Magnesium Sulfate Mitigates the Progression of Monocrotaline Pulmonary Hypertension in Rats
Abstract
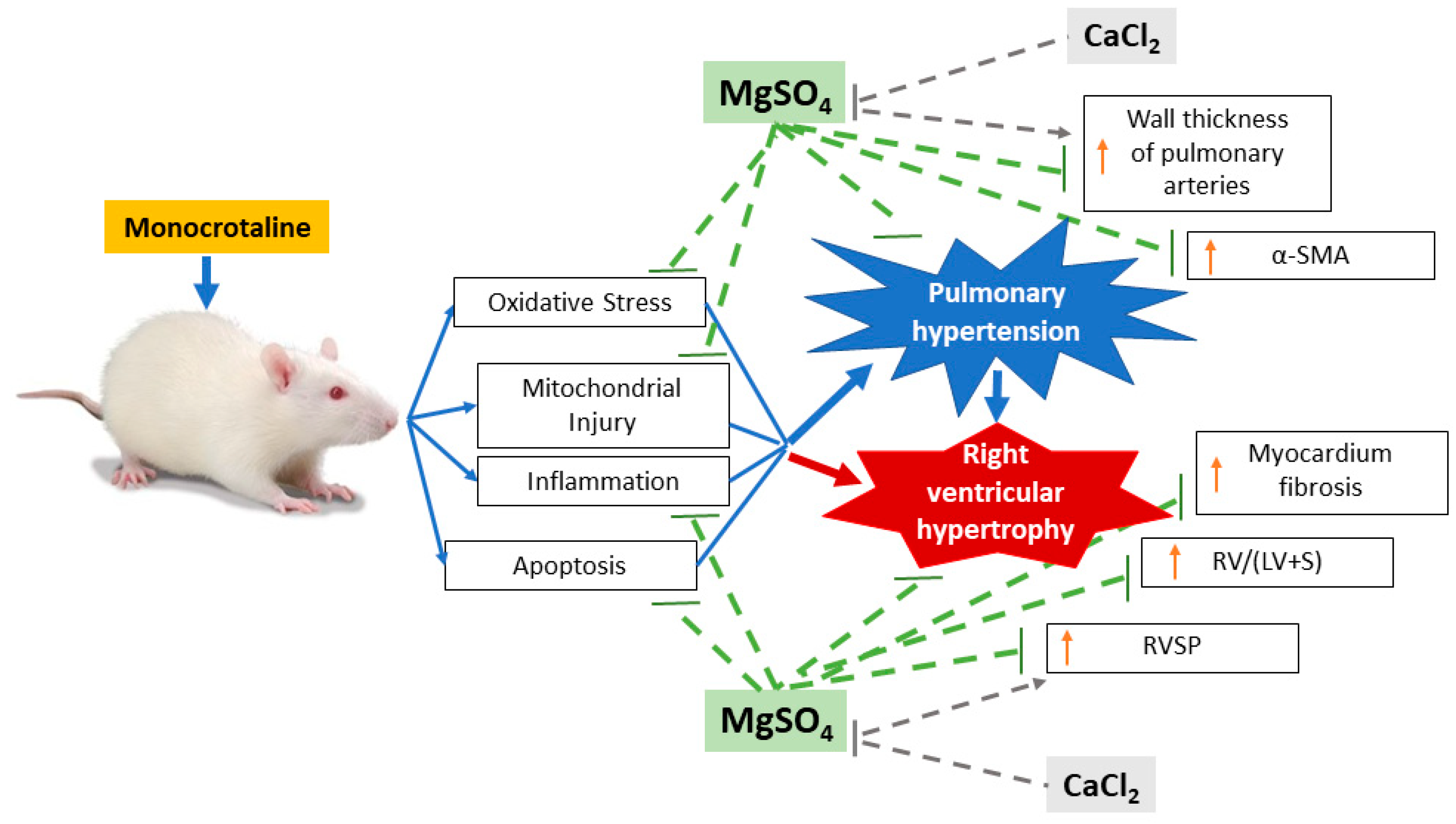
:1. Introduction
2. Results
2.1. MgSO4 Improves Survival in Monocrotaline-Treated Rats
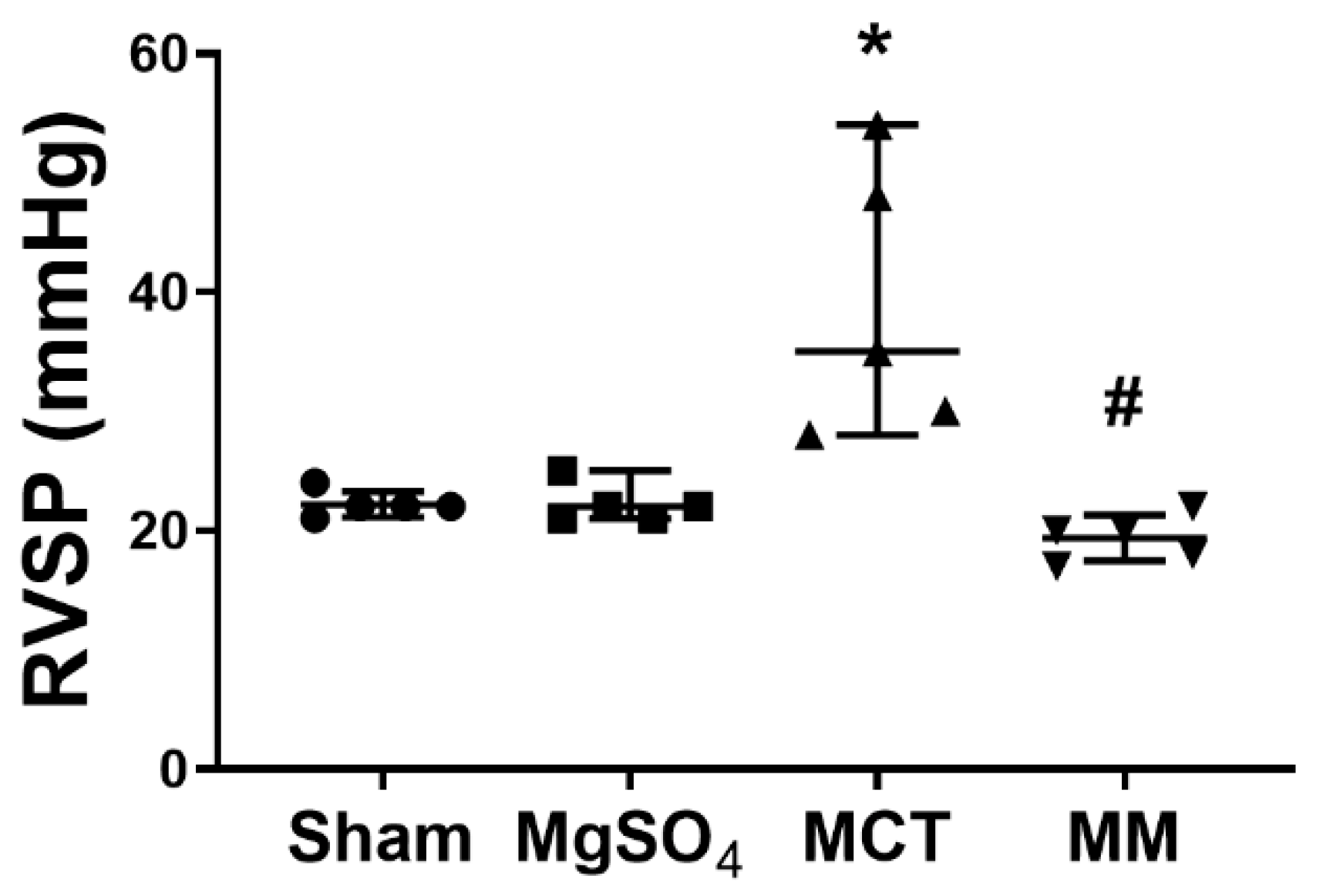
2.2. MgSO4 Mitigates Monocrotaline-Induced Pulmonary Hypertension
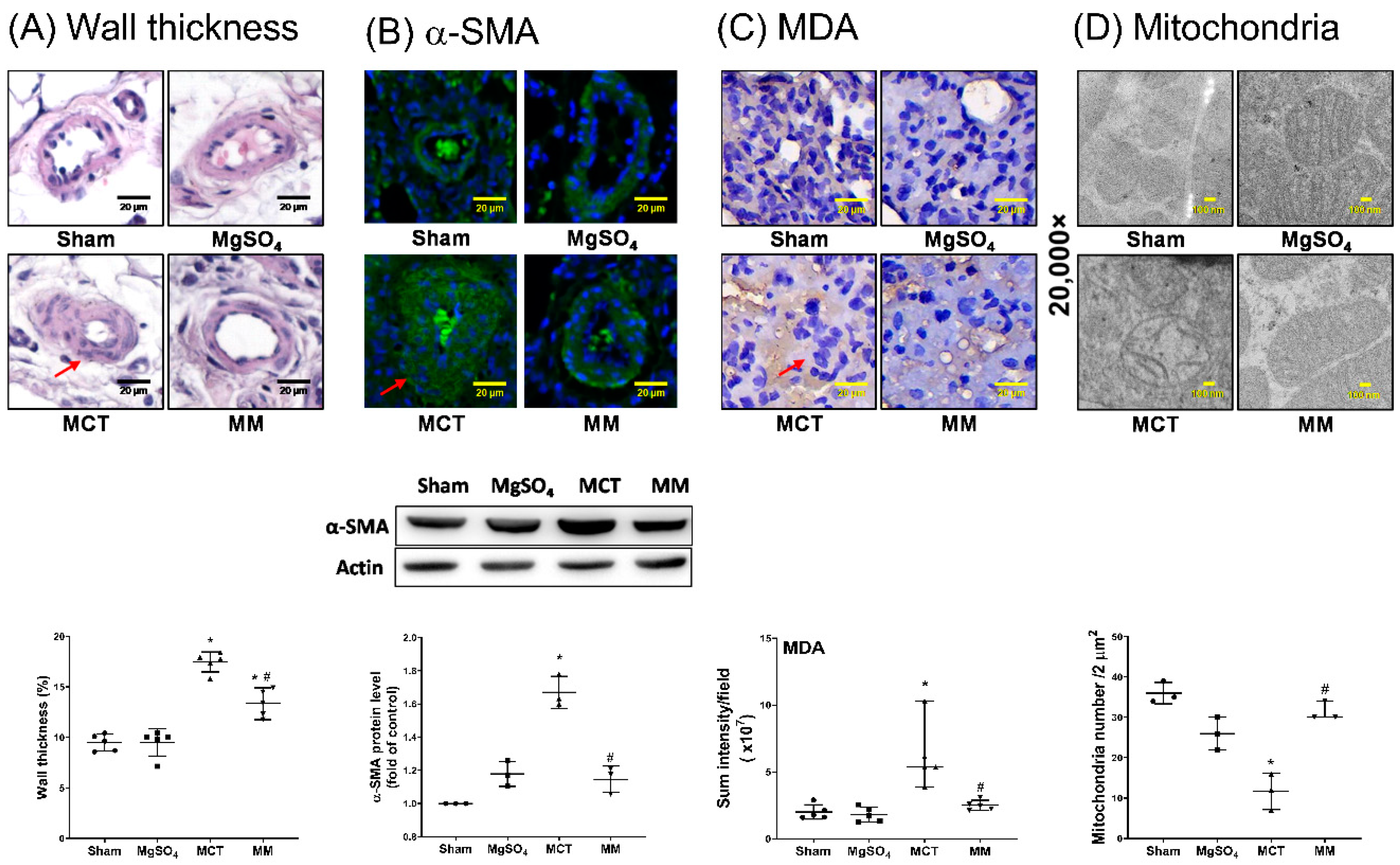
2.3. MgSO4 Mitigates Monocrotaline-Induced Pulmonary Artery Remodeling, Lung Tissue Lipid Peroxidation, and Pulmonary Artery Mitochondrial Injury
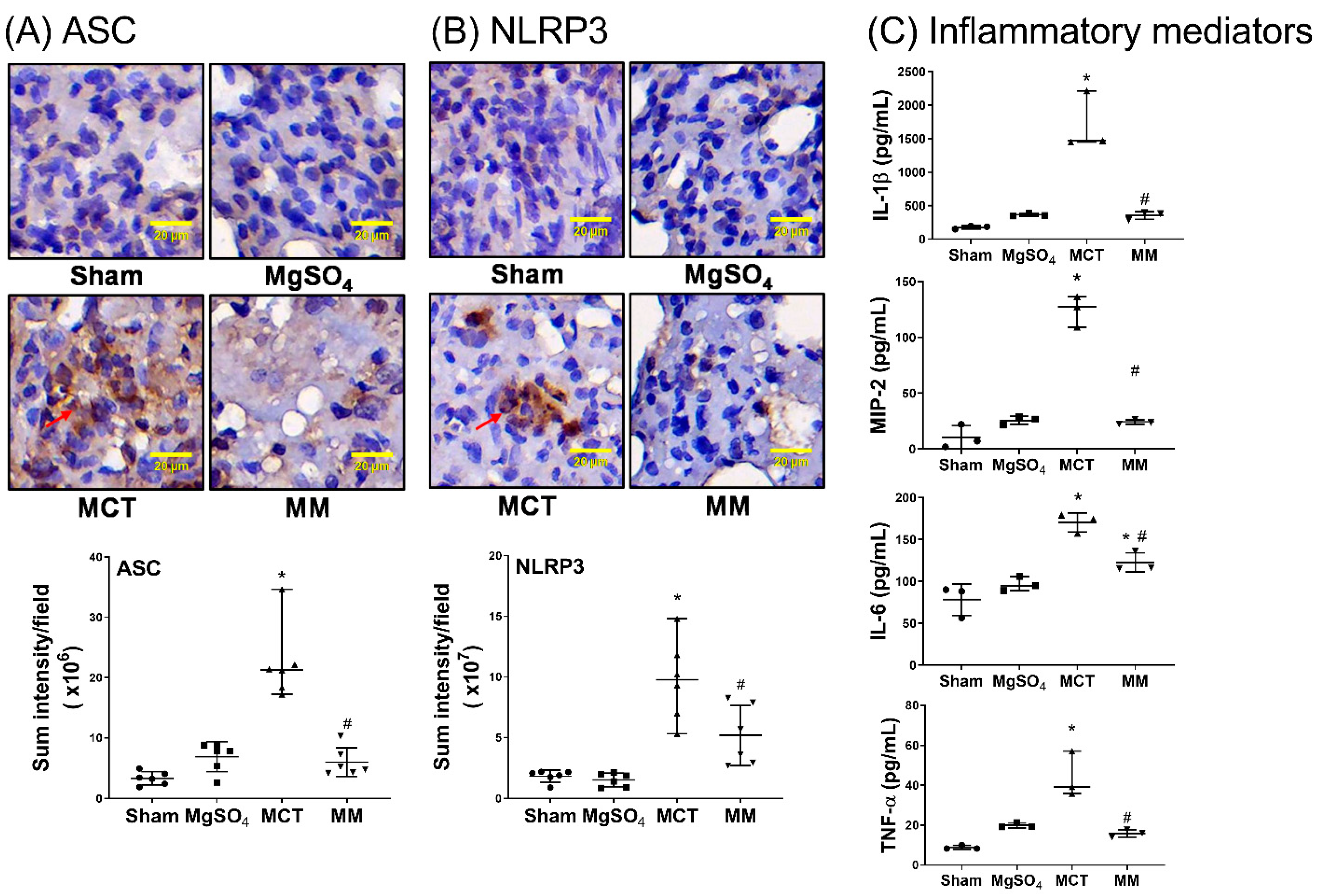
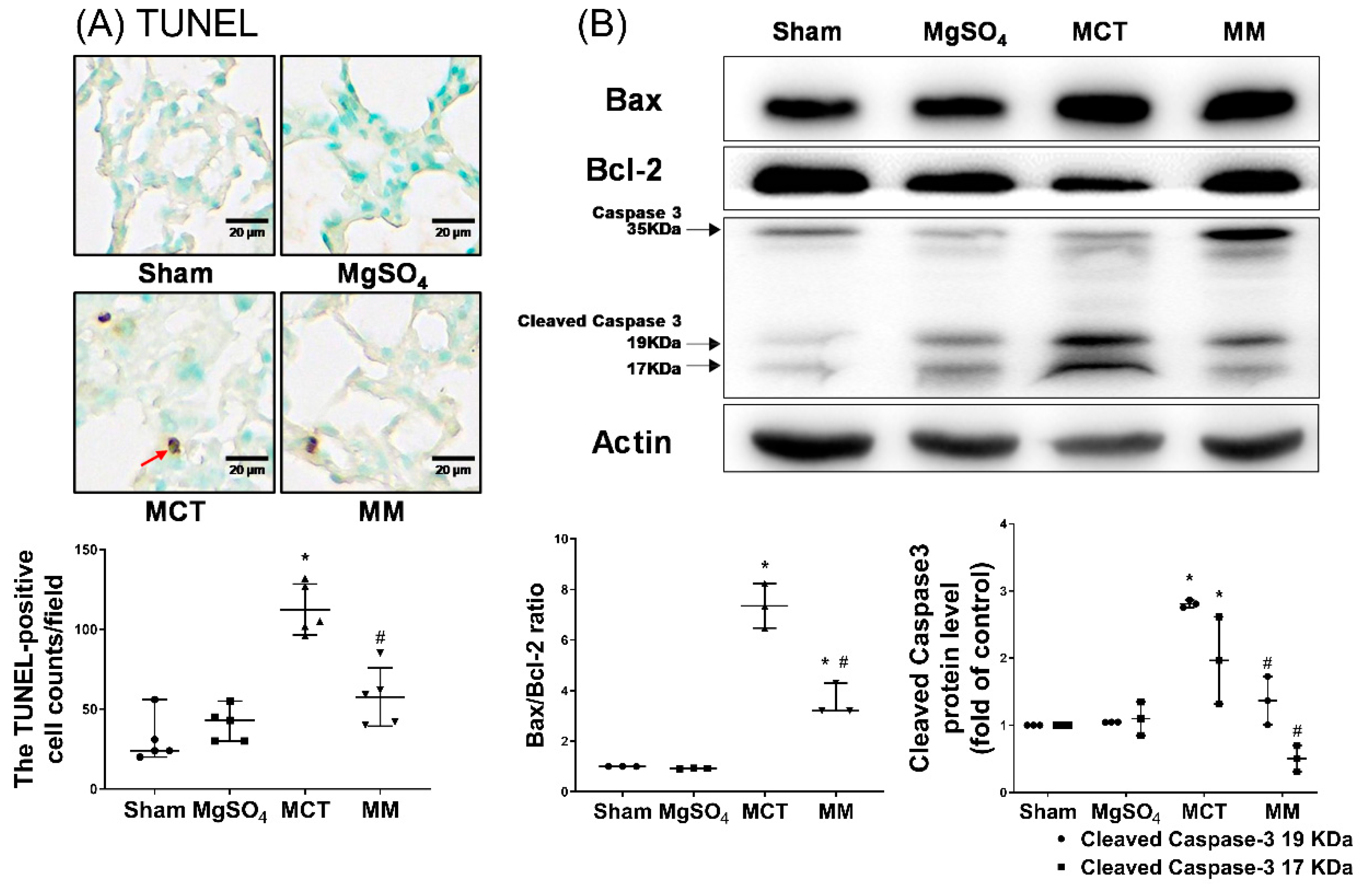
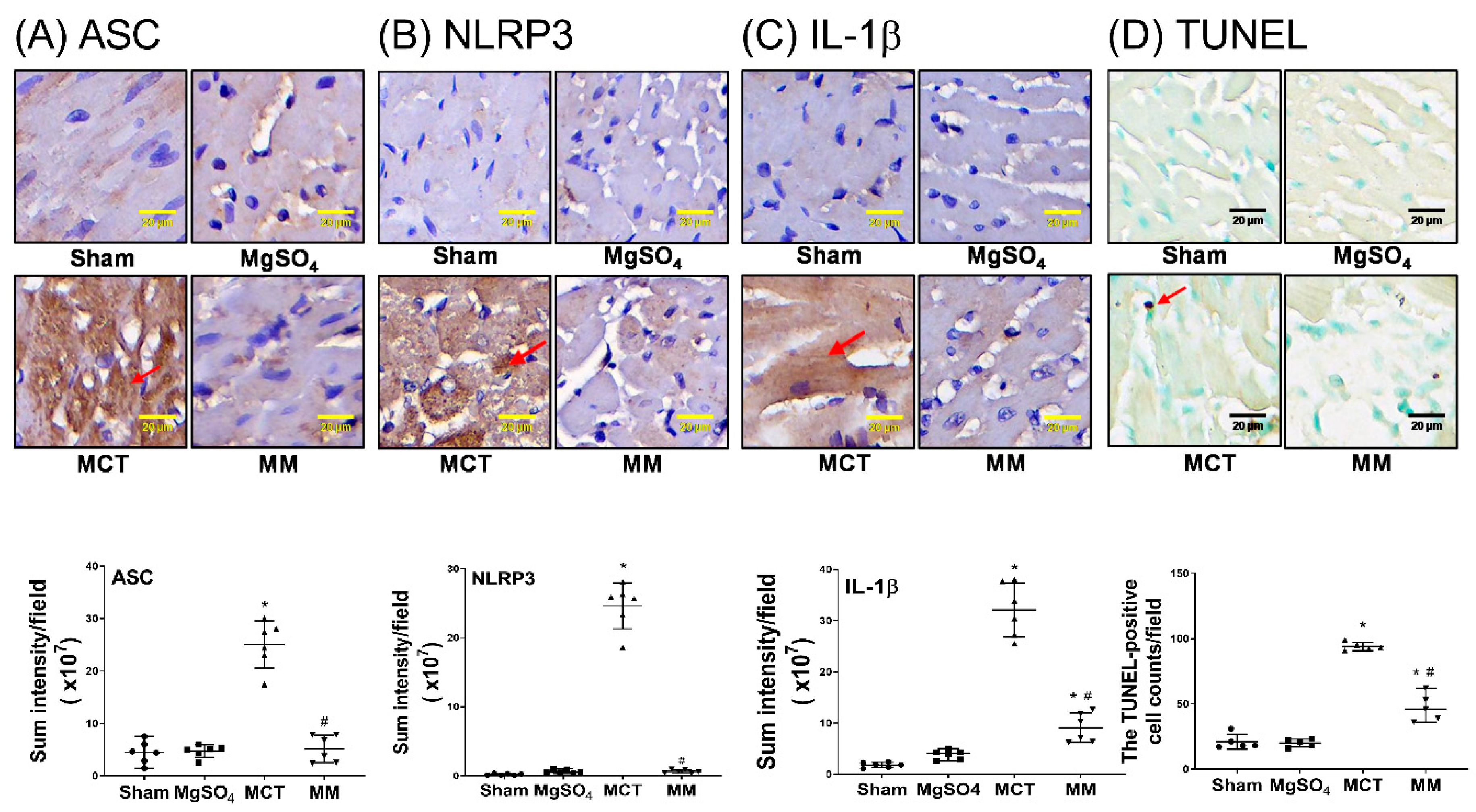
2.4. MgSO4 Mitigates Monocrotaline-Induced Inflammation and Apoptosis in Lung Tissues
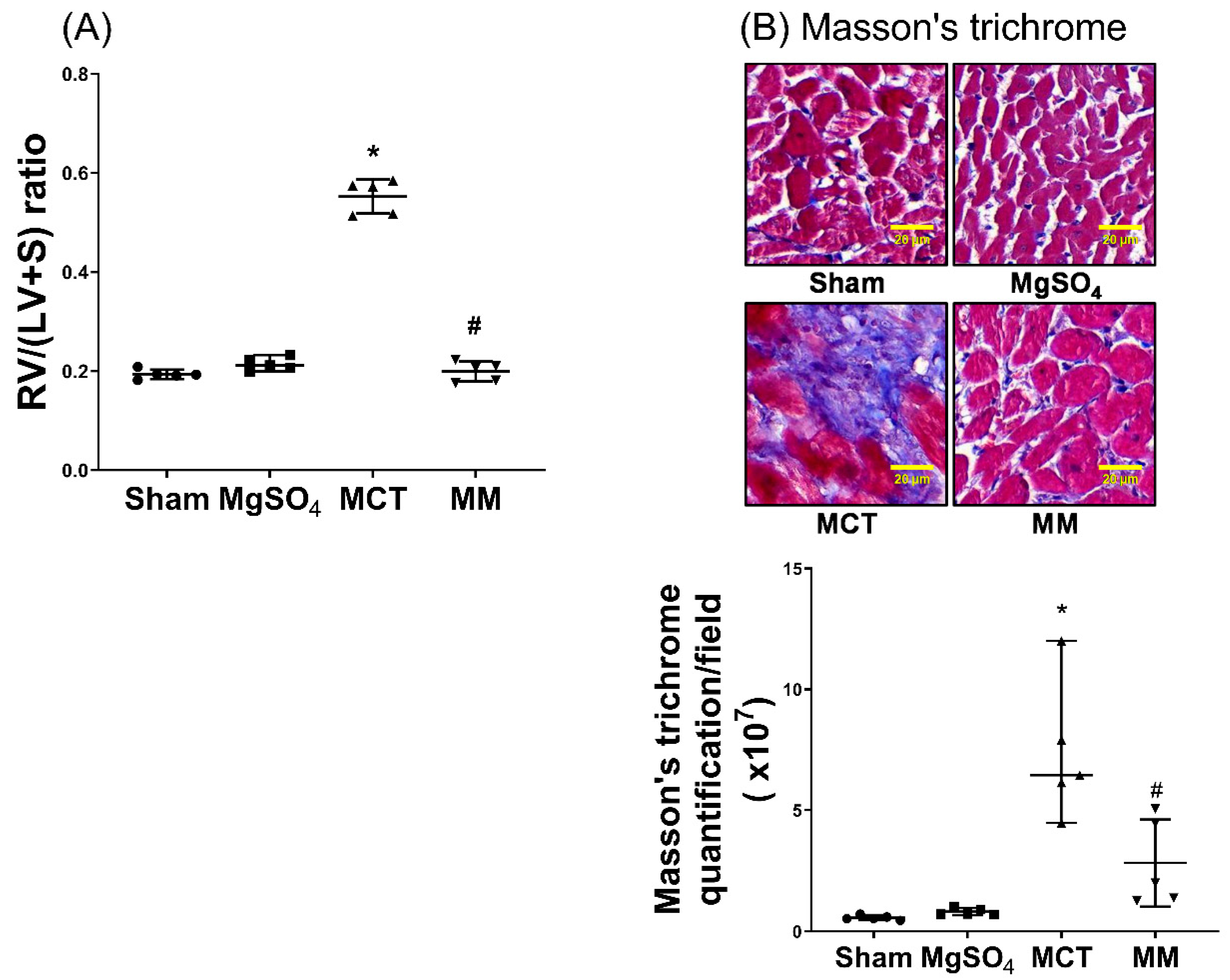
2.5. MgSO4 Mitigates Monocrotaline-Induced Right Ventricular Hypertrophy
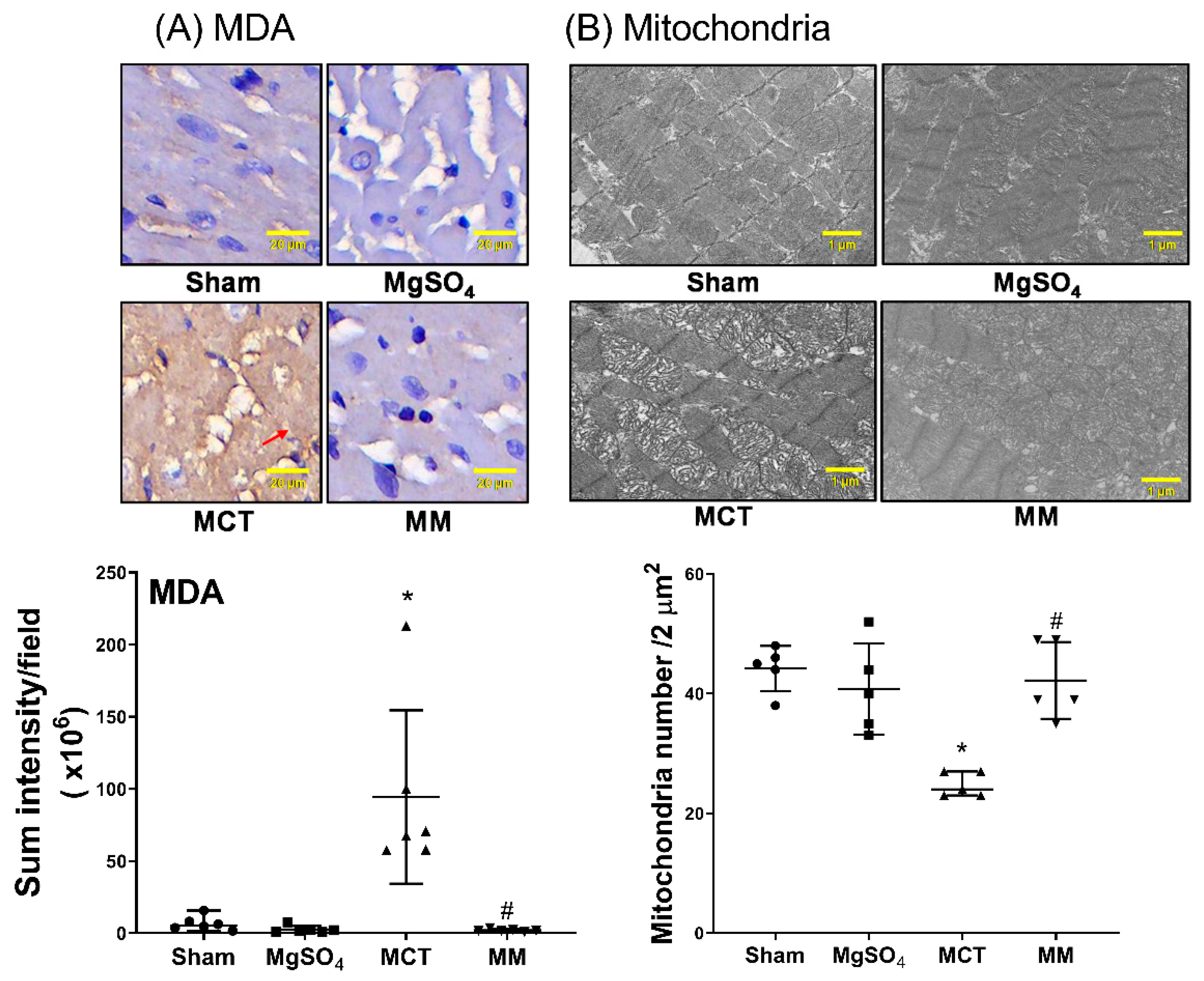
2.6. MgSO4 Mitigates Monocrotaline-Induced Lipid Peroxidation, Mitochondrial Injury, Inflammation, and Apoptosis in Myocardium
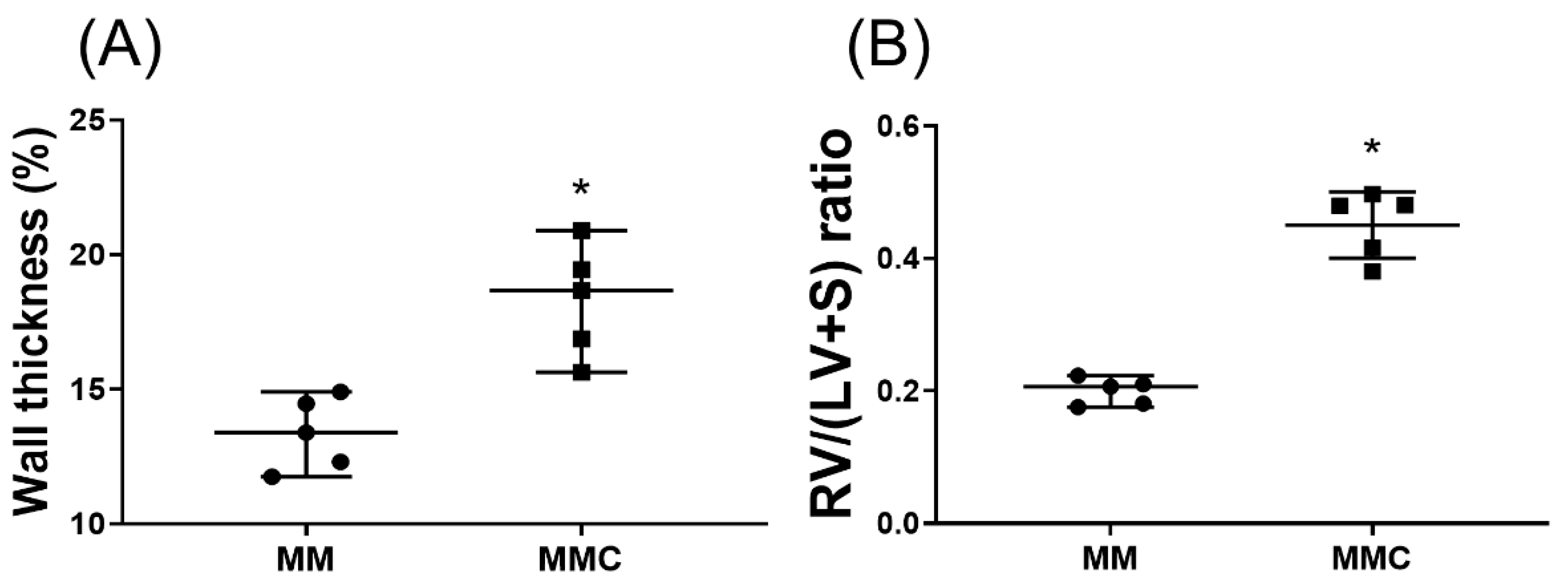
2.7. Calcium Chloride Counteracts the Effect of Mgso4 in Monocrotaline-Treated Rats
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Monocrotaline Pulmonary Hypertension Model and Dosages of Monocrotaline and MgSO4
4.3. Study Protocols
4.4. RVSP and RV/(LV+S) Ratio Measurements
4.5. Assay of Wall Thickness of Pulmonary Arteries
4.6. Immunofluorescence Staining
4.7. Immunoblotting Assay
4.8. Enzyme-linked Immunosorbent Assay
4.9. Terminal Deoxynucleotidyl Transferase dUTP Nick End Labeling
4.10. Immunohistochemistry Staining Assay
4.11. Masson’s Trichrome Staining
4.12. Transmission Electron Microscopic Analysis
4.13. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Farber, H.W.; Loscalzo, J. Pulmonary arterial hypertension. N. Engl. J. Med. 2004, 351, 1655–1665. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, V.; McGoon, M. Pulmonary arterial hypertension. Circulation 2006, 114, 1417–1431. [Google Scholar] [CrossRef] [PubMed]
