Maternal Exposure to Bisphenol A Combined with High-Fat Diet-Induced Programmed Hypertension in Adult Male Rat Offspring: Effects of Resveratrol
Abstract
1. Introduction
2. Results
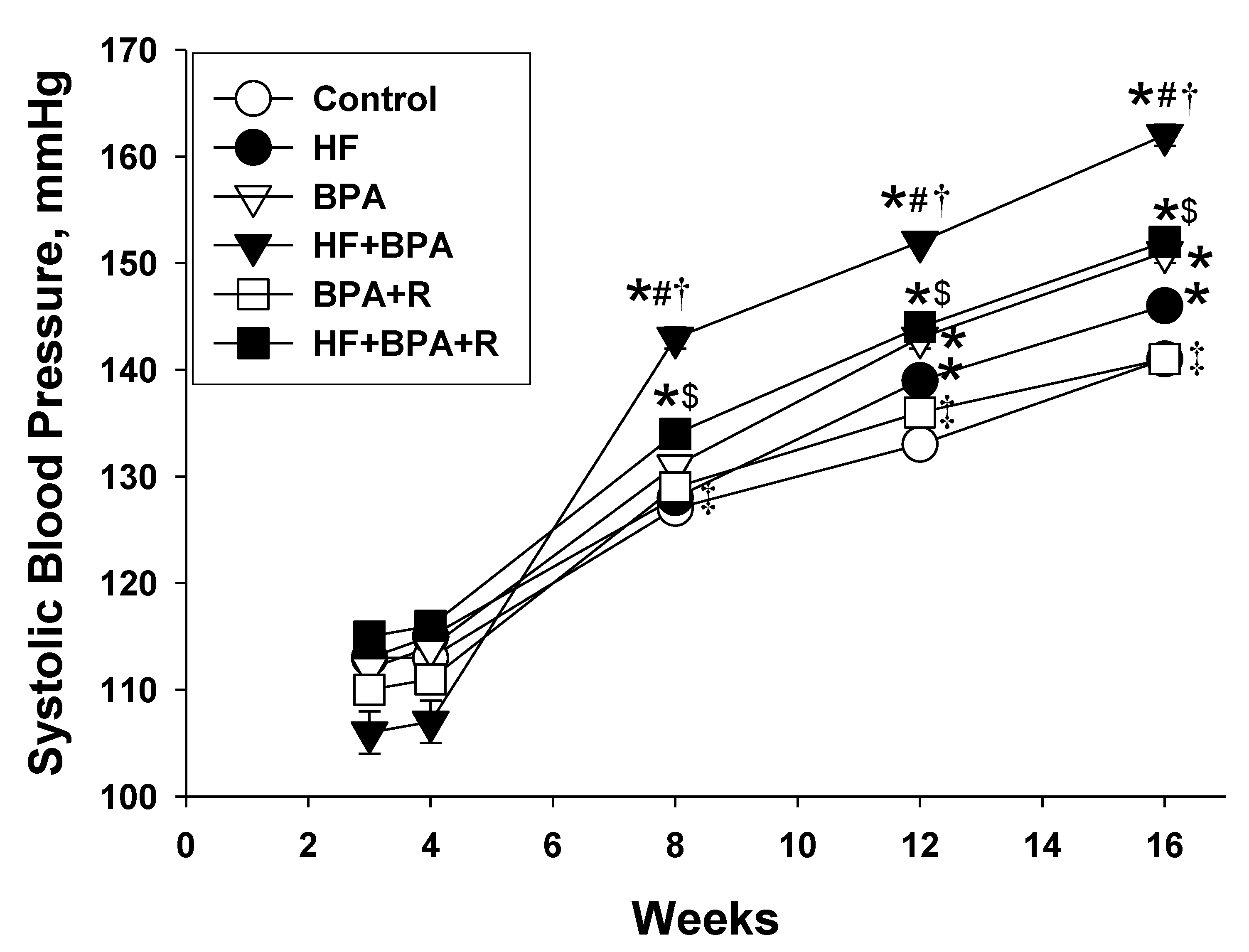
2.1. Morphometric Values and Blood Pressures
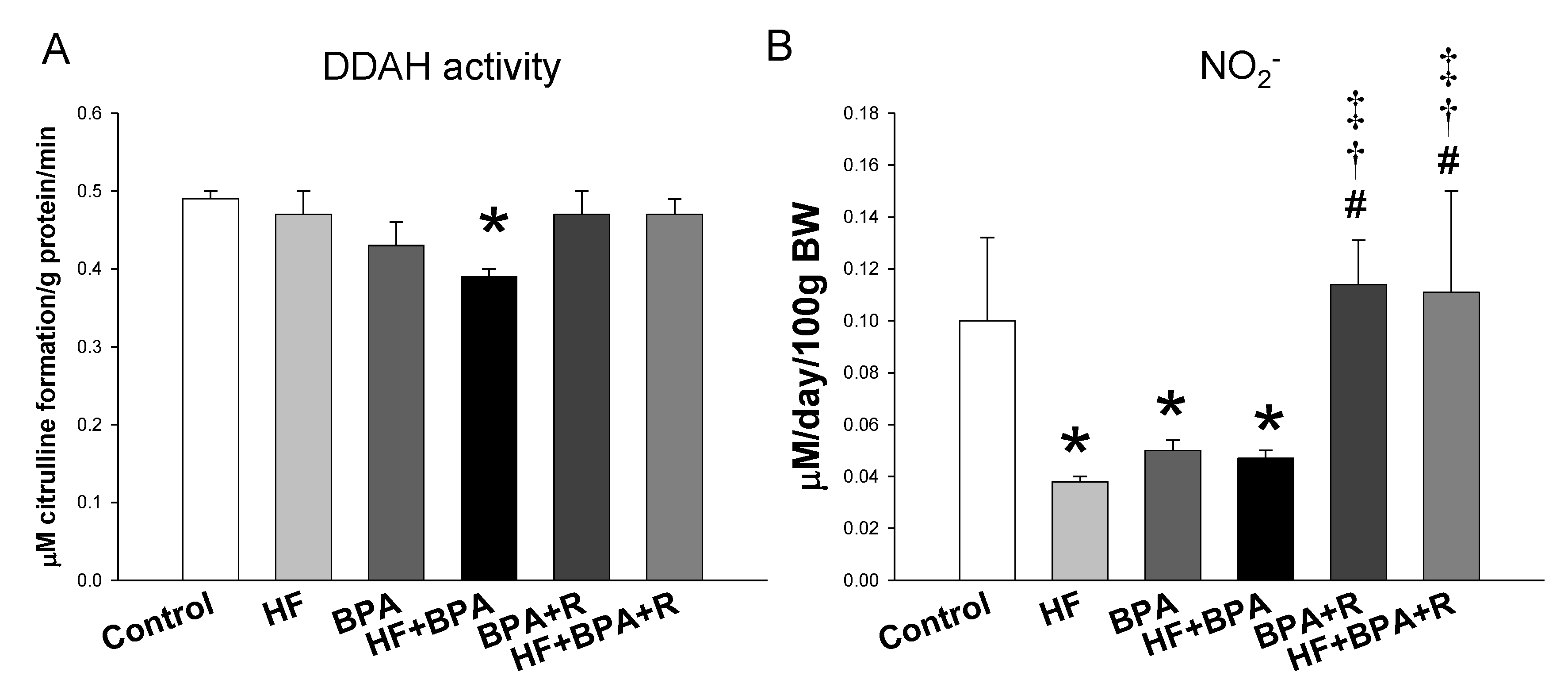
2.2. Plasma Levels of NO-Related Elements
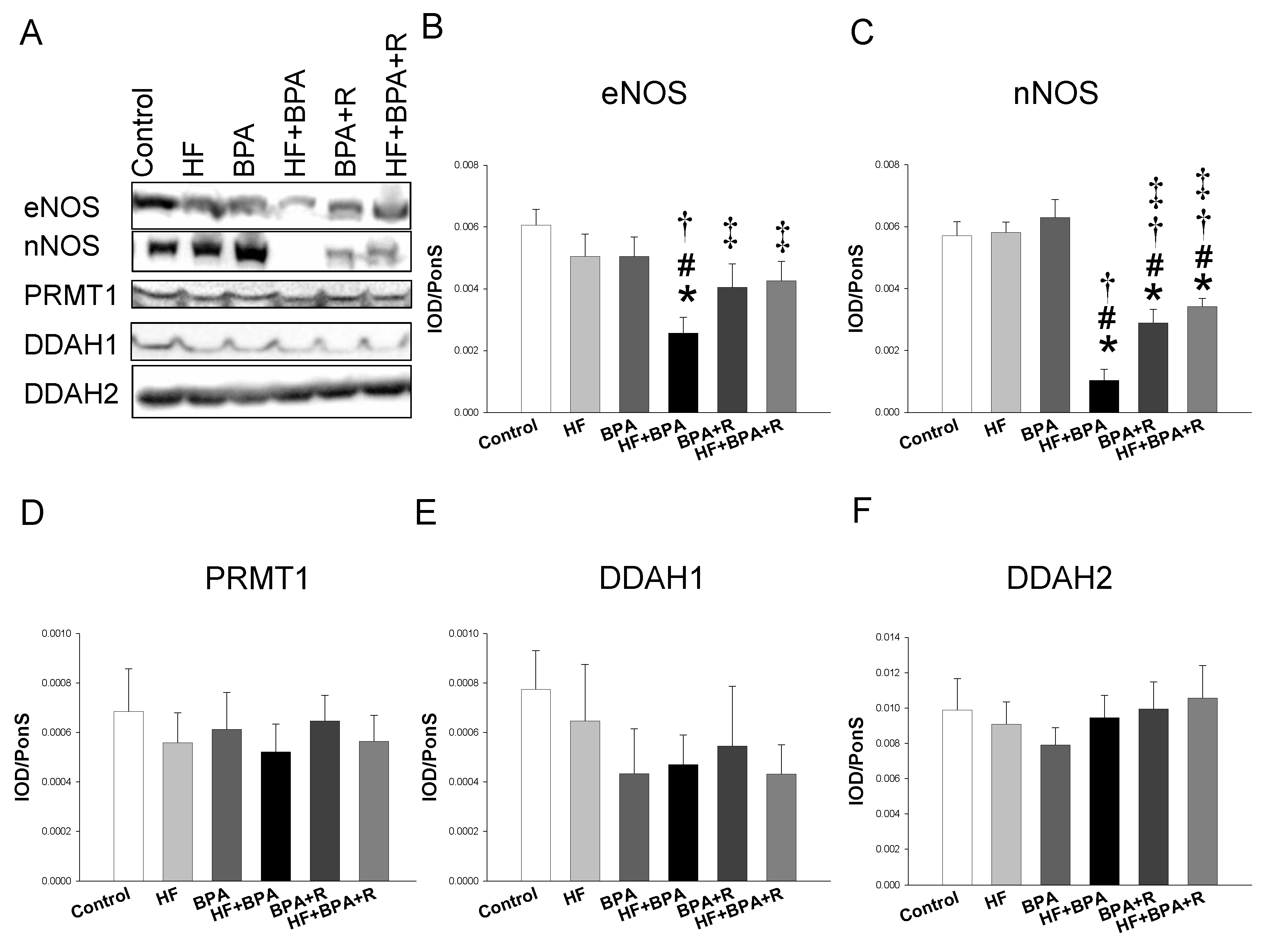
2.3. Protein Levels in the ADMA–NO Pathway
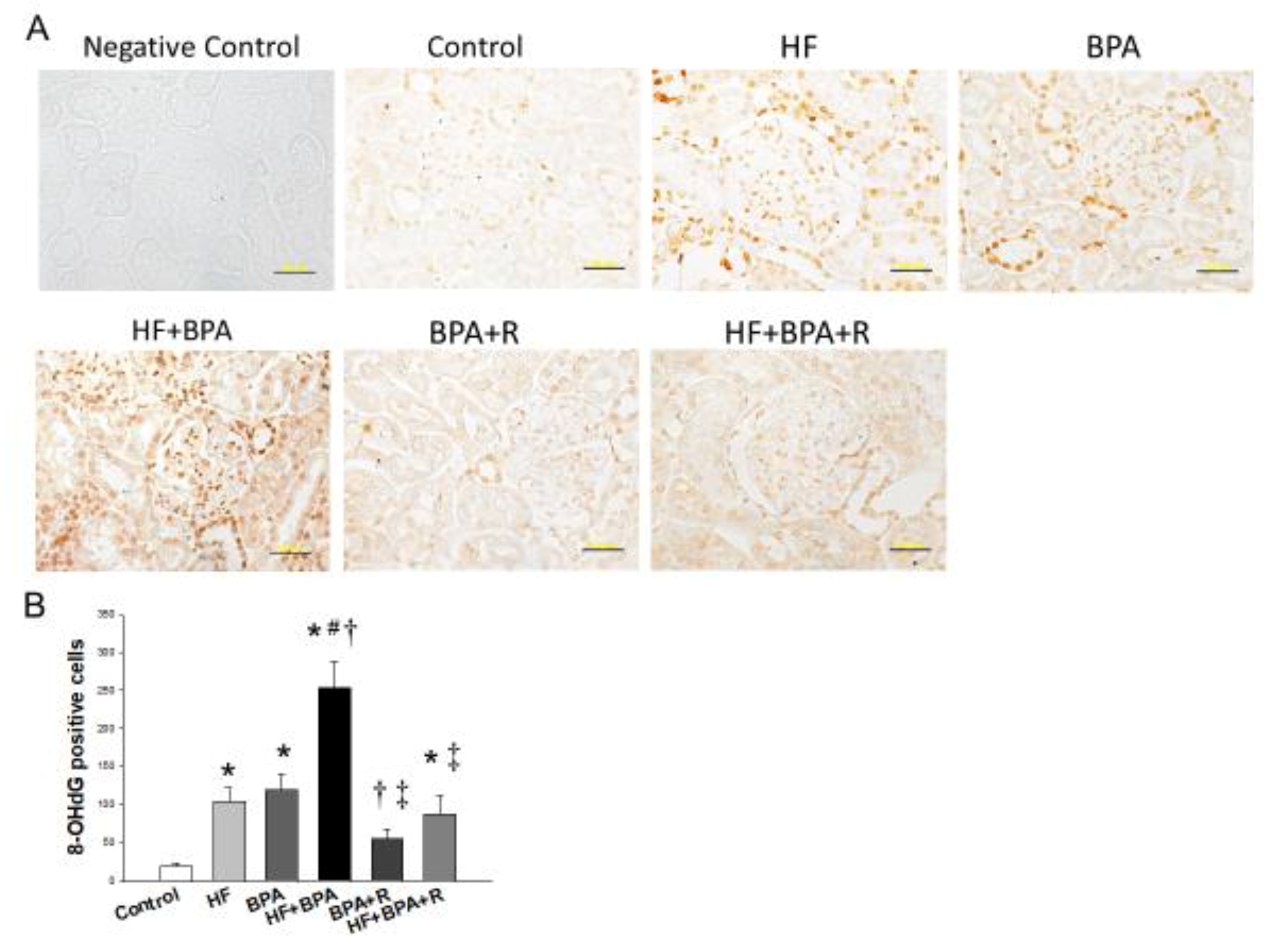
2.4. Immunohistochemistry Staining of 8-OHdG
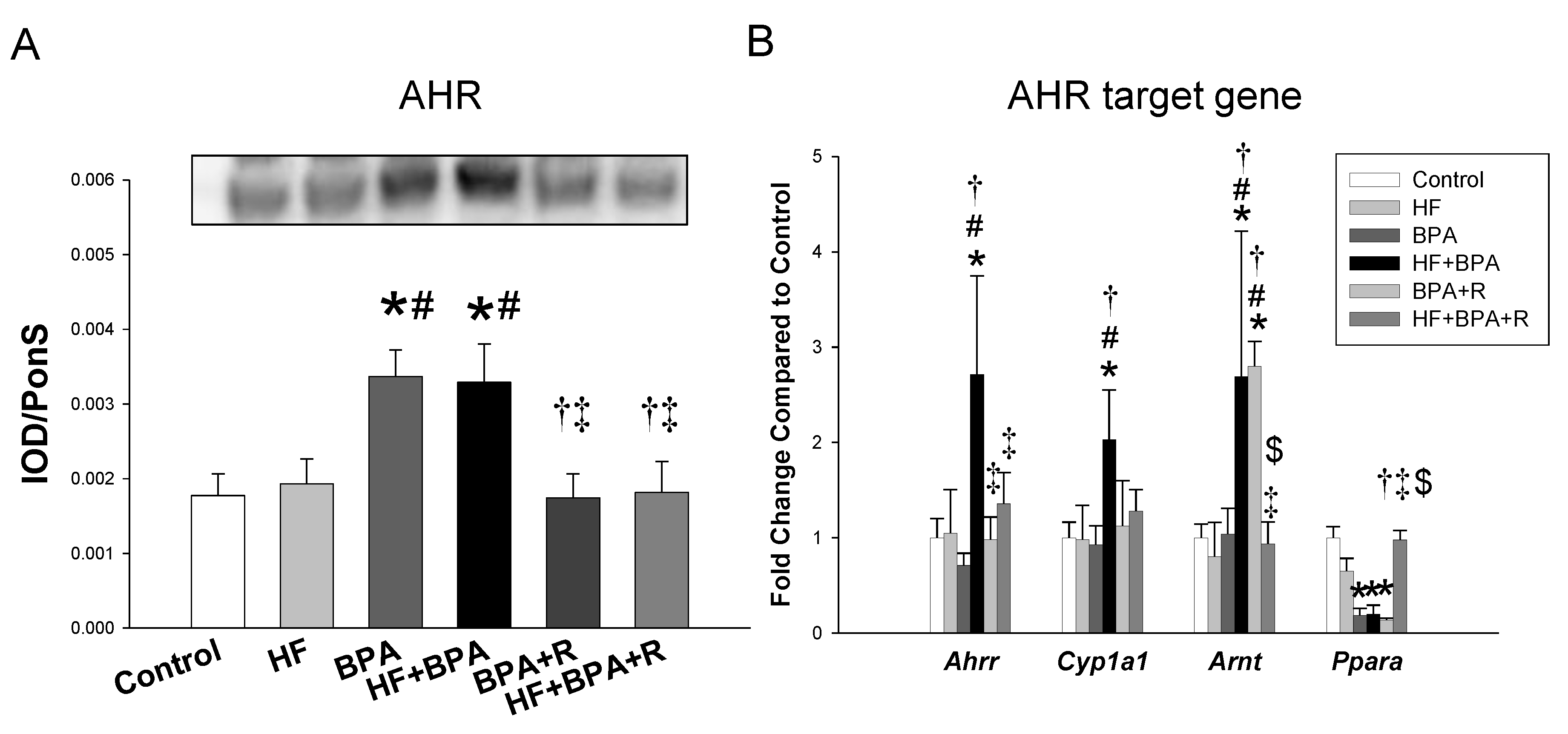
2.5. AHR and Its Target Genes
3. Discussion
4. Materials and Methods
4.1. Animal Models
4.2. High-Performance Liquid Chromatography (HPLC)
4.3. Quantitative Real-Time Polymerase Chain Reaction (PCR)
4.4. Western Blot
4.5. Immunohistochemistry Staining
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ADMA | Asymmetric dimethylarginine |
| AHR | Aryl hydrocarbon receptor |
| AHRR | Aryl hydrocarbon receptor repressor |
| ANRT | Aryl hydrocarbon receptor nuclear translocator |
| BPA | Bisphenol A |
| DDAH1 | Dimethylarginine dimethylamonihydrolase-1 |
| DDAH2 | Dimethylarginine dimethylamonihydrolase-2 |
| DOHaD | Developmental origins of health and disease |
| eNOS | Endothelial NO synthase |
| EDC | Endocrine disrupting chemical |
| nNOS | Neuronal NO synthase |
| NO | Nitric oxide |
| PRMT1 | Protein arginine methyltransferase-1 |
