Expression and Functional Analysis of Hepcidin from Mandarin Fish (Siniperca chuatsi)
Abstract
1. Introduction
2. Results
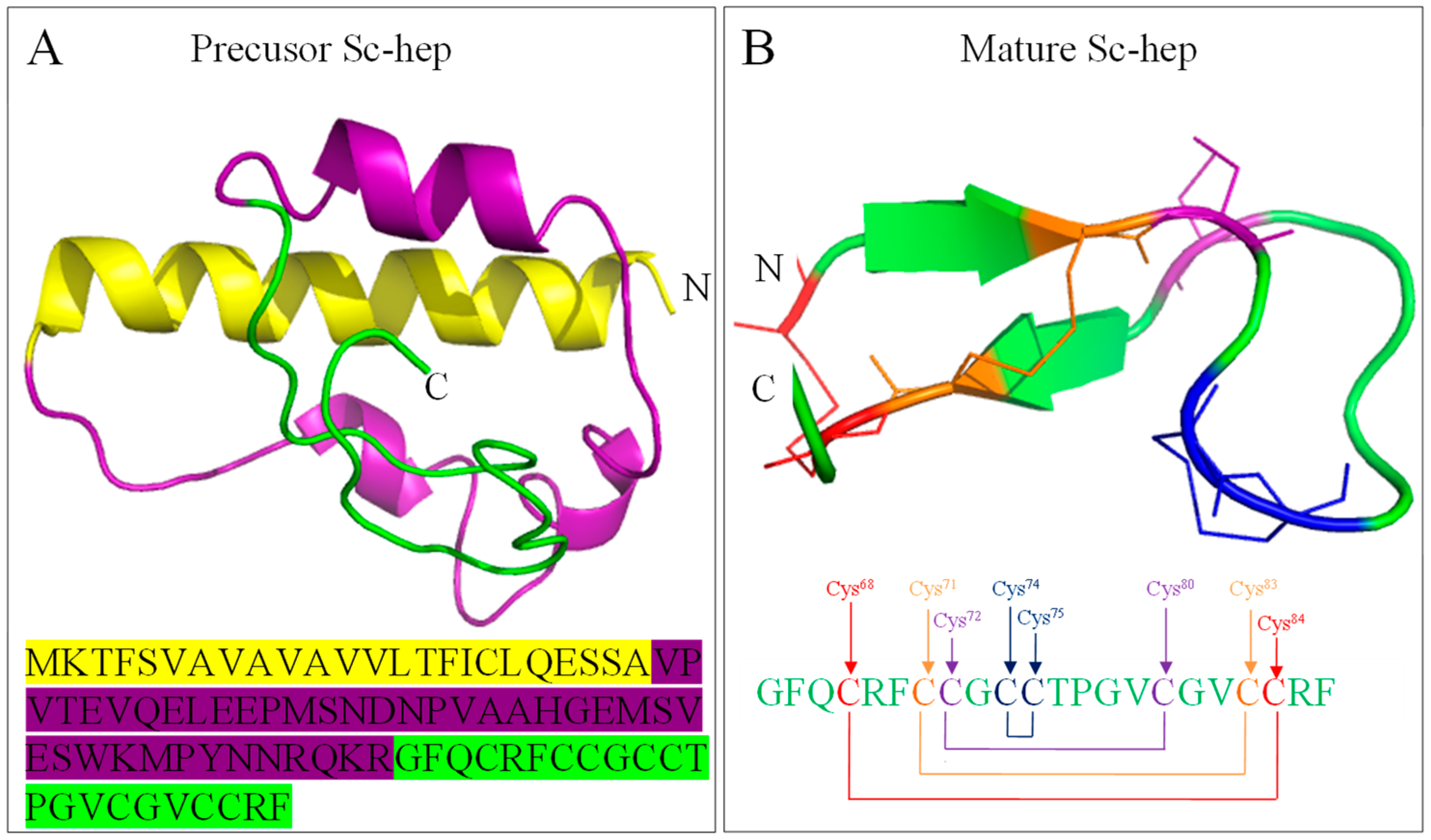
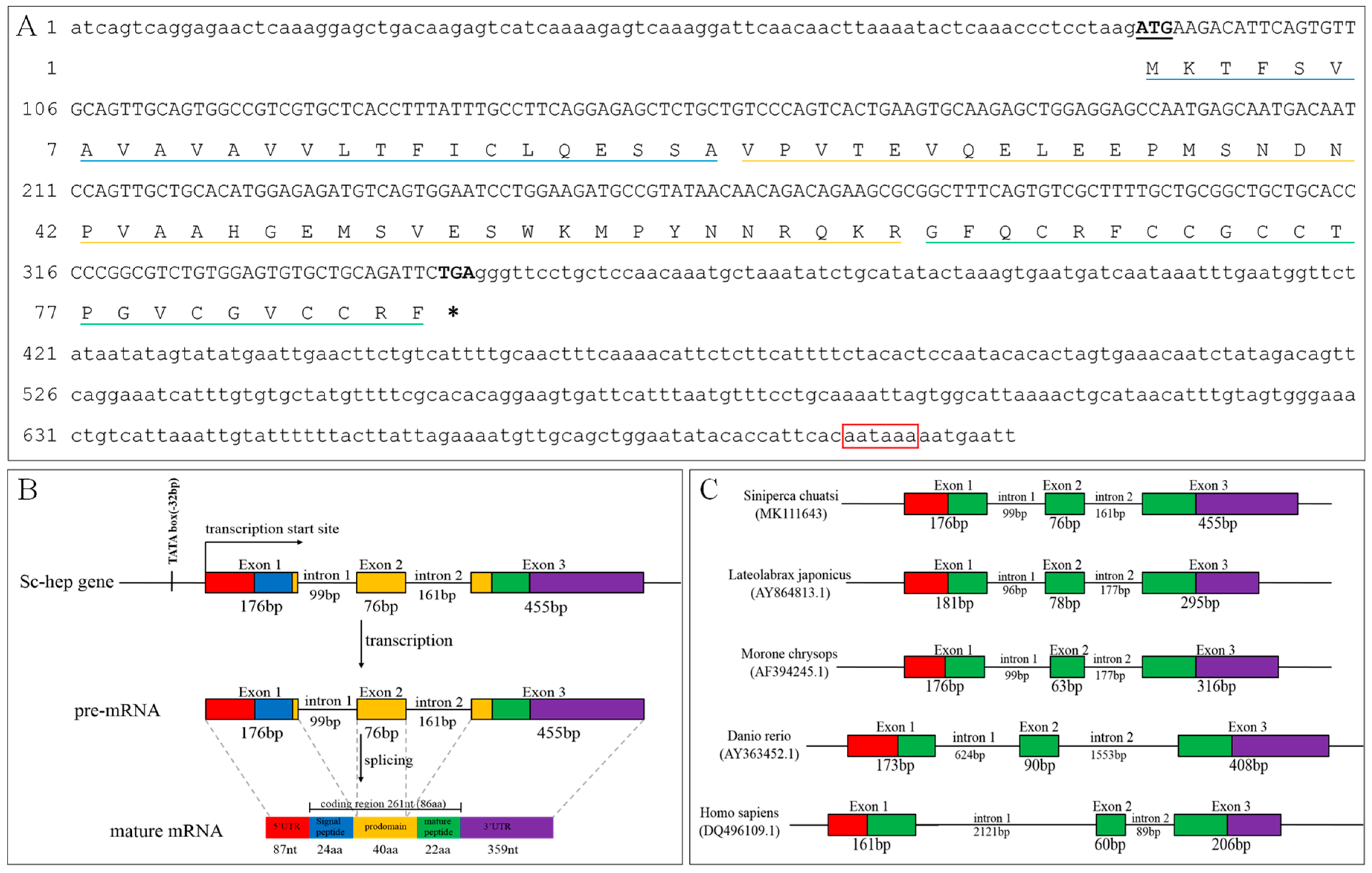
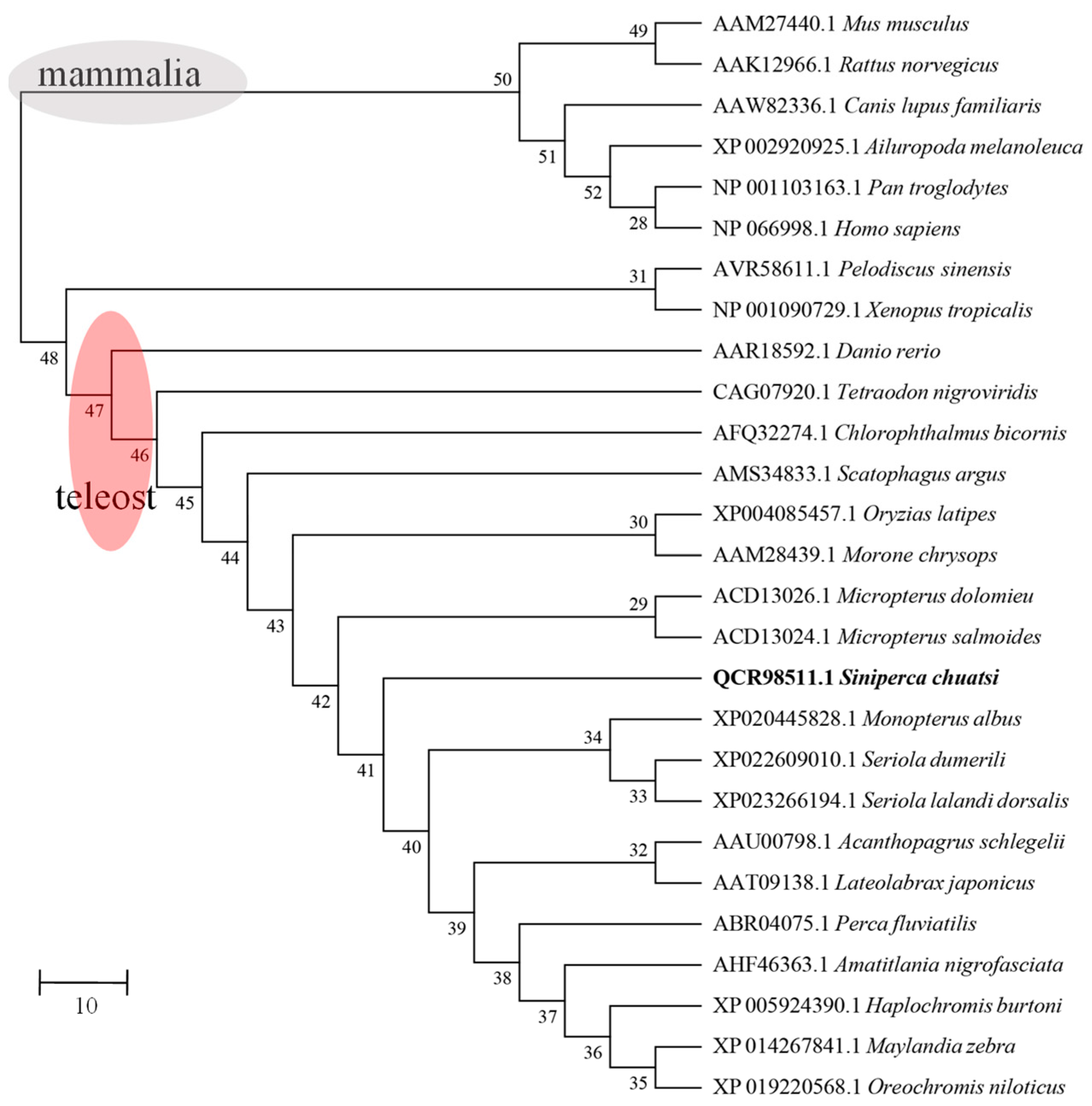
2.1. Sequence Analysis of Sc-Hep
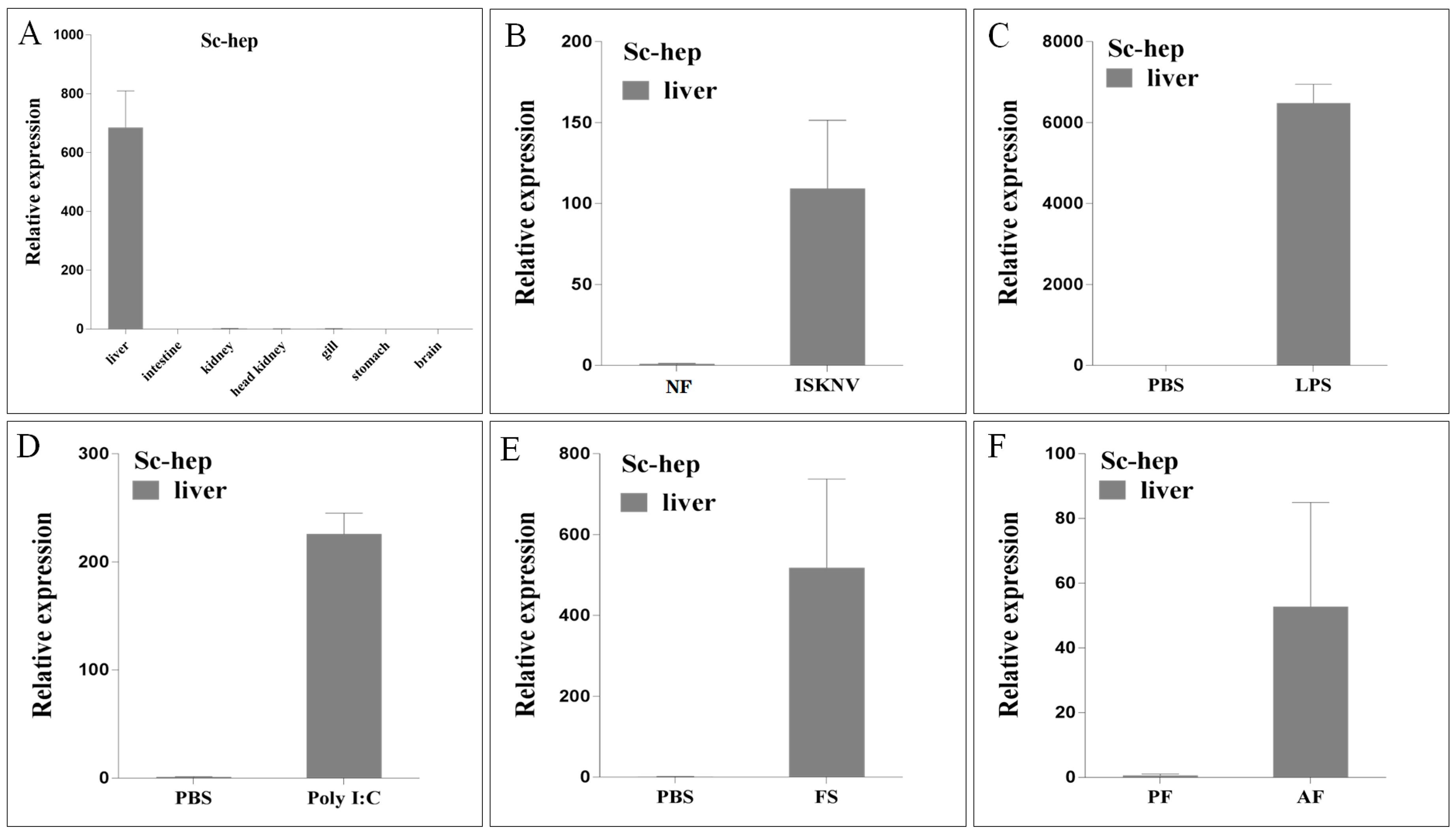
2.2. Sc-Hep Expression
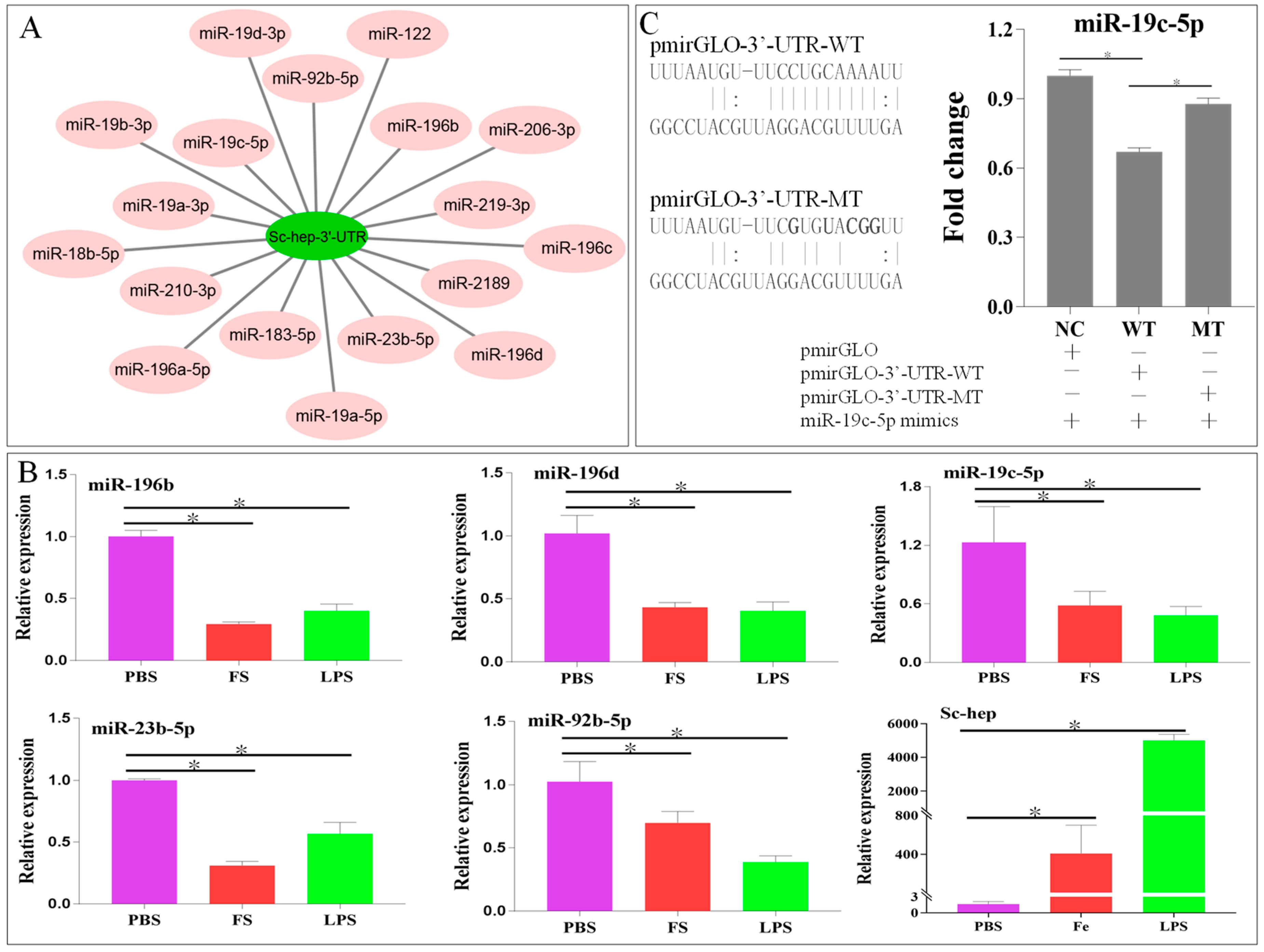
2.3. Relationship Between Sc-Hep and miR-19c-5p
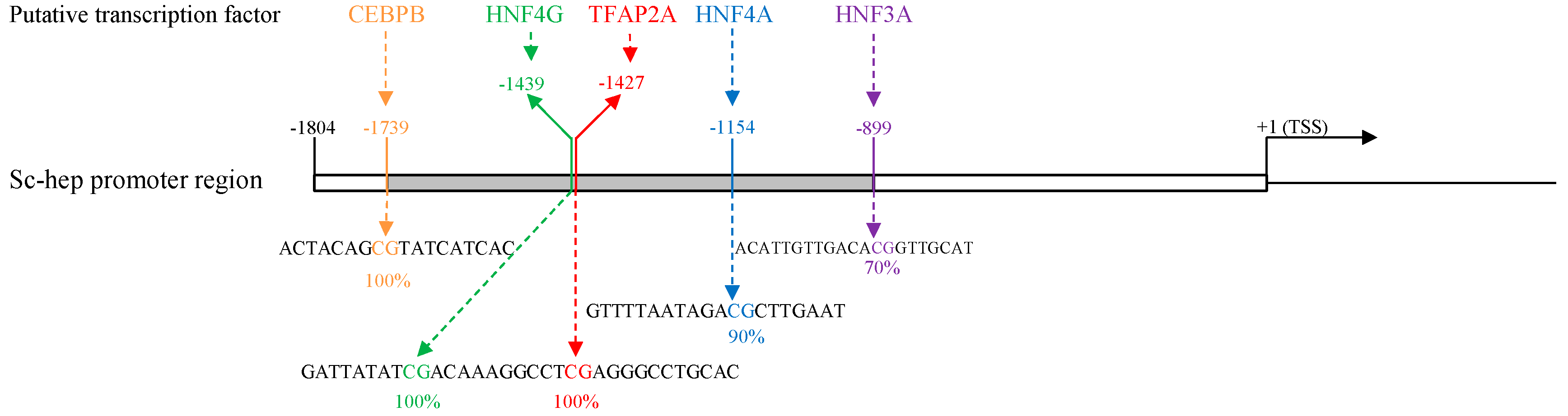
2.4. Sc-Hep DNA Methylation Analysis
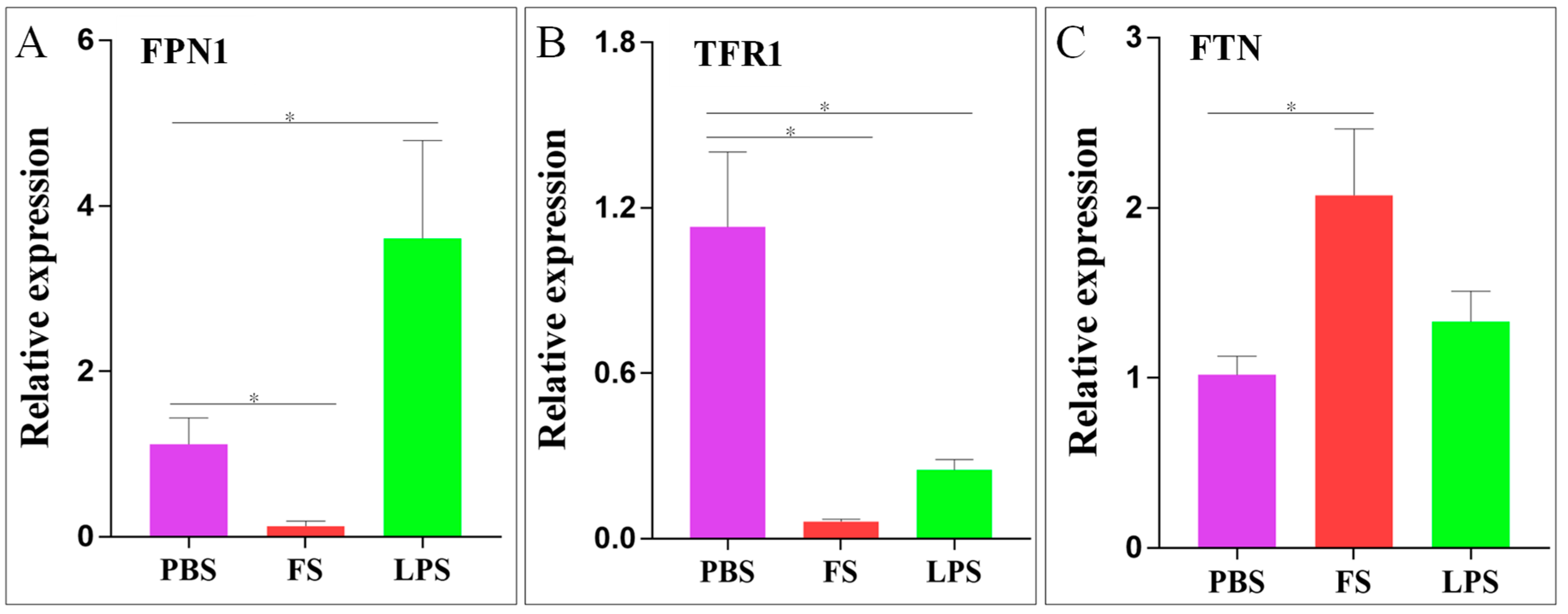
2.5. Expression of Iron Balance-Related Genes
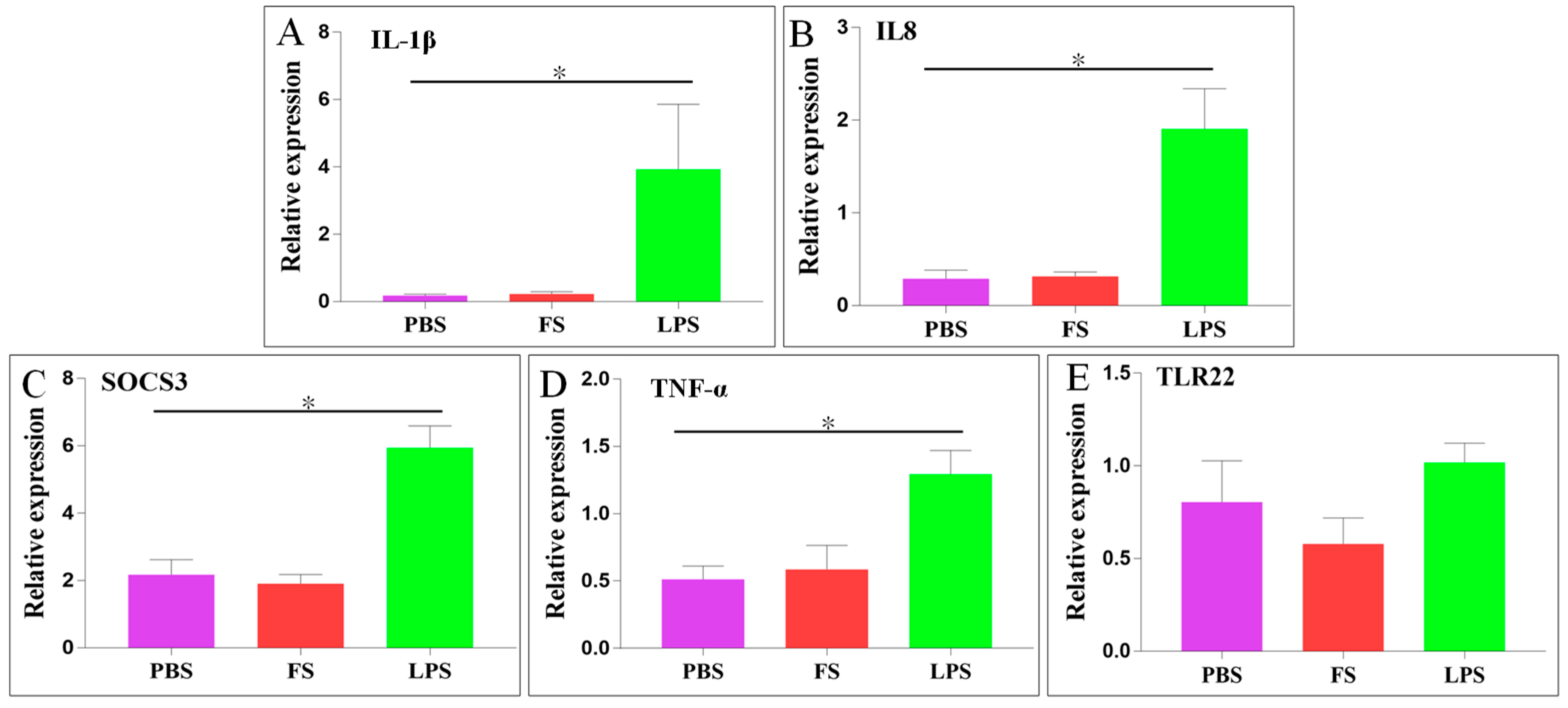
2.6. Expression of Immune-Related Genes
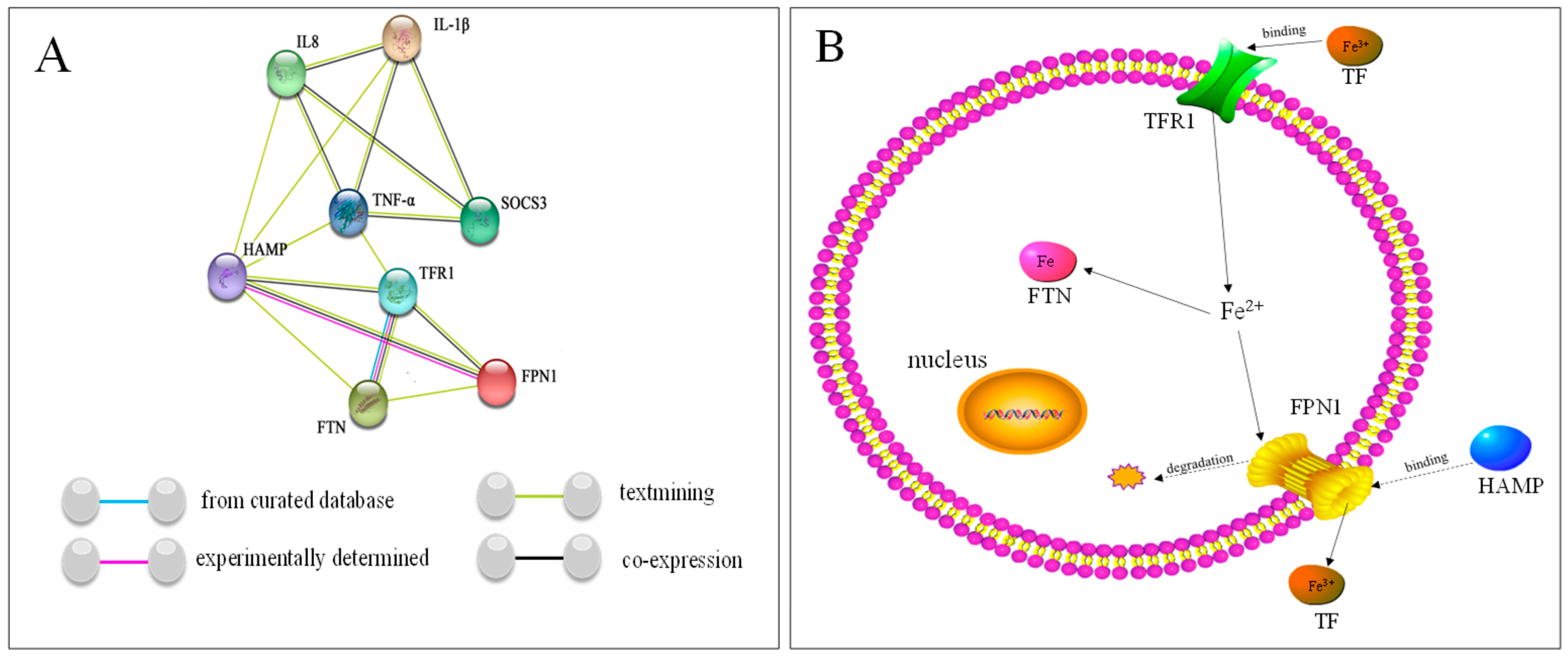
2.7. Analysis of Protein–Protein Interactions and Iron Balance Mechanism
3. Discussion
4. Materials and Methods
4.1. Fish and Sample Preparation
4.2. Total RNA Extraction and cDNA Synthesis
4.3. DNA Extraction and Identification of Sc-Hep DNA Sequences
4.4. Analysis of Sc-Hep Gene And Deduced Protein Sequence
4.5. qPCR Analysis of the Sc-Hep Gene
4.6. DNA Methylation Analysis of the Sc-Hep Promoter
4.7. qPCR Analysis of miRNA and Dual-Luciferase Reporter Assay
4.8. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Sc-hep | Siniperca chuatsi hepcidin |
| ISKNV | infectious spleen and kidney necrosis virus |
| LPS | lipopolysaccharide |
| Poly I:C | Polyinosinic–polycytidylic acid |
| FS | ferrous sulfate |
| PBS | phosphate buffered saline |
| PF | cultured with the live prey fish |
| AF | fed with artificially domesticated fish |
| BLM | basolateral membrane |
| HAMP | hepcidin |
| FPN1 | ferroportin 1 |
| TFR1 | transferrin receptor 1 |
| TF | transferrin |
| FTN | ferritin |
| IL-1β | interleukin-1 beta |
| IL8 | interleukin-8 |
| SOCS3 | suppressor of cytokine signaling 3 |
| TNF-α | tumor necrosis factor-alpha |
| TLR22 | toll-like receptor 22 |
| PPI | protein–protein interaction |
| C/EBP | CCAAT-enhancer-binding protein |
| HNF | Hepatocyte nuclear factor |
| NFKB1 | Nuclear factor NF-kappa-B p105 subunit |
| USF | Upstream stimulatory factor |
| GATA | GATA-binding factor |
| STAT | Signal transducer and activator of transcription |
| TFAP2A | Transcription factor AP-2 alpha |
| NC | Negative control |
| WT | Wild-type |
| MT | Mutated-type |
Appendix A
| Transcription Factors | Putative Binding Site |
|---|---|
| TATA box | −32 |
