Mice Exposed to Combined Chronic Low-Dose Irradiation and Modeled Microgravity Develop Long-Term Neurological Sequelae
Abstract
1. Introduction
2. Results
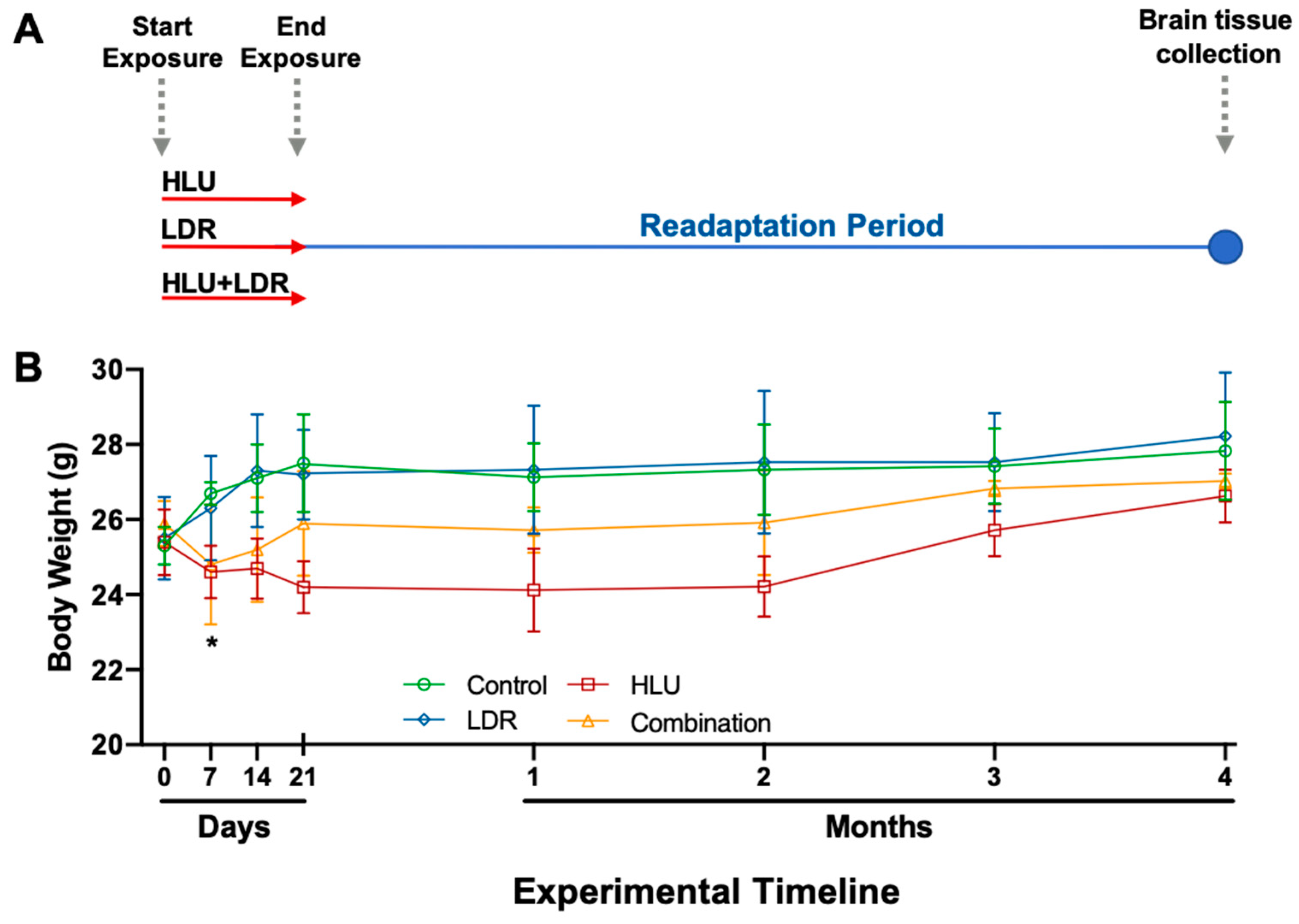
2.1. Experimental Design and Mouse Body Weights Across Experimental and Readaptation Timepoints
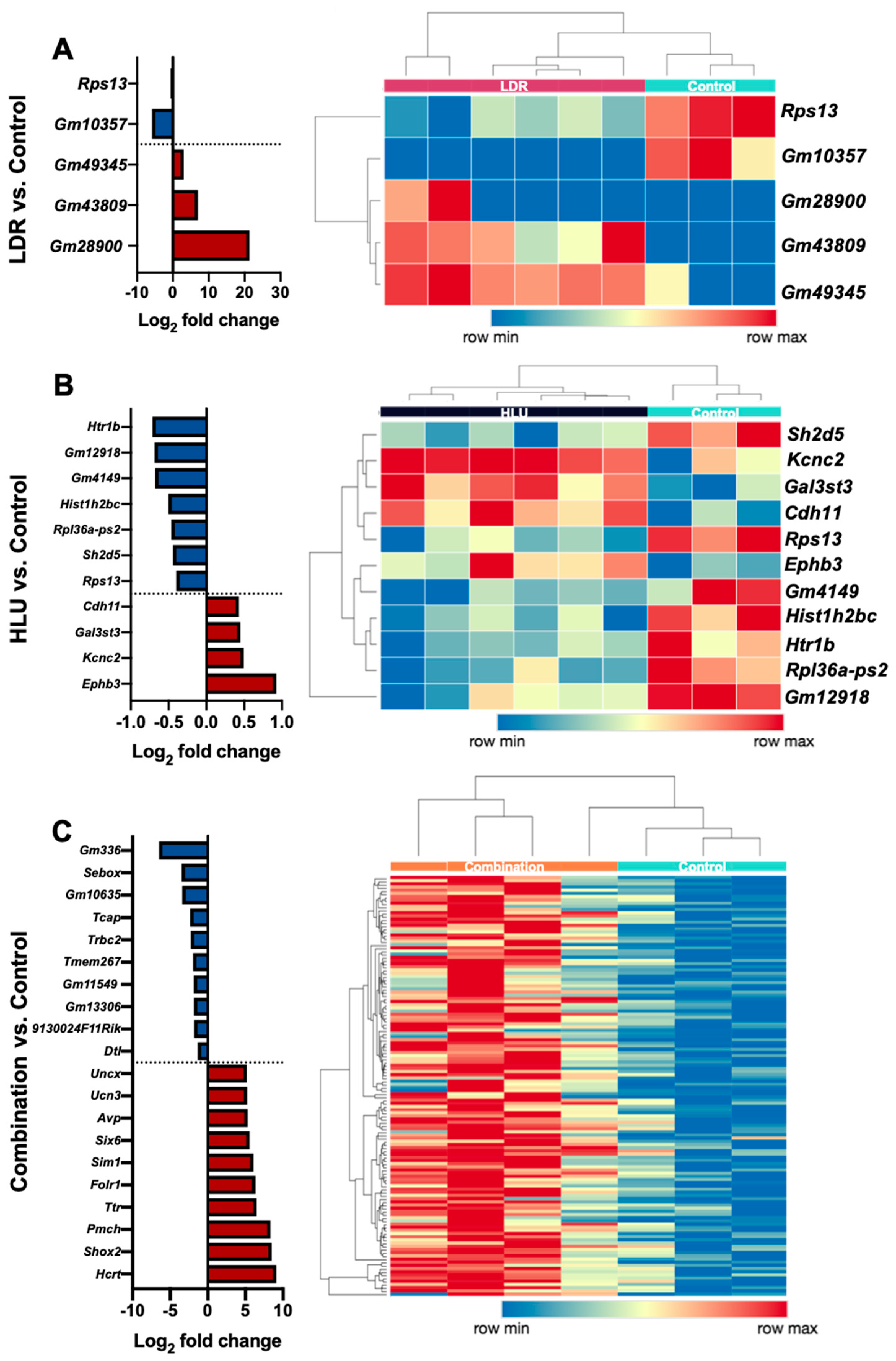
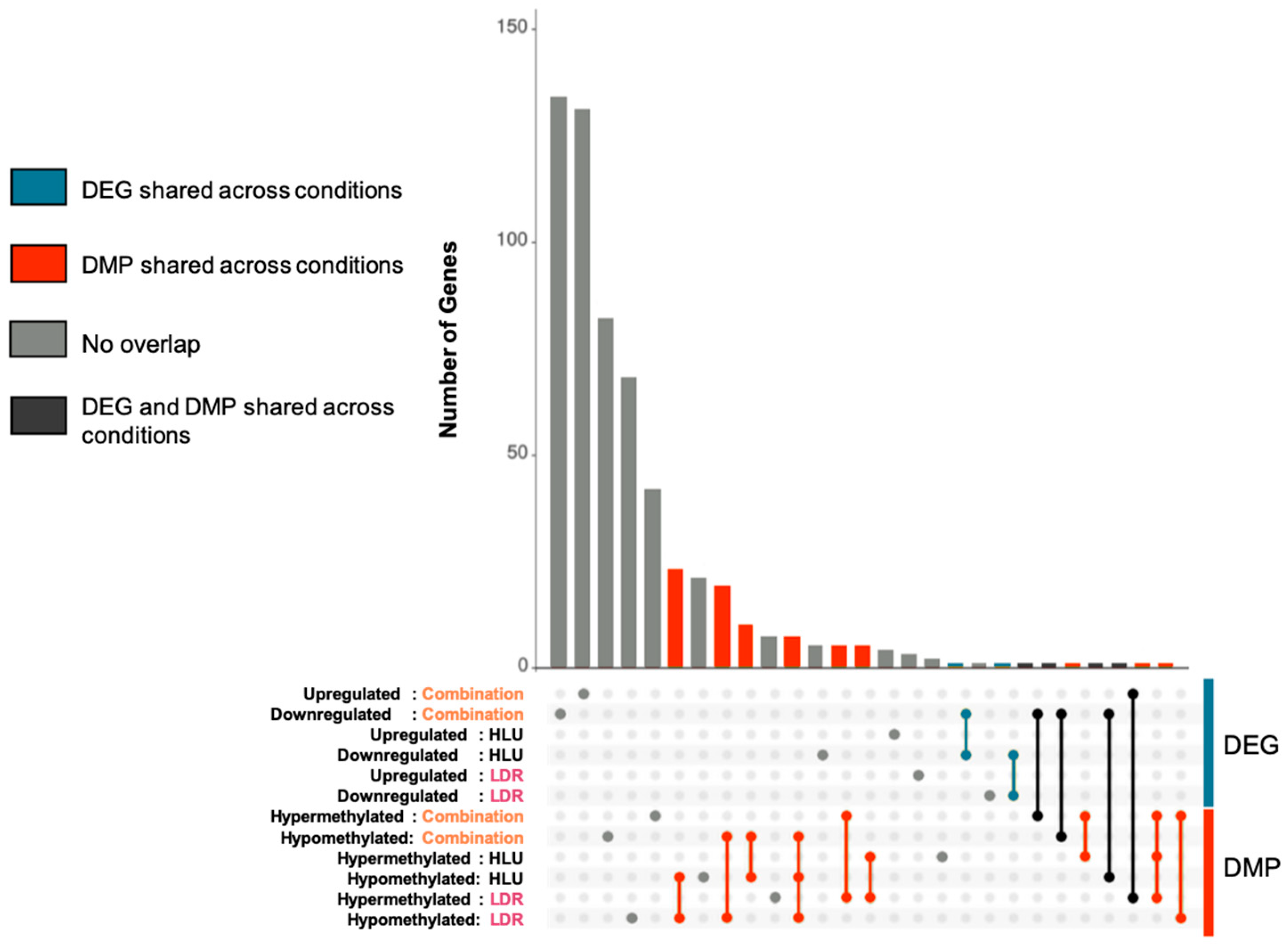
2.2. LDR, HLU and Combined HLU+LDR Exposures Displayed Minimal Gene and Promoter Methylation Overlap at 4 Months Readaptation
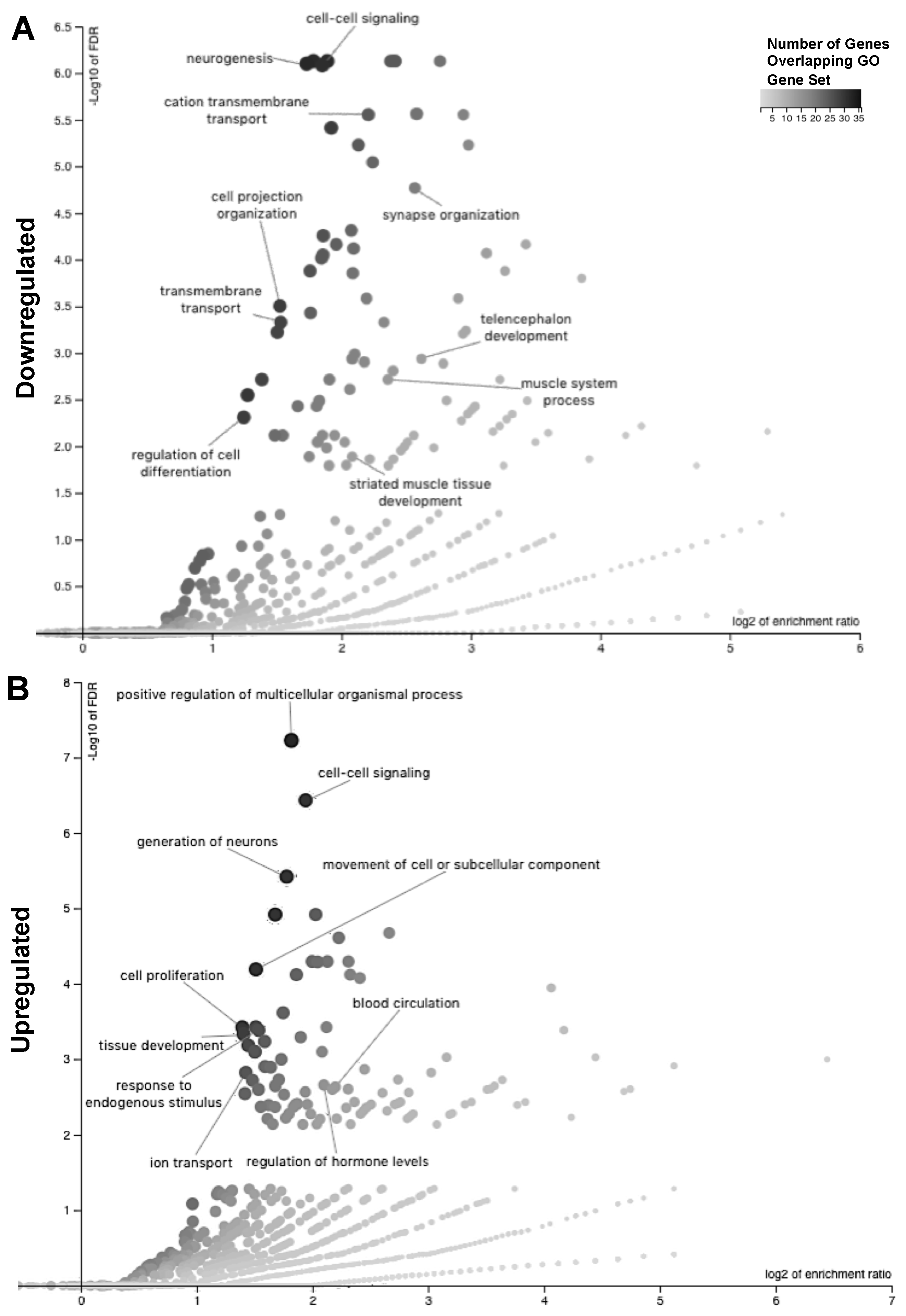
2.3. Readaptation at 4 Months Following Exposure to Combined HLU+LDR Involves Gene Expression Changes in Multiple Brain-Associated Pathways
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Experimental Conditions
4.3. Hindlimb Unloading and Whole-Body Irradiation
4.4. Euthanasia, Dissection, Tissue Storage, and Nucleic Acid Extraction
4.5. RNA Sequencing
4.6. Reduced Representation Bisulfite Sequencing (RRBS)
4.7. Differential Expression and Pathway Enrichment Analysis
4.8. Differential Methylation Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kovalchuk, A.; Kolb, B. Low dose radiation effects on the brain-from mechanisms and behavioral outcomes to mitigation strategies. Cell Cycle 2017, 16, 1266–1270. [Google Scholar] [CrossRef]
- Lawley, J.S.; Petersen, L.G.; Howden, E.J.; Sarma, S.; Cornwell, W.K.; Zhang, R.; Whitworth, L.A.; Williams, M.A.; Levine, B.D. Effect of gravity and microgravity on intracranial pressure. J. Physiol. 2017, 595, 2115–2127. [Google Scholar] [CrossRef]
- Chancellor, J.C.; Scott, G.B.I.; Sutton, J.P. Space Radiation: The Number One Risk to Astronaut Health beyond Low Earth Orbit. Life 2014, 4, 491–510. [Google Scholar] [CrossRef]
- Seawright, J.W.; Samman, Y.; Sridharan, V.; Mao, X.W.; Cao, M.; Singh, P.; Melnyk, S.; Koturbash, I.; Nelson, G.A.; Hauer-Jensen, M.; et al. Effects of low-dose rate γ-irradiation combined with simulated microgravity on markers of oxidative stress, DNA methylation potential, and remodeling in the mouse heart. PloS ONE 2017, 12, e0180594. [Google Scholar] [CrossRef]