| ROS | Reactive oxygen species |
| SDMA | Symmetric dimethylarginine |
| TCDD | 2,3,7,8-tetrachlorodibenzo-p-dioxin |
| 8-OHdG | 8-Hydroxydeoxyguanosine |
References
- Haugen, A.C.; Schug, T.T.; Collman, G.; Heindel, J.J. Evolution of DOHaD: the impact of environmental health sciences. J. Dev. Orig. Health Dis. 2015, 6, 55–64. [Google Scholar] [CrossRef]
- Han, C.; Hong, Y.C. Bisphenol A, Hypertension, and Cardiovascular Diseases: Epidemiological, Laboratory, and Clinical Trial Evidence. Curr. Hypertens. Rep. 2016, 18, 11. [Google Scholar] [CrossRef]
- Bae, S.; Lim, Y.H.; Lee, Y.A.; Shin, C.H.; Oh, S.Y.; Hong, Y.C. Maternal Urinary Bisphenol A Concentration During Midterm Pregnancy and Children’s Blood Pressure at Age 4. Hypertension 2017, 69, 367–374. [Google Scholar] [CrossRef]
- Damjanovic, M.; Barton, M. Fat intake and cardiovascular response. Curr. Hypertens. Rep. 2008, 10, 25–31. [Google Scholar] [CrossRef]
- Tain, Y.L.; Lin, Y.J.; Sheen, J.M.; Yu, H.R.; Tiao, M.M.; Chen, C.C.; Tsai, C.C.; Huang, L.T.; Hsu, C.N. High Fat Diets Sex-Specifically Affect the Renal Transcriptome and Program Obesity, Kidney Injury, and Hypertension in the Offspring. Nutrients 2017, 9, 57. [Google Scholar] [CrossRef]
- Alonso-Magdalena, P.; Ropero, A.B.; Carrera, M.P.; Cederroth, C.R.; Baquie, M.; Gauthier, B.R.; Nef, S.; Stefani, E.; Nadal, A. Pancreatic insulin content regulation by the estrogen receptor ER alpha. PLoS ONE 2008, 3, e2069. [Google Scholar] [CrossRef]
