Balneotherapy, Immune System, and Stress Response: A Hormetic Strategy?
Abstract
1. Introduction
2. Balneotherapy as a Strategy for Health
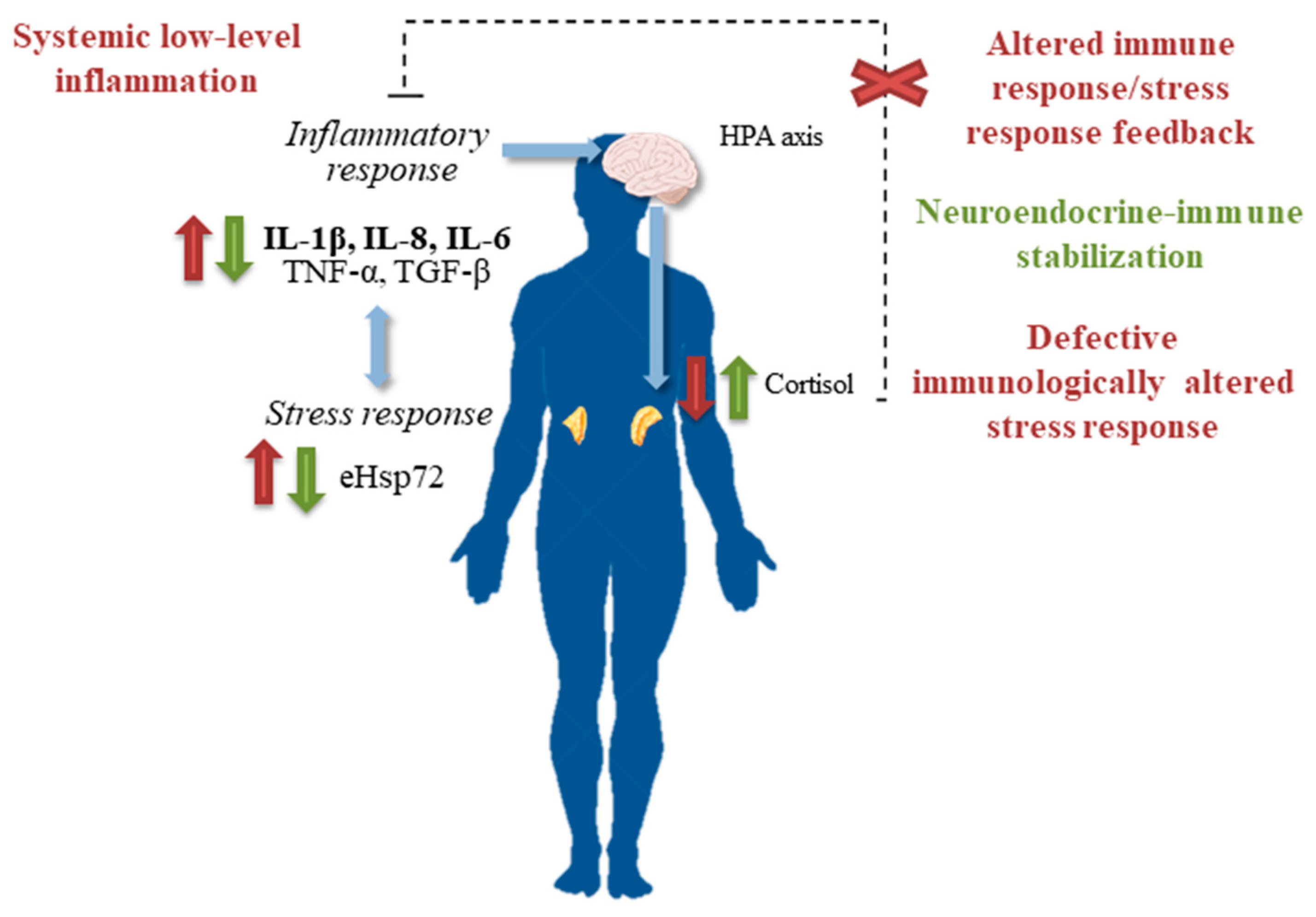
3. Balneotherapy and Immune System
4. Balneotherapy and Stress
5. Balneotherapy as a Hormetic Strategy
5.1. Heat Stress Hormetic Effects in Balneotherapy
5.2. Hydrogen Sulfide Hormetic Effects in Balneotherapy
5.3. Radon Hormetic Effects in Balneotherapy
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| ACTH | Adrenocorticotropic hormone |
| AGE | Advanced glycation end |
| Akt | Protein kinase B |
| AMPK | AMP-activated protein kinase |
| COMP | Cartilage oligomeric matrix protein |
| CRP | C-reactive protein |
| eHsp | Extracellular heat shock protein |
| FM | Fibromyalgia |
| GH | Growth hormone |
| GSH | Glutathione |
| H2S | Hydrogen sulfide |
| HDL | High-density lipoprotein |
| HPA | Hypothalamic-pituitary-adrenal |
| HS | Heat shock |
| IFN-γ | Interferon gamma |
| iHsp | Intracellular heat shock protein |
| LDL | Low-density lipoprotein |
| LTB4 | Leukotriene B4 |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MDA | Malondialdehyde |
| MMP | Matrix metalloproteinases |
| NA | Noradrenaline |
| NF-κβ | Nuclear factor kappa beta |
| NO | Nitric oxide |
| OA | Osteoarthritis |
| OARSI | Osteoarthritis Research Society International |
| PBMC | Peripheral blood mononuclear cells |
| PGE2 | Prostaglandin E2 |
| RA | Rheumatoid arthritis |
| RANTES | Regulated on Activation, Normal T-cell Expressed and Secreted |
| RNS | Reactive nitrogen species |
| ROS | Reactive oxygen species |
| SNS | Sympathetic nervous system |
| SOD | Superoxide dismutase |
| TC | Total cholesterol |
| TG | Triglycerides |
References
- Calabrese, E.J.; Bachmann, K.A.; Bailer, A.J.; Bolger, P.M.; Borak, J.; Cai, L.; Cedergreen, N.; Cherian, M.G.; Chiueh, C.C.; Clarkson, T.W.; et al. Biological stress response terminology: Integrating the concepts of adaptive response and preconditioning stress within a hormetic dose-response framework. Toxicol. Appl. Pharmacol. 2007, 222, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P. Hormesis defined. Ageing Res. Rev. 2008, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.; Demirovic, D. Hormesis can and does work in humans. Dose Response 2010, 8, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Ortega, E. The “bioregulatory effect of exercise” on the innate/inflammatory responses. J. Physiol. Biochem. 2016, 72, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.; Fernandes, R.A.; Demirovic, D.; Dymek, B.; Lima, C.F. Heat stress and hormetin-induced hormesis in human cells: Effects on aging, wound healing, angiogenesis, and differentiation. Dose Response 2009, 7, 90–103. [Google Scholar] [CrossRef] [PubMed]
- Gomes, C.; Carretero, M.I.; Pozo, M.; Maraver, F.; Cantista, P.; Armijo, F.; Legido, J.L.; Teixeira, F.; Rautureau, M.; Delgado, R. Peloids and pelotherapy: Historical evolution, classification and glossary. Appl. Clay Sci. 2013, 75–76, 28–38. [Google Scholar] [CrossRef]
- Van Tubergen, A.; van der Linden, S. A brief history of spa therapy. Ann. Rheum. Dis. 2002, 61, 273–275. [Google Scholar] [CrossRef] [PubMed]
- Gutenbrunner, C.; Bender, T.; Cantista, P.; Karagülle, Z. A proposal for a worldwide definition of health resort medicine, balneology, medical hydrology and climatology. Int. J. Biometeorol. 2010, 54, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Nasermoaddeli, A.; Kagamimori, S. Balneotherapy in medicine: A review. Environ. Health Prev. Med. 2005, 10, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Forestier, R.; Erol-Forestier, F.B.; Francon, A. Current role for spa therapy in rheumatology. Jt. Bone Spine 2017, 84, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, A.P.; Bierma-Zeinstra, S.M.; Boers, M.; Cardoso, J.R.; Lambeck, J.; de Bie, R.A.; de Vet, H.C. Balneotherapy for osteoarthritis. Cochrane Database Syst. Rev. 2007, CD006864. [Google Scholar] [CrossRef] [PubMed]
- Harzy, T.; Ghani, N.; Akasbi, N.; Bono, W.; Nejjari, C. Short- and long-term therapeutic effects of thermal mineral waters in knee osteoarthritis: A systematic review of randomized controlled trials. Clin. Rheumatol. 2009, 28, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Giannitti, C.; Bellisai, B.; Iacoponi, F.; Galeazzi, M. Efficacy of balneotherapy on pain, function and quality of life in patients with osteoarthritis of the knee. Int. J. Biometeorol. 2012, 56, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Espejo-Antúnez, L.; Cardero-Durán, M.A.; Garrido-Ardila, E.M.; Torres-Piles, S.; Caro-Puértolas, B. Clinical effectiveness of mud pack therapy in knee osteoarthritis. Rheumatology 2013, 52, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Forestier, R.; Erol Forestier, F.B.; Francon, A. Spa therapy and knee osteoarthritis: A systematic review. Ann. Phys. Rehabil. Med. 2016, 59, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zeng, C.; Gao, S.G.; Yang, T.; Luo, W.; Li, Y.S.; Xiong, Y.L.; Sun, J.P.; Lei, G.H. The effect of mud therapy on pain relief in patients with knee osteoarthritis: A meta-analysis of randomized controlled trials. J. Int. Med. Res. 2013, 41, 1418–1425. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, S.; Buskila, D.; Neumann, L.; Kleiner-Baumgarten, A. Mud pack therapy in rheumatoid arthritis. Clin. Rheumatol. 1992, 11, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Brosseau, L.; Robinson, V.; Léonard, G.; Casimiro, L.; Pelland, L.; Wells, G.; Tugwell, P. Efficacy of balneotherapy for rheumatoid arthritis: A meta-analysis. Phys. Ther. Rev. 2002, 7, 67–87. [Google Scholar] [CrossRef]
- Santos, I.; Cantista, P.; Vasconcelos, C. Balneotherapy in rheumatoid arthritis—A systematic review. Int. J. Biometeorol. 2016, 60, 1287–1301. [Google Scholar] [CrossRef] [PubMed]
- Evcik, D.; Kizilay, B.; Gökçen, E. The effects of balneotherapy on fibromyalgia patients. Rheumatol. Int. 2002, 22, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Perpignano, G.; Tirri, G.; Cardinale, G.; Gianniti, C.; Lanza, C.E.; Loi, A.; Tirri, E.; Sfriso, P.; Cozzi, F. Effects of mud-bath treatment on fibromyalgia patients: A randomized clinical trial. Rheumatol. Int. 2007, 27, 1157–1161. [Google Scholar] [CrossRef] [PubMed]
- Ozkurt, S.; Dönmez, A.; Zeki Karagülle, M.; Uzunoğlu, E.; Turan, M.; Erdoğan, N. Balneotherapy in fibromyalgia: A single blind randomized controlled clinical study. Rheumatol. Int. 2012, 32, 1949–1954. [Google Scholar] [CrossRef] [PubMed]
- Ablin, J.N.; Häuser, W.; Buskila, D. Spa Treatment (Balneotherapy) for Fibromyalgia—A Qualitative-Narrative Review and a Historical Perspective. Evid. Based Complement. Altern. Med. 2013, 2013, 638050. [Google Scholar] [CrossRef] [PubMed]
- Yurtkuran, M.; Ay, A.; Karakoç, Y. Improvement of the clinical outcome in Ankylosing spondylitis by balneotherapy. Jt. Bone Spine 2005, 72, 303–308. [Google Scholar] [CrossRef] [PubMed]
- McAlindon, T.E.; Bannuru, R.R.; Sullivan, M.C.; Arden, N.K.; Berenbaum, F.; Bierma-Zeinstra, S.M.; Hawker, G.A.; Henrotin, Y.; Hunter, D.J.; Kawaguchi, H.; et al. OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthr. Cartil. 2014, 22, 363–388. [Google Scholar] [CrossRef] [PubMed]
- Srámek, P.; Simecková, M.; Janský, L.; Savlíková, J.; Vybíral, S. Human physiological responses to immersion into water of different temperatures. Eur. J. Appl. Physiol. 2000, 81, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Cantarini, L.; Guidelli, G.M.; Galeazzi, M. Mechanisms of action of spa therapies in rheumatic diseases: What scientific evidence is there? Rheumatol. Int. 2011, 31, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, S.; Flusser, D.; Abu-Shakra, M. The role of spa therapy in various rheumatic diseases. Rheum. Dis. Clin. N. Am. 1999, 25, 883–897. [Google Scholar] [CrossRef]
- Shani, J.; Barak, S.; Levi, D.; Ram, M.; Schachner, E.R.; Schlesinger, T.; Robberecht, H.; Van Grieken, R.; Avrach, W.W. Skin penetration of minerals in psoriatics and guinea-pigs bathing in hypertonic salt solutions. Pharmacol. Res. Commun. 1985, 17, 501–512. [Google Scholar] [CrossRef]
- Halevy, S.; Giryes, H.; Friger, M.; Grossman, N.; Karpas, Z.; Sarov, B.; Sukenik, S. The role of trace elements in psoriatic patients undergoing balneotherapy with Dead Sea bath salt. Isr. Med. Assoc. J. 2001, 3, 828–832. [Google Scholar] [PubMed]
- Beer, A.M.; Junginger, H.E.; Lukanov, J.; Sagorchev, P. Evaluation of the permeation of peat substances through human skin in vitro. Int. J. Pharm. 2003, 253, 169–175. [Google Scholar] [CrossRef]
- Flusser, D.; Abu-Shakra, M.; Friger, M.; Codish, S.; Sukenik, S. Therapy with mud compresses for knee osteoarthritis: Comparison of natural mud preparations with mineral-depleted mud. J. Clin. Rheumatol. 2002, 8, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Kovács, I.; Bender, T. The therapeutic effects of Cserkeszölö thermal water in osteoarthritis of the knee: A double blind, controlled, follow-up study. Rheumatol. Int. 2002, 21, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Balogh, Z.; Ordögh, J.; Gász, A.; Német, L.; Bender, T. Effectiveness of balneotherapy in chronic low back pain—A randomized single-blind controlled follow-up study. Forsch. Komplementarmed Klass Naturheilkd 2005, 12, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Yurtkuran, M.; Yurtkuran, M.; Alp, A.; Nasircilar, A.; Bingöl, U.; Altan, L.; Sarpdere, G. Balneotherapy and tap water therapy in the treatment of knee osteoarthritis. Rheumatol. Int. 2006, 27, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Bálint, G.P.; Buchanan, W.W.; Adám, A.; Ratkó, I.; Poór, L.; Bálint, P.V.; Somos, E.; Tefner, I.; Bender, T. The effect of the thermal mineral water of Nagybaracska on patients with knee joint osteoarthritis—A double blind study. Clin. Rheumatol. 2007, 26, 890–894. [Google Scholar] [CrossRef] [PubMed]
