Dental Pulp Stem Cell-Derived, Scaffold-Free Constructs for Bone Regeneration
Abstract
1. Introduction
2. Results
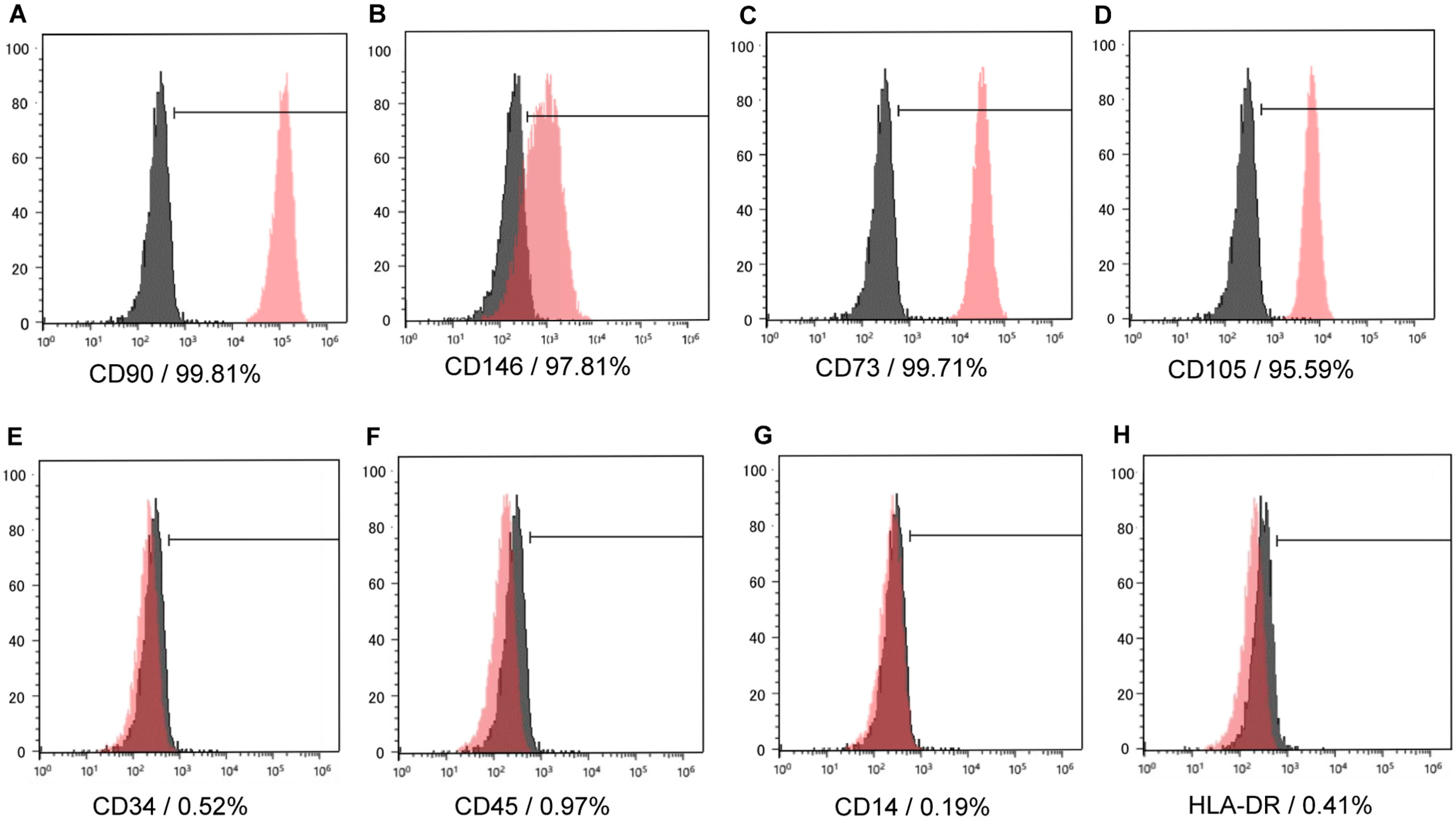
2.1. Characterization of Human Dental Pulp Stem Cells (hDPSCs)
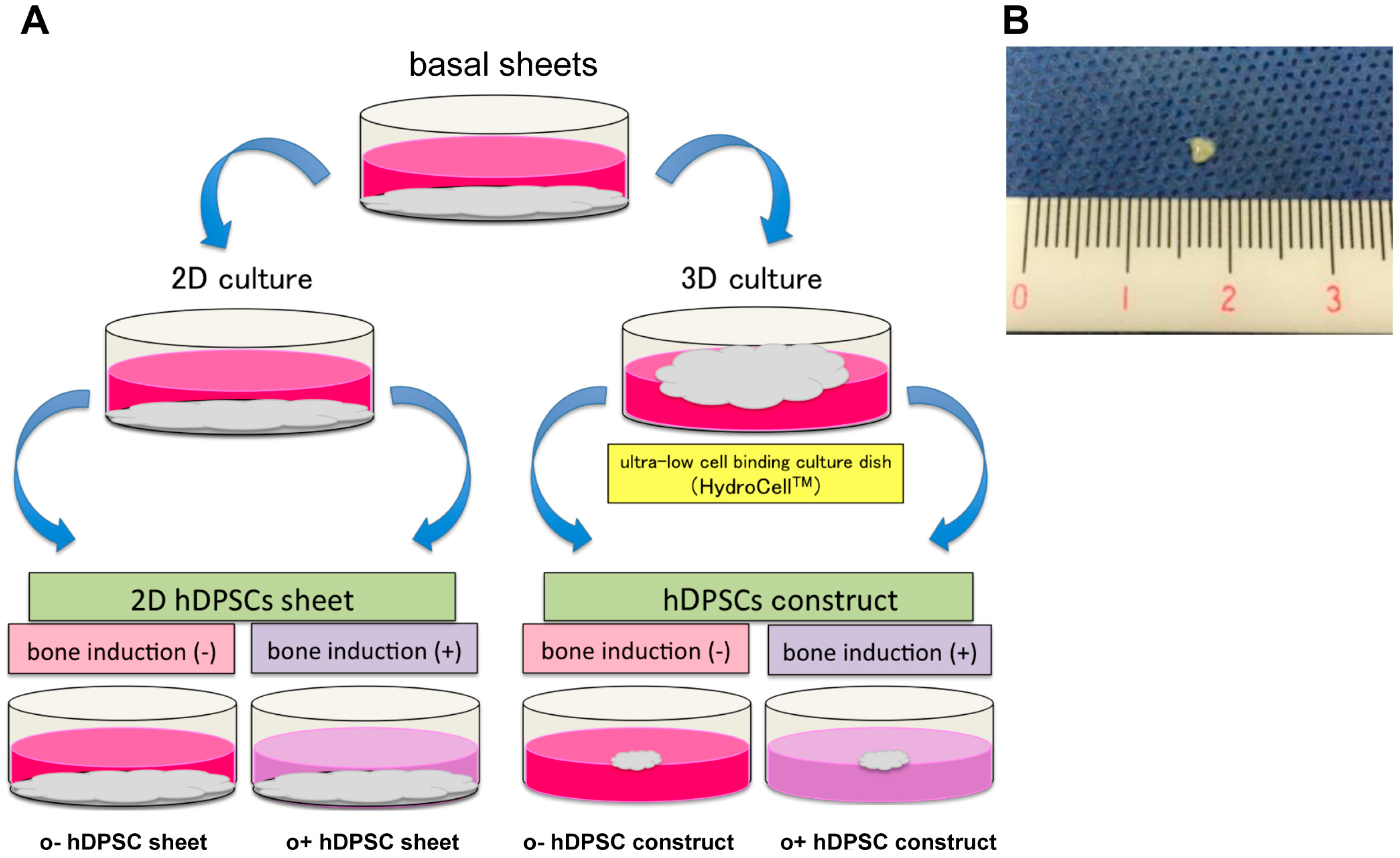
2.2. Findings of hDPSC Construct
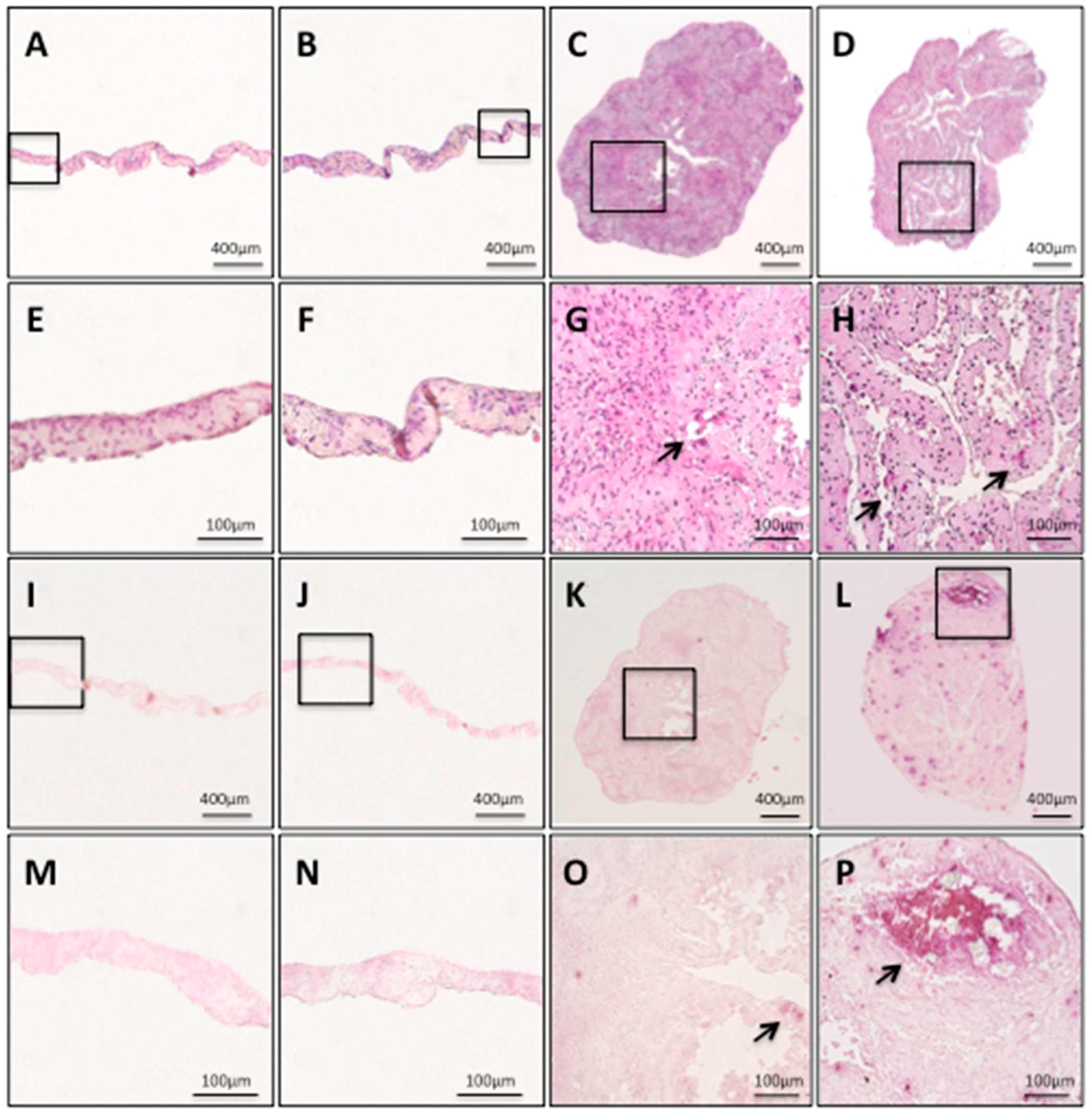
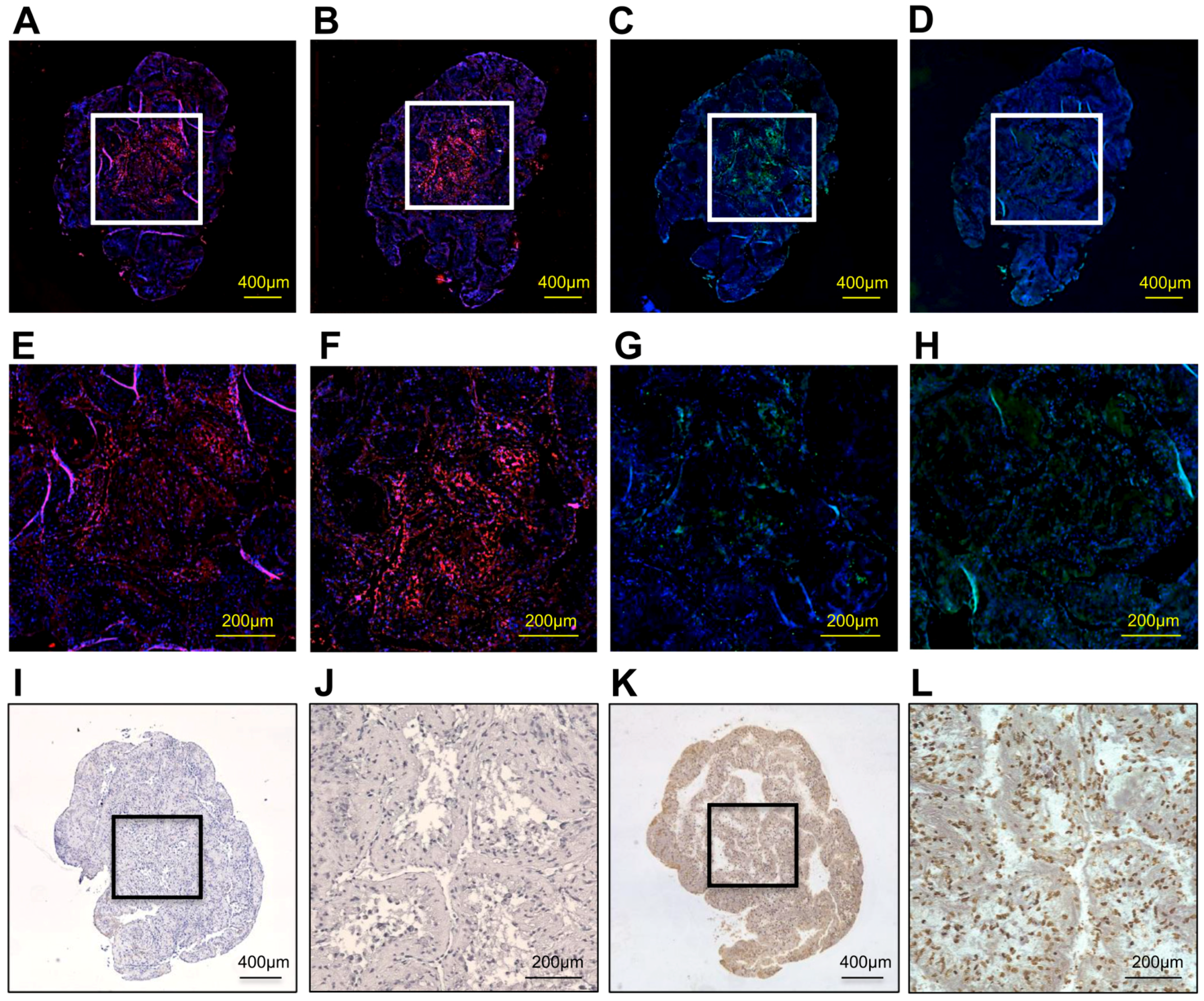
2.3. Histological Findings
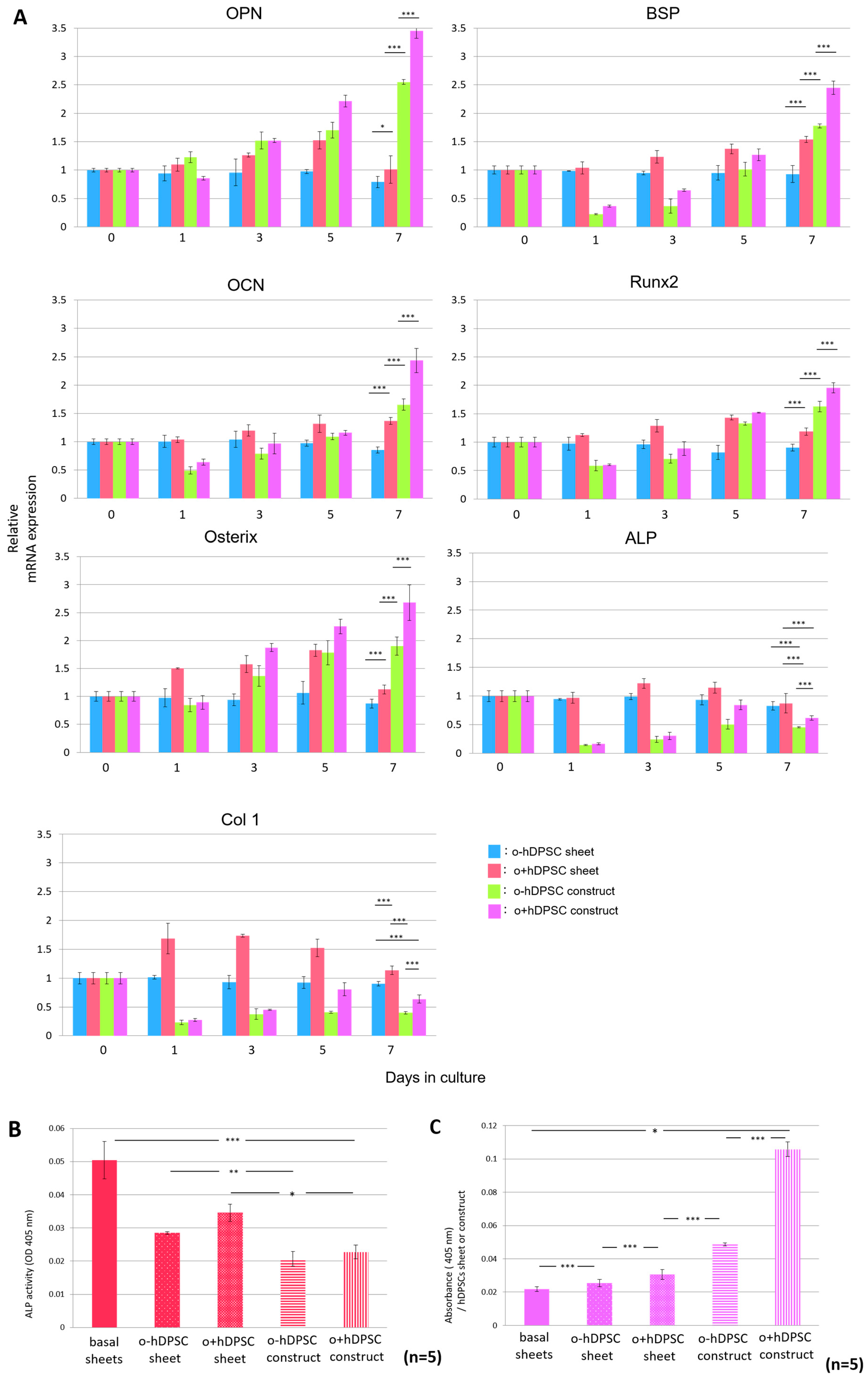
2.4. Expression of Bone-Related Genes and Calcium Content in hDPSC Construct
3. Discussion
4. Materials and Methods
4.1. Cell Isolation and Culture from Human Dental Pulp Tissue
4.2. Flow Cytometry Analysis
4.3. Preparation of hDPSC Constructs
- hDPSC sheet: cultured in basal medium under 2D culture.
- o+hDPSC sheet: cultured in osteogenic medium under 2D culture.
- hDPSC construct: cultured in basal medium under 3D culture.
- o+hDPSC construct: cultured in osteogenic medium under 3D culture.
4.4. Characterization of the hDPSC Construct
4.5. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| 3D | three-dimensional |
| 2D | monolayer culture |
| TEC | tissue-engineered constructs |
| DPSC | dental pulp stem cells |
| DPC | dental pulp cells |
| hDPSC | human dental pulp stem cells |
| PBS | phosphate-buffered saline |
| PCR | polymerase chain reaction |
| TUNEL | terminal deoxynucleotidyl transferase dUTP nick-end labeling |
| OPN | osteopontin |
| BSP | bone sialoprotein |
| OCN | osteocalcin |
| Col 1 | type I collagen |
| FBS | fetal bovine serum |
References
- Laurencin, C.; Khan, Y.; El-Amin, S.F. Bone graft substitutes. Expert Rev. Med. Devices 2006, 3, 49–57. [Google Scholar] [CrossRef] [PubMed]