- Muka, T.; Vargas, K.G.; Jaspers, L.; Wen, K.X.; Dhana, K.; Vitezova, A.; Nano, J.; Brahimaj, A.; Colpani, V.; Bano, A.; et al. Estrogen receptor β actions in the female cardiovascular system: A systematic review of animal and human studies. Maturitas 2016, 86, 28–43. [Google Scholar] [CrossRef]
- Zhang, N. The role of endogenous aryl hydrocarbon receptor signaling in cardiovascular physiology. J. Cardiovasc. Dis. Res. 2011, 2, 91–95. [Google Scholar] [CrossRef]
- Hsu, C.N.; Lin, Y.J.; Lu, P.C.; Tain, Y.L. Maternal Resveratrol Therapy Protects Male Rat Offspring against Programmed Hypertension Induced by TCDD and Dexamethasone Exposures: Is It Relevant to Aryl Hydrocarbon Receptor? Int. J. Mol. Sci. 2018, 19, 2459. [Google Scholar] [CrossRef]
- Tain, Y.L.; Lin, Y.J.; Sheen, J.M.; Lin, I.C.; Yu, H.R.; Huang, L.T.; Hsu, C.N. Resveratrol prevents the combined maternal plus postweaning high-fat-diets-induced hypertension in male offspring. J. Nutr. Biochem. 2017, 48, 120–127. [Google Scholar] [CrossRef]
- Tain, Y.L.; Joles, J.A. Reprogramming: A preventive strategy in hypertension focusing on the kidney. Int. J. Mol. Sci. 2016, 17, 23. [Google Scholar] [CrossRef]
- Tain, Y.L.; Hsu, C.N. Developmental Programming of the Metabolic Syndrome: Can We Reprogram with Resveratrol? Int. J. Mol. Sci. 2018, 19, 2584. [Google Scholar] [CrossRef]
