Absence of MCP-induced Protein 1 Enhances Blood–Brain Barrier Breakdown after Experimental Stroke in Mice
Abstract
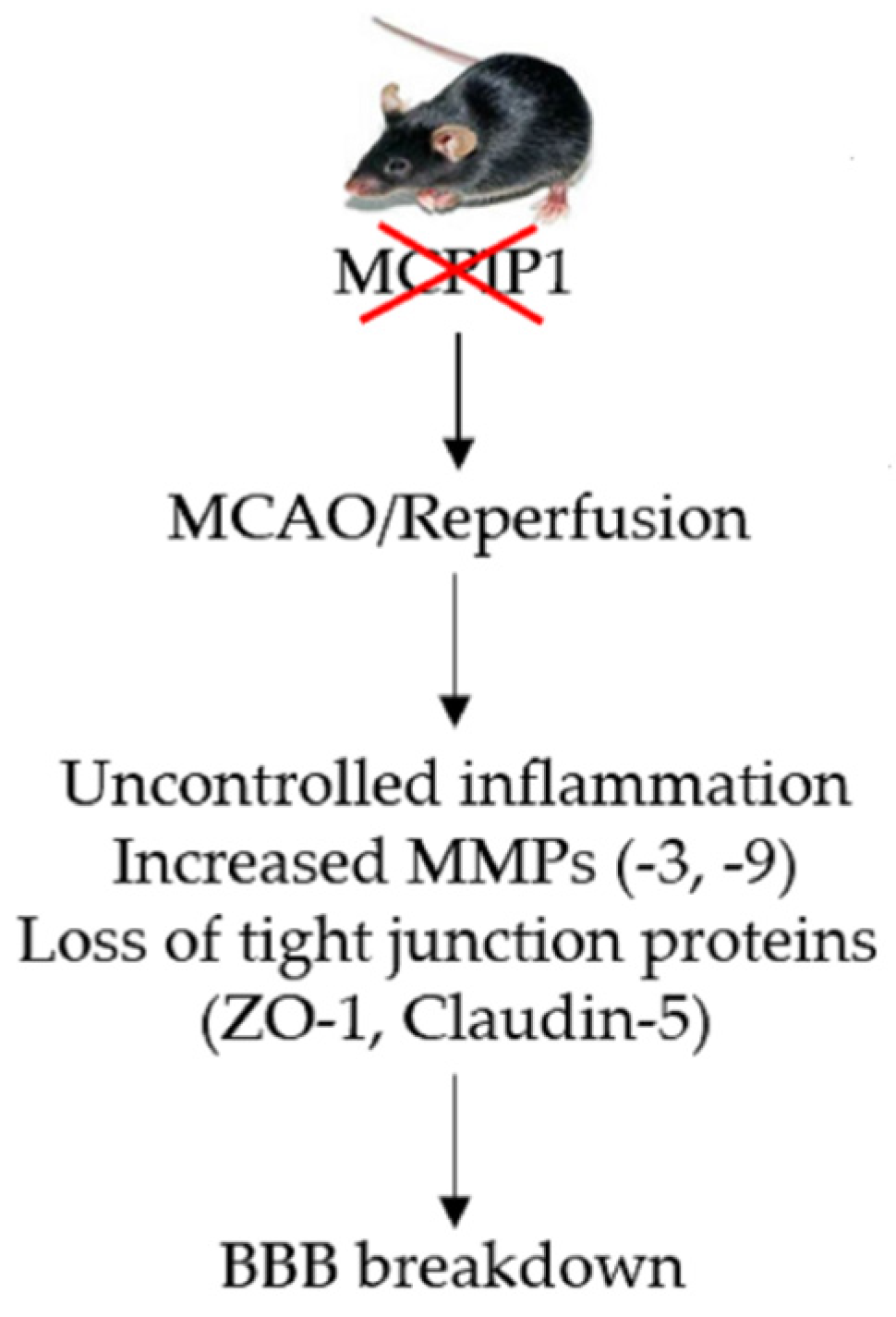
:1. Introduction
2. Results
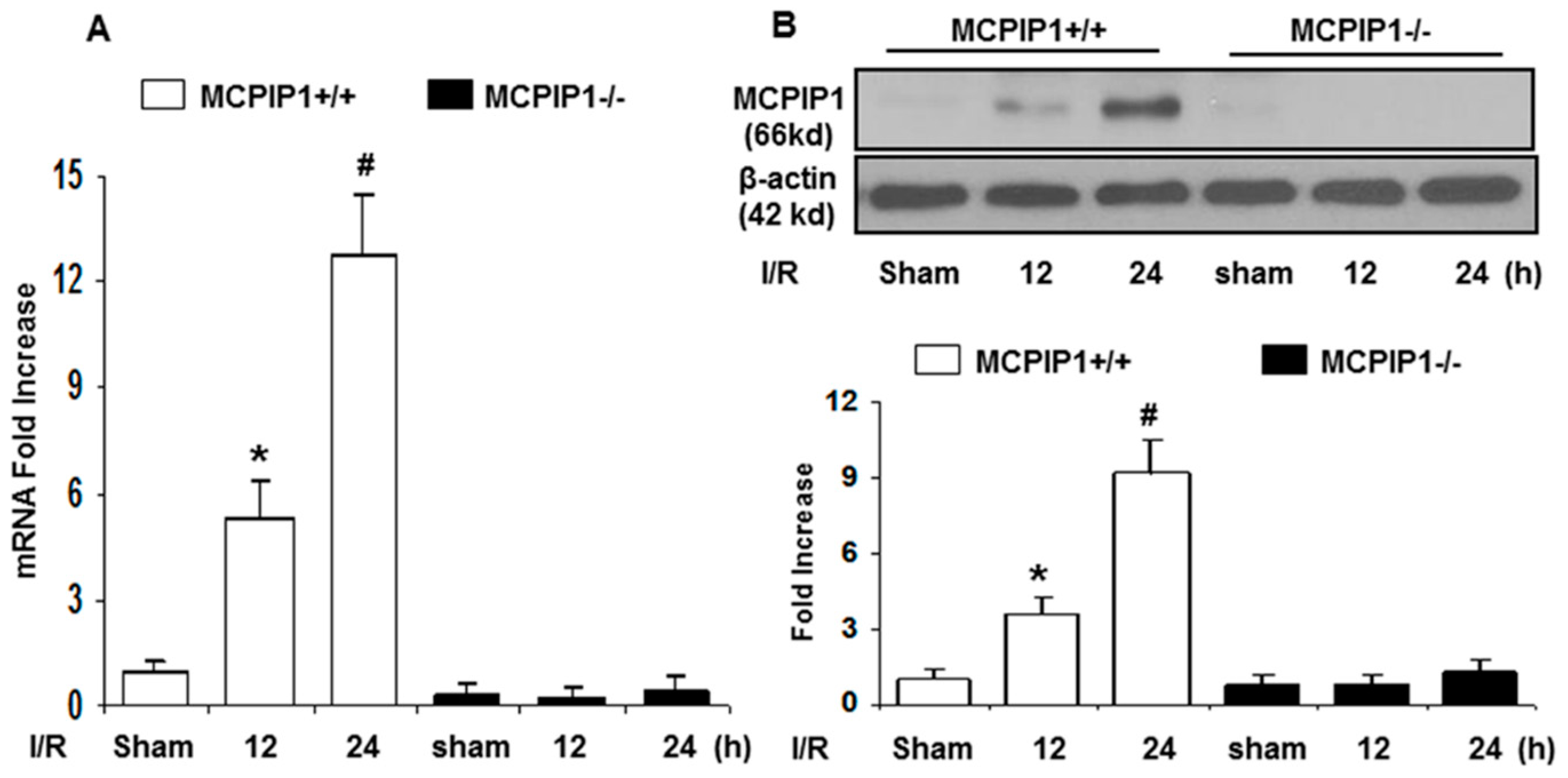
2.1. MCPIP1 Is Up-Regulated in the Murine Brain Subjected to Transient Focal Ischemia/Reperfusion (I/R) Injury
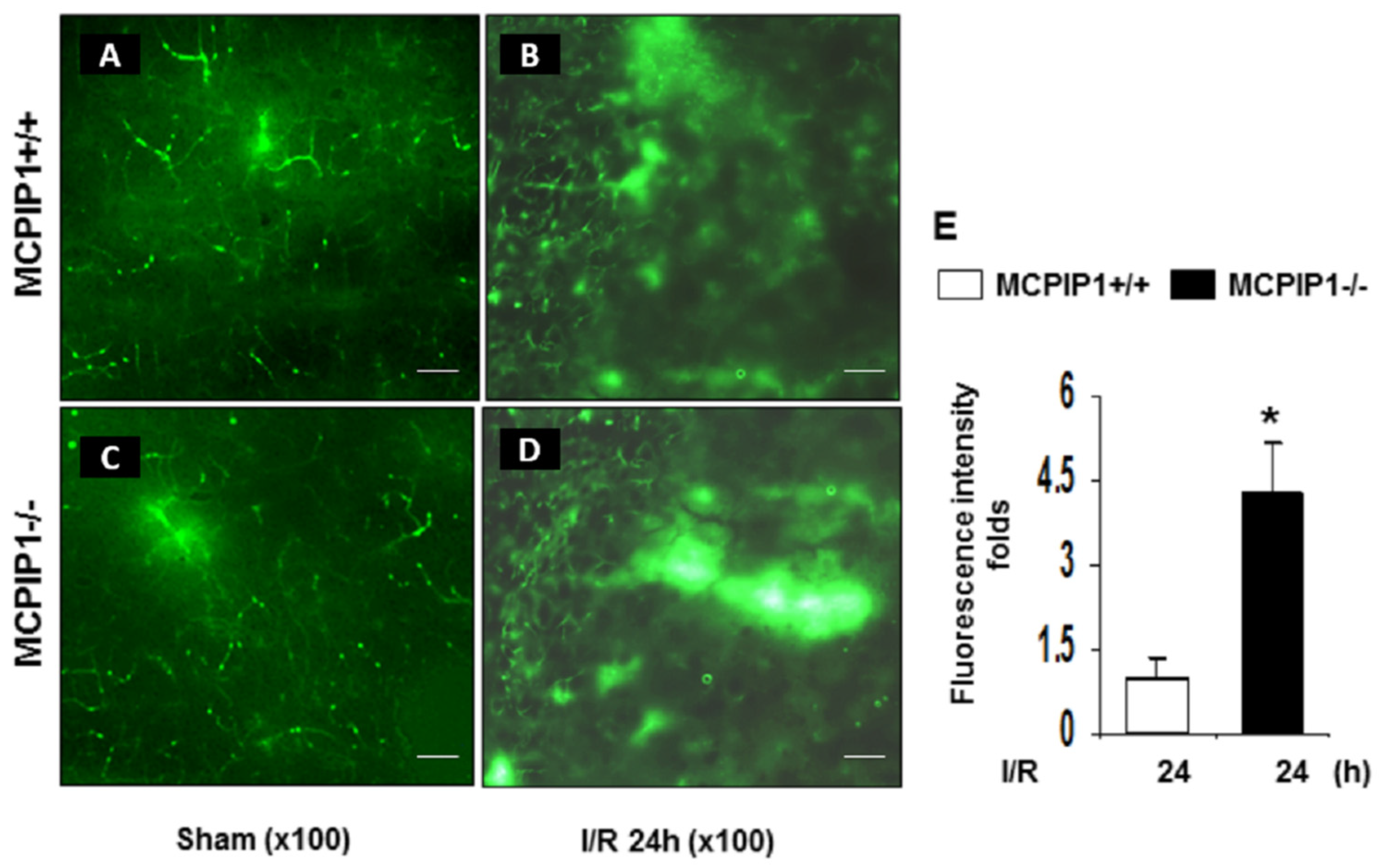
2.2. Extravasation of FITC-Dextran Is Markedly Increased in the Brains of MCPIP1–/– Mice Subjected to Transient Focal I/R Injury
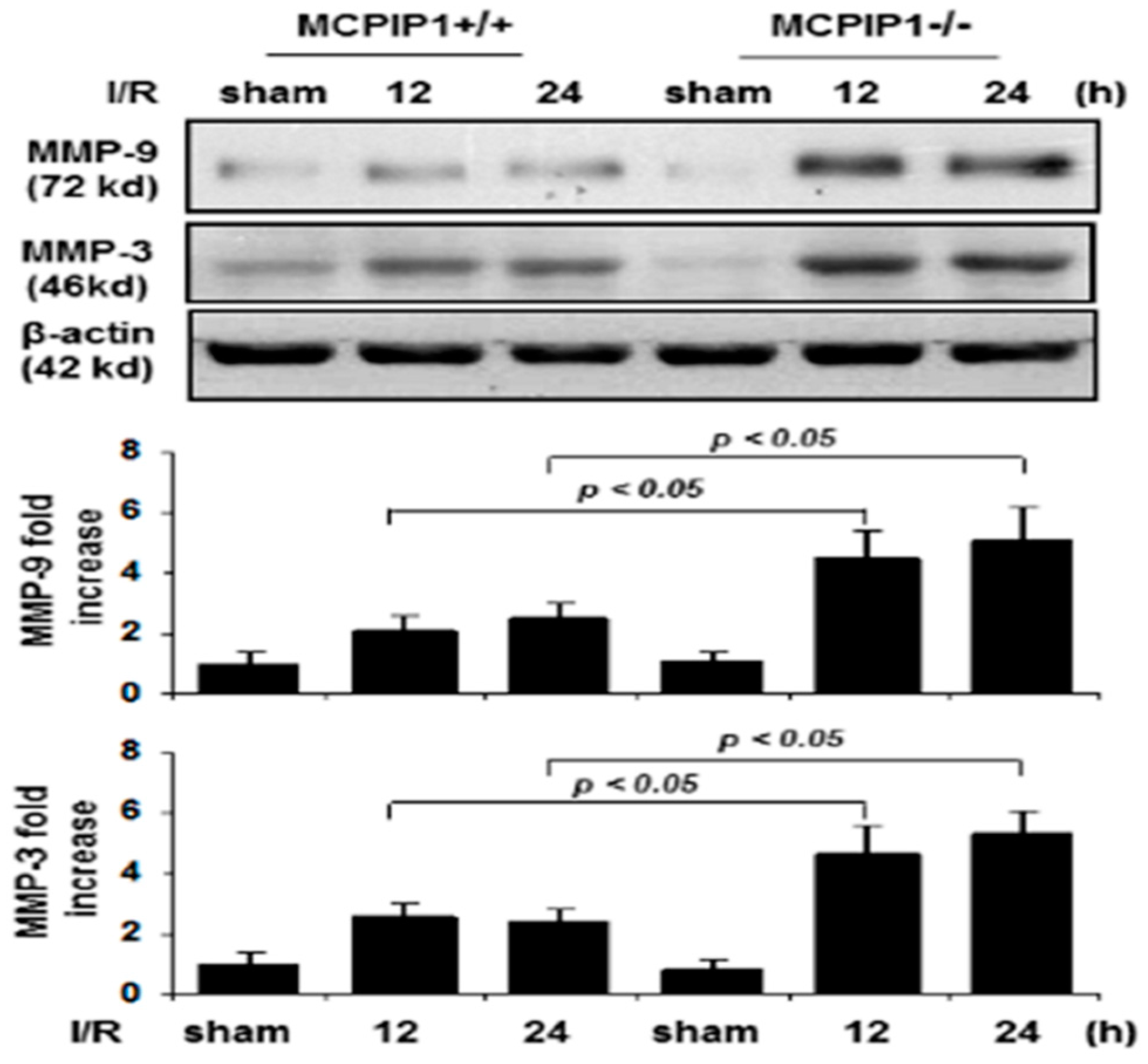
2.3. Matrix Metalloproteinase Expression in MCPIP1–/–Mice Subjected to Transient Focal I/R Injury
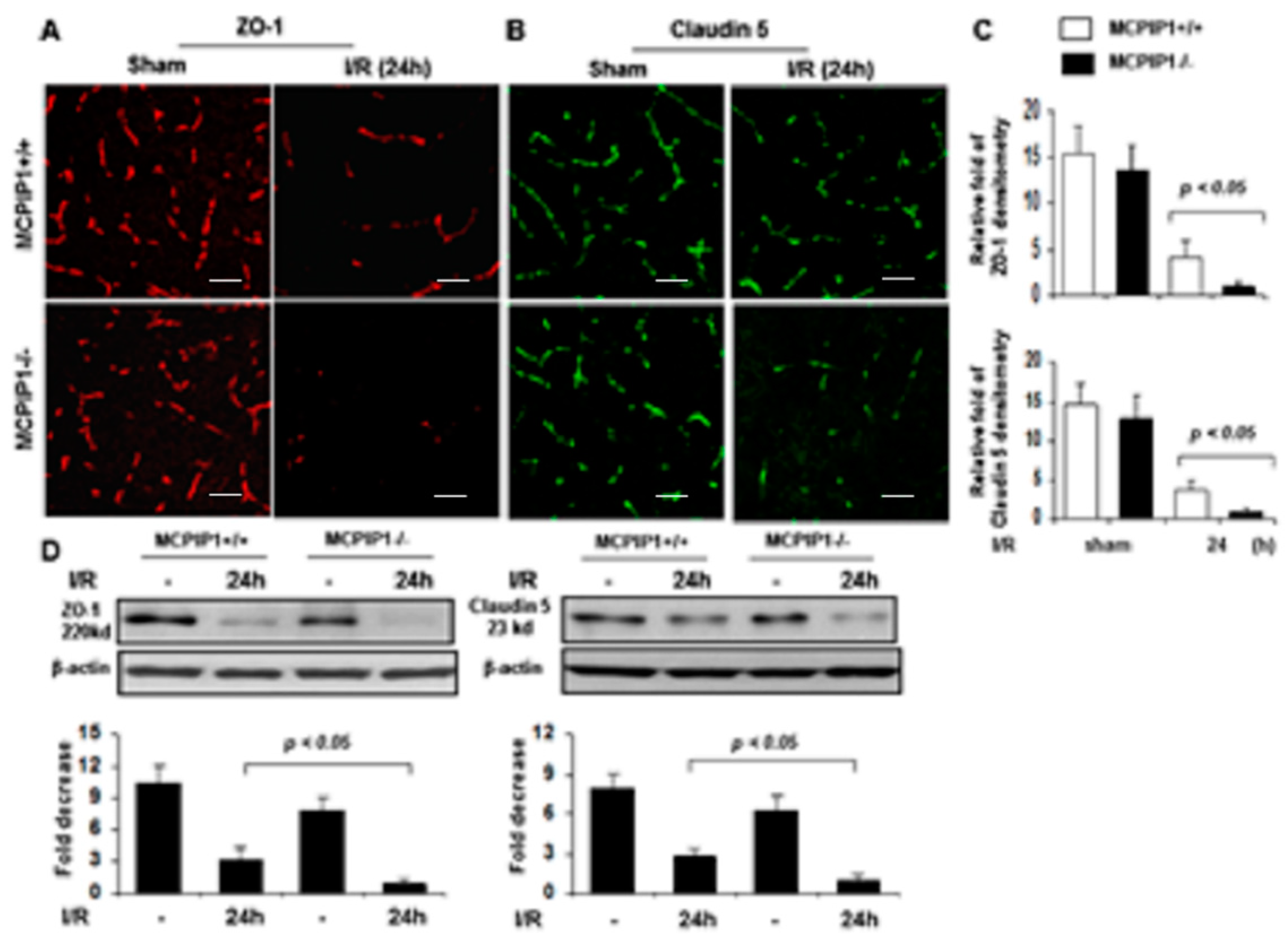
2.4. Altered Expression of Tight Junctions in MCPIP1−/− Mice Subjected to Transient Focal I/R Injury
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Transient Focal Brain Ischemia/Reperfusion in Mice
4.3. Assessment of BBB Breakdown Using FITC-Dextran
4.4. Quantitative Real-Time PCR
4.5. Western Blots
4.6. Immunohistochemistry
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| I/R | ischemia/reperfusion |
| MCAO | middle cerebral artery occlusion |
| MMPs | matrix metalloproteinases |
