Cartilage Regeneration in Humans with Adipose Tissue-Derived Stem Cells and Adipose Stromal Vascular Fraction Cells: Updated Status
Abstract
1. Introduction
2. ASCs in the Form of Adipose SVF and Cultured Expansion
3. Potential Biological Mechanisms of Cartilage Regeneration by MSCs
3.1. Direct Engraftment
3.2. Trophic Bioactive Factors
3.2.1. Cytokines and Growth Factors
3.2.2. Extracellular Vesicles
4. PRP, HA, and ECM
5. Clinical Applications of ASCs in the Form of Adipose SVF and Culture-Expanded Cells
5.1. Retrospective Cohort Study by Kim et al.
5.2. Case Series by Fodor and Paulseth
5.3. A Phase 1 Dose Escalation Trial by Pers et al.
5.4. Placebo-Controlled Prospective Comparative Study by Nguyen et al.
5.5. Case Report by Pak et al.
5.6. A Randomized, Double-Blinded Pilot Study by Song et al.
5.7. Retrospective Comparative Study by Kim and Koh
5.8. Prospective Cohort Study by Jo et al.
5.9. Case Series by Pak et al.
5.10. Randomized, Double-Blind, Placebo-Controlled Study by Kuah et al.
6. Discussion
7. Method
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Buckwalter, J.A. Articular cartilage injuries. Clin. Orthop. Relat. Res. 2002, 402, 21–37. [Google Scholar] [CrossRef]
- Simon, L.S. Osteoarthritis. Curr. Rheumatol. Rep. 1999, 1, 45–47. [Google Scholar] [CrossRef] [PubMed]
- Aynardi, M.; Pulido, L.; Parvizi, J.; Sharkey, P.F.; Rothman, R.H. Early mortality after modern total hip arthroplasty. Clin. Orthop. Relat. Res. 2009, 467, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Belmont, P.J., Jr.; Goodman, G.P.; Waterman, B.R.; Bader, J.O.; Schoenfeld, A.J. Thirty-day postoperative complications and mortality following total knee arthroplasty: Incidence and risk factors among a national sample of 15,321 patients. J. Bone Jt. Surg. Am. 2014, 96, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Glyn-Jones, S.; Palmer, A.J.; Agricola, R.; Price, A.J.; Vincent, T.L.; Weinans, H.; Carr, A.J. Osteoarthritis. Lancet 2015, 386, 376–387. [Google Scholar] [CrossRef]
- Huang, G.T.; Gronthos, S.; Shi, S. Mesenchymal stem cells derived from dental tissues vs. those from other sources: Their biology and role in regenerative medicine. J. Dent. Res. 2009, 88, 792–806. [Google Scholar] [CrossRef] [PubMed]
- Orbay, H.; Tobita, M.; Mizuno, H. Mesenchymal stem cells isolated from adipose and other tissues: Basic biological properties and clinical applications. Stem Cells Int. 2012, 2012, 461718. [Google Scholar] [CrossRef] [PubMed]
- Via, A.G.; Frizziero, A.; Oliva, F. Biological properties of mesenchymal stem cells from different sources. Muscles Ligaments Tendons J. 2012, 2, 154–162. [Google Scholar] [PubMed]
- Arnoczky, S.P. Building a meniscus. Biologic considerations. Clin. Orthop. Relat. Res. 1999, 367, S244–S253. [Google Scholar] [CrossRef]
- Caplan, A.I. Mesenchymal stem cells. J. Orthop. Res. 1991, 9, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Szilvassy, S.J. The biology of hematopoietic stem cells. Arch. Med. Res. 2003, 34, 446–460. [Google Scholar] [CrossRef] [PubMed]
- Carter, D.R.; Beaupre, G.S.; Giori, N.J.; Helms, J.A. Mechanobiology of skeletal regeneration. Clin. Orthop. Relat. Res. 1998, 355, S41–S55. [Google Scholar] [CrossRef]
- Johnstone, B.; Yoo, J.U. Autologous mesenchymal progenitor cells in articular cartilage repair. Clin. Orthop. Relat. Res. 1999, 367, S156–S162. [Google Scholar] [CrossRef]
- Pak, J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose-tissue-derived stem cells: A case series. J. Med. Case Rep. 2011, 5, 296. [Google Scholar] [CrossRef] [PubMed]
- Pak, J.; Lee, J.H.; Kartolo, W.A.; Lee, S.H. Cartilage regeneration in human with adipose tissue-derived stem cells: Current status in clinical implications. BioMed Res. Int. 2016, 2016, 4702674. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef] [PubMed]
- Pak, J.; Lee, J.H.; Park, K.S.; Jeong, B.C.; Lee, S.H. Regeneration of cartilage in human knee osteoarthritis with autologous adipose tissue-derived stem cells and autologous extracellular matrix. Biores. Open Access 2016, 5, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Food and Drug Safety (MFDS). Cell Therapy: Rules and Regulations; MFDS: Seoul, Korea, 2009. Available online: http://www.mfds.go.kr/index.do?mid=1013&seq=9618&cmd=v (accessed on 22 October 2015).