- De la Torre, G.G. Cognitive neuroscience in space. Life 2014, 4, 281–294. [Google Scholar] [CrossRef]
- Garrett-Bakelman, F.E.; Darshi, M.; Green, S.J.; Gur, R.C.; Lin, L.; Macias, B.R.; McKenna, M.J.; Meydan, C.; Mishra, T.; Nasrini, J.; et al. The NASA Twins Study: A multidimensional analysis of a year-long human spaceflight. Science 2019, 364, eaau8650. [Google Scholar]
- Lee, J.K.; Koppelmans, V.; Riascos, R.F.; Hasan, K.M.; Pasternak, O.; Mulavara, A.P.; Bloomberg, J.J.; Seidler, R.D. Spaceflight-Associated Brain White Matter Microstructural Changes and Intracranial Fluid Redistribution. JAMA Neurol. 2019, 76, 412–419. [Google Scholar] [CrossRef]
- Ronca, A.E.; Moyer, E.L.; Talyansky, Y.; Lowe, M.; Padmanabhan, S.; Choi, S.; Gong, C.; Cadena, S.M.; Stodieck, L.; Globus, R.K. Behavior of mice aboard the International Space Station. Sci. Rep. 2019, 9, 4717. [Google Scholar] [CrossRef]
- Bellone, J.A.; Gifford, P.S.; Nishiyama, N.C.; Hartman, R.E.; Mao, X.W. Long-term effects of simulated microgravity and/or chronic exposure to low-dose gamma radiation on behavior and blood-brain barrier integrity. NPJ Microgravity 2016, 2, 16019. [Google Scholar] [CrossRef]
- Mao, X.W.; Nishiyama, N.C.; Pecaut, M.J.; Campbell-Beachler, M.; Gifford, P.; Haynes, K.E.; Becronis, C.; Gridley, D.S. Simulated Microgravity and Low-Dose/Low-Dose-Rate Radiation Induces Oxidative Damage in the Mouse Brain. Radiat. Res. 2016, 185, 647–657. [Google Scholar] [CrossRef]
- Mao, X.W.; Nishiyama, N.C.; Campbell-Beachler, M.; Gifford, P.; Haynes, K.E.; Gridley, D.S.; Pecaut, M.J. Role of NADPH Oxidase as a Mediator of Oxidative Damage in Low-Dose Irradiated and Hindlimb-Unloaded Mice. Radiat. Res. 2017, 188, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Smart, D. Radiation Toxicity in the Central Nervous System: Mechanisms and Strategies for Injury Reduction. Semin. Radiat. Oncol. 