- Taichman, D.B.; Ornelas, J.; Chung, L.; Klinger, J.R.; Lewis, S.; Mandel, J.; Paleversusky, H.I.; Rich, S.; Sood, N.; Rosenzweig, E.B.; et al. Pharmacologic therapy for pulmonary arterial hypertension in adults: Chest guideline and expert panel report. Chest 2014, 146, 449–475. [Google Scholar] [CrossRef] [PubMed]
- Thenappan, T.; Ormiston, M.L.; Ryan, J.J.; Archer, S.L. Pulmonary arterial hypertension: Pathogenesis and clinical management. BMJ 2018, 360, j5492. [Google Scholar] [CrossRef] [PubMed]
- Hoeper, M.M.; Kramer, T.; Pan, Z.; Eichstaedt, C.A.; Spiesshoefer, J.; Benjamin, N.; Olsson, K.M.; Meyer, K.; Vizza, C.D.; Vonk-Noordegraaf, A.; et al. Mortality in pulmonary arterial hypertension: Prediction by the 2015 european pulmonary hypertension guidelines risk stratification model. Eur. Respir. J. 2017, 50, 1700740. [Google Scholar] [CrossRef] [PubMed]
- Tuder, R.M. Pulmonary vascular remodeling in pulmonary hypertension. Cell. Tissue. Res. 2017, 367, 643–649. [Google Scholar] [CrossRef]
- Crosswhite, P.; Sun, Z. Molecular mechanisms of pulmonary arterial remodeling. Mol. Med. 2014, 20, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Dromparis, P.; Sutendra, G.; Michelakis, E.D. The role of mitochondria in pulmonary vascular remodeling. J. Mol. Med. 2010, 8, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- McMurtry, M.S.; Archer, S.L.; Altieri, D.C.; Bonnet, S.; Haromy, A.; Harry, G.; Bonnet, S.; Puttagunta, L.; Michelakis, E.D. Gene therapy targeting survivin selectively induces pulmonary vascular apoptosis and reverses pulmonary arterial hypertension. J. Clin. Invest. 2005, 115, 1479–1491. [Google Scholar] [CrossRef]
- Galiè, N.; Manes, A.; Negro, L.; Palazzini, M.; Bacchi-Reggiani, M.L.; Branzi, A. A meta-analysis of randomized controlled trials in pulmonary arterial hypertension. Eur. Heart. J. 2009, 30, 394–403. [Google Scholar] [CrossRef]