- Pearle, A.D.; Warren, R.F.; Rodeo, S.A. Basic science of articular cartilage and osteoarthritis. Clin. Sports Med. 2005, 24, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sophia Fox, A.J.; Bedi, A.; Rodeo, S.A. The basic science of articular cartilage: Structure, composition, and function. Sports Health 2009, 1, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Haslauer, C.M.; Elsaid, K.A.; Fleming, B.C.; Proffen, B.L.; Johnson, V.M.; Murray, M.M. Loss of extracellular matrix from articular cartilage is mediated by the synovium and ligament after anterior cruciate ligament injury. Osteoarthr. Cartil. 2013, 21, 1950–1957. [Google Scholar] [CrossRef] [PubMed]
- Heard, B.J.; Barton, K.I.; Chung, M.; Achari, Y.; Shrive, N.G.; Frank, C.B.; Hart, D.A. Single intra-articular dexamethasone injection immediately post-surgery in a rabbit model mitigates early inflammatory responses and post-traumatic osteoarthritis-like alterations. J. Orthop. Res. 2015, 33, 1826–1834. [Google Scholar] [CrossRef] [PubMed]
- Toh, W.S.; Brittberg, M.; Farr, J.; Foldager, C.B.; Gomoll, A.H.; Hui, J.H.; Richardson, J.B.; Roberts, S.; Spector, M. Cellular senescence in aging and osteoarthritis. Acta Orthop. 2016, 87, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Romero, C.; Calamia, V.; Mateos, J.; Carreira, V.; Martinez-Gomariz, M.; Fernandez, M.; Blanco, F.J. Mitochondrial dysregulation of osteoarthritic human articular chondrocytes analyzed by proteomics: A decrease in mitochondrial superoxide dismutase points to a redox imbalance. Mol. Cell. Proteom. 2009, 8, 172–189. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhao, X.; Lotz, M.; Terkeltaub, R.; Liu-Bryan, R. Mitochondrial biogenesis is impaired in osteoarthritis chondrocytes but reversible via peroxisome proliferator-activated receptor γ coactivator 1α. Arthritis Rheum. 2015, 67, 2141–2153. [Google Scholar] [CrossRef] [PubMed]
- Corciulo, C.; Lendhey, M.; Wilder, T.; Schoen, H.; Cornelissen, A.S.; Chang, G.; Kennedy, O.D.; Cronstein, B.N. Endogenous adenosine maintains cartilage homeostasis and exogenous adenosine inhibits osteoarthritis progression. Nat. Commun. 2017, 8, 15019. [Google Scholar] [CrossRef] [PubMed]
- Terkeltaub, R.; Johnson, K.; Murphy, A.; Ghosh, S. Invited review: The mitochondrion in osteoarthritis. Mitochondrion 2002, 1, 301–319. [Google Scholar] [CrossRef]
- Loeser, R.F. Aging and osteoarthritis. Curr. Opin. Rheumatol. 2011, 23, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Vaamonde-Garcia, C.; Riveiro-Naveira, R.R.; Valcarcel-Ares, M.N.; Hermida-Carballo, L.; Blanco, F.J.; Lopez-Armada, M.J. Mitochondrial dysfunction increases inflammatory responsiveness to cytokines in normal human chondrocytes. Arthritis Rheum. 2012, 64, 2927–2936. [Google Scholar] [CrossRef] [PubMed]
- Arslan, F.; Lai, R.C.; Smeets, M.B.; Akeroyd, L.; Choo, A.; Aguor, E.N.; Timmers, L.; van Rijen, H.V.; Doevendans, P.A.; Pasterkamp, G.; et al. Mesenchymal stem cell-derived exosomes increase ATP levels, decrease oxidative stress and activate PI3K/AKT pathway to enhance myocardial viability and prevent adverse remodeling after myocardial ischemia/reperfusion injury. Stem Cell Res. 2013, 10, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Zwolanek, D.; Satue, M.; Proell, V.; Godoy, J.R.; Odorfer, K.I.; Flicker, M.; Hoffmann, S.C.; Rulicke, T.; Erben, R.G. Tracking mesenchymal stem cell contributions to regeneration in an immunocompetent cartilage regeneration model. JCI Insight 2017, 2, 87322. [Google Scholar] [CrossRef] [PubMed]
- Chute, J.P. Stem cell homing. Curr. Opin. Hematol. 2006, 13, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Khaldoyanidi, S. Directing stem cell homing. Cell Stem Cell 2008, 2, 198–200. [Google Scholar] [CrossRef] [PubMed]
- Sohni, A.; Verfaillie, C.M. Mesenchymal stem cells migration homing and tracking. Stem Cells Int. 2013, 2013, 130763. [Google Scholar] [CrossRef] [PubMed]