- von Arx, T.; Cochran, D.L.; Hermann, J.S.; Schenk, R.K.; Buser, D. Lateral ridge augmentation using different bone fillers and barrier membrane application. A histologic and histomorphometric pilot study in the canine mandible. Clin. Oral Implants Res. 2001, 12, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Kostakis, G.C. An investigation of post-operative morbidity following iliac crest graft harvesting. Br. Dent. J. 2004, 196, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Harada, N.; Sato, K.; Abe, S.; Yamanaka, K.; Matushita, T. Stem cell therapy: Is there a future for reconstruction of large bone defect? Injury 2016, 47, S47–S51. [Google Scholar] [CrossRef]
- Yamamoto, M.; Kawashima, N.; Takashino, N.; Koizumi, Y.; Takimoto, K.; Suzuki, N.; Saito, M.; Suda, H. Three-dimensional spheroid culture promotes odonto/osteoblastic differentiation of dental pulp cells. Arch. Oral Biol. 2014, 59, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Elbert, D.L. Bottom-up tissue engineering. Curr. Opin. Biotechnol. 2011, 22, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Cukierman, E.; Pankov, R.; Yamada, K.M. Cell interactions with three-dimensional matrices. Curr. Opin. Cell Biol. 2002, 14, 633–639. [Google Scholar] [CrossRef]
- Curcio, E.; Salerno, S.; Barbieri, G.; De Bartolo, L.; Drioli, E.; Bader, A. Mass transfer and metabolic reactions in hepatocyte spheroids cultured in rotating wall gas-permeable membrane system. Biomaterials 2007, 28, 5487–5497. [Google Scholar] [CrossRef] [PubMed]
- De Jong, W.H.; Eelco Bergsma, J.; Robinson, J.E.; Bos, R.R. Tissue response to partially in vitro predegraded poly-l-lactide implants. Biomaterials 2005, 26, 1781–1791. [Google Scholar] [CrossRef] [PubMed]