- Ryerson, C.J.; Nayar, S.; Swiston, J.R.; Sin, D.D. Pharmacotherapy in pulmonary arterial hypertension: A systematic review and meta-analysis. Respir. Res. 2010, 11, 12. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Tsai, P.S.; Hung, Y.C.; Huang, C.J. L-type calcium channels are involved in mediating the anti-inflammatory effects of magnesium sulphate. Br. J. Anaesth. 2010, 104, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Bar, J.; Ben-Haroush, A.; Feldberg, D.; Hod, M. The pharmacologic approach to the prevention of preeclampsia: From antiplatelet, antithrombosis and antioxidant therapy to anticonvulsants. Curr. Med. Chem. Cardiovasc. Hematol. Agents. 2005, 3, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.H.; Behr, E.R. Pharmacological treatment of acquired QT prolongation and torsades de pointes. Br. J. Clin. Pharmacol. 2016, 81, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Maselli, D.J.; Peters, J.I. Medication Regimens for Managing Acute Asthma. Respir. Care. 2018, 63, 783–796. [Google Scholar] [CrossRef] [PubMed]
- Kao, M.C.; Jan, W.C.; Tsai, P.S.; Wang, T.Y.; Huang, C.J. Magnesium sulfate mitigates lung injury induced by bilateral lower limb ischemia-reperfusion in rats. J. Surg. Res. 2011, 171, e97–e106. [Google Scholar] [CrossRef] [PubMed]
- Rochelson, B.; Dowling, O.; Schwartz, N.; Metz, C.N. Magnesium sulfate suppresses inflammatory responses by human umbilical vein endothelial cells (HuVECs) through the NFkappaB pathway. J. Reprod. Immunol. 2007, 73, 101–107. [Google Scholar] [CrossRef]
- Xu, M.; Dai, W.; Deng, X. Effects of magnesium sulfate on brain mitochondrial respiratory function in rats after experimental traumatic brain injury. Chin. J. Traumatol. 2002, 5, 361–364. [Google Scholar]
- Solaroglu, I.; Kaptanoglu, E.; Okutan, O.; Beskonakli, E.; Attar, A.; Kilinc, K. Magnesium sulfate treatment decreases caspase-3 activity after experimental spinal cord injury in rats. Surg. Neurol. 2005, 64, S17–S21. [Google Scholar] [CrossRef]