- Wynn, R.F.; Hart, C.A.; Corradi-Perini, C.; O’Neill, L.; Evans, C.A.; Wraith, J.E.; Fairbairn, L.J.; Bellantuono, I. A small proportion of mesenchymal stem cells strongly expresses functionally active CXCR4 receptor capable of promoting migration to bone marrow. Blood 2004, 104, 2643–2645. [Google Scholar] [CrossRef] [PubMed]
- Rüster, B.; Gottig, S.; Ludwig, R.J.; Bistrian, R.; Muller, S.; Seifried, E.; Gille, J.; Henschler, R. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood 2006, 108, 3938–3944. [Google Scholar] [CrossRef] [PubMed]
- Docheva, D.; Popov, C.; Mutschler, W.; Schieker, M. Human mesenchymal stem cells in contact with their environment: Surface characteristics and the integrin system. J. Cell. Mol. Med. 2007, 11, 21–38. [Google Scholar] [CrossRef] [PubMed]
- Teo, G.S.; Ankrum, J.A.; Martinelli, R.; Boetto, S.E.; Simms, K.; Sciuto, T.E.; Dvorak, A.M.; Karp, J.M.; Carman, C.V. Mesenchymal stem cells transmigrate between and directly through tumor necrosis factor-α-activated endothelial cells via both leukocyte-like and novel mechanisms. Stem Cells 2012, 30, 2472–2486. [Google Scholar] [CrossRef] [PubMed]
- Ip, J.E.; Wu, Y.; Huang, J.; Zhang, L.; Pratt, R.E.; Dzau, V.J. Mesenchymal stem cells use integrinβ1 not CXC chemokine receptor 4 for myocardial migration and engraftment. Mol. Biol. Cell 2007, 18, 2873–2882. [Google Scholar] [CrossRef] [PubMed]
- Steingen, C.; Brenig, F.; Baumgartner, L.; Schmidt, J.; Schmidt, A.; Bloch, W. Characterization of key mechanisms in transmigration and invasion of mesenchymal stem cells. J. Mol. Cell. Cardiol. 2008, 44, 1072–1084. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, K.; Muneta, T.; Morito, T.; Ichinose, S.; Koga, H.; Nimura, A.; Mochizuki, T.; Sekiya, I. Exogenous synovial stem cells adhere to defect of meniscus and differentiate into cartilage cells. J. Med. Dent. Sci. 2008, 55, 101–111. [Google Scholar] [PubMed]
- Park, Y.B.; Ha, C.W.; Kim, J.A.; Han, W.J.; Rhim, J.H.; Lee, H.J.; Kim, K.J.; Park, Y.G.; Chung, J.Y. Single-stage cell-based cartilage repair in a rabbit model: Cell tracking and in vivo chondrogenesis of human umbilical cord blood-derived mesenchymal stem cells and hyaluronic acid hydrogel composite. Osteoarthr. Cartil. 2017, 25, 570–580. [Google Scholar] [CrossRef] [PubMed]
- De Windt, T.S.; Vonk, L.A.; Slaper-Cortenbach, I.C.M.; Nizak, R.; van Rijen, M.H.P.; Saris, D.B.F. Allogeneic mscs and recycled autologous chondrons mixed in a one-stage cartilage cell transplantion: A first-in-man trial in 35 patients. Stem Cells 2017, 35, 1984–1993. [Google Scholar] [CrossRef] [PubMed]
- Lai, R.C.; Arslan, F.; Lee, M.M.; Sze, N.S.; Choo, A.; Chen, T.S.; Salto-Tellez, M.; Timmers, L.; Lee, C.N.; El Oakley, R.M.; et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010, 4, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Lai, R.C.; Yeo, R.W.; Tan, K.H.; Lim, S.K. Mesenchymal stem cell exosome ameliorates reperfusion injury through proteomic complementation. Regen. Med. 2013, 8, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Lai, R.C.; Yeo, R.W.; Tan, S.S.; Zhang, B.; Yin, Y.; Sze, S.K.; Choo, A.; Lim, S.-K. Mesenchymal stem cell exosomes: The future MSC-based therapy. In Mesenchymal Stem Cell Therapy; Chase, L., Vemuri, M., Eds.; Humana Press: New York, NY, USA, 2013; pp. 39–61. [Google Scholar]
- Zhang, S.; Chu, W.C.; Lai, R.C.; Lim, S.K.; Hui, J.H.; Toh, W.S. Exosomes derived from human embryonic mesenchymal stem cells promote osteochondral regeneration. Osteoarthr. Cartil. 2016, 24, 2135–2140. [Google Scholar] [CrossRef] [PubMed]