- Odabasi, E.; Turan, M.; Erdem, H.; Tekbas, F. Does mud pack treatment have any chemical effect? A randomized controlled clinical study. J. Altern. Complement. Med. 2008, 14, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Sarsan, A.; Akkaya, N.; Ozgen, M.; Yildiz, N.; Atalay, N.S.; Ardic, F. Comparing the efficacy of mature mud pack and hot pack treatments for knee osteoarthritis. J. Back Musculoskelet. Rehabil. 2012, 25, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Morer, C.; Roques, C.F.; Françon, A.; Forestier, R.; Maraver, F. The role of mineral elements and other chemical compounds used in balneology: Data from double-blind randomized clinical trials. Int. J. Biometeorol. 2017, 61, 2159–2173. [Google Scholar] [CrossRef] [PubMed]
- Monasterio, A.M.; Armijo, F.; Maraver, F. Therapeutic effects of the mineral waters from Copahue spa. In Copahue Volcano; Tassi, F., Vaselli, O., Caselli, A.T., Eds.; Springer: Berlin, Germany, 2016; pp. 273–282. ISBN 978-3-662-48005-2. [Google Scholar]
- Maraver, F.; Armijo, F. Vademecum II de Aguas Mineromedicinales Españolas; Editorial Complutense: Madrid, Spain, 2010; pp. 13–343. ISBN 978-84-7491-998-1. [Google Scholar]
- Bellometti, S.; Cecchettin, M.; Galzigna, L. Mud pack therapy in osteoarthrosis. Changes in serum levels of chondrocyte markers. Clin. Chim. Acta 1997, 268, 101–106. [Google Scholar] [CrossRef]
- Cozzi, F.; Carrara, M.; Sfriso, P.; Todesco, S.; Cima, L. Anti-inflammatory effect of mud-bath applications on adjuvant arthritis in rats. Clin. Exp. Rheumatol. 2004, 22, 763–766. [Google Scholar] [PubMed]
- Mahboob, N.; Sousan, K.; Shirzad, A.; Amir, G.; Mohammad, V.; Reza, M.; Mansour, V.A.; Hadi, V. The efficacy of a topical gel prepared using Lake Urmia mud in patients with knee osteoarthritis. J. Altern. Complement. Med. 2009, 15, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, S.; Canino, C.; Tonti, G.; Medda, V.; Calcaterra, P.; Nappi, G.; Salaffi, F.; Canestrari, F. Biomarkers of oxidation, inflammation and cartilage degradation in osteoarthritis patients undergoing sulfur-based spa therapies. Clin. Biochem. 2010, 43, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Oyama, J.; Kudo, Y.; Maeda, T.; Node, K.; Makino, N. Hyperthermia by bathing in a hot spring improves cardiovascular functions and reduces the production of inflammatory cytokines in patients with chronic heart failure. Heart Vessels 2013, 28, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Ardiç, F.; Ozgen, M.; Aybek, H.; Rota, S.; Cubukçu, D.; Gökgöz, A. Effects of balneotherapy on serum IL-1, PGE2 and LTB4 levels in fibromyalgia patients. Rheumatol. Int. 2007, 27, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Basili, S.; Martini, F.; Ferroni, P.; Grassi, M.; Sili Scavalli, A.; Streva, P.; Cusumano, G.; Musca, A.; Battista Rini, G. Effects of mud-pack treatment on plasma cytokine and soluble adhesion molecule levels in healthy volunteers. Clin. Chim. Acta 2001, 314, 209–214. [Google Scholar] [CrossRef]
- Ortega, E.; Gálvez, I.; Hinchado, M.D.; Guerrero, J.; Martín-Cordero, L.; Torres-Piles, S. Anti-inflammatory effect as a mechanism of effectiveness underlying the clinical benefits of pelotherapy in osteoarthritis patients: Regulation of the altered inflammatory and stress feedback response. Int. J. Biometeorol. 2017, 61, 1777–1785. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Cantarini, L.; Bacarelli, M.R.; de Lalla, A.; Ceccatelli, L.; Blardi, P. Effects of spa therapy on serum leptin and adiponectin levels in patients with knee osteoarthritis. Rheumatol. Int. 2011, 31, 879–882. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Giannitti, C.; Cheleschi, S.; Simpatico, A.; Pascarelli, N.A.; Galeazzi, M. Circulating levels of adiponectin, resistin, and visfatin after mud-bath therapy in patients with bilateral knee osteoarthritis. Int. J. Biometeorol. 2015, 59, 1691–1700. [Google Scholar] [CrossRef] [PubMed]
- Berenbaum, F.; Eymard, F.; Houard, X. Osteoarthritis, inflammation and obesity. Curr. Opin. Rheumatol. 2013, 25, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, S.; Gamba, P.; Poli, G.; Leonarduzzi, G. Metalloproteinases and metalloproteinase inhibitors in age-related diseases. Curr. Pharm. Des. 2014, 20, 2993–3018. [Google Scholar] [CrossRef] [PubMed]
- Bellometti, S.; Richelmi, P.; Tassoni, T.; Bertè, F. Production of matrix metalloproteinases and their inhibitors in osteoarthritic patients undergoing mud bath therapy. Int. J. Clin. Pharmacol. Res. 2005, 25, 77–94. [Google Scholar] [PubMed]