- Chang, Y.Y.; Kao, M.C.; Lin, J.A.; Chen, T.Y.; Cheng, C.F.; Wong, C.S.; Tzeng, I.S.; Huang, C.J. Effects of MgSO4 on inhibiting Nod-like receptor protein 3 inflammasome involve decreasing intracellular calcium. J. Surg. Res. 2018, 221, 257–265. [Google Scholar] [CrossRef]
- Wu, T.J.; Teng, R.J.; Tsou, K.I. Persistent pulmonary hypertension of the newborn treated with magnesium sulfate in premature neonates. Pediatrics 1995, 96, 472–474. [Google Scholar] [PubMed]
- Raimondi, F.; Migliaro, F.; Capasso, L.; Ausanio, G.; Bisceglia, M.; Giliberti, P.; Messina, F.; Salvia, G.; Paludetto, R. Intravenous magnesium sulphate versus inhaled nitric oxide for moderate, persistent pulmonary hypertension of the newborn. A Multicentre, retrospective study. J. Trop. Pediatr. 2008, 54, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, H.; Hirota, K.; Sato, T.; Hashimoto, Y.; Ishihara, H.; Matsuki, A. Spasmolytic effect of magnesium sulfate on serotonin-induced pulmonary hypertension and bronchoconstriction in dogs. Acta Anaesthesiol. Scand. 2001, 45, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.E.; Burnette, T.M.; Geiser, D.R.; Janjindamai, W. Magnesium attenuates pulmonary hypertension due to hypoxia and group B streptococci. J. Appl. Physiol. 1994, 77, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Mathew, R.; Gloster, E.S.; Altura, B.T.; Altura, B.M. Magnesium aspartate hydrochloride attenuates monocrotaline-induced pulmonary artery hypertension in rats. Clin. Sci. 1988, 75, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Umar, S.; Nadadur, R.D.; Li, J.; Maltese, F.; Partownavid, P.; van der Laarse, A.; Eghbali, M. Intralipid prevents and rescues fatal pulmonary arterial hypertension and right ventricular failure in rats. Hypertension 2011, 58, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, Y.; Kato, T.; Ito, M.; Azuma, Y.; Fukasawa, Y.; Ohno, K.; Kojima, S. Hydrogen ameliorates