1. Introduction
The intestinal epithelium not only plays a key role in digestion and absorption of nutrients, but also has an important function in preventing pathogen invasion and dissemination of commensals [
1]. However, the intestinal epithelial health status, especially in young animals, could be easily injured by many factors, such as inflammation and infection [
2]. Inflammation often results in intestinal mucosal damage and dysfunction, which negatively affects animal performance and health [
3]. In order to alleviate the inflammation and maintain health and function, the intestine needs a high level of energy and amino acids (AAs) [
4,
5].
Glycine, whose structure is the simplest of all AAs, is regarded as a conditionally essential AA for young mammals, and a nutritionally essential amino acid for fetal and neonatal development of poultry [
6,
7]. In addition, glycine is the most abundant AA in the body [
8], and it is highly required for neonatal growth and development [
7,
9]. Recently, some reports showed that glycine could alleviate colitis induced by chemicals, small intestine injury induced by endotoxins, and inhibit overproduction of pro-inflammatory cytokines in rats [
10,
11,
12].
Activation of inflammatory signaling pathways can lead to intestinal damage [
13]. Toll-like receptors (TLRs) and nucleotide-binding oligomerization domain proteins (NODs) are important protein families of inflammatory signaling pathways [
14,
15]. These proteins are expressed in many tissues, including the intestine [
16,
17,
18], and play key roles in induction of inflammatory responses by recognition of pathogen-associated molecular patterns (PAMPs) [
16,
19]. Interactions of TLRs or NODs with their specific PAMPs trigger downstream signaling events that lead to activation of nuclear factor-κB (NF-κB), which could further induce the expression of genes related to pro-inflammatory cytokines, such as interleukin-1β (IL-1β), IL-6 and tumor necrosis factor-α (TNF-α) [
20]. As a consequence, these pro-inflammatory cytokines adjust the host’s defense against invading pathogens. However, the intestine can be easily injured by overproduction of these cytokines, especially TNF-α. Adenosine monophosphate-activated protein kinase (AMPK) is a serine/threonine protein kinase, which widely exists in eukaryotic cells [
21]. AMPK can directly mediate metabolic adaptations to a change of energy status [
22]. The overproduction of cytokines, such as TNF-α, increases energy consumption which could activate AMPK [
23]. The activated AMPK could further inhibit the mammalian target of rapamycin (mTOR) signaling pathway to reduce the synthesis of protein in tissues, including the intestine [
24].
The aim of this study was to investigate whether glycine could mitigate lipopolysaccharide (LPS)-induced intestinal injury, and to explore its molecular mechanism(s). We hypothesized that dietary glycine addition could enhance energy status and protein synthesis by suppressing AMPK activation and activating the mTOR signaling pathway, and reduce the production of pro-inflammatory cytokines in the intestine through regulating inflammatory signaling pathways to maintain intestinal integrity. LPS, a component of Gram-negative bacteria, is responsible for neonatal mortality and sepsis, but low concentrations of LPS resulted in tissue protection in some studies [
25]. In the present study,
Escherichia coli (
E. coli) lipopolysaccharide (LPS;
E. coli serotype 055:B5; potency ≥ 5,000,000 EU/mg) was intraperitoneally injected at 100 μg/kg BW (body weight), aimed to establish the model of endotoxemia [
26]. Furthermore, we used the weanling piglet model, which is a suitable animal model for human nutrition research [
27,
28].
3. Discussion
In the present study, the effect of glycine on intestinal integrity after a 4 h E. coli LPS challenge was evaluated in a weaned piglet model. Dietary glycine supplementation improved the intestinal energy status and protein synthesis associated with inhibiting AMPK signaling and activating mTOR signaling, and simultaneously reduced the intestinal inflammatory response associated with inhibiting inflammatory signaling pathways (TLR4 and NOD), and as a consequence, improved intestinal integrity.