- Okano, T.; Yamada, N.; Sakai, H.; Sakurai, Y. A novel recovery system for cultured cells using plasma-treated polystyrene dishes grafted with poly(N-isopropylacrylamide). J. Biomed. Mater. Res. 1993, 27, 1243–1251. [Google Scholar] [CrossRef] [PubMed]
- Memon, I.A.; Sawa, Y.; Fukushima, N.; Matsumiya, G.; Miyagawa, S.; Taketani, S.; Sakakida, S.K.; Kondoh, H.; Aleshin, A.N.; Shimizu, T.; et al. Repair of impaired myocardium by means of implantation of engineered autologous myoblast sheets. J. Thorac. Cardiovasc. Surg. 2005, 130, 1333–1341. [Google Scholar] [CrossRef] [PubMed]
- Ohki, T.; Yamato, M.; Murakami, D.; Takagi, R.; Yang, J.; Namiki, H.; Okano, T.; Takasaki, K. Treatment of oesophageal ulcerations using endoscopic transplan- tation of tissue-engineered autologous oral mucosal epithelial cell sheets in a canine model. Gut 2006, 55, 1704–1710. [Google Scholar] [CrossRef] [PubMed]
- Nishida, K.; Yamato, M.; Murakami, D.; Watanabe, K.; Yamamoto, K.; Adachi, E.; Nagai, S.; Kikuchi, A.; Maeda, N.; Watanabe, H.; et al. Corneal reconstruction with tissue-engineered cell sheets composed of autologous oral mucosal epithelium. N. Engl. J. Med. 2004, 351, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Takewaki, M.; Kajiya, M.; Takeda, K.; Sasaki, S.; Motoike, S.; Komatsu, N.; Matsuda, S.; Ouhara, K.; Mizuno, N.; Fujita, T.; et al. MSC/ECM cellular complexes induce periodontal tissue regeneration. J. Dent. Res. 2017, 96, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Handschel, J.; Naujoks, C.; Depprich, R.; Lammers, L.; Kübler, N.; Meyer, U.; Wiesmann, H.P. Embryonic