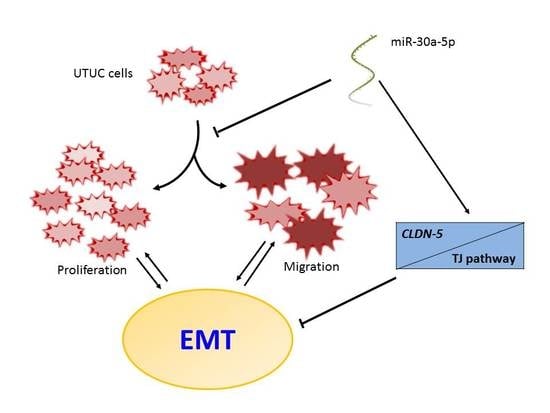
MiR-30a-5p Inhibits Epithelial-to-Mesenchymal Transition and Upregulates Expression of Tight Junction Protein Claudin-5 in Human Upper Tract Urothelial Carcinoma Cells
Abstract
1. Introduction
2. Results
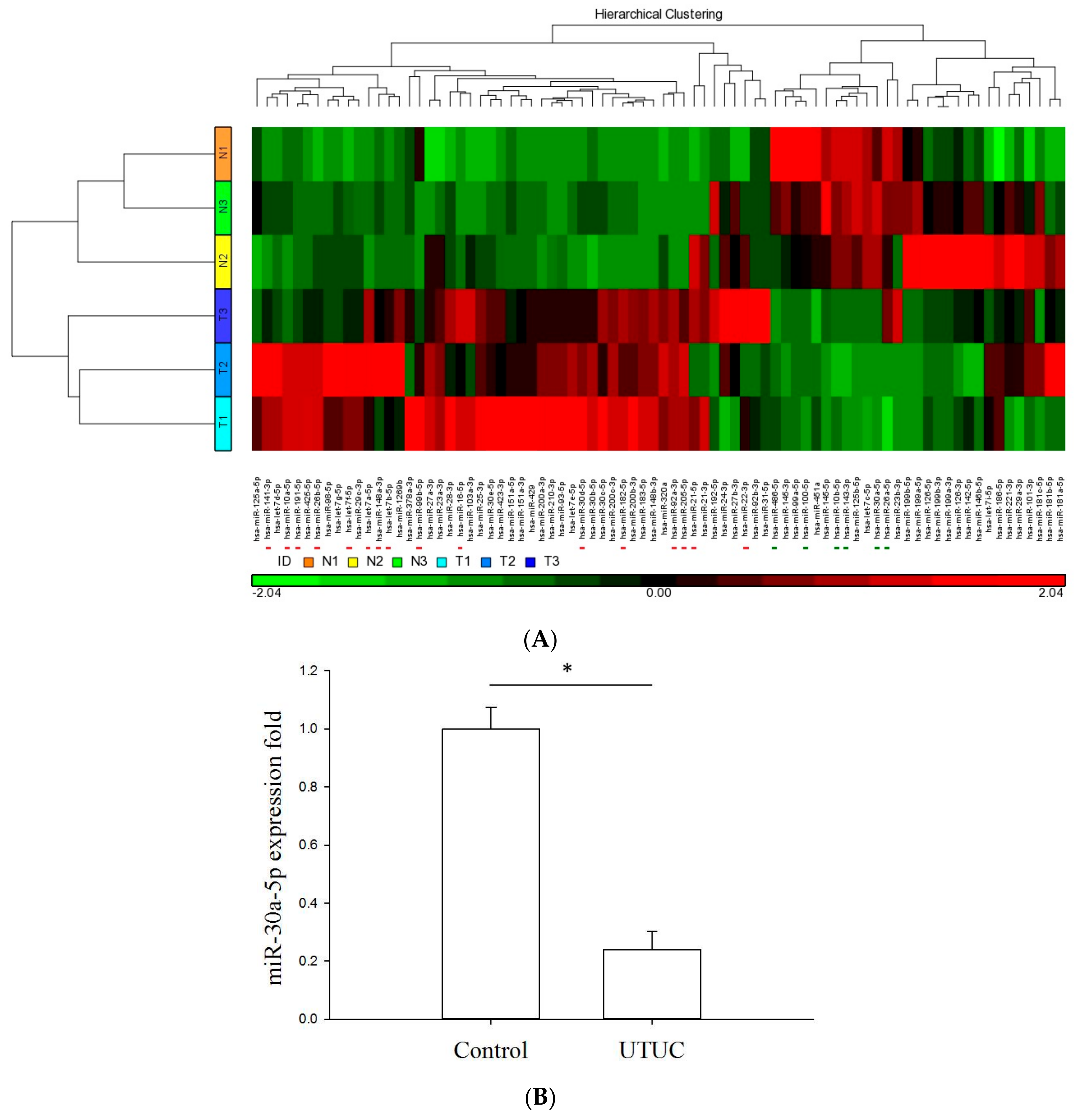
2.1. Profiling the miRNA Expression Signature of UTUC by NGS and qPCR
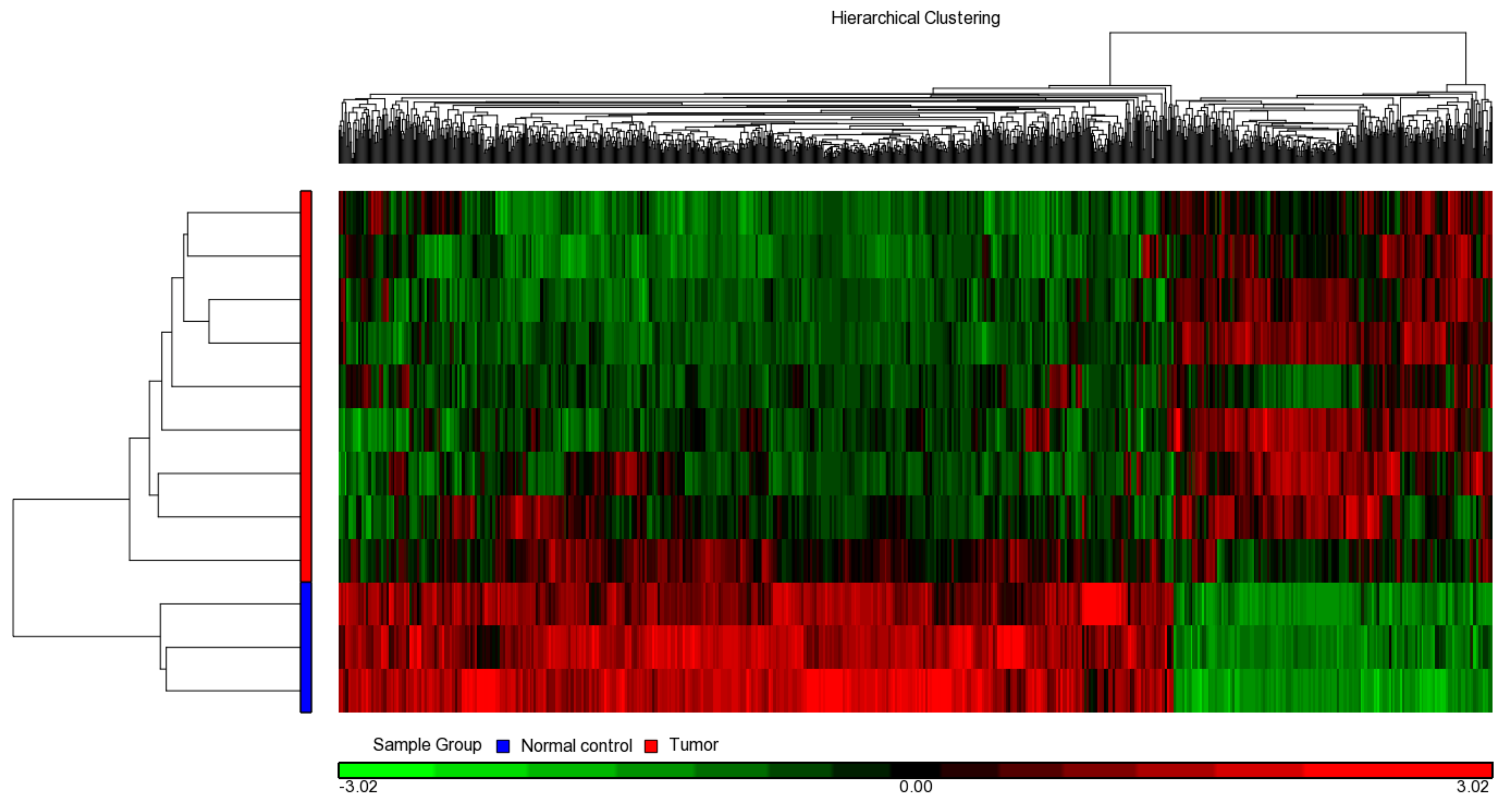
2.2. Transcriptome Profiles in UTUC Tissues on Illumina HT12 Microarray Chips
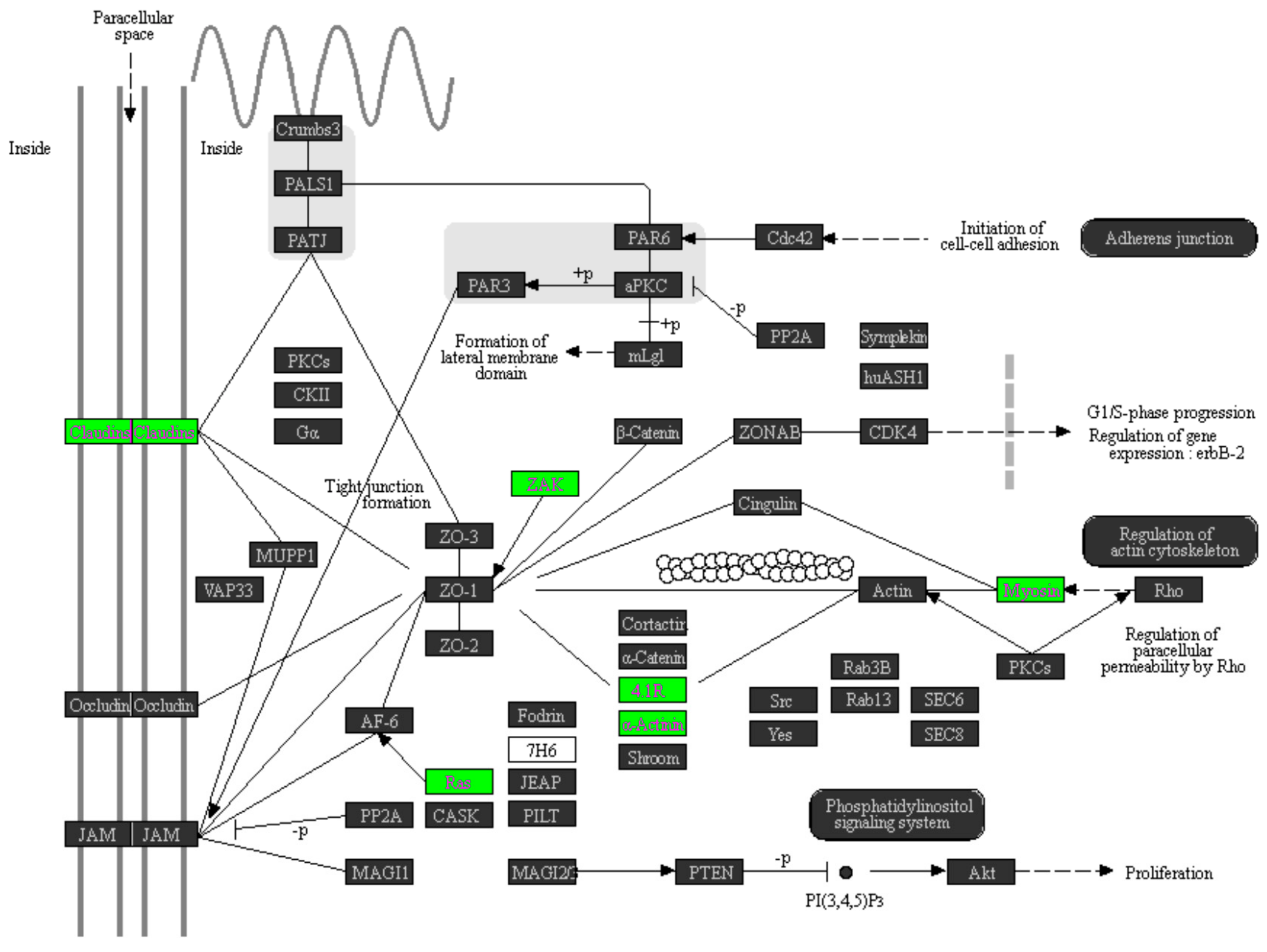
2.3. Pathway Analysis on the Involvement of miRNAs in the Tumorigenesis of UTUC
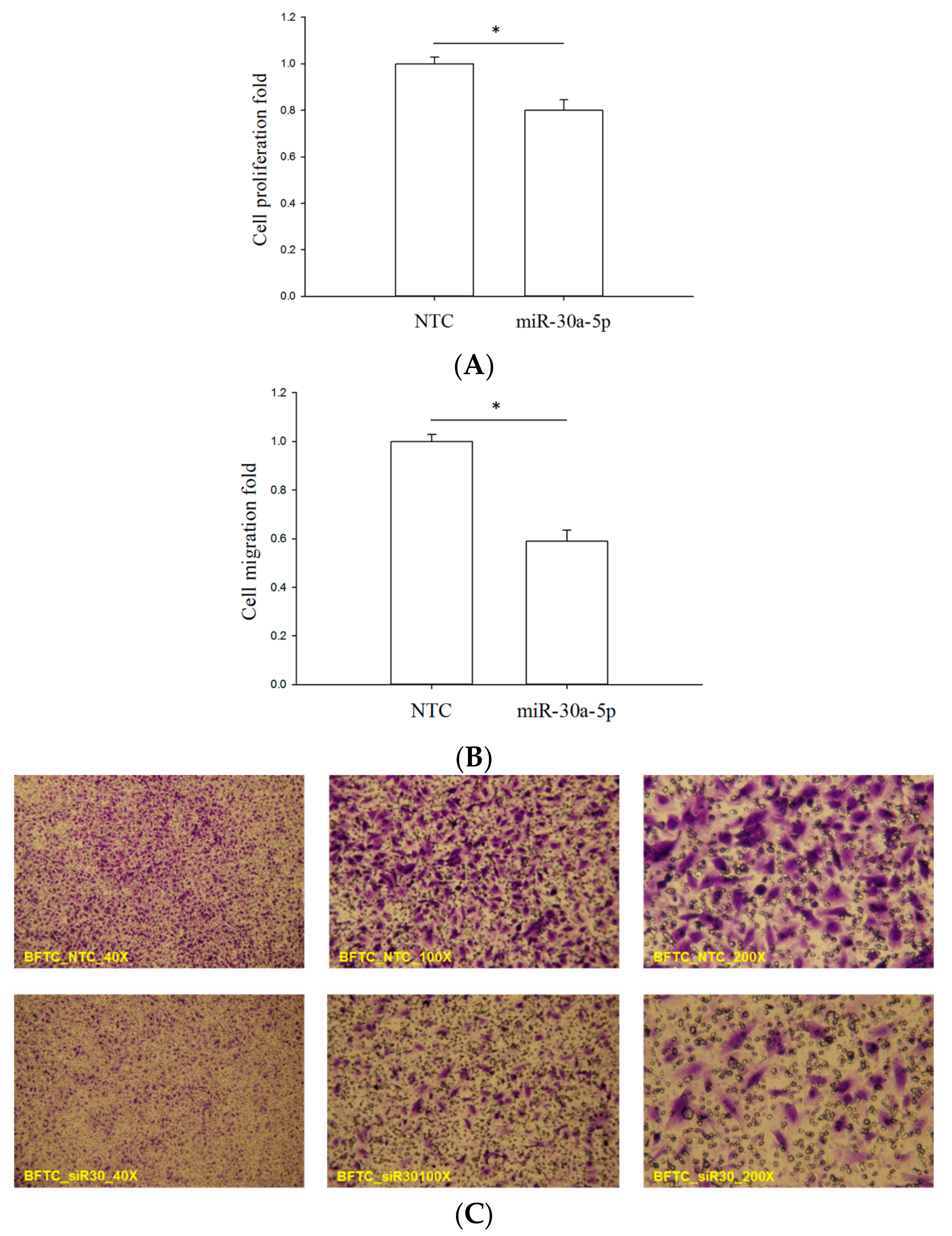
2.4. miR-30a-5p Overexpression Reduced Proliferation and Migration of BFTC-909 Cells
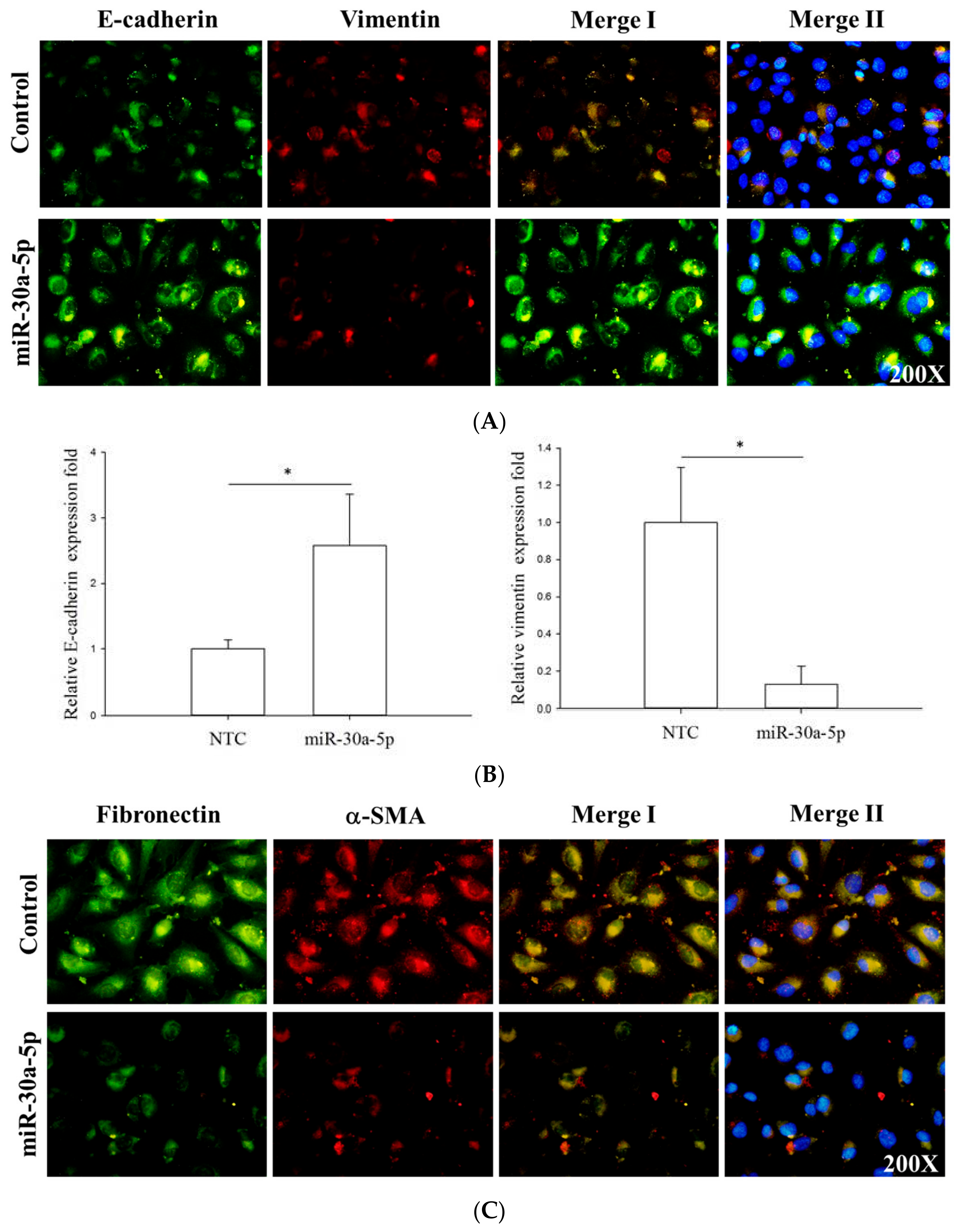
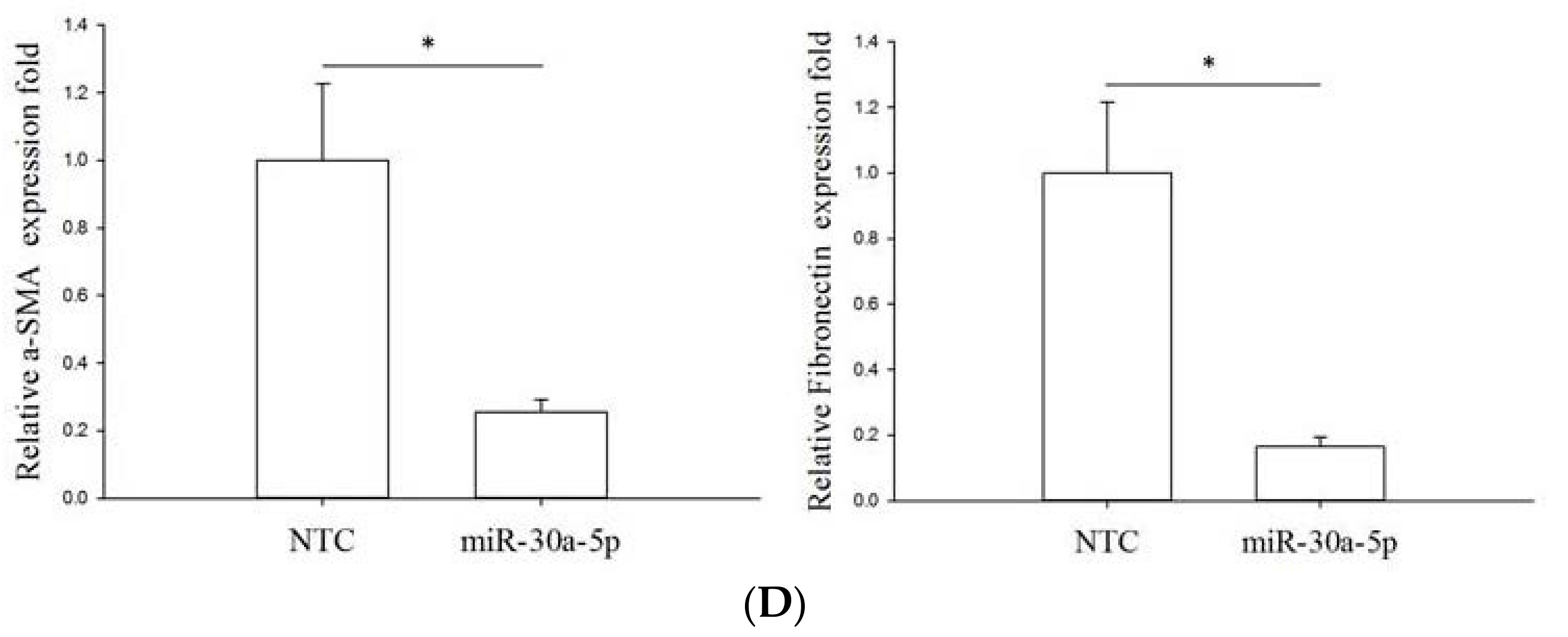
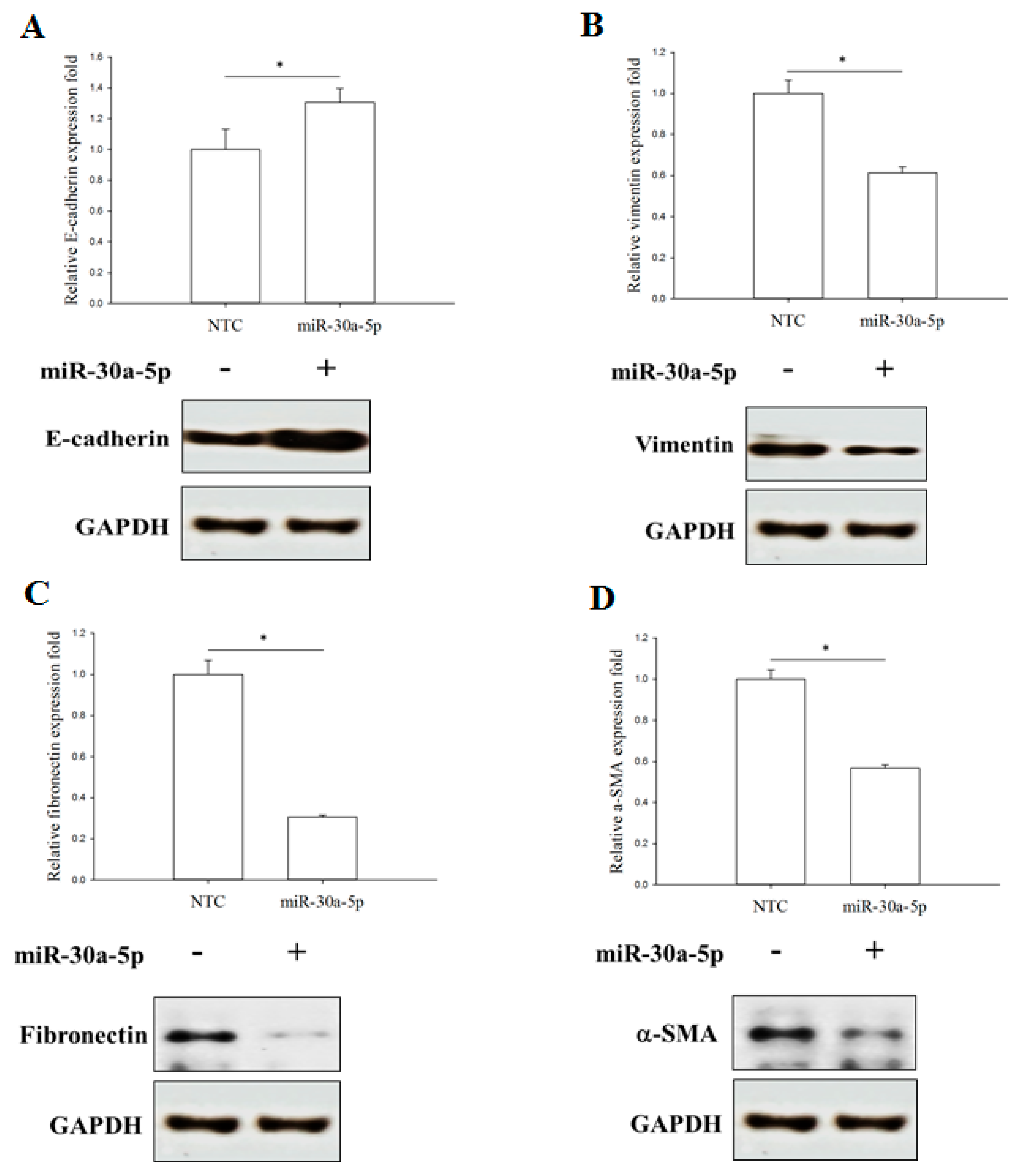
2.5. miR-30a-5p Overexpression Inhibits Epithelial-to-Mesenchymal Transition (EMT) in BFTC-909 Cells
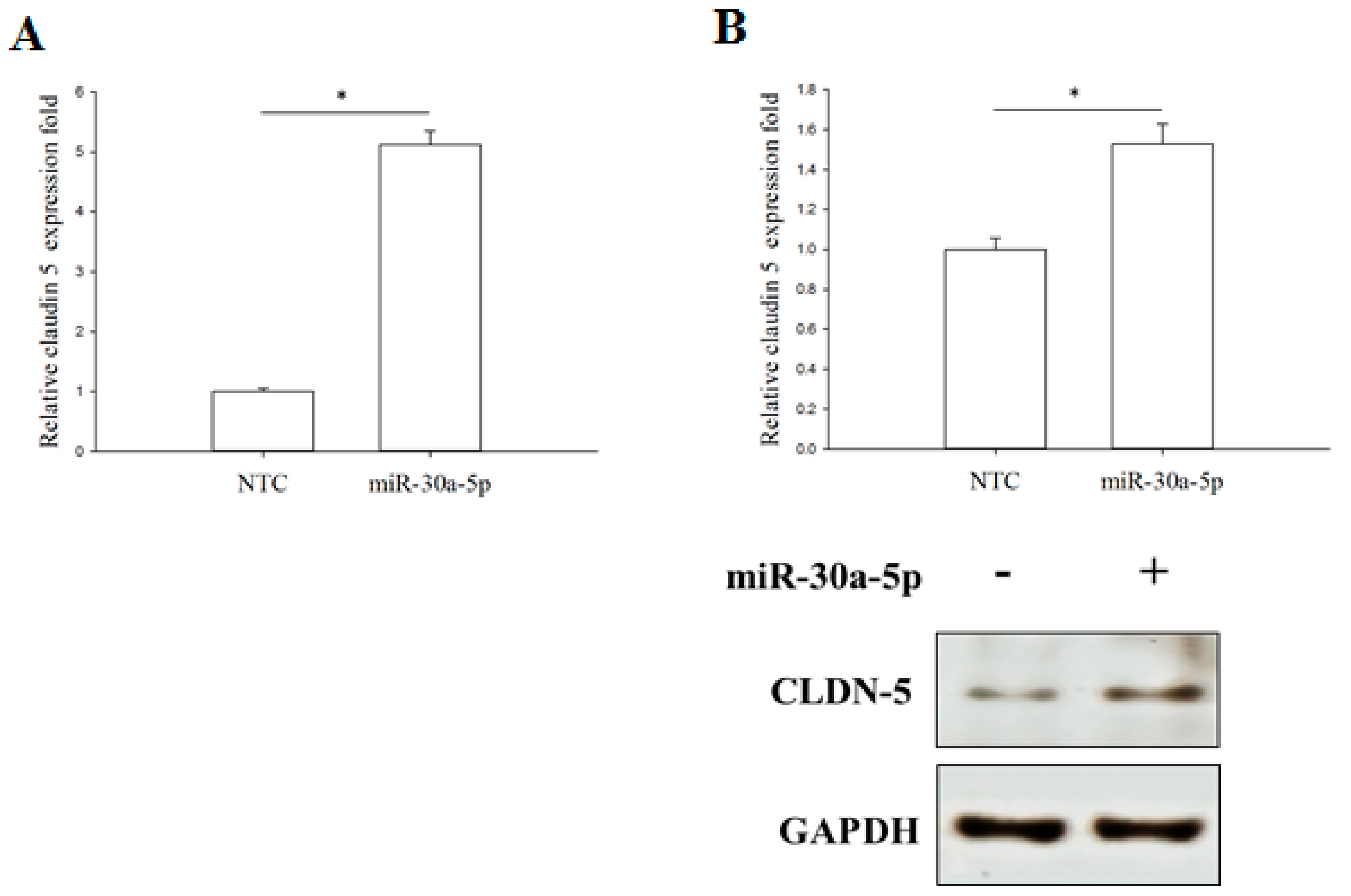
2.6. miR-30a-5p Overexpression Increased TJ Protein Claudin-5 Expression in BFTC-909 Cells
3. Discussion
4. Materials and Methods
4.1. Clinical Specimens
4.2. RNA Isolation
4.3. Library Preparation and Sequencing
4.4. Pathway Enrichment Analysis
4.5. Reverse Transcription (RT) and Real-Time PCR
4.6. Cell Culture and miRNA Treatment
4.7. Cell Proliferation Assay
4.8. Trans-Well Cell Migration Assay
4.9. Western Blot Analysis
4.10. Immunofluorescent Staining
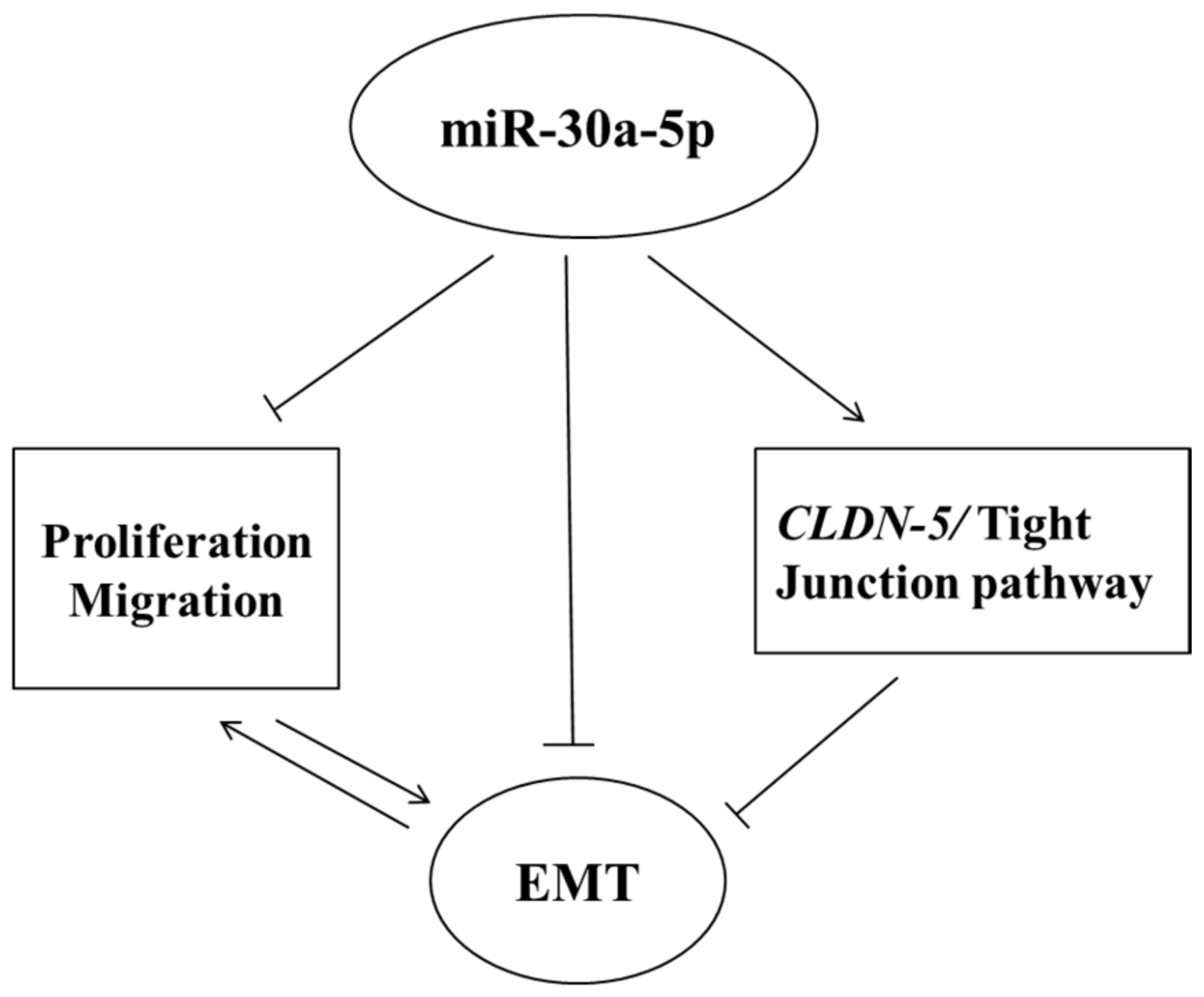