- Beer, L.; Mildner, M.; Ankersmit, H.J. Cell secretome based drug substances in regenerative medicine: When regulatory affairs meet basic science. Ann. Transl. Med. 2017, 5, 170. [Google Scholar] [CrossRef] [PubMed]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Puetzer, J.L.; Petitte, J.N.; Loboa, E.G. Comparative review of growth factors for induction of three-dimensional in vitro chondrogenesis in human mesenchymal stem cells isolated from bone marrow and adipose tissue. Tissue Eng. Part B Rev. 2010, 16, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Kim, J.; Lee, K.I.; Shin, J.M.; Chae, J.I.; Chung, H.M. Enhancement of wound healing by secretory factors of endothelial precursor cells derived from human embryonic stem cells. Cytotherapy 2011, 13, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Yi, T.; Song, S.U. Immunomodulatory properties of mesenchymal stem cells and their therapeutic applications. Arch. Pharm. Res. 2012, 35, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.A.; Han, J.; Kim, B.S. Stimulation of chondrogenic differentiation of mesenchymal stem cells. Int. J. Stem Cells 2012, 5, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Zagoura, D.S.; Roubelakis, M.G.; Bitsika, V.; Trohatou, O.; Pappa, K.I.; Kapelouzou, A.; Antsaklis, A.; Anagnou, N.P. Therapeutic potential of a distinct population of human amniotic fluid mesenchymal stem cells and their secreted molecules in mice with acute hepatic failure. Gut 2012, 61, 894–906. [Google Scholar] [CrossRef] [PubMed]
- Cantinieaux, D.; Quertainmont, R.; Blacher, S.; Rossi, L.; Wanet, T.; Noel, A.; Brook, G.; Schoenen, J.; Franzen, R. Conditioned medium from bone marrow-derived mesenchymal stem cells improves recovery after spinal cord injury in rats: An original strategy to avoid cell transplantation. PLoS ONE 2013, 8, e69515. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.; Liu, H.; Li, J.; Zhou, Y. Roles of hypoxia during the chondrogenic differentiation of mesenchymal stem cells. Curr. Stem Cell Res. Ther. 2014, 9, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, H.; Zeng, M.; He, W.; Li, M.; Huang, X.; Deng, D.Y.; Wu, J. Bone marrow mesenchymal stem cells protect alveolar macrophages from lipopolysaccharide-induced apoptosis partially by inhibiting the WNT/β-catenin pathway. Cell Biol. Int. 2015, 39, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Bermudez, M.A.; Sendon-Lago, J.; Seoane, S.; Eiro, N.; Gonzalez, F.; Saa, J.; Vizoso, F.; Perez-Fernandez, R. Anti-inflammatory effect of conditioned medium from human uterine cervical stem cells in uveitis. Exp. Eye Res. 2016, 149, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Amann, E.; Wolff, P.; Breel, E.; van Griensven, M.; Balmayor, E.R. Hyaluronic acid facilitates chondrogenesis and matrix deposition of human adipose derived mesenchymal stem cells and human chondrocytes co-cultures. Acta Biomater. 2017, 52, 130–144. [Google Scholar] [CrossRef] [PubMed]
- Van der Pol, E.; Boing, A.N.; Harrison, P.; Sturk, A.; Nieuwland, R. Classification, functions, and clinical relevance of extracellular vesicles. Pharmacol. Rev. 2012, 64, 676–705. [Google Scholar] [CrossRef] [PubMed]
- Rani, S.; Ryan, A.E.; Griffin, M.D.; Ritter, T. Mesenchymal stem cell-derived extracellular vesicles: Toward cell-free therapeutic applications. Mol. Ther. 2015, 23, 812–823. [Google Scholar] [CrossRef] [PubMed]
- Deregibus, M.C.; Cantaluppi, V.; Calogero, R.; Lo Iacono, M.; Tetta, C.; Biancone, L.; Bruno, S.; Bussolati, B.; Camussi, G. Endothelial progenitor cell derived microvesicles activate an angiogenic program in endothelial cells by a horizontal transfer of mrna. Blood 2007, 110, 2440–2448. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wu, X.; Zhang, X.; Sun, Y.; Yan, Y.; Shi, H.; Zhu, Y.; Wu, L.; Pan, Z.; Zhu, W.; et al. Human umbilical cord mesenchymal stem cell exosomes enhance angiogenesis through the WNT4/β-catenin pathway. Stem Cells Transl. Med. 2015, 4, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guan, J.; Niu, X.; Hu, G.; Guo, S.; Li, Q.; Xie, Z.; Zhang, C.; Wang, Y. Exosomes released from human induced pluripotent stem cells-derived mscs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J. Transl. Med. 2015, 13, 49. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.D.; Johansson, H.J.; Graham, C.S.; Vesterlund, M.; Pham, M.T.; Bramlett, C.S.; Montgomery, E.N.; Mellema, M.S.; Bardini, R.L.; Contreras, Z.; et al. Comprehensive proteomic analysis of mesenchymal stem cell exosomes reveals modulation of angiogenesis via nuclear factor-kappab signaling. Stem Cells 2016, 34, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, C.; Wei, L.; Li, Q.; Niu, X.; Xu, Y.; Wang, Y.; Zhao, J. Exosomes derived from endothelial progenitor cells attenuate vascular repair and accelerate reendothelialization by enhancing endothelial function. Cytotherapy 2016, 18, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Basu, J.; Ludlow, J.W. Exosomes for repair, regeneration and rejuvenation. Expert Opin. Biol. Ther. 2016, 16, 489–506. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles, and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Edgar, J.R. Q&A: What are exosomes, exactly? BMC Biol. 2016, 14, 46. [Google Scholar]
- Feng, D.; Zhao, W.L.; Ye, Y.Y.; Bai, X.C.; Liu, R.Q.; Chang, L.F.; Zhou, Q.; Sui, S.F. Cellular internalization of exosomes occurs through phagocytosis. Traffic 2010, 11, 675–687. [Google Scholar] [CrossRef] [PubMed]
- Lai, R.C.; Tan, S.S.; Teh, B.J.; Sze, S.K.; Arslan, F.; de Kleijn, D.P.; Choo, A.; Lim, S.K. Proteolytic potential of the MSC exosome proteome: Implications for an exosome-mediated delivery of therapeutic proteasome. Int. J. Proteom. 2012, 2012, 971907. [Google Scholar] [CrossRef] [PubMed]
- Svensson, K.J.; Christianson, H.C.; Wittrup, A.; Bourseau-Guilmain, E.; Lindqvist, E.; Svensson, L.M.; Morgelin, M.; Belting, M. Exosome uptake depends on ERK1/2-heat shock protein 27 signaling and lipid raft-mediated endocytosis negatively regulated by caveolin-1. J. Biol. Chem. 2013, 288, 17713–17724. [Google Scholar] [CrossRef] [PubMed]
- Luthje, J. Origin, metabolism and function of extracellular adenine nucleotides in the blood. Klin. Wochenschr. 1989, 67, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Aymeric, L.; Apetoh, L.; Ghiringhelli, F.; Tesniere, A.; Martins, I.; Kroemer, G.; Smyth, M.J.; Zitvogel, L. Tumor cell death and atp release prime dendritic cells and efficient anticancer immunity. Cancer Res. 2010, 70, 855–858. [Google Scholar] [CrossRef] [PubMed]
- Vitiello, L.; Gorini, S.; Rosano, G.; la Sala, A. Immunoregulation through extracellular nucleotides. Blood 2012, 120, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, K.A.; Gao, Z.G. Adenosine receptors as therapeutic targets. Nat. Rev. Drug Discov. 2006, 5, 247–264. [Google Scholar] [CrossRef] [PubMed]
- Chekeni, F.B.; Elliott, M.R.; Sandilos, J.K.; Walk, S.F.; Kinchen, J.M.; Lazarowski, E.R.; Armstrong, A.J.; Penuela, S.; Laird, D.W.; Salvesen, G.S.; et al. Pannexin 1 channels mediate "find-me" signal release and membrane permeability during apoptosis. Nature 2010, 467, 863–867. [Google Scholar] [CrossRef] [PubMed]
- Colgan, S.P.; Eltzschig, H.K.; Eckle, T.; Thompson, L.F. Physiological roles for ecto-5′-nucleotidase (CD73). Purinergic Signal. 2006, 2, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Beier, F.; Loeser, R.F. Biology and pathology of Rho GTPase, PI-3 kinase-AKT, and MAP kinase signaling pathways in chondrocytes. J. Cell. Biochem. 2010, 110, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Ouyang, G.; Bao, S. The activation of AKT/PKB signaling pathway and cell survival. J. Cell. Mol. Med. 2005, 9, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Chambard, J.C.; Lefloch, R.; Pouyssegur, J.; Lenormand, P. ERK implication in cell cycle regulation. Biochim. Biophys. Acta 2007, 1773, 1299–1310. [Google Scholar] [CrossRef] [PubMed]
- Fahy, N.; de Vries-van Melle, M.L.; Lehmann, J.; Wei, W.; Grotenhuis, N.; Farrell, E.; van der Kraan, P.M.; Murphy, J.M.; Bastiaansen-Jenniskens, Y.M.; van Osch, G.J. Human osteoarthritic synovium impacts chondrogenic differentiation of mesenchymal stem cells via macrophage polarisation state. Osteoarthr. Cartil. 2014, 22, 1167–1175. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Chen, B.; Lv, T.; Liu, X.; Fu, X.; Wang, Q.; Yan, L.; Kang, N.; Cao, Y.; Xiao, R. Bone marrow mesenchymal stem cell-based engineered cartilage ameliorates polyglycolic acid/polylactic acid scaffold-induced inflammation through M2 polarization of macrophages in a pig model. Stem Cells Transl. Med. 2016, 5, 1079–1089. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Chu, W.; Lai, R.; Hui, J.; Lee, E.; Lim, S.; Toh, W. Human mesenchymal stem cell-derived exosomes promote orderly cartilage regeneration in an immunocompetent rat osteochondral defect model. Cytotherapy 2016, 18, S13. [Google Scholar] [CrossRef]