| CEBPA | −1738, −1619, −1500, −1102, −1024, −896, −790, −607, −136, −107 |
| CEBPB | −1738, −1042, −896, −607, −136, −107 |
| HNF1A | −1793, −1605, −201 |
| HNF1B | −199, −330 |
| HNF3A | −911 |
| HNF3B | −1753, −1247, −597 |
| HNF4A | −1158 |
| HNF4G | −741, −1442 |
| NFKB1 | −164, −140 |
| USF1 | −1661, −1533, −1226, −783, −569 |
| USF2 | −1699, −904, −783, −655 |
| GATA1 | −1737, −1364, −986, −945, −321 |
| GATA3 | −943 |
| GATA4 | −324 |
| STAT1 | −288 |
| STAT3 TFAP2A | −1548, −288 −1434 |
| GenBank ID | Gene Name | Primer | Purpose |
|---|---|---|---|
| MK111643 | Sc-hep-F | AAGGAGCTGACAAGAGTCAT | Clone the cDNA sequence of Sc-hep |
| Sc-hep-R | GCTGCAACATTTTCTAATAAG | ||
| Sc-hep-qPCR-F | TGCTCACCTTTATTTGCCTTCA | Real-time quantitative PCR | |
| Sc-hep-qPCR-R | CGCTTCTGTCTGTTGTTATACG | ||
| MK605396 | Sc-FPN1-qPCR-F | CAGCCTCACCAGCGTTCTT | |
| Sc-FPN1-qPCR-R | CTCCGACTCTATCACATTCTCCT | ||
| MK605397 | Sc-FTN-qPCR-F | TCCTACACCTTCCTTGCTCTG | |
| Sc-FTN-qPCR-R | GACCACCTCTCCAATCCTCTC | ||
| MK605398 | Sc-TFR1-qPCR-F | GAATGCCTCTGCTGTGTTGAT | |
| Sc-TFR1-qPCR-R | GCCTGTCGTGATGGTCTGA | ||
| AY647430.1 | Sc-IL-1β-qPCR-F | TTCAGTTCAGGAGGACGGATG | |
| Sc-IL-1β-qPCR-R | TGTGGCAAGACAGGTAGAGATT | ||
| JN180845.1 | Sc-IL8-qPCR-F | GAAGAGCAGCAGAGTTATCATCA | |
| Sc-IL8-qPCR-R | ATCTCAGTCTCCTCGCAGTG | ||
| JN969981.1 | Sc-TLR22-qPCR-F | CCTACGCCTACTACTTCTTCTTG | |
| Sc-TLR22-qPCR-R | GGTCTTCCTGCTTCCATAGATG | ||
| MK605399 | Sc-TNF-α-qPCR-F | GCATACACAACCGCACTA | |
| Sc-TNF-α-qPCR-R | CTTCCATTCCAGCACCAA | ||
| KT895250.1 | Sc-SOCS3-qPCR-F | TCCTCACCACACTCCACAAG | |
| Sc-SOCS3-qPCR-F | CACATTGGATACGCAGGTTCTT | ||
| FJ436084 | β-actin-qPCR-F | GTGCGTGACATCAAGGAGAAG | |
| β-actin-qPCR-R | GGAAGGAAGGCTGGAAGAGG | ||
| Sc-hep-met-F1 | TGATGTGATTATATGTTAAGTATTTGTAAATATTA | Methylation | |
| Sc-hep-met-R1 | ACTAATATATTAATACTATATATTTATACAAAC | ||
| Sc-hep-met-F2 | ATTGTAAATGTTTTTTTAGATGTTGTTTTAATAG | ||
| Sc-hep-met-R2 | ATCATCAAACTCAAAAATCTAAATACAAATAATAC | ||
| Sc-hep-DNA-F1 | GGCAGGCTGTAGTTAATGTGT | Clone the DNA sequence of Sc-hep | |
| Sc-hep-DNA-R1 | AGTTCTCCTGACTGATGGTTGA | ||
| Sc-hep-DNA-F2 | CACACTCAACCATCAGTCAGG | ||
| Sc-hep-DNA-R2 | CAGTTTCCCACTACAAATGTTATGC | ||
| Sc-hep-3’-UTR-WT-F | CTCGAGCAGTGTTGCAGTTGCAGTGGCCGT | qPCR analysis of miRNA and Dual-Luciferase reporter assay | |
| Sc-hep-3’-UTR-WT-R | GTCGACAATTTTGCAGGAAACATTAAATGAAC | ||