| BBB | blood–brain barrier |
| MCPIP1 | monocyte chemotactic protein 1–induced protein 1 |
| FITC | fluorescein isothiocyanate |
| ZO-1 | zonula occludens-1 |
| MCP-1 | monocyte chemoattractant protein-1 |
| TNF-α | tumor necrosis factor-alpha |
| IL-1β | interleukin 1 beta |
| IL-6 | interleukin 6 |
| LPS | lipopolysaccharide |
| qRT-PCR | quantitative real time PCR |
References
- Edwards, J.D.; Kapral, M.K.; Fang, J.; Swartz, R.H. Trends in long-term mortality and morbidity in patients with no early complications after stroke and transient ischemic attack. J. Stroke. Cerebrovasc. Dis. 2017, 26, 1641–1645. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Rosenberg, G.A. Blood–brain barrier breakdown in acute and chronic cerebrovascular disease. Stroke. 2011, 42, 3323–3328. [Google Scholar] [CrossRef] [PubMed]
- Sifat, A.E.; Vaidya, B.; Abbruscato, T.J. Blood–brain Barrier Protection as a Therapeutic Strategy for Acute Ischemic Stroke. AAPS.J. 2017, 19, 957–972. [Google Scholar] [CrossRef] [PubMed]
- Ballabh, P.; Braun, A.; Nedergaard, M. The blood–brain barrier: An overview: structure, regulation, and clinical implications. Neurobiol. Dis. 2004, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Esenwa, C.C.; Elkind, M.S. Inflammatory risk factors, biomarkers and associated therapy in ischemic stroke. Nat. Rev. Neurol. 2016, 12, 594–604. [Google Scholar] [CrossRef] [PubMed]
- Amantea, D.; Micieli, G.; Tassorelli, C.; Cuartero, M.I.; Ballesteros, I.; Certo, M.; Moro, M.A.; Lizasoain, I.; Bagetta, G. Rational modulation of the innate immune system for neuroprotection in ischemic stroke. Front. Neurosci. 2015, 9, 147. [Google Scholar] [CrossRef] [PubMed]
- Svedin, P.; Hagberg, H.; Savman, K.; Zhu, C.; Mallard, C. Matrix metalloproteinase-9 gene knock-out protects the immature brain after cerebral hypoxia-ischemia. J. Neurosci. 2007, 27, 1511–1518. [Google Scholar] [CrossRef]
- Bauer, A.T.; Burgers, H.F.; Rabie, T.; Marti, H.H. Matrix metalloproteinase-9 mediates hypoxia-induced vascular leakage in the brain via tight junction rearrangement. J. Cereb. Blood. Flow. Metab. 2010, 30, 837–848. [Google Scholar] [CrossRef]
- Yang, Y.; Estrada, E.Y.; Thompson, J.F.; Liu, W.; Rosenberg, G.A. Matrix metalloproteinase-mediated disruption of tight junction proteins in cerebral vessels is reversed by synthetic matrix metalloproteinase inhibitor in focal ischemia in rat. J. Cereb. Blood Flow Metab. 2007, 27, 697–709. [Google Scholar] [CrossRef]