2017, 27, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Cucinotta, F.A.; Cacao, E. Risks of Cognitive Detriments after Low Dose Heavy Ion and Proton Exposures. Int. J. Radiat. Biol. 2019, 1–36. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.W. Low dose (0.04 Gy) irradiation (LDR) and hindlimb unloading (HLU) microgravity in mice: Brain transcriptomic and epigenomic data. In NASA GeneLab; 2018. Available online: https://genelab-data.ndc.nasa.gov/genelab/accession/GLDS-202 (accessed on 18 July 2019).
- Chatterjee, S.; Pietrofesa, R.A.; Park, K.; Tao, J.-Q.; Carabe-Fernandez, A.; Berman, A.T.; Koumenis, C.; Sielecki, T.; Christofidou-Solomidou, M. LGM2605 Reduces Space Radiation-Induced NLRP3 Inflammasome Activation and Damage in In Vitro Lung Vascular Networks. Int. J. Mol. Sci. 2019, 20, 176. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Mathelier, A. Intervene: A tool for intersection and visualization of multiple gene or genomic region sets. BMC Bioinformatics 2017, 18, 287. [Google Scholar] [CrossRef] [PubMed]
- Mizumatsu, S.; Monje, M.L.; Morhardt, D.R.; Rola, R.; Palmer, T.D.; Fike, J.R. Extreme sensitivity of adult neurogenesis to low doses of X-irradiation. Cancer Res. 2003, 63, 4021–4027. [Google Scholar] [PubMed]
- Najjar, S.; Pearlman, D.M.; Devinsky, O.; Najjar, A.; Zagzag, D. Neurovascular unit dysfunction with blood-brain barrier hyperpermeability contributes to major depressive disorder: A review of clinical and experimental evidence. J. Neuroinflammation 2013, 10, 142. [Google Scholar] [CrossRef] [PubMed]
- Song, T.-T.; Bi, Y.-H.; Gao, Y.-Q.; Huang, R.; Hao, K.; Xu, G.; Tang, J.-W.; Ma, Z.-Q.; Kong, F.-P.; Coote, J.H.; et al. Systemic pro-inflammatory response facilitates the development of cerebral edema during short hypoxia. J. Neuroinflammation 2016, 13, 63. [Google Scholar] [CrossRef]