| Sc-hep-3’-UTR-MT-F | CTCGAGGGAGAGCTCATCATCATCGTCACTGAAGTGCAAGAGCTGGA | ||
| Sc-hep-3’-UTR-MT-R | GTCGACAACCGTACACGAAACATTAAATGAATCACTTCCTGTGTGC | ||
| miR-196b-F | TAGGTAGTTTCAAGTTGTTGGG | ||
| miR-196d-F | TAGGTAGTTTTATGTTGTTGGG | ||
| miR-23b-5p-F | GGGTTCCTGGCGTGCTGATTT | ||
| miR-92b-5p-F | AGGTGTGGGATGTTGTGCAGTGTT | ||
| miR-19c-5p-F | AGTTTTGCAGGATTGCATCCGG | ||
| U6-F | TTGGAACGATACAGAGAAGATTAGCA | ||
| Universe-R | GCTGTCAACGATACGCTCG |
References
- Liu, L.; Liang, X.F.; Fang, J. The optimal stocking density for hybrid of Siniperca chuatsi (♀) × Siniperca scherzeri (♂) mandarin fish fed minced prey fish. Aquac. Res. 2017, 48, 1342–1345. [Google Scholar] [CrossRef]
- Liu, W.; Zhou, Y.; Fan, Y.; Jiang, N.; Cain, K.; Zeng, L. Development of cross-priming amplification coupled with vertical flow visualization for rapid detection of infectious spleen and kidney necrosis virus (ISKNV) in mandarin fish, Siniperca chuatsi. J. Virol. Methods 2018, 253, 38–42. [Google Scholar] [CrossRef]
- Bismuth, H. Surgical anatomy and anatomical surgery of the liver. World J. Surg. 1982, 6, 3–9. [Google Scholar] [CrossRef]
- Candiotto, F.; Freitas-Júnior, A.; Neri, R.; Bezerra, R.; Rodrigues, R.; Sampaio, L.; Tesser, M. Characterization of digestive enzymes from captive Brazilian flounder Paralichthys orbignyanus. Braz. J. Biol. 2018, 78, 281–288. [Google Scholar] [CrossRef]
- Wu, N.; Song, Y.-L.; Wang, B.; Zhang, X.-Y.; Zhang, X.-J.; Wang, Y.-L.; Cheng, Y.-Y.; Chen, D.-D.; Xia, X.-Q.; Lu, Y.-S.; et al. Fish gut-liver immunity during homeostasis or inflammation revealed by integrative transcriptome and proteome studies. Sci. Rep. 2016, 6, 36048. [Google Scholar] [CrossRef]
- Kumar, R.; Sahoo, P.K.; Barat, A. Transcriptome profiling and expression analysis of immune responsive genes in the liver of Golden mahseer (Tor putitora) challenged with Aeromonas hydrophila. Fish Shellfish Immunol. 2017, 67, 655–666. [Google Scholar] [CrossRef]
- Zhang, X.; Rovin, B.H. Beyond anemia: Hepcidin, monocytes and inflammation. Boil. Chem. 2013, 394, 1–10. [Google Scholar] [CrossRef][Green Version]
- Krause, A.; Neitz, S.; Mägert, H.-J.; Schulz, A.; Forssmann, W.-G.; Schulz-Knappe, P.; Adermann, K. LEAP-1, a novel highly disulfide-bonded human peptide, exhibits antimicrobial activity. FEBS Lett. 2000, 480, 147–150. [Google Scholar] [CrossRef]