pulmonary hypertension in rats by anti-inflammatory and antioxidant effects. J. Thorac. Cardiovasc. Surg. 2015, 150, 645–654.e3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, K.A.; Ayon, R.J.; Tang, H.; Makino, A.; Yuan, J.X. Calcium-Sensing Receptor Regulates Cytosolic [Ca 2+] and Plays a Major Role in the Development of Pulmonary Hypertension. Front. Physiol. 2016, 7, 517. [Google Scholar] [CrossRef]
- Sankhe, S.; Manousakidi, S.; Antigny, F.; Arthur Ataam, J.; Bentebbal, S.; Ruchon, Y.; Lecerf, F.; Sabourin, J.; Price, L.; Fadel, E.; et al. T-type Ca2+ channels elicit pro-proliferative and anti-apoptotic responses through impaired PP2A/Akt1 signaling in PASMCs from patients with pulmonary arterial hypertension. Biochim. Biophys. Acta Mol. Cell. Res. 2017, 1864, 1631–1641. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, X.; Kubo, H.; Harris, D.M.; Mills, G.D.; Moyer, J.; Berretta, R.; Potts, S.T.; Marsh, J.D.; Houser, S.R. Ca2+ influx-induced sarcoplasmic reticulum Ca2+ overload causes mitochondrial-dependent apoptosis in ventricular myocytes. Circ. Res. 2005, 97, 1009–1017. [Google Scholar] [CrossRef]
- Suresh, K.; Servinsky, L.; Jiang, H.; Bigham, Z.; Yun, X.; Kliment, C.; Huetsch, J.; Damarla, M.; Shimoda, L.A. Reactive oxygen species induced Ca2+ influx via TRPV4 and microvascular endothelial dysfunction in the SU5416/hypoxia model of pulmonary arterial hypertension. Am. J. Physiol. Lung. Cell. Mol. Physiol. 2018, 314, L893–L907. [Google Scholar] [CrossRef] [PubMed]
- Sitbon, O.; Humbert, M.; Jaïs, X.; Ioos, V.; Hamid, A.M.; Provencher, S.; Garcia, G.; Parent, F.; Hervé, P.; Simonneau, G. Long-term response to calcium channel blockers in idiopathic pulmonary arterial hypertension. Circulation 2005, 111, 3105–3111. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Chen, Y.; Liu, H. Calcium channel blockers for pulmonary arterial hypertension. Cochrane. Database. Syst. Rev. 2015, 9, CD010066. [Google Scholar] [CrossRef]