- Zhang, B.; Yin, Y.; Lai, R.C.; Tan, S.S.; Choo, A.B.; Lim, S.K. Mesenchymal stem cells secrete immunologically active exosomes. Stem Cells Dev. 2014, 23, 1233–1244. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Andia, I.; Ardanza, B.; Nurden, P.; Nurden, A.T. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb. Haemost. 2004, 91, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Centeno, C.J.; Busse, D.; Kisiday, J.; Keohan, C.; Freeman, M.; Karli, D. Increased knee cartilage volume in degenerative joint disease using percutaneously implanted, autologous mesenchymal stem cells. Pain Physician 2008, 11, 343–353. [Google Scholar] [PubMed]
- Eppley, B.L.; Pietrzak, W.S.; Blanton, M. Platelet-rich plasma: A review of biology and applications in plastic surgery. Plast Reconstr. Surg. 2006, 118, 147e–159e. [Google Scholar] [CrossRef] [PubMed]
- Fufa, D.; Shealy, B.; Jacobson, M.; Kevy, S.; Murray, M.M. Activation of platelet-rich plasma using soluble type i collagen. J. Oral Maxillofac. Surg. 2008, 66, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Pak, J.; Chang, J.J.; Lee, J.H.; Lee, S.H. Safety reporting on implantation of autologous adipose tissue-derived stem cells with platelet-rich plasma into human articular joints. BMC Musculoskelet. Disord. 2013, 14, 337. [Google Scholar] [CrossRef] [PubMed]
- Uzuki, M.; Sawai, T. A comparison of the affinity of sodium hyaluronate of various molecular weights for degenerated cartilage: A histochemical study using hyaluronic acid binding protein. Int. Congr. Ser. 2001, 1223, 279–284. [Google Scholar] [CrossRef]
- Benders, K.E.; van Weeren, P.R.; Badylak, S.F.; Saris, D.B.; Dhert, W.J.; Malda, J. Extracellular matrix scaffolds for cartilage and bone regeneration. Trends Biotechnol. 2013, 31, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Lee, M.; Koh, Y.G. Additional mesenchymal stem cell injection improves the outcomes of marrow stimulation combined with supramalleolar osteotomy in varus ankle osteoarthritis: Short-term clinical results with second-look arthroscopic evaluation. J. Exp. Orthop. 2016, 3, 12. [Google Scholar] [CrossRef] [PubMed]
- Fodor, P.B.; Paulseth, S.G. Adipose derived stromal cell (ADSC) injections for pain management of osteoarthritis in the human knee joint. Aesthet. Surg. J. 2016, 36, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Baer, P.C.; Geiger, H. Adipose-derived mesenchymal stromal/stem cells: Tissue localization, characterization, and heterogeneity. Stem Cells Int. 2012, 2012, 812693. [Google Scholar] [CrossRef] [PubMed]
- Jo, C.H.; Lee, Y.G.; Shin, W.H.; Kim, H.; Chai, J.W.; Jeong, E.C.; Kim, J.E.; Shim, H.; Shin, J.S.; Shin, I.S.; et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: A proof-of-concept clinical trial. Stem Cells 2014, 32, 1254–1266. [Google Scholar] [CrossRef] [PubMed]
- Pers, Y.M.; Rackwitz, L.; Ferreira, R.; Pullig, O.; Delfour, C.; Barry, F.; Sensebe, L.; Casteilla, L.; Fleury, S.; Bourin, P.; et al. Adipose mesenchymal stromal cell-based therapy for severe osteoarthritis of the knee: A phase i dose-escalation trial. Stem Cells Transl. Med. 2016, 5, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.D.; Tran, T.D.; Nguyen, H.T.; Vu, H.T.; Le, P.T.; Phan, N.L.; Vu, N.B.; Phan, N.K.; van Pham, P. Comparative clinical observation of arthroscopic microfracture in the presence and absence of a stromal vascular fraction injection for osteoarthritis. Stem Cells Transl. Med. 2017, 6, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Pak, J.; Lee, J.H.; Park, K.S.; Lee, S.H. Efficacy of autologous adipose tissue-derived stem cells with extracellular matrix and hyaluronic acid on human hip osteoarthritis. Biomed. Res. 2017, 28, 1654–1658. [Google Scholar]