- Rameshrad, M.; Imenshahidi, M.; Razavi, B.M.; Iranshahi, M.; Hosseinzadeh, H. Bisphenol A vascular toxicity: Protective effect of Vitis vinifera (grape) seed extract and resveratrol. Phytother. Res. 2018, 32, 2396–2407. [Google Scholar] [CrossRef]
- Tain, Y.L.; Hsu, C.N. Targeting on asymmetric dimethylarginine related nitric oxide-reactive oxygen species imbalance to reprogram the development of hypertension. Int. J. Mol. Sci. 2016, 17, 2020. [Google Scholar] [CrossRef]
- Bode-Böger, S.M.; Scalera, F.; Ignarro, L.J. The l-arginine paradox: importance of the l-arginine/asymmetrical dimethylarginine ratio. Pharmacol. Ther. 2007, 114, 295–306. [Google Scholar]
- Rezg, R.; El-Fazaa, S.; Gharbi, N.; Mornagui, B. Bisphenol A and human chronic diseases: current evidences, possible mechanisms, and future perspectives. Environ. Int. 2014, 64, 83–90. [Google Scholar] [CrossRef]
- Song, Y.; Chou, E.L.; Baecker, A.; You, N.Y.; Song, Y.; Sun, Q.; Liu, S. Endocrine-disrupting chemicals, risk of type 2 diabetes, and diabetes-related metabolic traits: A systematic review and meta-analysis. J. Diabetes 2015, 8, 516–532. [Google Scholar] [CrossRef]
- Shelnutt, S.; Kind, J.; Allaben, W. Bisphenol A: Update on newly developed data and how they address NTP’s 2008 finding of “Some Concern”. Food Chem. Toxicol. 2013, 57, 284–295. [Google Scholar] [CrossRef]