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hsieh, M.C.; Chiang, P.H.; Rau, K.M.; Chen, Y.Y.; Su, Y.L.; Huang, C.H. The comparison of oncologic outcomes between metastatic upper tract urothelial carcinoma and urothelial carcinoma of the bladder after cisplatin-based chemotherapy. Urol. Oncol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Baek, D.; Villen, J.; Shin, C.; Camargo, F.D.; Gygi, S.P.; Bartel, D.P. The impact of microRNAs on protein output. Nature 2008, 455, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Friedman, R.C.; Farh, K.K.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef] [PubMed]
- He, L.; He, X.; Lim, L.P.; de Stanchina, E.; Xuan, Z.; Liang, Y.; Xue, W.; Zender, L.; Magnus, J.; Ridzon, D.; et al. A microRNA component of the p53 tumour suppressor network. Nature 2007, 447, 1130–1134. [Google Scholar] [CrossRef] [PubMed]
- Croce, C.M. Oncogenes and cancer. N. Engl. J. Med. 2008, 358, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Getz, G.; Miska, E.A.; Alvarez-Saavedra, E.; Lamb, J.; Peck, D.; Sweet-Cordero, A.; Ebert, B.L.; Mak, R.H.; Ferrando, A.A.; et al. MicroRNA expression profiles classify human cancers. Nature 2005, 435, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Calin, G.A.; Croce, C.M. MicroRNA-cancer connection: The beginning of a new tale. Cancer Res. 2006, 66, 7390–7394. [Google Scholar] [CrossRef] [PubMed]
- Esquela-Kerscher, A.; Slack, F.J. Oncomirs-microRNAs with a role in cancer. Nat. Rev. Cancer 2006, 6, 259–269. [Google Scholar] [CrossRef] [PubMed]
- McManus, M.T. MicroRNAs and cancer. Semin. Cancer Biol. 2003, 13, 253–258. [Google Scholar] [CrossRef]
- Tao, J.; Yang, X.; Li, P.; Wei, J.; Deng, X.; Cheng, Y.; Qin, C.; Ju, X.; Meng, X.; Li, J.; et al. Identification of circulating microRNA signatures for upper tract urothelial carcinoma detection. Mol. Med. Rep. 2015, 12, 6752–6760. [Google Scholar] [CrossRef] [PubMed]