- Oláh, M.; Koncz, A.; Fehér, J.; Kálmánczhey, J.; Oláh, C.; Balogh, S.; Nagy, G.; Bender, T. The effect of balneotherapy on C-reactive protein, serum cholesterol, triglyceride, total antioxidant status and HSP-60 levels. Int. J. Biometeorol. 2010, 54, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Oláh, M.; Koncz, Á.; Fehér, J.; Kálmánczhey, J.; Oláh, C.; Nagy, G.; Bender, T. The effect of balneotherapy on antioxidant, inflammatory, and metabolic indices in patients with cardiovascular risk factors (hypertension and obesity)—A randomised, controlled, follow-up study. Contemp. Clin. Trials 2011, 32, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Salmon, J.A.; Higgs, G.A. Prostaglandins and leukotrienes as inflammatory mediators. Br. Med. Bull. 1987, 43, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Bellometti, S.; Galzigna, L. Serum levels of a prostaglandin and a leukotriene after thermal mud pack therapy. J. Investig. Med. 1998, 46, 140–145. [Google Scholar] [PubMed]
- Zhang, J.M.; An, J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Gálvez, I.; Torres-Piles, S.; Hinchado, M.D.; Álvarez-Barrientos, A.; Torralbo-Jiménez, P.; Guerrero, J.; Martín-Cordero, L.; Ortega, E. Immune-Neuroendocrine Dysregulation in Patients with Osteoarthritis: A Revision and a Pilot Study. Endocr. Metab. Immune Disord. Drug Targets 2017, 17, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, A.C. Peripheral beta-endorphin and pain modulation. Anesth. Prog. 1991, 38, 75–78. [Google Scholar] [PubMed]
- Khansari, D.N.; Murgo, A.J.; Faith, R.E. Effects of stress on the immune system. Immunol. Today 1990, 11, 170–175. [Google Scholar] [CrossRef]
- Del Rey, A.; Besedovsky, H.O. The cytokine-HPA axis circuit contributes to prevent or moderate autoimmune processes. Z. Rheumatol. 2000, 59, 31–35. [Google Scholar] [CrossRef]
- Matsumoto, T.; Nishiyama, T.; Nishimura, N.; Kato, M.; Inukai, Y.; Sugenoya, J.; Yamauchi, M.; Kosaka, M. Endocrine responses to heat and cold stress. In Thermotherapy for Neoplasia, Inflammation, and Pain; Kosaka, M., Sugahara, T., Schmidt, K.L., Simon, E., Eds.; Springer: Tokyo, Japan, 2001; pp. 228–241. ISBN 978-4-431-67035-3. [Google Scholar]
- De la Fuente, M.; Cruces, J.; Hernández, O.; Ortega, E. Strategies to improve the functions and redox state of the immune system in aged subjects. Curr. Pharm. Des. 2011, 17, 3966–3993. [Google Scholar] [CrossRef] [PubMed]
- Vescovi, P.P.; Gerra, G.; Pioli, G.; Pedrazzoni, M.; Maninetti, L.; Passeri, M. Circulating opioid peptides during thermal stress. Horm. Metab. Res. 1990, 22, 44–46. [Google Scholar] [CrossRef] [PubMed]
- Jezová, D.; Kvetnansky, R.; Vigas, M. Sex differences in endocrine response to hyperthermia in sauna. Acta Physiol. Scand. 1994, 150, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Møller, N.; Beckwith, R.; Butler, P.C.; Christensen, N.J.; Orskov, H.; Alberti, K.G. Metabolic and hormonal responses to exogenous hyperthermia in man. Clin. Endocrinol. 1989, 30, 651–660. [Google Scholar] [CrossRef]
- Weeke, J.; Gundersen, H.J. The effect of heating and central cooling on serum TSH, GH, and norepinephrine in resting normal man. Acta Physiol. Scand. 1983, 117, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Laatikainen, T.; Salminen, K.; Kohvakka, A.; Pettersson, J. Response to plasma endorphins, prolactin and catecholamines in women to intense heat in a sauna. Eur. J. Appl. Physiol. 1988, 57, 98–102. [Google Scholar] [CrossRef]
- Brisson, G.R.; Peronnet, F.; Perrault, H.; Boisvert, P.; Massicotte, D.; Gareau, R. Prolactinotrophic effect of endogenous and exogenous heat loads in human male adults. J. Appl. Physiol. 1991, 70, 1351–1355. [Google Scholar] [CrossRef] [PubMed]
- Kubota, K.; Kurabayashi, H.; Tamura, K.; Kawada, E.; Tamura, J.; Shirakura, T. A transient rise in plasma beta-endorphin after a traditional 47 degrees C hot-spring bath in Kusatsu-spa, Japan. Life Sci. 1992, 51, 1877–1880. [Google Scholar] [CrossRef]
- Tatár, P.; Vigas, M.; Jurcovicova, J.; Kvetnanský, R.; Strec, V. Increased glucagon secretion during hyperthermia in a sauna. Eur. J. Appl. Physiol. 1986, 55, 315–317. [Google Scholar] [CrossRef]
- Bote, M.E.; García, J.J.; Hinchado, M.D.; Ortega, E. Inflammatory/stress feedback dysregulation in women with fibromyalgia. Neuroimmunomodulation 2012, 19, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Kuczera, M.; Kokot, F. Effect of spa therapy on the endocrine system. I. Stress reaction hormones. Polskie Arch. Med. Wewn. 1996, 95, 11–20. [Google Scholar]
- Bellometti, S.; Galzigna, L. Function of the hypothalamic adrenal axis in patients with fibromyalgia syndrome undergoing mud-pack treatment. Int. J. Clin. Pharmacol. Res. 1999, 19, 27–33. [Google Scholar] [PubMed]
- Verbeke, P.; Fonager, J.; Clark, B.F.; Rattan, S.I. Heat shock response and ageing: Mechanisms and applications. Cell Biol. Int. 2001, 25, 845–857. [Google Scholar] [CrossRef] [PubMed]