stem cells in scaffold-free three-dimensional cell culture: Osteogenic differentiation and bone generation. Head Face Med. 2011, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Dang, P.N.; Herberg, S.; Varghai, D.; Riazi, H.; Varghai, D.; McMillan, A.; Awadallah, A.; Phillips, L.M.; Jeon, O.; Nguyen, M.K.; et al. Endochondral ossification in critical-sized bone defects via readily implantable scaffold-free stem cell constructs. Stem Cells Transl. Med. 2017, 6, 1644–1659. [Google Scholar] [CrossRef] [PubMed]
- Shimomura, K.; Ando, W.; Moriguchi, Y.; Sugita, N.; Yasui, Y.; Koizumi, K.; Fujie, H.; Hart, D.; Yoshikawa, H.; Nakamura, N. Next generation mesenchymal stem cell (MSC)–based cartilage repair using scaffold-free tissue engineered constructs generated with synovial mesenchymal stem cells. Cartilage 2015, 6, 13S–29S. [Google Scholar] [CrossRef] [PubMed]
- Gronthos, S.; Mankani, M.; Brahim, J.; Robey, P.G.; Shi, S. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2000, 97, 13625–13630. [Google Scholar] [CrossRef] [PubMed]
- Gronthos, S.; Brahim, J.; Li, W.; Fisher, L.W.; Cherman, N.; Boyde, A.; DenBesten, P.; Robey, P.G.; Shi, S. Stem cell properties of human dental pulp stem cells. J. Dent. Res. 2002, 81, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Ponnaiyan, D.; Jegadeesan, V. Comparison of phenotype and differentiation marker gene expression profiles in human dental pulp and bone marrow mesenchymal stem cells. Eur. J. Dent. 2014, 8, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.N.; Mar, K.; Chang, H.C.; Chiang, Y.L.; Hu, H.Y.; Lai, C.C.; Chu, R.M.; Ma, C.M. A comparison between adipose tissue and dental pulp as sources of MSCs for tooth regeneration. Biomaterials 2011, 32, 6995–7005. [Google Scholar] [CrossRef] [PubMed]