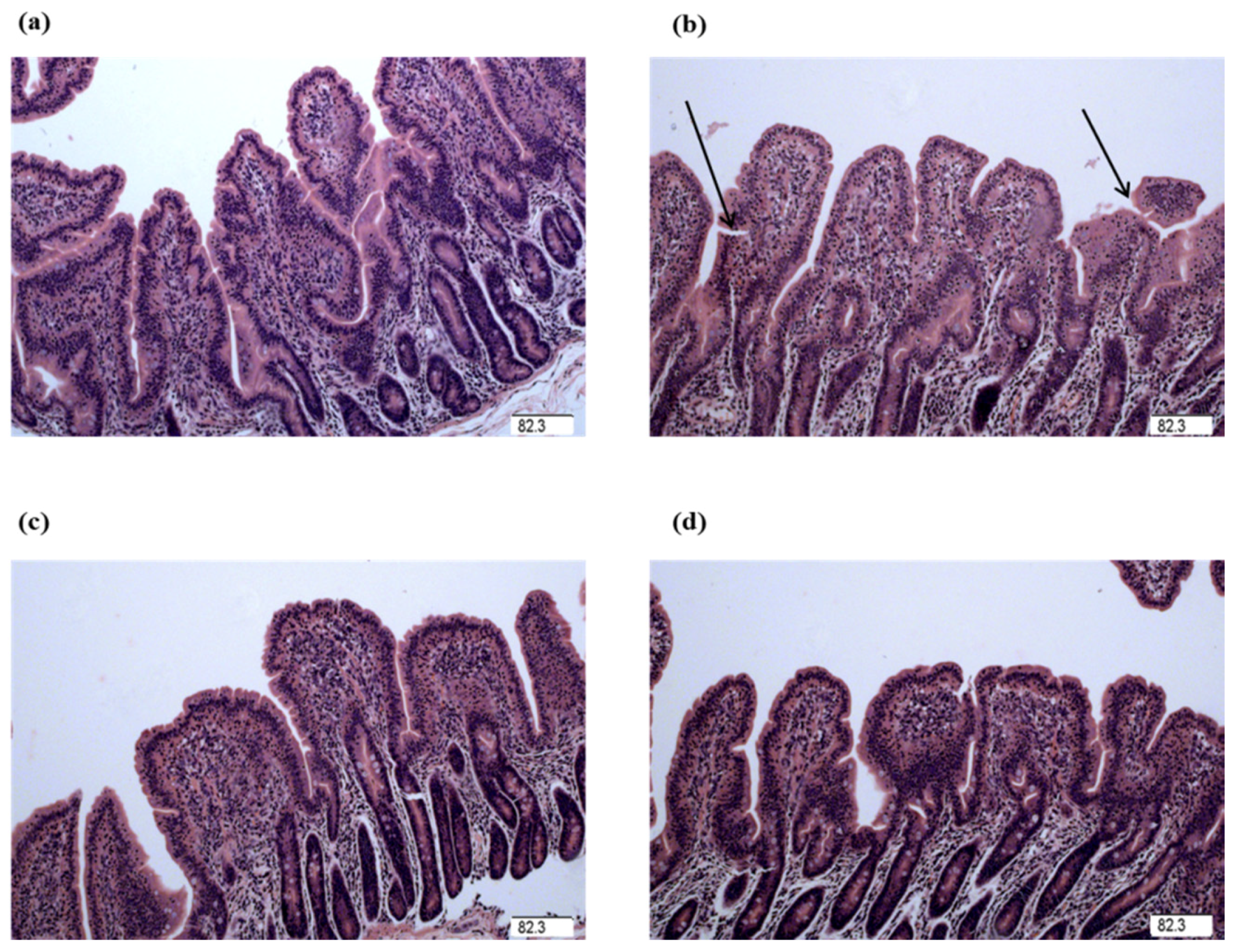
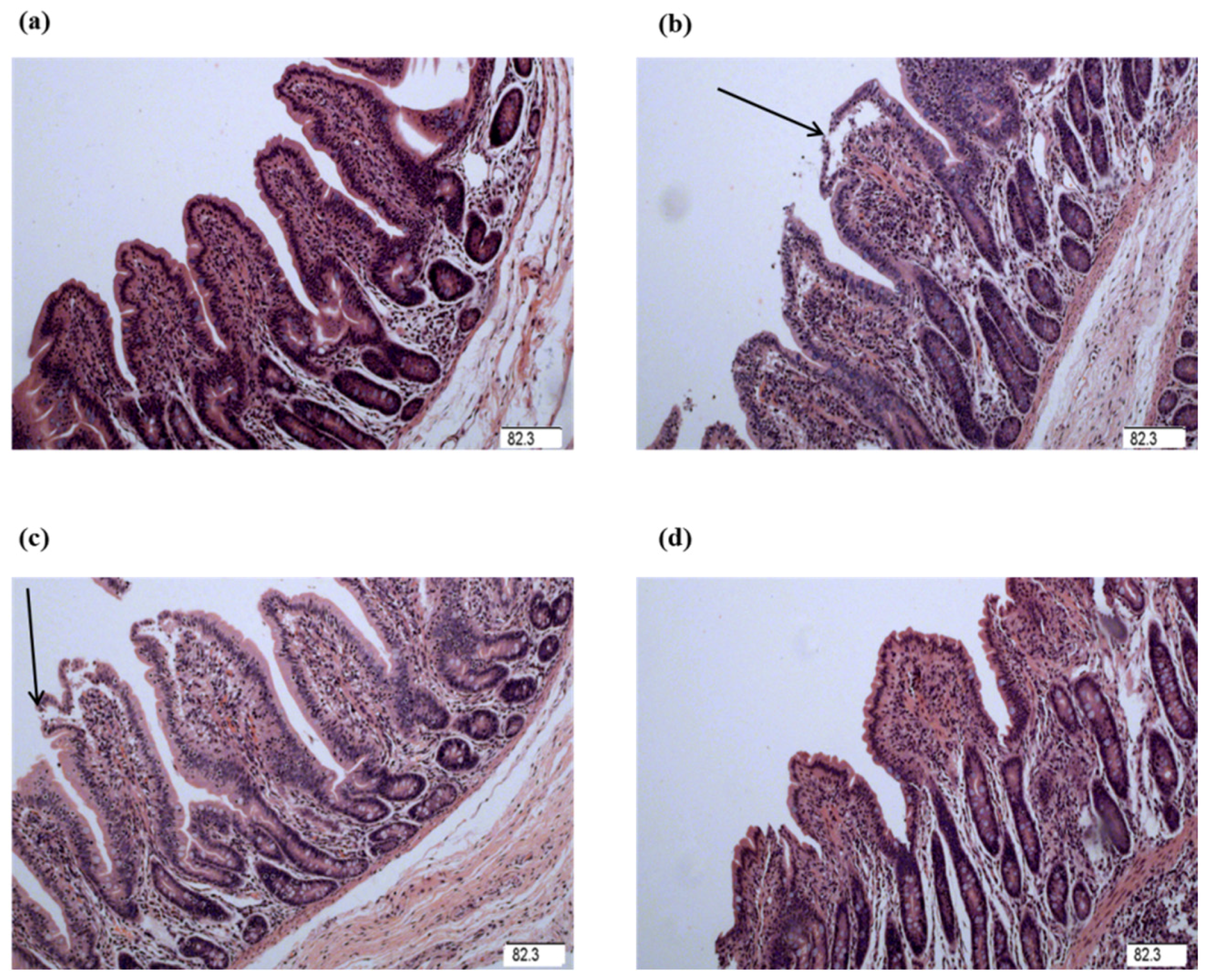
The intestinal mucosal integrity of weanling piglets is closely related to physical health and nutrient digestion and absorption capability [
24,
29]. Villus height, crypt depth, and VCR were determined to indicate gross intestinal morphology [
3]. In this study, the decreased jejunal crypt depth and increased jejunal VCR of the piglets fed glycine indicated that the digestive juice can be secreted easily into intestinal lumen, which showed glycine played a role in maintaining the integrity in mucosal structure in weanling piglets. Wang et al. reported supplementation with glycine increased intestinal villus height in nursery piglets [
7]. Effenberger-Neidnicht et al. found that supplementation with glycine improved intestinal architecture and reduced the LPS-induced intestinal accumulation of blood in rats [
11]. The above studies indicated that glycine had a positive effect on the intestinal mucosal integrity.
Protein and DNA are the basis for repair and proliferation of epithelial cells. The ratio of RNA/DNA is an indicator reflecting the cell capacity for protein synthesis [
30]. The ratio of protein/DNA is a sensitive indicator of protein mass and cell size [
24]. The decreased ileal RNA/DNA ratio in LPS-challenged piglets indicated that the mucosal cell capacity for repair was decreased, affected by LPS. The piglets fed diets containing glycine had increased jejunal and ileal protein mass, protein/DNA ratio, and RNA/DNA ratio in the present study. These results illustrated that glycine is beneficial to intestine mucosal repair after LPS-induced injury. Similar to our data, Lee et al. [
31] reported that glycine supplementation increased intestinal protein mass in the rats under ischemia–reperfusion injury. In addition, Stoll et al. [
32] demonstrated that glycine could be utilized directly to synthesize protein in the intestinal tract. Furthermore, Wang et al. [
7] demonstrated that glycine was the precursor of purines, which were involved in protein synthesis and cell proliferation.
