Google Scholar] [CrossRef]
- Rizvi, R.; Cochrane, B.; Naguib, H.; Lee, P.C. Fabrication and characterization of melt-blended polylactide-chitin composites and their foams. J. Cell. Plast. 2011, 47, 283–300. [Google Scholar] [CrossRef]
- Herrera, N.; Singh, A.A.; Salaberria, A.M.; Labidi, J.; Mathew, A.P.; Oksman, K. Triethyl Citrate (TEC) as a Dispersing Aid in Polylactic Acid/Chitin Nanocomposites Prepared via Liquid-Assisted Extrusion. Polymers 2017, 9, 406. [Google Scholar] [CrossRef]
- Guan, Q.; Naguib, H.E. Fabrication and Characterization of PLA/PHBV-Chitin Nanocomposites and Their Foams. J. Polym. Environ. 2014, 22, 119–130. [Google Scholar] [CrossRef]
- Herrera, N.; Salaberria, A.M.; Mathew, A.P.; Oksman, K. Plasticized polylactic acid nanocomposite films with cellulose and chitin nanocrystals prepared using extrusion and compression molding with two cooling rates: Effects on mechanical, thermal and optical properties. Compos. Part A 2016, 83, 89–97. [Google Scholar] [CrossRef]
- Herrera, N.; Roch, H.; Salaberria, A.M.; Pino-Orellana, M.A.; Labidi, J.; Fernandes, S.C.M.; Radic, D.; Leiva, A.; Oksman, K. Functionalized blown films of plasticized polylactic acid/chitin nanocomposite: Preparation and characterization. Mater. Des. 2016, 92, 846–852. [Google Scholar] [CrossRef]
- Fehri, M.K.; Mugoni, C.; Cinelli, P.; Anguillesi, I.; Coltelli, M.B.; Fiori, S.; Montorsi, M.; Lazzeri, A. Composition dependence of the synergistic effect of nucleating agent and plasticizer in poly(lactic acid): A Mixture Design study. eXPRESS Polym. Lett. 2016, 10, 274–288. [Google Scholar] [CrossRef]
- Coltelli, M.B.; Della Maggiore, I.; Bertoldo, M.; Signori, F.; Bronco, S.; Ciardelli, F. Poly(lactic acid) Properties as a Consequence of Poly(butylene adipate-co-terephthalate) Blending and Acetyl Tributyl Citrate Plasticization. J. Appl. Polym. Sci. 2008, 110, 1250–1262. [Google Scholar] [CrossRef]
- Quero, E.; Müller, A.J.; Signori, F.; Coltelli, M.B.; Bronco, S. Isothermal Cold-Crystallization of PLA/PBAT Blends with and without the Addition of Acetyl Tributyl Citrate. Macromol. Chem. Phys. 2012, 213, 36–48. [Google Scholar] [CrossRef]
- Scatto, M.; Salmini, E.; Castiello, S.; Coltelli, M.B.; Conzatti, L.; Stagnaro, P.; Andreotti, L.; Bronco, S. Plasticized and nanofilled poly(lactic acid)-based cast films: Effect of plasticizer and organoclay on processability and final properties. J. Appl. Polym. Sci. 2013, 127, 4947–4956. [Google Scholar] [CrossRef]
- Coltelli, M.B.; Cinelli, P.; Anguillesi, I.; Salvadori, S.; Lazzeri, A. Structure and properties of extruded composites based on bio-polyesters and nano-chitin, Session M: Functional Textiles-from research and development to innovations and industrial uptake. In Proceedings of the Symposium E-MRS Fall Meeting, Warsaw University of Technology, Warsaw, Poland, 15–18 September 2014; p. 192. [Google Scholar]
- Cinelli, P.; Coltelli, M.B.; Mallegni, N.; Morganti, P.; Lazzeri, A. Degradability and sustainability of nanocomposites based on polylactic acid and chitin nano fibrils. Chem. Eng. Trans. 2017, 60, 1–6. [Google Scholar]
- Coltelli, M.B.; Gigante, V.; Panariello, L.; Aliotta, L.; Morganti, P.; Danti, S.; Cinelli, P.; Lazzeri, A. Chitin nanofibrils in renewable materials for packaging and personal care applications. Adv. Mater. Lett. 2019, in press. [Google Scholar]