- Takebe, Y.; Tatehara, S.; Fukushima, T.; Tokuyama-Toda, R.; Yasuhara, R.; Mishima, K.; Satomura, K. Cryopreservation method for the effective collection of dental pulp stem cells. Tissue Eng. Part C Methods 2017, 23, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Annibali, S.; Bellavia, D.; Ottolenghi, L.; Cicconetti, A.; Cristalli, M.P.; Quaranta, R.; Pilloni, A. Micro-CT and PET analysis of bone regeneration induced by biodegradable scaffolds as carriers for dental pulp stem cells in a rat model of calvarial “critical size” defect: Preliminary data. J. Biomed. Mater. Res. B Appl. Biomater. 2014, 102, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.C.; E, L.L.; Wang, D.S.; Su, F.; Wu, X.; Shi, Z.P.; Lv, Y.; Wang, J.Z. Reconstruction of alveolar bone defects using bone morphogenetic protein 2 mediated rabbit dental pulp stem cells seeded on nano-hydroxyapatite/collagen/poly(l-lactide). Tissue Eng. Part A 2011, 17, 2417–2433. [Google Scholar] [CrossRef] [PubMed]
- Bosnakovski, D.; Mizuno, M.; Kim, G.; Ishiguro, T.; Okumura, M.; Iwanaga, T.; Kadosawa, T.; Fujinaga, T. Chondrogenic differentiation of bovine bone marrow mesenchymal stem cells in pellet culture system. Exp. Hematol. 2004, 32, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Richards, D.J.; Trusk, C.T.; Visconti, R.P.; Yost, M.J.; Kindy, M.S.; Drake, C.J.; Argraves, W.S.; Markwald, R.R.; Mei, Y. 3D printing facilitated scaffold-free tissue unit fabrication. Biofabrication 2014, 6, 024111. [Google Scholar] [CrossRef] [PubMed]