- Ho, J.J.; Rasa, G. Magnesium sulfate for persistent pulmonary hypertension of the newborn. Cochrane. Database. Syst. Rev 2007, 3, CD005588. [Google Scholar] [CrossRef]
- West, J.; Hemnes, A. Experimental and transgenic models of pulmonary hypertension. Compr. Physiol. 2011, 1, 769–782. [Google Scholar] [CrossRef] [PubMed]
- Nogueira-Ferreira, R.; Vitorino, R.; Ferreira, R.; Henriques-Coelho, T. Exploring the monocrotaline animal model for the study of pulmonary arterial hypertension: A network approach. Pulm. Pharm. Ther. 2015, 35, 8–16. [Google Scholar] [CrossRef]
- Gomez-Arroyo, J.; Mizuno, S.; Szczepanek, K.; Van Tassell, B.; Natarajan, R.; dos Remedios, C.G.; Drake, J.I.; Farkas, L.; Kraskauskas, D.; Wijesinghe, D.S.; et al. Metabolic gene remodeling and mitochondrial dysfunction in failing right ventricular hypertrophy secondary to pulmonary arterial hypertension. Circ. Heart. Fail. 2013, 6, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Lambert, M.; Capuano, V.; Olschewski, A.; Sabourin, J.; Nagaraj, C.; Girerd, B.; Weatherald, J.; Humbert, M.; Antigny, F. Ion Channels in Pulmonary Hypertension: A Therapeutic Interest? Int. J. Mol. Sci. 2018, 19, 3162. [Google Scholar] [CrossRef]
- Roth, R.A.; Dotzlaf, L.A.; Baranyi, B.; Kuo, C.H.; Hook, J.B. Effect of monocrotaline ingestion on liver, kidney, and lung of rats. Toxicol. Appl. Pharm. 1981, 60, 193–203. [Google Scholar] [CrossRef]
- Liu, R.; Zhang, Q.; Luo, Q.; Qiao, H.; Wang, P.; Yu, J.; Cao, Y.; Lu, B.; Qu, L. Norepinephrine stimulation of alpha1D-adrenoceptor promotes proliferation of pulmonary artery smooth muscle cells via ERK-1/2 signaling. Int. J. Biochem. Cell. Biol. 2017, 88, 100–112. [Google Scholar] [CrossRef]