The intestinal epithelial barrier can reduce the ability of luminal pathogens and their toxins to invade the mucosa, preventing the penetration of luminal bacteria into the mucosa, to maintain gut homeostasis [
33]. The disordered function of the intestinal epithelial barrier often leads to inflammatory disease in the intestine [
34]. The integrity of the intestinal epithelial barrier is maintained by cohesive interactions between epithelial cells to form tight junctions [
34]. Claudin-1, which is an important protein in formation of tight junctions, determines permeability characteristics in many tissues, especially the intestine [
35]. Therefore, the greater abundance of claudin-1 often reflects improved function of the epithelial barrier [
36]. In the present study, LPS injection decreased the ileal protein abundance of claudin-1, which showed that LPS impaired the function of intestinal epithelial barrier. Similarly, some previous reports demonstrated that LPS challenge decreased the abundance of claudin-1 [
37]. However, supplementation with glycine in the diets did not affect the abundance of claudin-1 in the piglets in our study. Few reports studied the effect of glycine on the intestinal tight junctions, so the pathway to repair injured tight junction in the intestine should be further studied in the future.
Citrate synthase, ICD, and α-KGDHC are key enzymes involved in the TCA cycle, which is a central route for energy production [
38]. CS catalyzes the first step of the TCA cycle by attaching molecules of acetate and attaching them to oxaloacetate [
39]. ICD, which exists in mitochondria and cytoplasm, is responsible for catalyzing the oxidative decarboxylation of isocitrate into α-ketoglutarate and CO
2 [
40]. α-KGDHC is a multi-enzymatic complex which converts α-ketoglutarate into succinyl-CoA [
41]. In our experiment, LPS challenge reduced the activities of ICD in jejunum and CS, and α-KGDHC in ileum, which indicated that the energy production efficiency decreased. This is consistent with a previous report which showed that LPS challenge decreased the activities of jejunal CS, ICD, and α-KGDHC, and ileal ICD in weanling piglets [