- Moldovan, L.; Barnard, A.; Gil, C.H.; Lin, Y.; Grant, M.B.; Yoder, M.C.; Prasain, N.; Moldovan, N.I. iPSC-Derived Vascular Cell Spheroids as Building Blocks for Scaffold-Free Biofabrication. Biotechnol. J. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Kizawa, H.; Nagao, E.; Shimamura, M.; Zhang, G.; Torii, H. Scaffold-free 3D bio-printed human liver tissue stably maintains metabolic functions useful for drug discovery. Biochem. Biophys. Rep. 2017, 10, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yamato, M.; Shimizu, T.; Sekine, H.; Ohashi, K.; Kanzaki, M.; Ohki, T.; Nishida, K.; Okano, T. Reconstruction of functional tissues with cell sheet engineering. Biomaterials 2007, 28, 5033. [Google Scholar] [CrossRef] [PubMed]
- Kutuna, T.; Sato, M.; Ishihara, M.; Furukawa, K.S.; Nagai, T.; Kikuchi, M.; Ushida, T.; Mochida, J. Noninvasive evaluation of tissue-engineered cartilage with time-resolved laser-induced fluorescence spectroscopy. Tissue Eng. Part C Methods 2010, 16, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Czajka, C.A.; Mehesz, A.N.; Trusk, T.C.; Yost, M.J.; Drake, C.J. Scaffold-free tissue engineering: Organization of the tissue cytoskeleton and its effects on tissue shape. Ann. Biomed. Eng. 2014, 42, 1049–1061. [Google Scholar] [CrossRef] [PubMed]
- Yannas, I.V.; Tzeranis, D.S.; Harley, B.A.; So, P.T. Biologically active collagen-based scaffolds: Advances in processing and characterization. Philos. Trans. Ser. A Math Phys. Eng. Sci. 2010, 368, 2123–2139. [Google Scholar] [CrossRef] [PubMed]