- Komaki, F.; Akiyama, T.; Yamazaki, T.; Kitagawa, H.; Nosaka, S.; Shirai, M. Effects of intravenous magnesium infusion on in vivo release of acetylcholine and catecholaminein rat adrenal medulla. Auton. Neurosci. 2013, 177, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Humbert, M.; Guignabert, C.; Bonnet, S.; Dorfmüller, P.; Klinger, J.R.; Nicolls, M.R.; Olschewski, A.J.; Pullamsetti, S.S.; Schermuly, R.T.; Stenmark, K.R.; et al. Pathology and pathobiology of pulmonary hypertension: State of the art and research perspectives. Eur. Respir. J. 2019, 53, 1801887. [Google Scholar] [CrossRef] [PubMed]
- Provencher, S.; Archer, S.L.; Ramirez, F.D.; Hibbert, B.; Paulin, R.; Boucherat, O.; Lacasse, Y.; Bonnet, S. Standards and methodological rigor in pulmonary arterial hypertension preclinical and translational research. Circ. Res. 2018, 122, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.; Jan, W.C.; Tsai, P.S.; Huang, C.J. Magnesium sulfate mitigates acute lung injury in endotoxemia rats. J. Trauma. 2011, 70, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Cheng, Y.; Zong, L.; Huang, L.; Qiao, C.; Li, W.; Gong, B.; Hu, J.; Liu, H.; Wang, X.; et al. Aspirin attenuates monocrotaline-induced pulmonary arterial hypertension in rats by suppressing the erk/mapk pathway. Clin. Exp. Hypertens. 2017, 39, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Peng, T.C.; Chang, C.Y.; Huang, I.T.; Kao, M.C.; Chang, Y.Y.; Huang, C.J. Platonin mitigates vascular hyporeactivity of thoracic aorta in septic rats. J. Surg. Res. 2018, 221, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Stumm, C.L.; Halcsik, E.; Landgraf, R.G.; Camara, N.O.; Sogayar, M.C.; Jancar, S. Lung remodeling in a mouse model of asthma involves a balance between tgf-beta1 and bmp-7. PLoS ONE 2014, 9, e95959. [Google Scholar] [CrossRef] [PubMed]
- Mogilner, J.G.; Lurie, M.; Coran, A.G.; Nativ, O.; Shiloni, E.; Sukhotnik, I. Effect of diclofenac on germ cell apoptosis following testicular ischemia-reperfusion injury in a rat. Pediatr. Surg. Int. 2006, 22, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Christia, P.; Bujak, M.; Gonzalez-Quesada, C.; Chen, W.; Dobaczewski, M.; Reddy, A.; Frangogiannis, N.G. Systematic characterization of myocardial inflammation, repair, and remodeling in a mouse model of reperfused myocardial infarction. J. Histochem. Cytochem. 2013, 61, 555–570. [Google Scholar] [CrossRef]