- Scapagnini, G.; Davinelli, S.; Fortunati, N.A.; Zella, D.; Vitale, M. Thermal Hydrotherapy as Adaptive Stress Response: Hormetic Significance, Mechanisms, and Therapeutic Implications. In Hormesis in Health and Disease; Rattan, S., Le Bourg, E., Eds.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2014; pp. 153–165. ISBN 978-1-4822-0545-9. [Google Scholar]
- Schöffl, F.; Prandl, R.; Reindl, A. Regulation of the Heat-Shock Response. Plant Physiol. 1998, 117, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Mao, H.P.; Ruchalski, K.L.; Wang, Y.H.; Choy, W.; Schwartz, J.H.; Borkan, S.C. Heat stress prevents mitochondrial injury in ATP-depleted renal epithelial cells. Am. J. Physiol. Cell Physiol. 2002, 283, C917–C926. [Google Scholar] [CrossRef] [PubMed]
- Asea, A.; Kraeft, S.K.; Kurt-Jones, E.A.; Stevenson, M.A.; Chen, L.B.; Finberg, R.W.; Koo, G.C.; Calderwood, S.K. HSP70 stimulates cytokine production through a CD14-dependant pathway, demonstrating its dual role as a chaperone and cytokine. Nat. Med. 2000, 6, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Campisi, J.; Leem, T.H.; Fleshner, M. Stress-induced extracellular Hsp72 is a functionally significant danger signal to the immune system. Cell Stress Chaperones 2003, 8, 272–286. [Google Scholar] [CrossRef]
- Calderwood, S.K.; Murshid, A.; Prince, T. The shock of aging: Molecular chaperones and the heat shock response in longevity and aging—A mini-review. Gerontology 2009, 55, 550–558. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F.; Barroso-Aranda, J.; Contreras, F. Regular thermal therapy may promote insulin sensitivity while boosting expression of endothelial nitric oxide synthase—Effects comparable to those of exercise training. Med. Hypotheses 2009, 73, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Hooper, P.L. Hot-tub therapy for type 2 diabetes mellitus. N. Engl. J. Med. 1999, 341, 924–925. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, N.; Hoshida, S.; Taniguchi, N.; Kuzuya, T.; Hori, M. Whole-body hyperthermia provides biphasic cardioprotection against ischemia/reperfusion injury in the rat. Circulation 1998, 98, 1414–1421. [Google Scholar] [CrossRef] [PubMed]
- Okada, M.; Hasebe, N.; Aizawa, Y.; Izawa, K.; Kawabe, J.; Kikuchi, K. Thermal treatment attenuates neointimal thickening with enhanced expression of heat-shock protein 72 and suppression of oxidative stress. Circulation 2004, 109, 1763–1768. [Google Scholar] [CrossRef] [PubMed]
- Bathaie, S.Z.; Jafarnejad, A.; Hosseinkhani, S.; Nakhjavani, M. The effect of hot-tub therapy on serum Hsp70 level and its benefit on diabetic rats: A preliminary report. Int. J. Hyperth. 2010, 26, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, K.; Davis, A.T.; Jenkins, K.A.; Flynn, D.M. Effects of heated hydrotherapy on muscle HSP70 and glucose metabolism in old and young vervet monkeys. Cell Stress Chaperones 2016, 21, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Krause, M.; Ludwig, M.S.; Heck, T.G.; Takahashi, H.K. Heat shock proteins and heat therapy for type 2 diabetes: Pros and cons. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Uzunoğlu, E.; Yentur, S.; Kayar, A.H.; Turan, M.; Dönmez, A.; Direskeneli, G.S.; Erdoğan, N. Effect of mild heat stress on heat shock protein 70 in a balneotherapy model. Eur. J. Integr. Med. 2017, 9, 86–90. [Google Scholar] [CrossRef]
- Asea, A. Initiation of the immune response by extracellular Hsp72: Chaperokine activity of Hsp72. Curr. Immunol. Rev. 2006, 2, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Amorim, F.; Moseley, P.L. Heat shock protein and inflammation. In Heat Shock Proteins and Whole Body Physiology; Asea, A., Pedersen, B.K., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 57–83. ISBN 978-90-481-3381-9. [Google Scholar]
- Ortega, E.; Hinchado, M.D.; Martín-Cordero, L.; Asea, A. The effect of stress-inducible extracellular Hsp72 on human neutrophil chemotaxis: A role during acute intense exercise. Stress 2009, 12, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Giraldo, E.; Martín-Cordero, L.; García, J.J.; Gehrmann, M.; Multhoff, G.; Ortega, E. Exercise-induced extracellular 72 kDa heat shock protein (Hsp72) stimulates neutrophil phagocytic and fungicidal capacities via TLR-2. Eur. J. Appl. Physiol. 2010, 108, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Ortega, E.; Bote, M.E.; Besedovsky, H.O.; del Rey, A. Hsp72, inflammation, and aging: Causes, consequences, and perspectives. Ann. N. Y. Acad. Sci. 1261, 1261, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Krause, M.; Heck, T.G.; Bittencourt, A.; Scomazzon, S.P.; Newsholme, P.; Curi, R.; Homem de Bittencourt, P.I., Jr. The chaperone balance hypothesis: The importance of the extracellular to intracellular HSP70 ratio to inflammation-driven type 2 diabetes, the effect of exercise, and the implications for clinical management. Mediat. Inflamm. 2015, 2015, 249205. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, V.; Cornelius, C.; Dinkova-Kostova, A.T.; Calabrese, E.J.; Mattson, M.P. Cellular stress responses, the hormesis paradigm, and vitagenes: Novel targets for therapeutic intervention in neurodegenerative disorders. Antioxid. Redox Signal. 2010, 13, 1763–1811. [Google Scholar] [CrossRef] [PubMed]