- Liu, J.; Jin, X.; Liu, J.; Liu, W. Matrix metalloproteinase-2-mediated occludin degradation and caveolin-1-mediated claudin-5 redistribution contribute to blood–brain barrier damage in early ischemic stroke stage. J. Neuroscience. 2012, 32, 3044–3057. [Google Scholar] [CrossRef]
- Dimitrijevic, O.B.; Stamatovic, S.M.; Keep, R.F.; Andjelkovic, A.V. Effects of the chemokine CCL2 on blood–brain barrier permeability during ischemia-reperfusion injury. J. Cereb. Blood. Flow. Metab. 2006, 26, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Strecker, J.K.; Minnerup, J.; Gess, B.; Ringelstein, E.B.; Schabitz, W.R.; Schilling, M. Monocyte chemoattractant protein-1-deficiency impairs the expression of IL-6, IL-1beta and G-CSF after transient focal ischemia in mice. PLoS. One. 2011, 6, e25863. [Google Scholar] [CrossRef] [PubMed]
- Stamatovic, S.M.; Shakui, P.; Keep, R.F.; Moore, B.B.; Kunkel, S.L.; Van Rooijen, N.; Andjelkovic, A.V. Monocyte chemoattractant protein-1 regulation of blood–brain barrier permeability. J. Cereb. Blood. Flow. Metab. 2005, 25, 593–606. [Google Scholar] [CrossRef] [PubMed]