- Li, J.; Gao, Y.; Zhao, J.; Sun, J.; Li, D. Homogeneous dispersion of chitin nanofibers in polylactic acid with different pretreatment methods. Cellulose 2017, 24, 1705–1715. [Google Scholar] [CrossRef]
- Nakagaito, A.N.; Yamada, K.; Ifuku, S.; Morimoto, M.; Saimoto, H. Fabrication of Chitin Nanofiber-Reinforced Polylactic Acid Nanocomposites by an Environmentally Friendly Process. J. Biobased Mater. Bioenergy 2013, 7, 152–156. [Google Scholar] [CrossRef]
- Shamshina, J.L.; Zavgorodnya, O.; Berton, P.; Chhotaray, P.K.; Choudhary, H.; Rogers, R.D. An Ionic Liquid Platform for Spinning Composite Chitin-Poly(lactic acid) Fibers. ACS Sustain. Chem. Eng. 2018, 6, 10241–10251. [Google Scholar] [CrossRef]
- Jolanta Kumirska, J.; Czerwicka, M.; Kaczyński, Z.; Bychowska, A.; Brzozowski, K.; Thöming, J.; Stepnowski, P. Application of Spectroscopic Methods for Structural Analysis of Chitin and Chitosan. Mar. Drugs 2010, 8, 1567–1636. [Google Scholar] [CrossRef]
- Pearson, F.G.; Marchessault, R.H.; Liang, C.Y. Infrared spectra of crystalline polysaccharides. V. Chitin. J. Polym. Sci. 1960, 13, 101–116. [Google Scholar] [CrossRef]
- Brunner, E.; Ehrlich, H.; Schupp, P.; Hedrich, R.; Hunoldt, S.; Kammer, M.; Machill, S.; Paasch, S.; Bazhenov, V.V.; Kurek, D.V.; et al. Chitin-based scaffolds are an integral part of the skeleton of the marine demosponge Ianthella basta. J. Struct. Biol. 2009, 168, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Focher, B.; Naggi, A.; Torri, G.; Cosani, A.; Terbojevich, M. Structural differences between chitin polymorphs and their precipitates from solutions-evidence from CP-MAS 13CNMR, FT-IR and FT-Raman spectroscopy. Carbohydr. Polym. 1992, 17, 97–102. [Google Scholar] [CrossRef]
- Cheng, D.; Wen, Y.; Wang, L.; An, X.; Zhua, X.; Ni, Y. Adsorption of polyethylene glycol (PEG) onto cellulose nano-crystals to improve its dispersity. Carbohydr. Polym. 2015, 123, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Paul, D.R.; Robeson, L.M. Polymer nanotechnology: Nanocomposites. Polymer 2008, 49, 3187–3204. [Google Scholar] [CrossRef]
- Biswas, S.K.; Shams, M.I.; Das, A.K.; Islam, M.N.; Nazhad, M.M. Flexible and Transparent Chitin/Acrylic Nanocomposite Films with High Mechanical Strength. Fibers Polym. 2015, 16, 774–781. [Google Scholar] [CrossRef]
- Mikesová, J.; Hasek, J.; Tishchenko, G.; Morganti, P. Rheological study of chitosan acetate solutions containing chitin nanofibrils. Carbohydr. Polym. 2014, 112, 753–757. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Wang, H.; Jiang, L.; Wang, X.; Payne, S.A.; Zhu, J.Y.; Li, R. Comparison between Cellulose Nanocrystal and Cellulose Nanofibril Reinforced Poly(ethylene oxide) Nanofibers and Their Novel Shish-Kebab-Like Crystalline Structures. Macromolecules 2014, 47, 3409–3416. [Google Scholar] [CrossRef]
- Cardinaels, R.; Moldenaers, P. Morphology development in immiscible polymer blends. In Polymer Morphology. Principles, Characterization and Properties; Guo, Q., Ed.; John Wiley and Sons: Hoboken, NJ, USA, 2016; Chapter 19; pp. 348–373. [Google Scholar]
- Potschke, P.; Paul, D.R. Formation of co-continuous structures in met-mixed immiscible polymer blends. J. Macromol. Sci. C Polym. Rev. 2003, 43, 87–141. [Google Scholar] [CrossRef]
- Muzzarelli, C.; Morganti, P. Preparation of Chitin and Derivatives Thereof for Cosmetic and Therapeutic Use. U.S. Patent 8383157 B2, 26 February 2013. [Google Scholar]