- Kew, S.J.; Gwynne, J.H.; Enea, D.; Abu-Rub, M.; Pandit, A.; Zeugolis, D.; Brooks, R.A.; Rushton, N.; Best, S.M.; Cameron, R.E. Regeneration and repair of tendon and ligament tissue using collagen fibre biomaterials. Acta Biomater. 2011, 7, 3237–3247. [Google Scholar] [CrossRef] [PubMed]
- Leeming, D.J.; Henriksen, K.; Byrjalsen, I.; Qvist, P.; Madsen, S.H.; Garnero, P.; Karsdal, M.A. Is bone quality associated with collagen age? Osteoporos. Int. 2009, 20, 1461–1470. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhu, Y.Q.; Jiang, L.; Peng, W.; Ritchie, H.H. Hypoxia promotes mineralization of human dental pulp stem cells. J. Endod. 2011, 36, 799–802. [Google Scholar] [CrossRef] [PubMed]
- Amemiya, K.; Kaneko, Y.; Muramatsu, T.; Shimono, M.; Inoue, T. Pulp cell responses during hypoxia and reoxygenation in vitro. Eur. J. Oral Sci. 2003, 111, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Sakdee, J.B.; White, R.R.; Pagonis, T.C.; Hauschka, P.V. Hypoxia-amplified proliferation of human dental pulp cells. J. Endod. 2009, 35, 818–823. [Google Scholar] [CrossRef] [PubMed]
- Iida, K.; Takeda-Kawaguchi, T.; Tezuka, Y.; Kunisada, T.; Shibata, T.; Tezuka, K. Hypoxia enhances colony formation and proliferation but inhibits differentiation of human dental pulp cells. Arch. Oral Biol. 2010, 55, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Shida, T.; Takahashi, H.; Tago, K.; Deguchi, S. Novel method for preparation of cell sheets using human alveolar bone periosteal cells. Nihon Shishubyo Gakkai Kaishi 2013, 55, 24–36. [Google Scholar] [CrossRef]
- Seibel, M.J.; Woitge, H.W. Basic principles and clinical applications of biochemical markers of bone metabo-lism: Biochemical and technical aspects. J. Clin. Densitom. 1999, 2, 299–321. [Google Scholar] [CrossRef]