- Carbajo, J.M.; Maraver, F. Sulphurous Mineral Waters: New Applications for Health. Evid. Based Complement. Altern. Med. 2017, 2017, 8034084. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Chan, H.; Ning, J.; Lu, K.; Ma, D. The role of hydrogen sulfide in pathologies of the vital organs and its clinical application. J. Physiol. Pharmacol. 2015, 66, 169–179. [Google Scholar] [PubMed]
- Olas, B. Hydrogen sulfide as a “double-faced” compound: One with pro- and antioxidant effect. Adv. Clin. Chem. 2017, 78, 187–196. [Google Scholar] [PubMed]
- Braga, P.C.; Sambataro, G.; dal Sasso, M.; Culici, M.; Alfieri, M.; Nappi, G. Antioxidant effect of sulphurous thermal water on human neutrophil bursts: Chemiluminescence evaluation. Respiration 2008, 75, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Braga, P.C.; dal Sasso, M.; Culici, M.; Falchi, M.; Spallino, A.; Nappi, G. Free radical-scavenging activity of sulfurous water investigated by electron paramagnetic resonance (EPR) spectroscopy. Exp. Lung Res. 2012, 38, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Braga, P.C.; Ceci, C.; Marabini, L.; Nappi, G. The antioxidant activity of sulphurous thermal water protects against oxidative DNA damage: A comet assay investigation. Drug Res. 2013, 63, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Lamboglia, A.; Pascarelli, N.A.; Cheleschi, S.; Manica, P.; Galeazzi, M.; Collodel, G. Thermal water of Vetriolo, Trentino, inhibits the negative effect of interleukin-1β on nitric oxide production and apoptosis in human osteoarthritic chondrocyte. J. Biol. Regul. Homeost. Agents 2013, 27, 891–902. [Google Scholar] [PubMed]
- Guzmán, R.; Campos, C.; Yuguero, R.; Masegù, C.; Gil, P.; Moragón, Á.C. Protective effect of sulfurous water in peripheral blood mononuclear cells of Alzheimer’s disease patients. Life Sci. 2015, 132, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Sieghart, D.; Liszt, M.; Wanivenhaus, A.; Bröll, H.; Kiener, H.; Klösch, B.; Steiner, G. Hydrogen sulphide decreases IL-1β-induced activation of fibroblast-like synoviocytes from patients with osteoarthritis. J. Cell. Mol. Med. 2015, 19, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Prandelli, C.; Parola, C.; Buizza, L.; Delbarba, A.; Marziano, M.; Salvi, V.; Zacchi, V.; Memo, M.; Sozzani, S.; Calza, S.; et al. Sulphurous thermal water increases the release of the anti-inflammatory cytokine IL-10 and modulates antioxidant enzyme activity. Int. J. Immunopathol. Pharmacol. 2013, 26, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Burguera, E.F.; Vela-Anero, A.; Magalhães, J.; Meijide-Faílde, R.; Blanco, F.J. Effect of hydrogen sulfide sources on inflammation and catabolic markers on interleukin 1β-stimulated human articular chondrocytes. Osteoarthr. Cartil. 2014, 22, 1026–1035. [Google Scholar] [CrossRef] [PubMed]
- Ekmekcioglu, C.; Strauss-Blasche, G.; Holzer, F.; Marktl, W. Effect of sulfur baths on antioxidative defense systems, peroxide concentrations and lipid levels in patients with degenerative osteoarthritis. Forsch. Komplementarmed Klass Naturheilkd 2002, 9, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Jokić, A.; Sremcević, N.; Karagülle, Z.; Pekmezović, T.; Davidović, V. Oxidative stress, hemoglobin content, superoxide dismutase and catalase activity influenced by sulphur baths and mud packs in patients with osteoarthritis. Vojnosanit. Pregl. 2010, 67, 573–578. [Google Scholar] [CrossRef] [PubMed]
- Kovács, C.; Pecze, M.; Tihanyi, Á.; Kovács, L.; Balogh, S.; Bender, T. The effect of sulphurous water in patients with osteoarthritis of hand. Double-blind, randomized, controlled follow-up study. Clin. Rheumatol. 2012, 31, 1437–1442. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, S.; Buskila, D.; Neumann, L.; Kleiner-Baumgarten, A.; Zimlichman, S.; Horowitz, J. Sulphur bath and mud pack treatment for rheumatoid arthritis at the Dead Sea area. Ann. Rheum. Dis. 1990, 49, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, S.; Benvenuti, F.; Nappi, G.; Fortunati, N.A.; Marino, L.; Aureli, T.; De Luca, S.; Pagliarani, S.; Canestrari, F. Antioxidative effects of sulfurous mineral water: Protection against lipid and protein oxidation. Eur. J. Clin. Nutr. 2009, 63, 106–112. [Google Scholar] [CrossRef] [PubMed]