- Park, C.H.; Valore, E.V.; Waring, A.J.; Ganz, T. Hepcidin, a urinary antimicrobial peptide synthesized in the liver. J. Biol. Chem. 2001, 276, 7806–7810. [Google Scholar] [CrossRef]
- Hunter, H.N.; Fulton, D.B.; Ganz, T.; Vogel, H.J. The Solution Structure of Human Hepcidin, a Peptide Hormone with Antimicrobial Activity That Is Involved in Iron Uptake and Hereditary Hemochromatosis. J. Boil. Chem. 2002, 277, 37597–37603. [Google Scholar] [CrossRef] [PubMed]
- Shike, H.; Lauth, X.; Westerman, M.E.; Ostland, V.E.; Carlberg, J.M.; Van Olst, J.C.; Shimizu, C.; Bulet, P.; Burns, J.C. Bass hepcidin is a novel antimicrobial peptide induced by bacterial challenge. JBIC J. Boil. Inorg. Chem. 2002, 269, 2232–2237. [Google Scholar] [CrossRef] [PubMed]
- Roth, M.-P.; Meynard, D.; Coppin, H. Regulators of hepcidin expression. Vitam. Horm. 2019, 110, 101–111. [Google Scholar] [PubMed]
- Ward, D.M.; Kaplan, J. Ferroportin-mediated iron transport: Expression and regulation. Biochim. Biophys. Acta 2012, 1823, 1426–1433. [Google Scholar] [CrossRef] [PubMed]
- Fillebeen, C.; Charlebois, E.; Wagner, J.; Katsarou, A.; Mui, J.; Vali, H.; Garcia-Santos, D.; Ponka, P.; Presley, J.; Pantopoulos, K. Transferrin receptor 1 controls systemic iron homeostasis by fine-tuning hepcidin expression to hepatocellular iron load. Blood 2019, 133, 344–355. [Google Scholar] [CrossRef]
- Hentze, M.W.; Muckenthaler, M.U.; Andrews, N.C. Balancing acts: Molecular control of mammalian iron metabolism. Cell 2004, 117, 285–297. [Google Scholar] [CrossRef]
- Drakesmith, H.; Prentice, A.M. Hepcidin and the iron-infection axis. Science 2012, 338, 768–772. [Google Scholar] [CrossRef]
- Nemeth, E.; Tuttle, M.S.; Powelson, J.; Vaughn, M.B.; Donovan, A.; Ward, D.M.; Ganz, T.; Kaplan, J. Hepcidin Regulates Cellular Iron Efflux by Binding to Ferroportin and Inducing Its Internalization. Science 2004, 306, 2090–2093. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Babitt, J.L. Hepcidin regulation in the anemia of inflammation. Curr. Opin. Hematol. 2016, 23, 189–197. [Google Scholar] [CrossRef]
- Guida, C.; Altamura, S.; Klein, F.A.; Galy, B.; Boutros, M.; Ulmer, A.J.; Hentze, M.W.; Muckenthaler, M.U. A novel inflammatory pathway mediating rapid hepcidin-independent hypoferremia. Blood 2015, 125, 2265–2275. [Google Scholar] [CrossRef]
- Michels, K.; Nemeth, E.; Ganz, T.; Mehrad, B. Hepcidin and Host Defense against Infectious Diseases. PLoS Pathog. 2015, 11, e1004998. [Google Scholar] [CrossRef]