- Tain, Y.L.; Lee, W.C.; Leu, S.; Wu, K.; Chan, J. High salt exacerbates programmed hypertension in maternal fructose-fed male offspring. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 1146–1151. [Google Scholar] [CrossRef]
- Bonnefont-Rousselot, D. Resveratrol and Cardiovascular Diseases. Nutrients 2016, 8, 250. [Google Scholar] [CrossRef]
- Avila, J.G.; Echeverri, I.; de Plata, C.A.; Castillo, A. Impact of oxidative stress during pregnancy on fetal epigenetic patterns and early origin of vascular diseases. Nutr. Rev. 2015, 73, 12–21. [Google Scholar] [CrossRef]
- Kobroob, A.; Peerapanyasut, W.; Chattipakorn, N.; Wongmekiat, O. Damaging Effects of Bisphenol A on the Kidney and the Protection by Melatonin: Emerging Evidences from In Vivo and In Vitro Studies. Oxid. Med. Cell Longev. 2018, 2018, 3082438. [Google Scholar] [CrossRef]
- Noguchi, S.; Nakatsuka, M.; Asagiri, K.; Habara, T.; Takata, M.; Konishi, H.; Kudo, T. Bisphenol A stimulates NO synthesis through a non-genomic estrogen receptor-mediated mechanism in mouse endothelial cells. Toxicol. Lett. 2002, 135, 95–101. [Google Scholar] [CrossRef]
- Huang, L.T.; Hsieh, C.S.; Chang, K.A.; Tain, Y.L. Roles of nitric oxide and asymmetric dimethylarginine in pregnancy and fetal programming. Int. J. Mol. Sci. 2012, 13, 14606–14622. [Google Scholar] [CrossRef]
- Mulero-Navarro, S.; Fernandez-Salguero, P.M. New trends in aryl hydrocarbon receptor biology. Front. Cell Dev. Biol. 2016, 4, 45. [Google Scholar] [CrossRef]