- Hughes, P.M.; Allegrini, P.R.; Rudin, M.; Perry, V.H.; Mir, A.K.; Wiessner, C. Monocyte chemoattractant protein-1 deficiency is protective in a murine stroke model. J. Cereb. Blood. Flow. Metab. 2002, 22, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Azfer, A.; Niu, J.; Graham, S.; Choudhury, M.; Adamski, F.M.; Younce, C.; Binkley, P.F.; Kolattukudy, P.E. Monocyte chemoattractant protein-1 induces a novel transcription factor that causes cardiac myocyte apoptosis and ventricular dysfunction. Circ. Res. 2006, 98, 1177–1185. [Google Scholar] [CrossRef]
- Liang, J.; Saad, Y.; Lei, T.; Wang, J.; Qi, D.; Yang, Q.; Kolattukudy, P.E.; Fu, M. MCP-induced protein 1 deubiquitinates TRAF proteins and negatively regulates JNK and NF-kappaB signaling. J. Exp. Med. 2010, 207, 2959–2973. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, K.; Takeuchi, O.; Standley, D.M.; Kumagai, Y.; Kawagoe, T.; Miyake, T.; Satoh, T.; Kato, H.; Tsujimura, T.; Nakamura, H.; et al. Zc3h12a is an RNase essential for controlling immune responses by regulating mRNA decay. Nature. 2009, 458, 1185–1190. [Google Scholar] [CrossRef]