- El-Seweidy, M.M.; Sadik, N.A.; Shaker, O.G. Role of sulfurous mineral water and sodium hydrosulfide as potent inhibitors of fibrosis in the heart of diabetic rats. Arch. Biochem. Biophys. 2011, 506, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Sadik, N.A.; El-Seweidy, M.M.; Shaker, O.G. The antiapoptotic effects of sulphurous mineral water and sodium hydrosulphide on diabetic rat testes. Cell. Physiol. Biochem. 2011, 28, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Safar, M.M.; Abdelsalam, R.M. H2S donors attenuate diabetic nephropathy in rats: Modulation of oxidant status and polyol pathway. Pharmacol. Rep. 2015, 67, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Erickson, B.E. The therapeutic use of radon: A biomedical treatment in Europe; an “alternative” remedy in the United States. Dose Response 2007, 5, 48–62. [Google Scholar] [CrossRef] [PubMed]
- Feinendegen, L.E. Evidence for beneficial low level radiation effects and radiation hormesis. Br. J. Radiol. 2005, 78, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Ibuki, Y.; Hayashi, A.; Suzuki, A.; Goto, R. Low-dose irradiation induces expression of heat shock protein 70 mRNA and thermo- and radio-resistance in myeloid leukemia cell line. Biol. Pharm. Bull. 1998, 21, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, K.; Mitsunobu, F.; Hanamoto, K.; Shibuya, K.; Mori, S.; Tanizaki, Y.; Sugita, K. Biochemical comparison between radon effects and thermal effects on humans in radon hot spring therapy. J. Radiat. Res. 2004, 45, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, K.; Mitsunobu, F.; Hanamoto, K.; Mori, S.; Tanizaki, Y.; Sugita, K. Study on biologic effects of radon and thermal therapy on osteoarthritis. J. Pain 2004, 5, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Nagy, K.; Berhés, I.; Kovács, T.; Kávási, N.; Somlai, J.; Bender, T. Does balneotherapy with low radon concentration in water influence the endocrine system? A controlled non-randomized pilot study. Radiat. Environ. Biophys. 2009, 48, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Calabrese, V. Reduction of arthritic symptoms by low dose radiation therapy (LD-RT) is associated with an anti-inflammatory phenotype. Int. J. Radiat. Biol. 2013, 89, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Calabrese, V. Low dose radiation therapy (LD-RT) is effective in the treatment of arthritis: Animal model findings. Int. J. Radiat. Biol. 2013, 89, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Franke, A.; Reiner, L.; Pratzel, H.G.; Franke, T.; Resch, K.L. Long-term efficacy of radon spa therapy in rheumatoid arthritis—A randomized, sham-controlled study and follow-up. Rheumatology 2000, 39, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Franke, A.; Reiner, L.; Resch, K.L. Long-term benefit of radon spa therapy in the rehabilitation of rheumatoid arthritis: A randomised, double-blinded trial. Rheumatol. Int. 2007, 27, 703–713. [Google Scholar] [CrossRef] [PubMed]
- Annegret, F.; Thomas, F. Long-term benefits of radon spa therapy in rheumatic diseases: Results of the randomised, multi-centre IMuRa trial. Rheumatol. Int. 2013, 33, 2839–2850. [Google Scholar] [CrossRef] [PubMed]
- Falkenbach, A.; Kovacs, J.; Franke, A.; Jörgens, K.; Ammer, K. Radon therapy for the treatment of rheumatic diseases—review and meta-analysis of controlled clinical trials. Rheumatol. Int. 2005, 25, 205–210. [Google Scholar] [CrossRef] [PubMed]
| Study | Treatment(s) | Main Hormetic Mechanism | Experimental Subjects | Pathology | Biomarkers | Clinical Benefits |
|---|---|---|---|---|---|---|
| Yamashita et al., 1998 [86] | Hydrotherapy (40–42 °C, 1 session of 5–15 min) | Heat stress | Rats | Myocardial ischemia | Increased Hsp72 and manganese-SOD myocardial levels | Biphasic reduction in the incidence of ventricular fibrillation and in the size of the myocardial infarction |
| Okada et al., 2004 [87] | Hydrotherapy (41 °C, 28 daily sessions of 15 min) | Heat stress | Rats | Inflammatory arterial lesions | Reduced monocyte/macrophage infiltration and MCP-1 expression in the adventitia of arteries; increased expression of Hsp72 in the adventitia and media of arteries | Suppression of neointimal thickening |
| Bathaie et al. 2010 [88] | Hydrotherapy (42 °C, 60 daily sessions of 30 min) | Heat stress | Rats | Diabetes | Serum HDL increased whereas LDL, TG, and TC decreased; insulin and eHsp72 serum levels increased; AGE products serum levels decreased; serum antioxidant capacity improved | Prevention of diabetes complications and increased survival |
| Kavanagh et al. 2016 [89] | Hydrotherapy (40 °C, 10 daily sessions of 30 min) | Heat stress | Monkeys | Insulin resistance | Increased muscle Hsp70 levels; reduction in plasma glucose concentration; improved insulin secretion and normalized responses to glucose challenge | Improved blood pressure and glucose metabolism |
| Hooper 1999 [85] | Hydrotherapy (38–41 °C, 18 daily sessions of 30 min) | Heat stress | Humans | Type 2 diabetes mellitus | Fasting plasma glucose and glycated hemoglobin levels decreased | Body weight decreased and glucose metabolism improved |