| Samples | PEG (%) | MW of PEG | CN |
|---|---|---|---|
| P | − | − | − |
| P2CN* a | − | − | 2 |
| P10low | 10 | 400 | 0 |
| P10low2CN | 10 | 400 | 2 |
| P10high2CN | 10 | 8000 | 2 |
| P1low2CN | 1 | 400 | 2 |
| P5low2CN | 5 | 400 | 2 |
| P10low5CN | 10 | 400 | 5 |
| P10low12CN | 10 | 400 | 12 |
| Samples | E (GPa) | σy (MPa) | σb (MPa) | εb (%) |
|---|---|---|---|---|
| P | 3.5 ± 0.1 | 60.4 ± 0.3 | 57 ± 1 | 4.1± 0.5 |
| P2CN* a | 2.9 ± 0.1 | − | 58 ± 2 | 2.3 ± 0.4 |
| P10low | 2.3 ± 0.3 | 26 ± 0.3 | 33 ± 2 | 180± 10 |
| P10low2CN | 1.8 ± 0.3 | 23 ± 5 | 32 ± 2 | 160 ± 10 |
| P10high2CN | 2.5 ± 0.1 | 45 ± 5 | 34 ± 2 | 160 ± 10 |
| P1low2CN | 3.2 ± 0.8 | 52 ± 6 | 40 ± 7 | 10 ± 2 |
| P5low2CN | 2.8 ± 0.8 | 47 ± 3 | 32 ± 2 | 11.4 ± 0.9 |
| P10low5CN | 1.8 ± 0.3 | 34 ± 2 | 23 ± 2 | 160 ± 10 |
| P10low12CN | 1.7 ± 0.3 | 23 ± 5 | 22 ± 6 | 181 ± 6 |
| Tg (°C) | Tcc (°C) | ΔHcc (J/g) | Tm (°C) | ΔHm (J/g) | Xc (%) | |
|---|---|---|---|---|---|---|
| P | 57.7 | 106.9 | 21.24 | 149.2/157.4 | 21.4 | 0.2 |
| P2CN* | 57.5 | 107.7 | 23.21 | 148.8/155.9 | 25.5 | 2.4 |
| P10low | 42.1 | 77.6 | 17.8 | 154.7 | 27.1 | 9.9 |
| P10low2CN | 40.0 | 74.4 | 18.3 | 152.1 | 26.6 | 7.8 |
| P10high2CN | 43.8 | 76.6 | 6.7 | 154.7 | 33.3 | 28.4 |
| P1low2CN | 54.9 | 100.9 | 18.6 | 147.9/157.9 | 23.7 | 5.5 |
| P5low2CN | 47.7 | 88.6 | 15.6 | (142.3)/155.9 | 19.2 | 3.8 |
| P10low5CN | 42.8 | 74.1 | 15.5 | 154.2 | 29.0 | 14.5 |
| P10low12CN | 40.8 | 75.5 | 11.4 | 152.3 | 29.1 | 19.0 |
| Tg (°C) | Tcc (°C) | ΔHcc (J/g) | Tm (°C) | ΔHm (J/g) | Xc (%) | |
|---|---|---|---|---|---|---|
| P | 59.1 | 109.4 | 23.8 | 148.3/157.4 | 26.3 | 2.8 |
| P2CN* | 55.2 | 108.8 | 23.3 | 147.9/155.8 | 24.5 | 1.3 |
| P10low | 36.1 | 83.8 | 21.3 | 153.7 | 32.6 | 12.1 |
| P10low2CN | 32.7 | 79.8 | 17.1 | 152.1 | 29.2 | 13.0 |
| P10high2CN | 31.4 | 80.5 | 17.5 | 154.2 | 29.3 | 12.7 |
| P1low2CN | 55.2 | 104.6 | 22.2 | 147.2/156.7 | 30.2 | 8.3 |
| P5low2CN | 45.3 | 92.4 | 24.6 | 141.4/157.4 | 27.2 | 2.7 |
| P10low5CN | 36.3 | 78.8 | 19.1 | 153.6 | 31.1 | 12.8 |
| P10low12CN | 29.8 | 73.3 | 20.4 | 151.6 | 28.8 | 9.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coltelli, M.-B.; Cinelli, P.; Gigante, V.; Aliotta, L.; Morganti, P.; Panariello, L.; Lazzeri, A. Chitin Nanofibrils in Poly(Lactic Acid) (PLA) Nanocomposites: Dispersion and Thermo-Mechanical Properties. Int. J. Mol. Sci. 2019, 20, 504. https://doi.org/10.3390/ijms20030504
Coltelli M-B, Cinelli P, Gigante V, Aliotta L, Morganti P, Panariello L, Lazzeri A. Chitin Nanofibrils in Poly(Lactic Acid) (PLA) Nanocomposites: Dispersion and Thermo-Mechanical Properties. International Journal of Molecular Sciences. 2019; 20(3):504. https://doi.org/10.3390/ijms20030504
Chicago/Turabian StyleColtelli, Maria-Beatrice, Patrizia Cinelli, Vito Gigante, Laura Aliotta, Pierfrancesco Morganti, Luca Panariello, and Andrea Lazzeri. 2019. "Chitin Nanofibrils in Poly(Lactic Acid) (PLA) Nanocomposites: Dispersion and Thermo-Mechanical Properties" International Journal of Molecular Sciences 20, no. 3: 504. https://doi.org/10.3390/ijms20030504
APA StyleColtelli, M.-B., Cinelli, P., Gigante, V., Aliotta, L., Morganti, P., Panariello, L., & Lazzeri, A. (2019). Chitin Nanofibrils in Poly(Lactic Acid) (PLA) Nanocomposites: Dispersion and Thermo-Mechanical Properties. International Journal of Molecular Sciences, 20(3), 504. https://doi.org/10.3390/ijms20030504