- Kriebel, S.; Schmidt, D.; Holdenrieder, S.; Goltz, D.; Kristiansen, G.; Moritz, R.; Fisang, C.; Muller, S.C.; Ellinger, J. Analysis of tissue and serum microRNA expression in patients with upper urinary tract urothelial cancer. PLoS ONE 2015, 10, e0117284. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, L.; Ingelmo-Torres, M.; Mallofre, C.; Lozano, J.J.; Verhasselt-Crinquette, M.; Leroy, X.; Colin, P.; Comperat, E.; Roupret, M.; Alcaraz, A.; et al. Prognostic value of microRNA expression pattern in upper tract urothelial carcinoma. BJU Int. 2014, 113, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Baraniskin, A.; Birkenkamp-Demtroder, K.; Maghnouj, A.; Zollner, H.; Munding, J.; Klein-Scory, S.; Reinacher-Schick, A.; Schwarte-Waldhoff, I.; Schmiegel, W.; Hahn, S.A. miR-30a-5p suppresses tumor growth in colon carcinoma by targeting dtl. Carcinogenesis 2012, 33, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Yang, Y.; Cai, J.; Cui, K.; Li, R.X.; Wang, H.; Shang, X.; Wei, D. miR-30a-5p suppresses tumor metastasis of human colorectal cancer by targeting ITGB3. Cell. Physiol. Biochem. 2016, 39, 1165–1176. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, C.; Chen, H.; Li, L.; Tu, Y.; Liu, C.; Shi, S.; Zen, K.; Liu, Z. Evaluation of microRNAs miR-196a, miR-30a-5p, and miR-490 as biomarkers of disease activity among patients with FSGS. Clin. J. Am. Soc. Nephrol. 