- Jiang, X.-F.; Liu, Z.-F.; Lin, A.-F.; Xiang, L.-X.; Shao, J.-Z. Coordination of Bactericidal and Iron Regulatory Functions of Hepcidin in Innate Antimicrobial Immunity in a Zebrafish Model. Sci. Rep. 2017, 7, 4265. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Babu, V.S.; Lin, L.; Hu, Y.; Zhang, Y.; Liu, X.; Su, J.; Li, J.; Zhao, L.; Yuan, G. Hepcidin protects grass carp (Ctenopharyngodon idellus) against Flavobacterium columnare infection via regulating iron distribution and immune gene expression. Fish Shellfish Immunol. 2018, 75, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Cuesta, A.; Meseguer, J.; Esteban, M.Á. The antimicrobial peptide hepcidin exerts an important role in the innate immunity against bacteria in the bony fish gilthead seabream. Mol. Immunol. 2008, 45, 2333–2342. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.-J.; Cai, J.-J.; Cai, L.; Qu, H.-D.; Yang, M.; Zhang, M. Cloning and expression of a hepcidin gene from a marine fish (Pseudosciaena crocea) and the antimicrobial activity of its synthetic peptide. Peptides 2009, 30, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, P.N.; Vázquez-Dorado, S.; Neves, J.V.; Wilson, J.M. Dual function of fish hepcidin: Response to experimental iron overload and bacterial infection in sea bass (Dicentrarchus labrax). Dev. Comp. Immunol. 2006, 30, 1156–1167. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Han, X.; Chen, X.; Yu, S.; Chai, Y.; Zhai, T.; Zhu, Q. Molecular characterization and functional analysis of the hepcidin gene from roughskin sculpin (Trachidermus fasciatus). Fish Shellfish Immunol. 2017, 68, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Huang, T.; Gu, W.; Zhang, Y.; Yao, Z.; Zhao, C.; Wang, B. Characterization, expression, and functional analysis of the hepcidin gene from Brachymystax lenok. Dev. Comp. Immunol. 2018, 89, 131–140. [Google Scholar] [CrossRef]
- Huang, T.; Gu, W.; Wang, B.; Zhang, Y.; Cui, L.; Yao, Z.; Zhao, C.; Xu, G. Identification and expression of the hepcidin gene from brown trout (Salmo trutta) and functional analysis of its synthetic peptide. Fish Shellfish Immunol. 2019, 87, 243–253. [Google Scholar] [CrossRef]