- Wada, T.; Sunaga, H.; Miyata, K.; Shirasaki, H.; Uchiyama, Y.; Shimba, S. Aryl Hydrocarbon Receptor Plays Protective Roles against High Fat Diet (HFD)-induced Hepatic Steatosis and the Subsequent Lipotoxicity via Direct Transcriptional Regulation of Socs3 Gene Expression. J. Biol. Chem. 2016, 291, 7004–7016. [Google Scholar] [CrossRef]
- Cottart, C.H.; Nivet-Antoine, V.; Laguillier-Morizot, C.; Beaudeux, J.L. Resveratrol bioavailability and toxicity in humans. Mol. Nutr. Food Res. 2010, 54, 7–16. [Google Scholar] [CrossRef]
- Acconcia, F.; Pallottini, V.; Marino, M. Molecular Mechanisms of Action of BPA. Dose Response 2015, 13, 1559325815610582. [Google Scholar] [CrossRef]
- Sandberg, K.; Ji, H. Sex differences in primary hypertension. Biol. Sex Differ. 2012, 3, 7. [Google Scholar] [CrossRef]
- Wei, J.; Sun, X.; Chen, Y.; Li, Y.; Song, L.; Zhou, Z.; Xu, B.; Lin, Y.; Xu, S. Perinatal exposure to bisphenol A exacerbates nonalcoholic steatohepatitis-like phenotype in male rat offspring fed on a high-fat diet. J. Endocrinol. 2014, 222, 313–325. [Google Scholar] [CrossRef]
- Chen, H.E.; Lin, Y.J.; Lin, I.C.; Yu, H.R.; Sheen, J.M.; Tsai, C.C.; Huang, L.T.; Tain, Y.L. Resveratrol prevents combined prenatal N(G)-nitro-l-arginine-methyl ester (l-NAME) treatment plus postnatal high-fat diet induced programmed hypertension in adult rat offspring: interplay between nutrient-sensing signals, oxidative stress and gut microbiota. J. Nutr. Biochem. 2019, 70, 28–37. [Google Scholar]