2014, 9, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Kumarswamy, R.; Mudduluru, G.; Ceppi, P.; Muppala, S.; Kozlowski, M.; Niklinski, J.; Papotti, M.; Allgayer, H. MicroRNA-30a inhibits epithelial-to-mesenchymal transition by targeting Snai1 and is downregulated in non-small cell lung cancer. Int. J. Cancer 2012, 130, 2044–2053. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.L.; Zhang, X.H.; Zhang, X.; Chu, J.K. miR-30a increases cisplatin sensitivity of gastric cancer cells through suppressing epithelial-to-mesenchymal transition (EMT). Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1733–1739. [Google Scholar] [PubMed]
- Chang, C.W.; Yu, J.C.; Hsieh, Y.H.; Yao, C.C.; Chao, J.I.; Chen, P.M.; Hsieh, H.Y.; Hsiung, C.N.; Chu, H.W.; Shen, C.Y.; et al. MicroRNA-30a increases tight junction protein expression to suppress the epithelial-mesenchymal transition and metastasis by targeting slug in breast cancer. Oncotarget 2016, 7, 16462–16478. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Wang, X.; Huo, Q.; Sun, M.; Cai, C.; Liu, Z.; Hu, G.; Yang, Q. MicroRNA-30a suppresses breast tumor growth and metastasis by targeting metadherin. Oncogene 2014, 33, 3119–3128. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.T.; Tsai, K.W.; Hung, T.M.; Lin, W.C.; Pan, C.Y.; Yu, H.R.; Li, S.C. Mirseq: A user-friendly standalone toolkit for sequencing quality evaluation and miRNA profiling. Biomed. Res. Int. 2014, 2014, 462135. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.T.; Li, S.C.; Ho, M.R.; Pan, H.W.; Ger, L.P.; Hu, L.Y.; Yu, S.Y.; Li, W.H.; Tsai, K.W. Comprehensive analysis of microRNAs in breast cancer. BMC Genom. 2012, 13, S18. [Google Scholar]