4]. After supplementation with glycine in the diets, the activities of α-KGDHC in jejunum and CS in ileum were increased. Therefore, it is possible that glycine could improve the energy production efficiency in intestinal mucosa by enhancing the key enzyme activities of the TCA cycle.
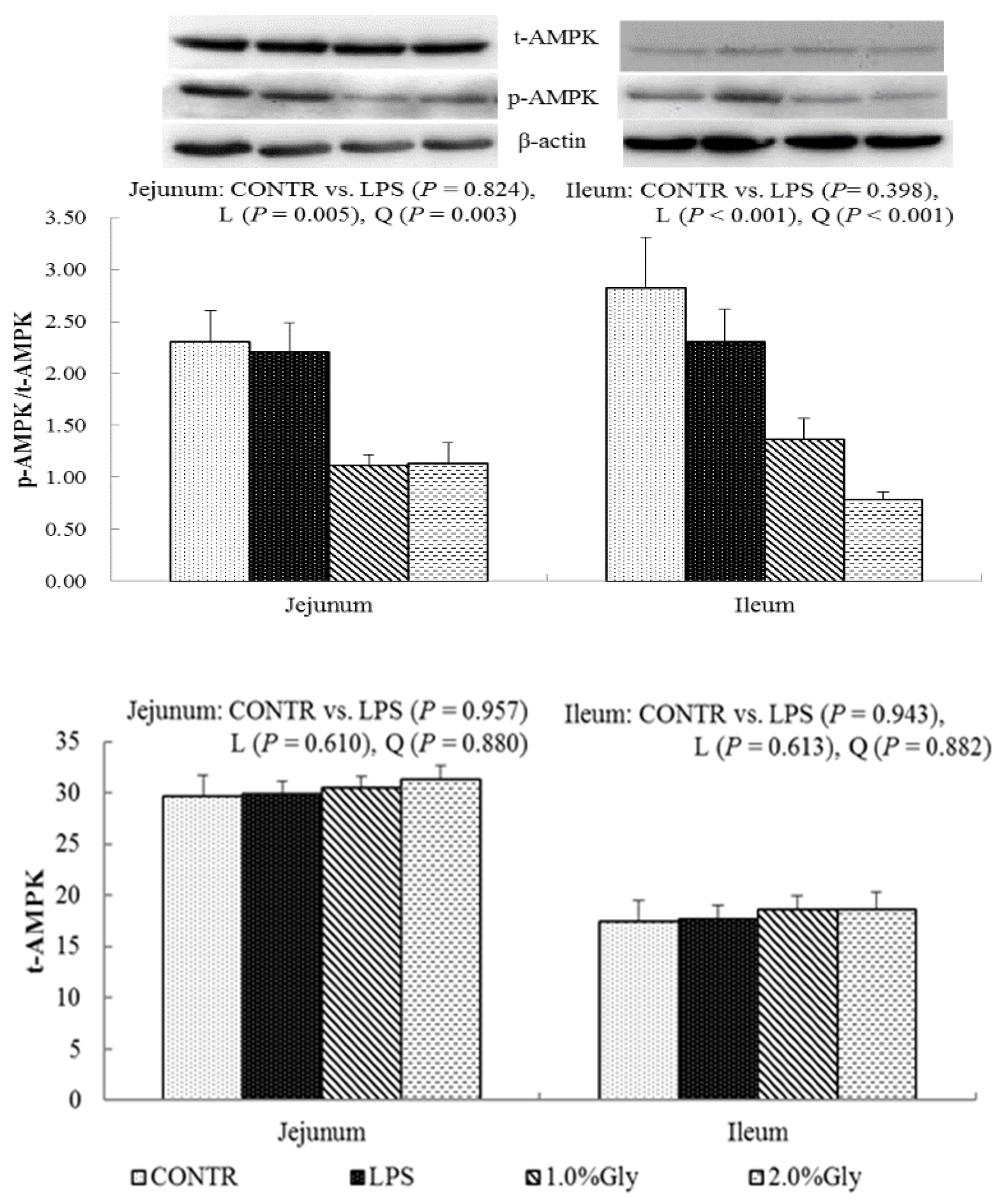
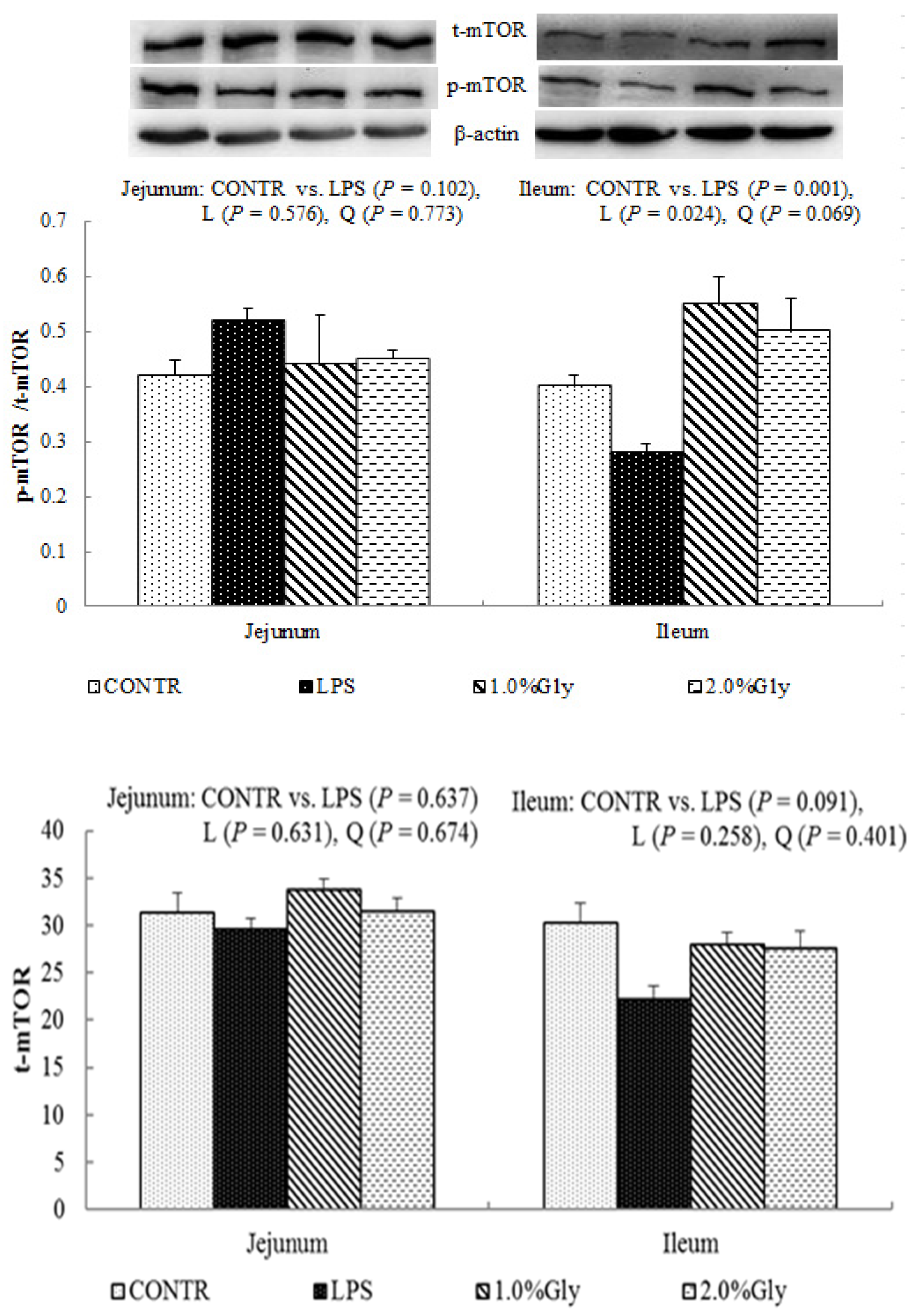
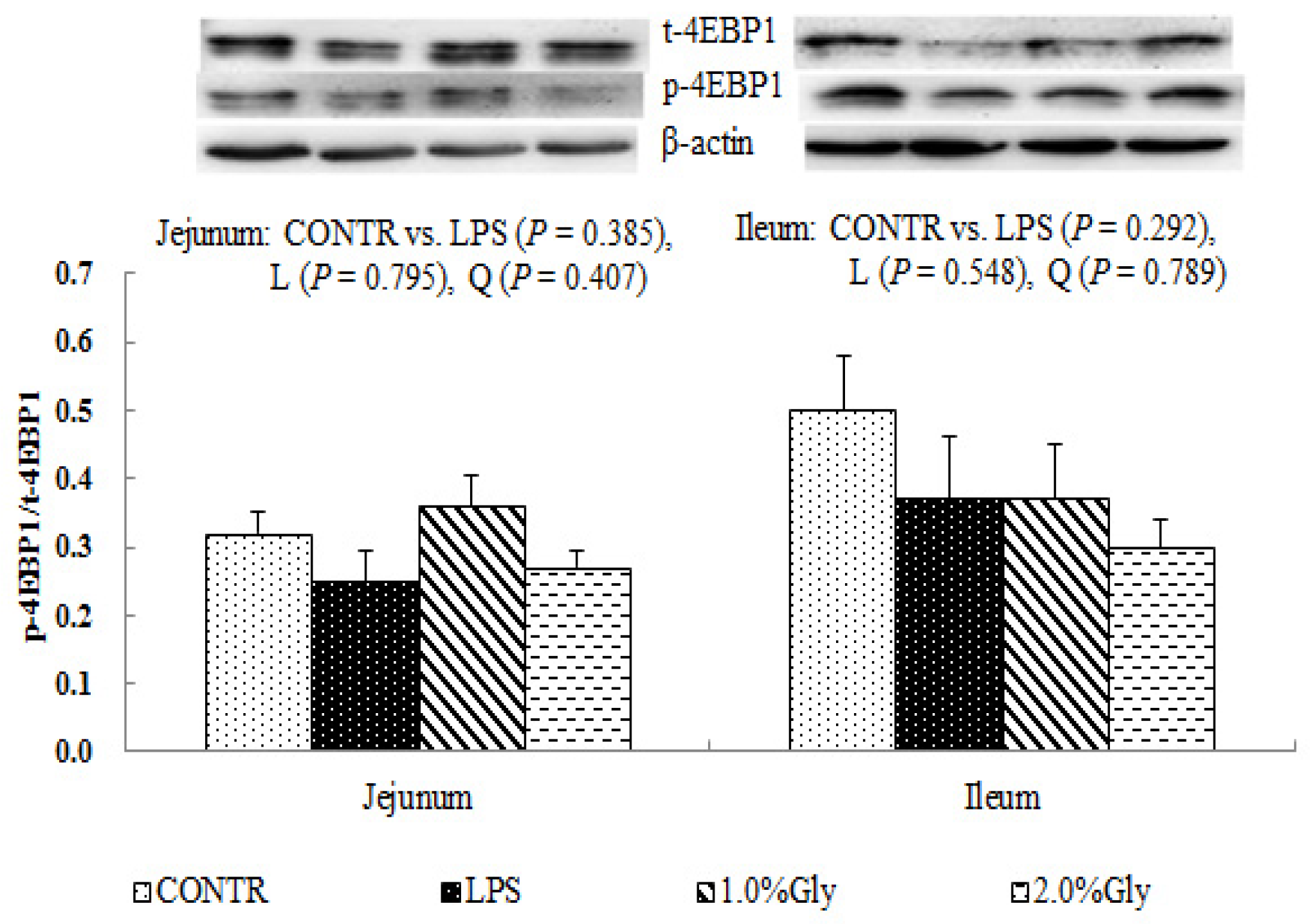
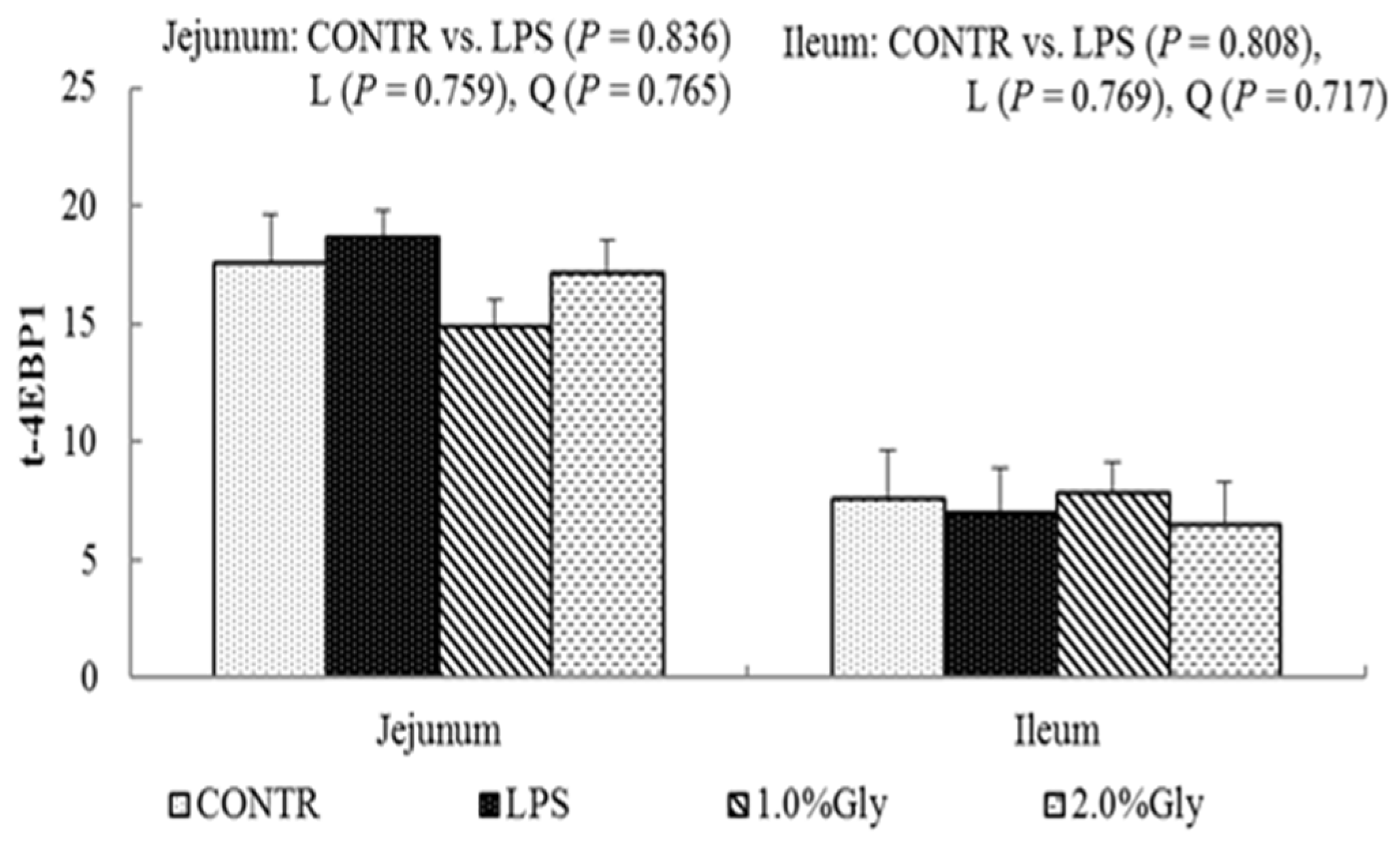
We hypothesized that glycine had a beneficial effect on intestinal integrity through enhancing energy production and protein synthesis in epithelial cells. AMPK, as an energy regulator, maintains the intracellular energy balance in eukaryons [
42]. When the intracellular AMP/ATP ratio increases, AMPK is activated by phosphorylation [
43]. The activated AMPK can switch on ATP-producing processes while synchronously switching off ATP-consuming processes to restore the cellular energy status [
44]. Supplementation with glycine decreased jejunal and ileal p-AMPK/t-AMPK ratio, which illustrated dietary glycine could potentially enhance ATP-consumption to synthesize protein. mTOR is a serine–threonine kinase which controls many important aspects in mammalian cell functions, such as protein synthesis [
45]. Its activity is modulated by various intracellular and extracellular factors (especially AAs and energy), meanwhile, mTOR adjusts rates of translation, transcription, protein degradation, cell signaling, and metabolism [
44]. Specially, mTOR signaling plays a critical role in maintaining intestinal health [
45,
46]. Similar to our results, a previous report showed that LPS injection decreased jejunal mRNA expression of mTOR [
47]. It is known that LPS could inhibit intestinal protein synthesis through suppressing activation of mTOR. The piglets fed diets supplemented with glycine had increased ileal p-mTOR/t-mTOR ratio, which demonstrated glycine could relieve the reduction of mucosal protein synthesis caused by LPS injury in the ileum. These results are in agreement with the decreased p-AMPK/t-AMPK ratio in piglets fed the diets supplemented with glycine. Therefore, we propose that the mechanism may be that glycine improves the intestinal mucosal energy status, and further activates mTOR signaling pathway to enhance protein synthesis and mucosal repair.