| Control | HF | BPA | HF + BPA | BPA + R | HF + BPA + R | |
|---|---|---|---|---|---|---|
| n | 8 | 8 | 8 | 8 | 8 | 8 |
| Mortality | 0% | 0% | 0% | 12.5% | 0% | 0% |
| Body weight (BW) (g) | 553 ± 12 | 533 ± 21 | 501 ± 11 | 558 ± 16 | 553 ± 11 | 525 ± 13 |
| Left kidney weight (g) | 2.22 ± 0.1 | 1.96 ± 0.07 | 2.06 ± 0.09 | 2.21 ± 0.06 | 2.26 ± 0.08 | 2.00 ± 0.06 |
| Left kidney weight/100g BW | 0.4 ± 0.02 | 0.37 ± 0.01 | 0.41 ± 0.01 | 0.40 ± 0.02 | 0.41 ± 0.01 | 0.38 ± 0.01 |
| Systolic blood pressure (mmHg) | 141 ± 1 | 146 ± 0.4 * | 151 ± 1 *# | 162 ± 1 *#† | 141 ± 1 #†‡ | 152 ± 1 *#$ |
| Control | HF | BPA | HF + BPA | BPA + R | HF + BPA + R | |
|---|---|---|---|---|---|---|
| l-citrulline (μM) | 56.4 ± 5.8 | 45 ± 2 | 52.9 ± 3.6 | 42.8 ± 4.5 | 48.8 ± 8.6 | 67.3 ± 4.3 ‡ |
| l-arginine (μM) | 153.6 ± 12.8 | 125.7 ± 9.6 | 137.1 ± 12.9 | 141.9 ± 7.2 | 175 ± 17 | 191.7 ± 11.7 #† |
| ADMA (μM) | 1.66 ± 0.21 | 1.36 ± 0.1 | 2.14 ± 0.22 | 2.67 ± 0.27 *#† | 2.26 ± 0.21 #‡ | 2.8 ± 0.2 *# |
| SDMA (μM) | 0.38 ± 0.07 | 0.31 ± 0.05 | 1.01 ± 0.12 *# | 0.74 ± 0.16*# | 0.58 ± 0.1 † | 0.7 ± 0.1 # |
| l-arginine-to-ADMA ratio (μM/μM) | 99.3 ± 10.8 | 97.2 ± 12.9 | 66.2 ± 6.6 | 58.2 ± 9.4*# | 79.5 ± 7 | 69.9 ± 6.6 |
| Gene | Forward (5′–3′) | Reverse (5′–3′) |
|---|---|---|