| Primer | Forward Primer | Reverse Primer | Size (bp) |
|---|---|---|---|
| GAPDH | GAGTCAACGGATTTGGTCGT | TTGATTTTGGAGGGATCTCG | 451 |
| ALP | ACCATTCCCACGTCTTCACATTT | AGACATTCTCTCGTTCACCGCC | 476 |
| Col 1 | CACTGGTGATGCTGGTCCTG | CGAGGTCACGGTCACGAAC | 335 |
| OPN | TGAAACGAGTCAGCTGGATG | TGAAATTCATGGCTGTGGAA | 162 |
| BSP | CAACAGCACAGAGGCAGAAA | CGTACTCCCCCTCGTATTCA | 247 |
| OCN | GTGCAGAGTCCAGCAAAGGT | TCAGCCAACTCGTCACAGTC | 175 |
| Runx2 | ACTTCCTGTGCTCGGTGCT | GACGGTTATGGTCAAGGTGAA | 289 |
| Osterix | CCAGCCAACACTCCTACTCC | GCCTTGCCATACACTTGC | 255 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tatsuhiro, F.; Seiko, T.; Yusuke, T.; Reiko, T.-T.; Kazuhito, S. Dental Pulp Stem Cell-Derived, Scaffold-Free Constructs for Bone Regeneration. Int. J. Mol. Sci. 2018, 19, 1846. https://doi.org/10.3390/ijms19071846
Tatsuhiro F, Seiko T, Yusuke T, Reiko T-T, Kazuhito S. Dental Pulp Stem Cell-Derived, Scaffold-Free Constructs for Bone Regeneration. International Journal of Molecular Sciences. 2018; 19(7):1846. https://doi.org/10.3390/ijms19071846
Chicago/Turabian StyleTatsuhiro, Fukushima, Tatehara Seiko, Takebe Yusuke, Tokuyama-Toda Reiko, and Satomura Kazuhito. 2018. "Dental Pulp Stem Cell-Derived, Scaffold-Free Constructs for Bone Regeneration" International Journal of Molecular Sciences 19, no. 7: 1846. https://doi.org/10.3390/ijms19071846
APA StyleTatsuhiro, F., Seiko, T., Yusuke, T., Reiko, T.-T., & Kazuhito, S. (2018). Dental Pulp Stem Cell-Derived, Scaffold-Free Constructs for Bone Regeneration. International Journal of Molecular Sciences, 19(7), 1846. https://doi.org/10.3390/ijms19071846