- Zungu-Edmondson, M.; Shults, N.V.; Melnyk, O.; Suzuki, Y.J. Natural reversal of pulmonary vascular remodeling and right ventricular remodeling in SU5416/hypoxia-treated Sprague-Dawley rats. PLoS ONE 2017, 12, e0182551. [Google Scholar] [CrossRef]
- Karnati, S.; Graulich, T.; Oruqaj, G.; Pfreimer, S.; Seimetz, M.; Stamme, C.; Mariani, T.J.; Weissmann, N.; Muhlfeld, C.; Baumgart-Vogt, E. Postnatal development of the bronchiolar club cells of distal airways in the mouse lung: Stereological and molecular biological studies. Cell. Tissue. Res. 2016, 364, 543–557. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, C.-Y.; Shih, H.-J.; Huang, I.-T.; Tsai, P.-S.; Chen, K.-Y.; Huang, C.-J. Magnesium Sulfate Mitigates the Progression of Monocrotaline Pulmonary Hypertension in Rats. Int. J. Mol. Sci. 2019, 20, 4622. https://doi.org/10.3390/ijms20184622
Chang C-Y, Shih H-J, Huang I-T, Tsai P-S, Chen K-Y, Huang C-J. Magnesium Sulfate Mitigates the Progression of Monocrotaline Pulmonary Hypertension in Rats. International Journal of Molecular Sciences. 2019; 20(18):4622. https://doi.org/10.3390/ijms20184622
Chicago/Turabian StyleChang, Chao-Yuan, Hung-Jen Shih, I-Tao Huang, Pei-Shan Tsai, Kung-Yen Chen, and Chun-Jen Huang. 2019. "Magnesium Sulfate Mitigates the Progression of Monocrotaline Pulmonary Hypertension in Rats" International Journal of Molecular Sciences 20, no. 18: 4622. https://doi.org/10.3390/ijms20184622
APA StyleChang, C.-Y., Shih, H.-J., Huang, I.-T., Tsai, P.-S., Chen, K.-Y., & Huang, C.-J. (2019). Magnesium Sulfate Mitigates the Progression of Monocrotaline Pulmonary Hypertension in Rats. International Journal of Molecular Sciences, 20(18), 4622. https://doi.org/10.3390/ijms20184622