- Song, Y.; Du, H.; Dai, C.; Zhang, L.; Li, S.; Hunter, D.J.; Lu, L.; Bao, C. Human adipose-derived mesenchymal stem cells for osteoarthritis: A pilot study with long-term follow-up and repeated injections. Regen. Med. 2018, 13, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Koh, Y.G. Injection of mesenchymal stem cells as a supplementary strategy of marrow stimulation improves cartilage regeneration after lateral sliding calcaneal osteotomy for varus ankle osteoarthritis: Clinical and second-look arthroscopic results. Arthroscopy 2016, 32, 878–889. [Google Scholar] [CrossRef] [PubMed]
- Jo, C.H.; Chai, J.W.; Jeong, E.C.; Oh, S.; Shin, J.S.; Shim, H.; Yoon, K.S. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: A 2-year follow-up study. Am. J. Sports Med. 2017, 45, 2774–2783. [Google Scholar] [CrossRef] [PubMed]
- Kuah, D.; Sivell, S.; Longworth, T.; James, K.; Guermazi, A.; Cicuttini, F.; Wang, Y.; Craig, S.; Comin, G.; Robinson, D.; et al. Safety, tolerability and efficacy of intra-articular progenza in knee osteoarthritis: A randomized double-blind placebo-controlled single ascending dose study. J. Transl. Med. 2018, 16, 49. [Google Scholar] [CrossRef] [PubMed]
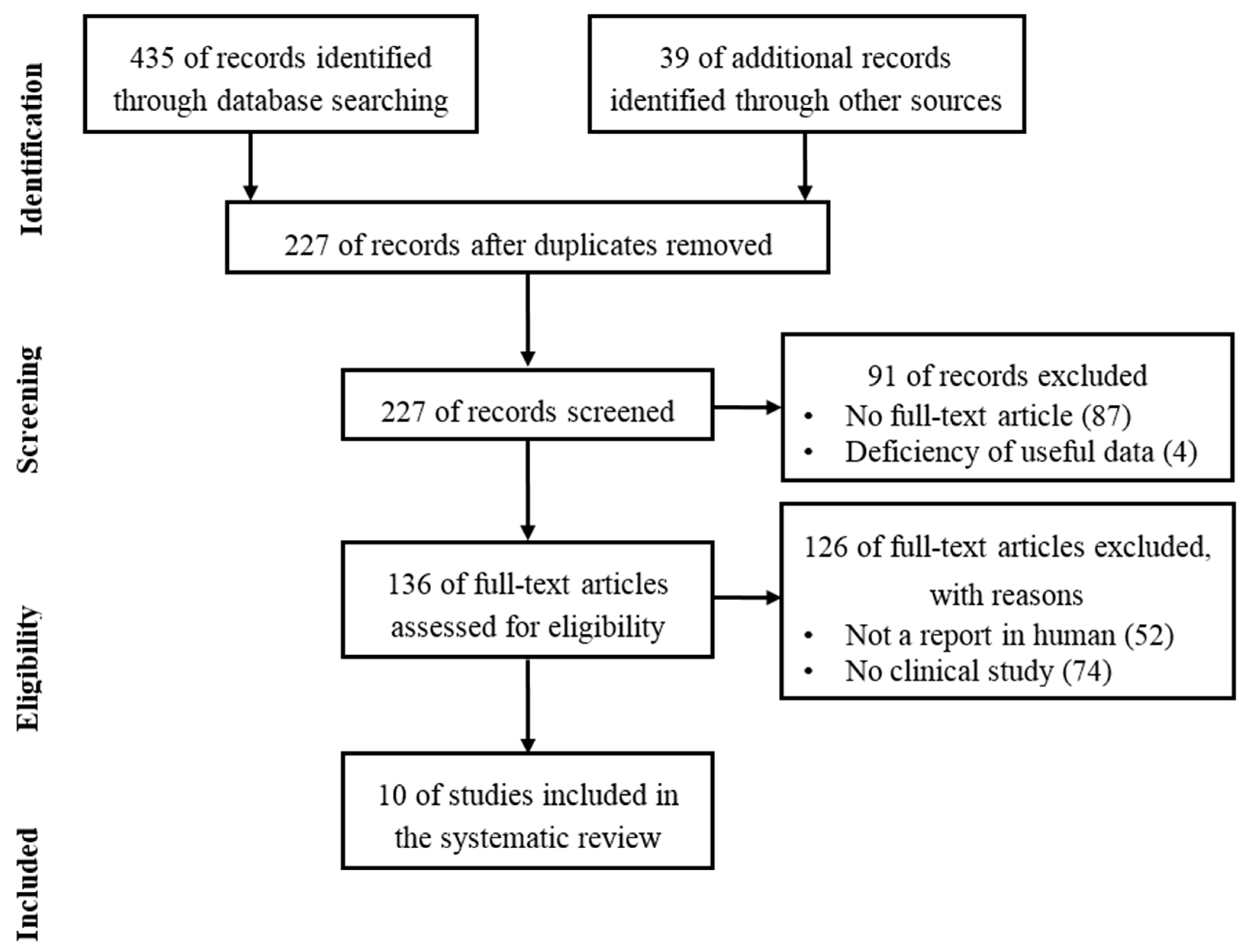
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The prisma statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [PubMed]
| Study (Year) | Intervention Treatment | Study Type | Number of Subjects/Age (Years) | Subject Characteristic | Concurrent Treatment | Follow-Up | Outcome Measures | Results |
|---|---|---|---|---|---|---|---|---|
| Kim et al. (2016) [94] | ASCs harvested from the patient’s buttock ASC injection Arthroscopic marrow stimulation and SMO alone vs. arthroscopic marrow stimulation and SMO + ASCs (4.0 × 106 stem cells) | Retrospective comparative study, level III | 62 patients (64 ankles)/51.8: 31 patients/33 ankles Marrow stimulation alone (Group I); 31 patients/31 ankles Marrow stimulation with ASCs injection (Group II) | Varus ankle OA | 12.8 months | VAS, AOFAS | The mean VAS and AOFAS scores improved significantly for both groups. There were significant differences in the mean VAS and AOFAS scores between groups at the final follow-up. At second-look arthroscopy, there were significant differences in ICRS grades between groups | |
| Fodor and Paulseth (2016) [95] | ASCs obtained through enzymatic disaggregation of lipoaspirate from the abdomen, flanks, or lateral thighs One intra-articularinjection of ASCs (14.1 million cells) | Case series, level IV | 6 patients (8 knees)/59 | OA knee | 12 months | WOMAC, VAS, ROM, TUG, MRI | Improvement in WOMAC and VAS scores at 3 months and maintained at 1 year. ROM and TUG both improved from preoperative to 3 months. MRI showed no detectable structural differences | |
| Pers et al. (2016) [98] | Autologous ASCs: one intra-articular injection, low dose (2 × 106 cells) vs. medium dose (10 × 106 cells) vs. high dose (50 × 106 cells) | Cohort study, level III | 18/64.6: 6 low dose, 6 medium dose, 6 high dose | OA knee | 6 months | VAS | Even the low-dose patients group experienced significant improvements in pain levels and function compared with the baseline | |
| Nguyen et al. (2016) [99] | Autologous ASCs harvested from the abdomen isolated arthroscopic microfracture vs. arthroscopic microfracture + ASCs (107 ASCs cells/mL) suspended in PRP | Prospective comparative study, level II | 30 patients: 15 patients placebo group/58.2; 15 patients treatment group/58.6 | Knee OA (Kellgren–Lawrence grade II–III) | Arthroscopic microfracture and ASC injection | 18 months | WOMAC, Lysholm, VAS, Outerbridge classification, MRI | WOMAC, Lysholm, and VAS scores improved; Outerbridge classification, measured with MRI, showed non-differences between the two group, but Outerbridge scores increased in the placebo group over time and decreased in the treatment group |