We hypothesized that supplementation with glycine improved intestinal integrity by inhibiting intestinal TLR4 and NODs, and their respective downstream signals, further reducing the inflammatory response. In the current study, LPS injection increased the mRNA abundance of TLR4 (TLR4, MyD88, and NF-κB in jejunum) and NOD signaling-related genes (NOD2 and RIPK2 in jejunum and ileum) which reflected LPS-induced intestinal inflammation by activation of TLR4 and NOD pathways. Supplementation with glycine reduced mRNA expressions of TLR4 (TLR4, LBP, MyD88, TRAF6, and NF-κB in jejunum) and NOD signaling-related genes (NOD2 and RIPK2 in jejunum and ileum) in the LPS-challenged piglets, which illustrated that glycine could relieve intestinal inflammation by inhibiting activation of inflammatory signaling pathways. In general, the inflammatory cytokines, such as TNF-α, IL-1β, and IL-6 can be overproduced when the inflammatory signaling pathways (TLR4 and NODs) are activated. Similar to our study, Tsune et al. [
10] reported that glycine relieved colitis in rats by reducing mRNA expression of TNF-α and IL-1β. Furthermore, Stoffels et al. [
48] demonstrated that injection of glycine before intestinal surgery could alleviate inflammation by decreasing mRNA expression of IL-6 and TNF-α.
Activation of TLR4 and NOD signaling could prevent against pathogens invading by triggering the production of pro-inflammatory cytokines and inflammatory response. However, over activation of inflammatory signaling pathways also lead to collateral host tissue injury [
49]. To prevent excessive and harmful inflammatory responses, these inflammatory signaling pathways are negatively controlled by multiple mechanisms. So far, many negative regulators of TLR4 signaling (Tollip and SOCS1) and NOD signaling (ERBB2IP and centaurin β1) have been identified and characterized [
50,
51]. The current results showed that the piglets challenged with LPS had decreased mRNA expression of Tollip both in jejunum and ileum, as well as ERBB2IP and centaurin β1 in ileum, which is in agreement with the report of Wang et al. [
52]. These results reflect that LPS challenge reduced the mRNA expression of TLR4 and NOD negative regulators, which is in agreement with increased mRNA expression of TLR4 and NOD signaling-related genes. Fujimoto et al. reported that excessive inflammatory cytokines enforced the expression of SOCS1, which resulted in decreased response of cells to TLR ligands [
53]. In accordance with this, our present study showed that LPS challenge increased mRNA expressions of jejunal SOCS1, which indicates that SOCS1 might play a key role in intestinal self-protection. Supplementation with glycine increased mRNA expression of ileal ERBB2IP. Previous studies showed that Tollip bound to IL-1 receptor-associated kinase (IRAK) and inhibited IRAK phosphorylation to downregulate TLR4 signaling [
54]. This indicates that supplementation with glycine could suppress the activity of IRAK by increasing the mRNA expression of Tollip, resulting in impairing the signaling from TLR4 to downstream pathways, and reducing the synthesis of pro-inflammatory cytokines.
In the present study, the effects of LPS challenge or glycine supplementation on some parameters were inconsistent in different sites of the intestine. This may be due to the difference in the anatomy and physiology among the different sites of the intestine [
37]. In addition, LPS caused dynamic changes in the physiological variables, and gene and protein expression of inflammatory signaling pathways [
55,
56]. After 28 days feeding of Gly, the efficacy of Gly has risen. However, having only one time point (4 h) selected, to measure the effect of LPS on various physiological variables and gene and protein expression, was not perfect. Therefore, in future studies, sample collections at more time points are needed to better understand the dynamic effect of LPS on intestinal injury. Furthermore, the maintenance of normal blood flow through microcirculation plays a fundamental role in the protection and healing of intestinal mucosa [
57]. Many previous studies reported that the intestinal injury induced by various factors can be alleviated through improving blood flow in microcirculation of duodenum and colon [
58,
59,
60,
61]. In the future, the blood flow of jejunal and ileal mucosa needs to be measured to better explore the mechanism of effects of LPS or glycine on the intestine.