- Li, L.; Zhang, H.; Varrin-Doyer, M.; Zamvil, S.S.; Verkman, A.S. Proinflammatory role of aquaporin-4 in autoimmune neuroinflammation. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2011, 25, 1556–1566. [Google Scholar] [CrossRef]
- Ikeshima-Kataoka, H. Neuroimmunological Implications of AQP4 in Astrocytes. Int. J. Mol. Sci. 2016, 17, 1306. [Google Scholar] [CrossRef]
- Meli, R.; Pirozzi, C.; Pelagalli, A. New Perspectives on the Potential Role of Aquaporins (AQPs) in the Physiology of Inflammation. Front. Physiol. 2018, 9, 101. [Google Scholar] [CrossRef] [PubMed]
- Kotas, M.E.; Medzhitov, R. Homeostasis, inflammation, and disease susceptibility. Cell 2015, 160, 816–827. [Google Scholar] [CrossRef]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef]
- Lowe, X.R.; Bhattacharya, S.; Marchetti, F.; Wyrobek, A.J. Early brain response to low-dose radiation exposure involves molecular networks and pathways associated with cognitive functions, advanced aging and Alzheimer’s disease. Radiat. Res. 2009, 171, 53–65. [Google Scholar] [CrossRef]
- Paul, A.M.; Acharya, D.; Duty, L.; Thompson, E.A.; Le, L.; Stokic, D.S.; Leis, A.A.; Bai, F. Osteopontin facilitates West Nile virus neuroinvasion via neutrophil “Trojan horse” transport. Sci. Rep. 2017, 7, 4722. [Google Scholar] [CrossRef]
- Yamazaki, Y.; Shinohara, M.; Shinohara, M.; Yamazaki, A.; Murray, M.E.; Liesinger, A.M.; Heckman, M.G.; Lesser, E.R.; Parisi, J.E.; Petersen, R.C.; et al. Selective loss of cortical endothelial tight junction proteins during Alzheimer’s disease progression. Brain, J. Neurol. 2019, 142, 1077–1092. [Google Scholar] [CrossRef]
- Zhao, C.; Deng, W.; Gage, F.H. Mechanisms and functional implications of adult neurogenesis. Cell 2008, 132, 645–660. [Google Scholar] [CrossRef]
- Zhang, J.; Jiao, J. Molecular Biomarkers for Embryonic and Adult Neural Stem Cell and Neurogenesis. BioMed Res. Int. 2015, 2015, 727542. [Google Scholar] [CrossRef]