- Truksa, J.; Lee, P.; Beutler, E. The role of STAT, AP-1, E-box and TIEG motifs in the regulation of hepcidin by IL-6 and BMP-9: Lessons from human HAMP and murine Hamp1 and Hamp2 gene promoters. Blood Cells Mol. Dis. 2007, 39, 255–262. [Google Scholar] [CrossRef]
- Ganz, T.; Nemeth, E. Iron balance and the role of hepcidin in chronic kidney disease. In Seminars in Nephrology; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Hilton, K.B.; Lambert, L.A. Molecular evolution and characterization of hepcidin gene products in vertebrates. Gene 2008, 415, 40–48. [Google Scholar] [CrossRef]
- Neves, J.V.; Caldas, C.; Vieira, I.; Ramos, M.F.; Rodrigues, P.N.S. Multiple Hepcidins in a Teleost Fish, Dicentrarchus labrax: Different Hepcidins for Different Roles. J. Immunol. 2015, 195, 2696–2709. [Google Scholar] [CrossRef] [PubMed]
- Neves, J.V.; Ramos, M.F.; Moreira, A.C.; Silva, T.; Gomes, M.S.; Rodrigues, P.N.S. Hamp1 but not Hamp2 regulates ferroportin in fish with two functionally distinct hepcidin types. Sci. Rep. 2017, 7, 14793. [Google Scholar] [CrossRef] [PubMed]
- Carmel, L.; Chorev, M. The function of introns. Front. Genet. 2012, 3, 55. [Google Scholar]
- Schübeler, D. Function and information content of DNA methylation. Nature 2015, 517, 321–326. [Google Scholar] [CrossRef]
- Udali, S.; Castagna, A.; Corbella, M.; Ruzzenente, A.; Moruzzi, S.; Mazzi, F.; Campagnaro, T.; Santis, D.D.; Franceschi, A.; Pattini, P. Hepcidin and DNA promoter methylation in hepatocellular carcinoma. Eur. J. Clin. Investig. 2018, 48, e12870. [Google Scholar] [CrossRef]
- Ha, M.; Kim, V.N. Regulation of microRNA biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509. [Google Scholar] [CrossRef]
- Zumbrennen-Bullough, K.B.; Wu, Q.; Core, A.B.; Canali, S.; Chen, W.; Theurl, I.; Meynard, D.; Babitt, J.L. MicroRNA-130a Is Up-regulated in Mouse Liver by Iron Deficiency and Targets the Bone Morphogenetic Protein (BMP) Receptor ALK2 to Attenuate BMP Signaling and Hepcidin Transcription. J. Boil. Chem. 2014, 289, 23796–23808. [Google Scholar] [CrossRef]
- Castoldi, M.; Spasić, M.V.; Altamura, S.; Elmén, J.; Lindow, M.; Kiss, J.; Stolte, J.; Sparla, R.; D’Alessandro, L.A.; Klingmüller, U.; et al. The liver-specific microRNA miR-122 controls systemic iron homeostasis in mice. J. Clin. Investig. 2011, 121, 1386–1396. [Google Scholar] [CrossRef]