- Jin, Z.; Liang, J.; Wang, J.; Kolattukudy, P.E. Delayed brain ischemia tolerance induced by electroacupuncture pretreatment is mediated via MCP-induced protein 1. J. Neuroinflammation. 2013, 10, 63. [Google Scholar] [CrossRef]
- Liang, J.; Wang, J.; Saad, Y.; Warble, L.; Becerra, E.; Kolattukudy, P.E. Participation of MCP-induced protein 1 in lipopolysaccharide preconditioning-induced ischemic stroke tolerance by regulating the expression of proinflammatory cytokines. J. Neuroinflammation. 2011, 8, 182. [Google Scholar] [CrossRef]
- Rempe, R.G.; Hartz, A.M.S.; Bauer, B. Matrix metalloproteinases in the brain and blood–brain barrier: Versatile breakers and makers. J. Cereb. Blood. Flow. Metab. 2016, 36, 1481–1507. [Google Scholar] [CrossRef]
- Clark, A.W.; Krekoski, C.A.; Bou, S.S.; Chapman, K.R.; Edwards, D.R. Increased gelatinase A (MMP-2) and gelatinase B (MMP-9) activities in human brain after focal ischemia. Neurosci. Lett. 1997, 238, 53–56. [Google Scholar] [CrossRef]
- Sole, S.; Petegnief, V.; Gorina, R.; Chamorro, A.; Planas, A.M. Activation of matrix metalloproteinase-3 and agrin cleavage in cerebral ischemia/reperfusion. J. Neuropathol. Exp. Neurol. 