- Li, S.C.; Wang, F.S.; Yang, Y.L.; Tiao, M.M.; Chuang, J.H.; Huang, Y.H. Microarray study of pathway analysis expression profile associated with microRNA-29a with regard to murine cholestatic liver injuries. Int. J. Mol. Sci. 2016, 17, 324. [Google Scholar] [CrossRef] [PubMed]
- Lamouille, S.; Xu, J.; Derynck, R. Molecular mechanisms of epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2014, 15, 178–196. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Yongjun, L.; Nan, D.; Junyun, W.; Qiong, Y.; Yaran, Y.; Yanming, L.; Xiangdong, F.; Hua, Z. Molecular networks and mechanisms of epithelial-mesenchymal transition regulated by mirnas in the malignant melanoma cell line. Yi Chuan 2015, 37, 673–682. [Google Scholar] [PubMed]
- Ikenouchi, J.; Matsuda, M.; Furuse, M.; Tsukita, S. Regulation of tight junctions during the epithelium-mesenchyme transition: Direct repression of the gene expression of claudins/occludin by Snail. J. Cell Sci. 2003, 116, 1959–1967. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, K.; Ogata, S.; Hiroi, S.; Tominaga, S.; Aida, S.; Kawai, T. Expression of occludin and claudins 1, 3, 4, and 7 in urothelial carcinoma of the upper urinary tract. Am. J. Clin. Pathol. 2008, 130, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.L.; Sung, M.T.; Tsai, E.M.; Lin, C.S.; Lee, N.L.; Chung, Y.H.; Chiang, P.H. Expression of estrogen receptor beta predicts oncologic outcome of pt3 upper urinary tract urothelial carcinoma better than aggressive pathological features. Sci. Rep. 2016, 6, 24263. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.Y.; Shieh, D.E.; Chen, C.C.; Yeh, C.S.; Dong, H.P. Linalool induces cell cycle arrest and apoptosis in leukemia cells and cervical cancer cells through CDKIS. Int. J. Mol. Sci. 2015, 16, 28169–28179. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yin, J.; Wang, X.; Shao, M.; Duan, F.; Wu, W.; Peng, P.; Jin, J.; Tang, Y.; Ruan, Y.; et al. C-type lectin-like receptor 2 suppresses AKT signaling and invasive activities of gastric cancer cells by blocking expression of phosphoinositide 3-kinase subunits. Gastroenterology 2016, 150, 1183–1195. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.G.; Yang, M.F.; Fan, J.X.; Wang, W. MiR-30a and miR-205 are downregulated in hypoxia and modulate radiosensitivity of prostate cancer cells by inhibiting autophagy via TP53INP1. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1501–1508. [Google Scholar] [PubMed]
- Li, W.F.; Dai, H.; Ou, Q.; Zuo, G.Q.; Liu, C.A. Overexpression of microRNA-30a-5p inhibits liver cancer cell proliferation and induces apoptosis by targeting MTDH/PTEN/AKT pathway. Tumour Biol. 2016, 37, 5885–5895. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.W.; Wang, H.W.; Chang, C.W.; Chu, H.W.; Chen, C.Y.; Yu, J.C.; Chao, J.I.; Liu, H.F.; Ding, S.L.; Shen, C.Y. MicroRNA-30a inhibits cell migration and invasion by downregulating vimentin expression and is a potential prognostic marker in breast cancer. Breast Cancer Res. Treat. 2012, 134, 1081–1093. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Ma, X.; Du, J.; Yao, Z.; Shi, T.; Ai, Q.; Chen, X.; Zhang, Z.; Zhang, X.; Yao, X. MicroRNA-30a as a prognostic factor in urothelial carcinoma of bladder inhibits cellular malignancy by antagonizing Notch1. BJU Int. 2016, 118, 578–589. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Wei, B.; Ye, Q.; Liu, W. Mir-30a-5p/UBE3C axis regulates breast cancer cell proliferation and migration. Biochem. Biophys. Res. Commun. 2016. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Ni, M.; Zhang, J.; Chen, Y.; Ma, H.; Qian, S.; Tang, L.; Tang, J.; Yao, H.; Zhao, C.; et al. MiR-30a can inhibit DNA replication by targeting RPA1 thus slows down the proliferation of cancer cells. Biochem. J. 2016, 473, 2131–2139. [Google Scholar] [CrossRef] [PubMed]
- Ewald, J.A.; Downs, T.M.; Cetnar, J.P.; Ricke, W.A. Expression microarray meta-analysis identifies genes associated with Ras/MAPK and related pathways in progression of muscle-invasive bladder transition cell carcinoma. PLoS ONE 2013, 8, e55414. [Google Scholar] [CrossRef] [PubMed]
- Meng, F.; Wang, F.; Wang, L.; Wong, S.C.; Cho, W.C.; Chan, L.W. MiR-30a-5p overexpression may overcome EGFR-inhibitor resistance through regulating PI3K/AKT signaling pathway in non-small cell lung cancer cell lines. Front Genet 2016, 7, 197. [Google Scholar] [CrossRef] [PubMed]
- Torzsok, P.; Riesz, P.; Kenessey, I.; Szekely, E.; Somoracz, A.; Nyirady, P.; Romics, I.; Schaff, Z.; Lotz, G.; Kiss, A. Claudins and ki-67: Potential markers to differentiate low- and high-grade transitional cell carcinomas of the urinary bladder. J. Histochem. Cytochem. 2011, 59, 1022–1030. [Google Scholar] [CrossRef] [PubMed]
- Akizuki, R.; Shimobaba, S.; Matsunaga, T.; Endo, S.; Ikari, A. Claudin-5, -7, and -18 suppress proliferation mediated by inhibition of phosphorylation of AKT in human lung squamous cell carcinoma. Biochim. Biophys. Acta 2017, 