- Sun, X.Z.; Inouye, M.; Fukui, Y.; Hisano, S.; Sawada, K.; Muramatsu, H.; Muramatsu, T. An immunohistochemical study of radial glial cells in the mouse brain prenatally exposed to gamma-irradiation. J. Neuropathol. Exp. Neurol. 1997, 56, 1339–1348. [Google Scholar] [CrossRef][Green Version]
- Kleindienst, A.; McGinn, M.J.; Harvey, H.B.; Colello, R.J.; Hamm, R.J.; Bullock, M.R. Enhanced hippocampal neurogenesis by intraventricular S100B infusion is associated with improved cognitive recovery after traumatic brain injury. J. Neurotrauma 2005, 22, 645–655. [Google Scholar] [CrossRef]
- Sun, D.; Bullock, M.R.; McGinn, M.J.; Zhou, Z.; Altememi, N.; Hagood, S.; Hamm, R.; Colello, R.J. Basic fibroblast growth factor-enhanced neurogenesis contributes to cognitive recovery in rats following traumatic brain injury. Exp. Neurol. 2009, 216, 56–65. [Google Scholar] [CrossRef]
- Jandial, R.; Hoshide, R.; Waters, J.D.; Limoli, C.L. Space-brain: The negative effects of space exposure on the central nervous system. Surg. Neurol. Int. 2018, 9, 9. [Google Scholar]
- Grammaticos, P.; Giannoula, E.; Fountos, G.P. Acute radiation syndrome and chronic radiation syndrome. Hell. J. Nucl. Med. 2013, 16, 56–59. [Google Scholar]
- Walker, S.A.; Townsend, L.W.; Norbury, J.W. Heavy ion contributions to organ dose equivalent for the 1977 galactic cosmic ray spectrum. Adv. Space Res. 2013, 51, 1792–1799. [Google Scholar] [CrossRef]
- Guide for the Care and Use of Laboratory Animals, 8th ed.; National Research Council (U.S.), Institute for Laboratory Animal Research (U.S.), National Academies Press (U.S.), Eds.; National Academies Press: Washington, DC, USA, 2011; ISBN 978-0-309-15400-0. [Google Scholar]