- Courselaud, B.; Pigeon, C.; Inoue, Y.; Inoue, J.; Gonzalez, F.J.; Leroyer, P.; Gilot, D.; Boudjema, K.; Guguen-Guillouzo, C.; Brissot, P. C/EBPα Regulates Hepatic Transcription of Hepcidin, an Antimicrobial Peptide and Regulator of Iron Metabolism CROSS-TALK between C/EBP pathway and iron metabolism. J. Biol. Chem. 2002, 277, 41163–41170. [Google Scholar] [CrossRef]
- Kanamori, Y.; Murakami, M.; Sugiyama, M.; Hashimotod, O.; Matsuia, T.; Funaba, M. Hepcidin and IL-1β. Vitam. Horm. 2019, 110, 143–156. [Google Scholar]
- Sow, F.B.; Alvarez, G.R.; Gross, R.P.; Satoskar, A.R.; Schlesinger, L.S.; Zwilling, B.S.; Lafuse, W.P. Role of STAT1, NF-κB, and C/EBPβ in the macrophage transcriptional regulation of hepcidin by mycobacterial infection and IFN-γ. J. Leukoc. Boil. 2009, 86, 1247–1258. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-O.; Cho, Y.-S.; Kim, H.-L.; Park, J.-W. ROS mediate the hypoxic repression of the hepcidin gene by inhibiting C/EBPα and STAT-3. Biochem. Biophys. Res. Commun. 2007, 356, 312–317. [Google Scholar] [CrossRef] [PubMed]
- Shike, H. Organization and expression analysis of the zebrafish hepcidin gene, an antimicrobial peptide gene conserved among vertebrates. Dev. Comp. Immunol. 2004, 28, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Darshan, D.; Anderson, G.J. Liver-gut axis in the regulation of iron homeostasis. World J. Gastroenterol. 2007, 13, 4737–4745. [Google Scholar] [CrossRef] [PubMed]
- Hocquellet, A.; Le Senechal, C.; Garbay, B. Importance of the disulfide bridges in the antibacterial activity of human hepcidin. Peptides 2012, 36, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-M.; Chen, J.; Lv, Y.-P.; Hu, Z.-H.; Dai, Q.-M.; Fan, X.-L. Molecular characterization of a hepcidin homologue in starry flounder (Platichthys stellatus) and its synergistic interaction with antibiotics. Fish Shellfish Immunol. 2018, 83, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Schaible, U.E.; Kaufmann, S.H. Iron and microbial infection. Nat. Rev. Microbiol. 2004, 2, 946. [Google Scholar] [CrossRef]
- Tymchuk, W.; Sakhrani, D.; Devlin, R. Domestication causes large-scale effects on gene expression in rainbow trout: Analysis of muscle, liver and brain transcriptomes. Gen. Comp. Endocrinol. 2009, 164, 175–183. [Google Scholar] [CrossRef]
- OIE. Manual of Diagnostic Tests for Aquatic Animals; Office International des Epizooties: Paris, France, 2006. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Boil. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Kelley, L.A.; Mezulis, S.; Yates, C.M.; Wass, M.N.; Sternberg, M.J.E. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protoc. 2015, 10, 845–858. [Google Scholar] [CrossRef]