| Ortega et al. 2017 [49] | Balneotherapy, using water rich in bicarbonate and calcium, and mud (38–42 °C, 10 daily sessions of 60 min) | Heat stress | Humans | Osteoarthritis | Levels of serum inflammatory cytokines (IL-1β, TNF-α, IL-8, IL-6, and TGF-β) decreased; cortisol serum levels increased and eHsp72 serum levels decreased | Pain reduction; improved knee flexion angle, stiffness and physical function; better health-related quality of life |
| Uzunoğlu et al. 2017 [91] | Balneotherapy, using water rich in bicarbonate and calcium (39–40 °C, 21 daily sessions of 15 min) | Heat stress | Humans | Osteoarthritis | Initial and transient increase in serum eHsp72 and IFN-γ levels after first session, but final decrease of these biomarkers at the end of the protocol | Not evaluated |
| Benedetti et al. 2010 [45] | Balneotherapy using sulfurous water at 37 °C and mud at 46–48 °C (12 daily sessions of 20 min); with (Group A) or without (Group B) drinking 400 mL of the water daily | Hydrogen sulfide | Humans | Osteoarthritis | Group A: increase in plasma thiol levels, decrease in plasma MDA and carbonyl levels, and in serum TNF-α and COMP levels; all of them at the end of the treatment and at 1-month follow-up. Plasma MMP-2 levels decreased only at the end of the treatment.Group B: plasma MDA and carbonyl levels, and serum TNF-α levels decreased only at the end of the therapy | Pain reduction |
| Benedetti et al. 2009 [114] | Balneotherapy consisting of drinking sulfurous water (500 mL daily for 2 weeks) | Hydrogen sulfide | Humans | Healthy | Decreased plasma MDA, carbonyls, and advanced oxidation protein products levels; increased plasma antioxidant capacity and thiol levels | Not evaluated |
| El-Seweidy et al. 2011 [115] | Balneotherapy consisting of drinking sulfurous water (ad libitum daily for 7 weeks) | Hydrogen sulfide | Rats | Diabetes | Serum concentrations of insulin, C-peptide and IGF-1 increased; glycemia and glycated hemoglobin levels decreased.Cardiac GSH and thiol levels increased; glutathione disulfide levels decreased; reduction in NF-κβ, MMP-2, TGF-β1, procollagen-1 and Fas-L gene expression in the left ventricle | Prevention of the development of diabetes-induced fibrosis in the heart: normal myocytes and absence of collagen |
| Sadik et al. 2011 [116] | Balneotherapy consisting of drinking sulfurous water (ad libitum daily for 7 weeks) | Hydrogen sulfide | Rats | Diabetes | Serum concentrations of insulin, C-peptide and IGF-1 increased; glycemia and glycated hemoglobin levels decreased.Testosterone serum levels and testicular GSH increased; testicular overexpression of Bax/Bcl-2, cytochrome c, caspase-9 and -3, and p53 was blocked. | Prevention of diabetes-induced testicular dysfunction: improved seminiferous tubule structure, number of spermatogenic cells and hormonal function |
| Safar et al. 2015 [117] | Balneotherapy consisting of drinking sulfurous water (ad libitum daily for 6 weeks) | Hydrogen sulfide | Rats | Diabetes | Glycemia and glycated hemoglobin levels decreased.Decreased creatinine and urea serum levels; decreased renal thiobarbituric acid reactive substances levels; increased renal GSH levels | Prevention of diabetes-induced nephropathy: improved kidney function and absence of histopathological alterations |
| Yamaoka et al. 2004 [121] | Spa therapy consisting of inhalating radon at 36 °C (Group A), or sauna bath at 48 °C in the absence of radon (Group B) (5 sessions of 40 min) | Radon | Humans | Healthy | Group A and B: SOD and catalase activity, and insulin and glucose-6-phosphate dehydrogenase levels increased; lipid peroxide levels and total cholesterol decreased.Group A only: decreased percentage of CD8+ cells and increased percentage of CD4+ cells. Increased α-atrial natriuretic polypeptide levels, ACTH, and β-endorphins; decreased vasopressin levels. | Not evaluated |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gálvez, I.; Torres-Piles, S.; Ortega-Rincón, E. Balneotherapy, Immune System, and Stress Response: A Hormetic Strategy? Int. J. Mol. Sci. 2018, 19, 1687. https://doi.org/10.3390/ijms19061687
Gálvez I, Torres-Piles S, Ortega-Rincón E. Balneotherapy, Immune System, and Stress Response: A Hormetic Strategy? International Journal of Molecular Sciences. 2018; 19(6):1687. https://doi.org/10.3390/ijms19061687
Chicago/Turabian StyleGálvez, Isabel, Silvia Torres-Piles, and Eduardo Ortega-Rincón. 2018. "Balneotherapy, Immune System, and Stress Response: A Hormetic Strategy?" International Journal of Molecular Sciences 19, no. 6: 1687. https://doi.org/10.3390/ijms19061687
APA StyleGálvez, I., Torres-Piles, S., & Ortega-Rincón, E. (2018). Balneotherapy, Immune System, and Stress Response: A Hormetic Strategy? International Journal of Molecular Sciences, 19(6), 1687. https://doi.org/10.3390/ijms19061687