- Morey-Holton, E.; Globus, R.K.; Kaplansky, A.; Durnova, G. The hindlimb unloading rat model: Literature overview, technique update and comparison with space flight data. Adv. Space Biol. Med. 2005, 10, 7–40. [Google Scholar]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet.journal 2011, 17, 10. [Google Scholar] [CrossRef]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 2011, 12, 323. [Google Scholar] [CrossRef]
- Soneson, C.; Love, M.I.; Robinson, M.D. Differential analyses for RNA-seq: Transcript-level estimates improve gene-level inferences. F1000Research 2016, 4, 1521. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Liao, Y.; Wang, J.; Jaehnig, E.J.; Shi, Z.; Zhang, B. WebGestalt 2019: Gene set analysis toolkit with revamped UIs and APIs. Nucleic Acids Res. 2019, 47, W199–W205. [Google Scholar] [CrossRef] [PubMed]
- Fabregat, A.; Jupe, S.; Matthews, L.; Sidiropoulos, K.; Gillespie, M.; Garapati, P.; Haw, R.; Jassal, B.; Korninger, F.; May, B.; et al. The Reactome Pathway Knowledgebase. Nucleic Acids Res. 2018, 46, D649–D655. [Google Scholar] [CrossRef]
- Krueger, F.; Andrews, S.R. Bismark: A flexible aligner and methylation caller for Bisulfite-Seq applications. Bioinformatics 2011, 27, 1571–1572. [Google Scholar] [CrossRef] [PubMed]
- Assenov, Y.; Müller, F.; Lutsik, P.; Walter, J.; Lengauer, T.; Bock, C. Comprehensive analysis of DNA methylation data with RnBeads. Nat. Methods 2014, 11, 1138–1140. [Google Scholar] [CrossRef] [PubMed]

| DMP | DEG | |||||
|---|---|---|---|---|---|---|
| Total DMP | Hypomethylated DMP | Hypermethylated DMP | TotalDEG | Upregulated DEG | Downregulated DEG | |
| Radiation vs. Control | 137 promoters | 118 promoters | 19 promoters | Gm28900 | Gm28900 | Rps13 |
| Gm43809 | Gm43809 | Gm10357 | ||||
| Gm49345 | Gm49345 | |||||
| Rps13 | ||||||
| Gm10357 | ||||||
| Unloading vs. Control | 71 promoters | 62 promoters | Gm35801 | Ephb3 | Ephb3 | Gm12918 |
| Gm13066 | Kcnc2 | Kcnc2 | Gm4149 | |||
| Cnpy1 | Cdh11 | Cdh11 | Hist1h2bc | |||
| Cbfa2t2-ps1 | Gal3st3 | Gal3st3 | Htr1b | |||
| Gm15700 | Gm12918 | Sh2d5 | ||||
| Gm27679 | Gm4149 | Rps13 | ||||