| Ahrr | cagcaacatggcttctttca | tgaagcactgcattccagac |
| Cyp1a1 | gcactctggacaaacacctg | atatccaccttctcgcctgg |
| Arnt | gtctccctcccagatgatga | gctggtagccaacagtagcc |
| Rn18s | gccgcggtaattccagctcca | cccgcccgctcccaagatc |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, C.-N.; Lin, Y.-J.; Tain, Y.-L. Maternal Exposure to Bisphenol A Combined with High-Fat Diet-Induced Programmed Hypertension in Adult Male Rat Offspring: Effects of Resveratrol. Int. J. Mol. Sci. 2019, 20, 4382. https://doi.org/10.3390/ijms20184382
Hsu C-N, Lin Y-J, Tain Y-L. Maternal Exposure to Bisphenol A Combined with High-Fat Diet-Induced Programmed Hypertension in Adult Male Rat Offspring: Effects of Resveratrol. International Journal of Molecular Sciences. 2019; 20(18):4382. https://doi.org/10.3390/ijms20184382
Chicago/Turabian StyleHsu, Chien-Ning, Yu-Ju Lin, and You-Lin Tain. 2019. "Maternal Exposure to Bisphenol A Combined with High-Fat Diet-Induced Programmed Hypertension in Adult Male Rat Offspring: Effects of Resveratrol" International Journal of Molecular Sciences 20, no. 18: 4382. https://doi.org/10.3390/ijms20184382
APA StyleHsu, C.-N., Lin, Y.-J., & Tain, Y.-L. (2019). Maternal Exposure to Bisphenol A Combined with High-Fat Diet-Induced Programmed Hypertension in Adult Male Rat Offspring: Effects of Resveratrol. International Journal of Molecular Sciences, 20(18), 4382. https://doi.org/10.3390/ijms20184382