2004, 63, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Zhang, R.; Liu, D.; Wang, J.; Yuan, C.; Zhao, X.; Li, Y.; Ji, X.; Chi, T.; Zou, L. Time-course investigation of blood–brain barrier permeability and tight junction protein changes in a rat model of permanent focal ischemia. J. Physiol. Sci. 2018, 68, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Rosenberg, G.A. MMP-Mediated Disruption of Claudin-5 in the Blood–Brain Barrier of Rat Brain After Cerebral Ischemia. Methods. Mol. Biol. 2011, 762, 333–345. [Google Scholar] [PubMed]
- Feng, S.; Cen, J.; Huang, Y.; Shen, H.; Yao, L.; Wang, Y.; Chen, Z. Matrix metalloproteinase-2 and -9 secreted by leukemic cells increase the permeability of blood–brain barrier by disrupting tight junction proteins. PLoS. One. 2011, 6, e20599. [Google Scholar] [CrossRef]
- Jiao, H.; Wang, Z.; Liu, Y.; Wang, P.; Xue, Y. Specific role of tight junction proteins claudin-5, occludin, and ZO-1 of the blood–brain barrier in a focal cerebral ischemic insult. J. Mol. Neurosci. 2011, 44, 130–139. [Google Scholar] [CrossRef]