| Hbq1a | Hist1h2bc | Rpl36a-ps2 | ||||
| 1700019N19Rik | Htr1b | |||||
| Gm6024 | Sh2d5 | |||||
| Rps13 | ||||||
| Rpl36a-ps2 | ||||||
| Combined vs. Control | 170 promoters | 119 promoters | 51 promoters | 270 genes | 132 genes | 138 genes |
| DEG/DEG Overlap | |
| Condition | Gene |
| Down HLU+LDR/Down HLU | Gm12918 |
| Down HLU/Down LDR | Rps13 |
| DMP/DMP Overlap | |
| Condition | Methylated promoter of gene |
| Hypo LDR/Hypo HLU | Gm38049; Zfp648; 1700018C11Rik; |
| B230104I21Rik; Cald1; Lmod3 | |
| Smco3; Angpt2; Gm28515 | |
| Sln; Gm27159; Gm15271 | |
| 4933405E24Rik; Ndel1; Gm11373 | |
| Gm5083; Cdhr2; Mir8097 | |
| 1700011A15Rik; Krt8; Mir8094 | |
| Pcdh12; Tmem180 | |
| Hypo LDR/Hypo HLU+LDR | Gm16564; Gm37267; Adamtsl2 |
| Gm13413; Bbox1; Gm11805 | |
| 1700071G01Rik; Prr33; Cdhr4 | |
| Slfn4; Krt19; Gm6401 | |
| Gm15516; Mx2; Gm10268 | |
| Gm25301; Armcx2; Gm15726 | |
| Gm15247 | |
| Hypo HLU/Hypo HLU+LDR | Gm29100; Nav1; Gm29282 |
| Cfi; Mypopos; Adgb | |
| Gm26535; Abi3; Gm6969 | |
| Lpar6 | |
| Hyper LDR/Hyper HLU+LDR | Otos; Sys1; Klhl34 |
| Crh; Pcdhb12 | |
| Hyper HLU/Hyper HLU+LDR | Gm35801 |
| Hypo LDR/Hyper HLU+LDR | Gm6987 |
| Hyper HLU/Hyper LDR | 1700019N19Rik; Cnpy1; Gm6024 |
| Gm15700; Gm27679 | |
| Hypo LDR/Hypo HLU/Hypo HLU+LDR | Trp53inp2; Gm22518; Dio1 |
| Mir704; Gm805; Gm24998 | |
| Gm15648 | |
| Hyper LDR/Hyper HLU/Hyper HLU+LDR | Cbfa2t2-ps1 |
| DEG/DMP Overlap | |
| Condition | Gene or Methylated promoter of gene |
| Down HLU+LDR/Hyper HLU+LDR | Gm7120 |
| Down HLU+LDR/Hypo HLU+LDR | Ephx4 |
| Down HLU+LDR/Hypo HLU | Blnk |
| Up HLU+LDR/Hyper LDR | Rgag4 |
| Pathway Name | Associated Mouse Genes | Mouse Pathway ID | Human Pathway ID |
|---|---|---|---|
| Neuronal System | Gria3, Cacnb3, Hcn1, Camkk2, Kcnh1, Kcnma1, Kcnn1, Stx1a, Kcnv1, Kcnh5, Grin2a, Slc17a7, Dlgap1, Homer1, Snap25, Nrgn, Kcnh7, Slitrk1 | R-MMU-112316 | R-HSA-112316 |