| Pak et al. (2017) [100] | Autologous adipose SVF + ECM + PRP + HA | Case report | 1 patient | Hip OA | 20 weeks | MRI, FRI, ROM, VAS | Along with MRI evidence, FRI, ROM, and VAS all improved | |
| Song et al. (2018) [101] | Autologous culture-expanded ASCs were injected for the low-dose, mid-dose, and high-dose groups, providing three injections and followed up for 96 weeks. | Double-blind, randomized pilot study | 18 patients divided into three dose groups: the low-dose (1 × 107), mid-dose (2 × 107), and high-dose group (5 × 107) cells | Knee OA | 96 weeks | WOMAC, NRS-11 and SF-36, MRI | Along with MRI evidence, autologous ASCs improved WOMAC, NRS-11, and SF-36 results. The dosage of 5 × 107 adipose MSCs exhibited the highest improvement | |
| Kim and Koh (2016) [102] | ASCs harvested from the patient’s buttock ASCs injection along with arthroscopic marrow stimulation Arthroscopic marrow stimulation vs. ASCs (4.1 × 106 stem cells) + marrow stimulation | Retrospective comparative study, level III | 49 patients/53.9: 23 ankles underwent marrow stimulation alone (Group 1), and 26 underwent marrow stimulation with ASC injection (Group 2). | Varus ankle OA | 27.6 months 12.5 second-look arthroscopies | VAS, AOFAS, Second-look-arthroscopy | The mean VAS and AOFAS scores improved significantly for both groups. The VAS and AOFAS scores were significantly better in Group 2. Significant differences in ICRS grades between the groups | |
| Jo et al. (2017) [103] | Autologous ASCs isolated from abdominal subcutaneous fat by liposuction and culture-expanded autologous ASCs in normal saline were injected intra-articularly | Cohort study; level of evidence, 3. | 18 patients: 3 male/61.8; 15 female/66.6 | Knee OA | 24 months | WOMAC, KSS, KOOS, VAS, MRI | WOMAC, KSS, KOOS, and VAS improved for up to 2 years regardless of the cell dosage. However, statistical significance was found mainly in the high-dose group. Clinical outcomes tended to deteriorate after 1 year in the low- and medium-dose groups, whereas those in the high-dose group plateaued until 2 years. The structural outcomes evaluated with MRI also showed similar trends. | |
| Pak et al. (2016) [18] | Autologous adipose SVF + ECM | Case series | 3 patients: 2 female/60 and 87; 1 male/68 | Knee OA | 6–22 weeks | MRI, FRI, ROM, VAS | Along with MRI evidence, FRI, ROM, and VAS all improved | |
| Kuah et al. (2018) [104] | Culture-expanded ASCs with culture media supernatant (CMS) | Randomized, double-blind, placebo-controlled Study | 20 patients/40–65 | Knee OA | None | 12 months | MRI, VAS, WOMAC | VAS and WOMAC improved in ASC + CMS groups, but MRI deteriorated in placebo and high-dose ASC + CMS group; no change in low-dose ASC + CMS group |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pak, J.; Lee, J.H.; Pak, N.; Pak, Y.; Park, K.S.; Jeon, J.H.; Jeong, B.C.; Lee, S.H. Cartilage Regeneration in Humans with Adipose Tissue-Derived Stem Cells and Adipose Stromal Vascular Fraction Cells: Updated Status. Int. J. Mol. Sci. 2018, 19, 2146. https://doi.org/10.3390/ijms19072146
Pak J, Lee JH, Pak N, Pak Y, Park KS, Jeon JH, Jeong BC, Lee SH. Cartilage Regeneration in Humans with Adipose Tissue-Derived Stem Cells and Adipose Stromal Vascular Fraction Cells: Updated Status. International Journal of Molecular Sciences. 2018; 19(7):2146. https://doi.org/10.3390/ijms19072146
Chicago/Turabian StylePak, Jaewoo, Jung Hun Lee, Natalie Pak, Yoon Pak, Kwang Seung Park, Jeong Ho Jeon, Byeong Chul Jeong, and Sang Hee Lee. 2018. "Cartilage Regeneration in Humans with Adipose Tissue-Derived Stem Cells and Adipose Stromal Vascular Fraction Cells: Updated Status" International Journal of Molecular Sciences 19, no. 7: 2146. https://doi.org/10.3390/ijms19072146
APA StylePak, J., Lee, J. H., Pak, N., Pak, Y., Park, K. S., Jeon, J. H., Jeong, B. C., & Lee, S. H. (2018). Cartilage Regeneration in Humans with Adipose Tissue-Derived Stem Cells and Adipose Stromal Vascular Fraction Cells: Updated Status. International Journal of Molecular Sciences, 19(7), 2146. https://doi.org/10.3390/ijms19072146