- Farré, D.; Martínez, J.; Albà, M.; Messeguer, X.; Escudero, R.; Núñez, O. PROMO: Detection of known transcription regulatory elements using species-tailored searches. Bioinformatics 2002, 18, 333–334. [Google Scholar]
- Khan, A.; Fornes, O.; Stigliani, A.; Gheorghe, M.; Castro-Mondragon, J.A.; Van Der Lee, R.; Bessy, A.; Chèneby, J.; Kulkarni, S.R.; Tan, G.; et al. JASPAR 2018: Update of the open-access database of transcription factor binding profiles and its web framework. Nucleic Acids Res. 2017, 46, D260–D266. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shen, Y.; Wu, M.; Zhao, J. Irf3 from mandarin fish thymus initiates interferon transcription. Fish Physiol. Biochem. 2019, 45, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Miranda, K.C.; Huynh, T.; Tay, Y.; Ang, Y.-S.; Tam, W.-L.; Thomson, A.M.; Lim, B.; Rigoutsos, I. A Pattern-Based Method for the Identification of MicroRNA Binding Sites and Their Corresponding Heteroduplexes. Cell 2006, 126, 1203–1217. [Google Scholar] [CrossRef]
- Enright, A.J.; John, B.; Gaul, U.; Tuschl, T.; Sander, C.; Marks, D.S. MicroRNA targets in Drosophila. Genome Biol. 2003, 5, R1. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, Y.; Zhao, Z.; Zhao, J.; Chen, X.; Cao, M.; Wu, M. Expression and Functional Analysis of Hepcidin from Mandarin Fish (Siniperca chuatsi). Int. J. Mol. Sci. 2019, 20, 5602. https://doi.org/10.3390/ijms20225602
Shen Y, Zhao Z, Zhao J, Chen X, Cao M, Wu M. Expression and Functional Analysis of Hepcidin from Mandarin Fish (Siniperca chuatsi). International Journal of Molecular Sciences. 2019; 20(22):5602. https://doi.org/10.3390/ijms20225602
Chicago/Turabian StyleShen, Yawei, Ziwei Zhao, Jinliang Zhao, Xiaowu Chen, Ming Cao, and Minglin Wu. 2019. "Expression and Functional Analysis of Hepcidin from Mandarin Fish (Siniperca chuatsi)" International Journal of Molecular Sciences 20, no. 22: 5602. https://doi.org/10.3390/ijms20225602
APA StyleShen, Y., Zhao, Z., Zhao, J., Chen, X., Cao, M., & Wu, M. (2019). Expression and Functional Analysis of Hepcidin from Mandarin Fish (Siniperca chuatsi). International Journal of Molecular Sciences, 20(22), 5602. https://doi.org/10.3390/ijms20225602