| Potassium Channels | Kcnn1, Kcnh5, Kcnma1, Hcn1, Kcnh1, Kcnv1, Kcnh7 | R-MMU-1296071 | R-HSA-1296071 |
| Voltage Gated Potassium Channels | Kcnv1, Kcnh1, Kcnh5, Kcnh7 | R-MMU-1296072 | R-HSA-1296072 |
| cGMP effects | Pde1a | R-MMU-418457 | R-HSA-418457 |
| p75 NTR receptor-mediated signaling | Rtn4r, Obscn, Lingo1, Kalrn | R-MMU-193704 | R-HSA-193704 |
| Nitric oxide stimulates guanylate cyclase | Pde1a | R-MMU-392154 | R-HSA-392154 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Overbey, E.G.; Paul, A.M.; da Silveira, W.A.; Tahimic, C.G.T.; Reinsch, S.S.; Szewczyk, N.; Stanbouly, S.; Wang, C.; Galazka, J.M.; Mao, X.W. Mice Exposed to Combined Chronic Low-Dose Irradiation and Modeled Microgravity Develop Long-Term Neurological Sequelae. Int. J. Mol. Sci. 2019, 20, 4094. https://doi.org/10.3390/ijms20174094
Overbey EG, Paul AM, da Silveira WA, Tahimic CGT, Reinsch SS, Szewczyk N, Stanbouly S, Wang C, Galazka JM, Mao XW. Mice Exposed to Combined Chronic Low-Dose Irradiation and Modeled Microgravity Develop Long-Term Neurological Sequelae. International Journal of Molecular Sciences. 2019; 20(17):4094. https://doi.org/10.3390/ijms20174094
Chicago/Turabian StyleOverbey, Eliah G., Amber M. Paul, Willian A. da Silveira, Candice G.T. Tahimic, Sigrid S. Reinsch, Nathaniel Szewczyk, Seta Stanbouly, Charles Wang, Jonathan M. Galazka, and Xiao Wen Mao. 2019. "Mice Exposed to Combined Chronic Low-Dose Irradiation and Modeled Microgravity Develop Long-Term Neurological Sequelae" International Journal of Molecular Sciences 20, no. 17: 4094. https://doi.org/10.3390/ijms20174094
APA StyleOverbey, E. G., Paul, A. M., da Silveira, W. A., Tahimic, C. G. T., Reinsch, S. S., Szewczyk, N., Stanbouly, S., Wang, C., Galazka, J. M., & Mao, X. W. (2019). Mice Exposed to Combined Chronic Low-Dose Irradiation and Modeled Microgravity Develop Long-Term Neurological Sequelae. International Journal of Molecular Sciences, 20(17), 4094. https://doi.org/10.3390/ijms20174094