- Natarajan, R.; Northrop, N.; Yamamoto, B. Fluorescein isothiocyanate (FITC)-dextran extravasation as a measure of blood–brain barrier permeability. Curr. Protoc. Neurosci. 2017, 79, 1–9. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, Z.; Liang, J.; Li, J.; Kolattukudy, P.E. Absence of MCP-induced Protein 1 Enhances Blood–Brain Barrier Breakdown after Experimental Stroke in Mice. Int. J. Mol. Sci. 2019, 20, 3214. https://doi.org/10.3390/ijms20133214
Jin Z, Liang J, Li J, Kolattukudy PE. Absence of MCP-induced Protein 1 Enhances Blood–Brain Barrier Breakdown after Experimental Stroke in Mice. International Journal of Molecular Sciences. 2019; 20(13):3214. https://doi.org/10.3390/ijms20133214
Chicago/Turabian StyleJin, Zhuqing, Jian Liang, Jiaqi Li, and Pappachan E. Kolattukudy. 2019. "Absence of MCP-induced Protein 1 Enhances Blood–Brain Barrier Breakdown after Experimental Stroke in Mice" International Journal of Molecular Sciences 20, no. 13: 3214. https://doi.org/10.3390/ijms20133214
APA StyleJin, Z., Liang, J., Li, J., & Kolattukudy, P. E. (2019). Absence of MCP-induced Protein 1 Enhances Blood–Brain Barrier Breakdown after Experimental Stroke in Mice. International Journal of Molecular Sciences, 20(13), 3214. https://doi.org/10.3390/ijms20133214