1864, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Kao, Y.H.; Chen, C.L.; Jawan, B.; Chung, Y.H.; Sun, C.K.; Kuo, S.M.; Hu, T.H.; Lin, Y.C.; Chan, H.H.; Cheng, K.H.; et al. Upregulation of hepatoma-derived growth factor is involved in murine hepatic fibrogenesis. J. Hepatol. 2010, 52, 96–105. [Google Scholar] [CrossRef] [PubMed]
| Pathway Name | p-Value | Pathway ID |
|---|---|---|
| MAPK signaling pathway | 0.00274259 | kegg_pathway_261 |
| PI3K-Akt signaling pathway | 0.00308062 | kegg_pathway_262 |
| Tight junction | 0.00451891 | kegg_pathway_257 |
| Protein digestion and absorption | 0.00458404 | kegg_pathway_279 |
| Notch signaling pathway | 0.00534759 | kegg_pathway_55 |
| Cytokine-cytokine receptor interaction | 0.00924043 | kegg_pathway_79 |
| Endocytosis | 0.0151682 | kegg_pathway_232 |
| Focal adhesion | 0.0158629 | kegg_pathway_188 |
| Regulation of actin cytoskeleton | 0.0164765 | kegg_pathway_139 |
| Morphine addiction | 0.0191389 | kegg_pathway_101 |
| Calcium signaling pathway | 0.022008 | kegg_pathway_237 |
| Amoebiasis | 0.0234208 | kegg_pathway_214 |
| Amino sugar and nucleotide sugar metabolism | 0.0254128 | kegg_pathway_203 |
| GABAergic synapse | 0.0258019 | kegg_pathway_235 |
| Ras signaling pathway | 0.0265302 | kegg_pathway_265 |
| Endocrine and other factor-regulated calcium reabsorption | 0.0266089 | kegg_pathway_174 |
| HTLV-I infection | 0.0280212 | kegg_pathway_190 |
| ECM-receptor interaction | 0.0314539 | kegg_pathway_242 |
| Jak-STAT signaling pathway | 0.0400457 | kegg_pathway_40 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chung, Y.-H.; Li, S.-C.; Kao, Y.-H.; Luo, H.-L.; Cheng, Y.-T.; Lin, P.-R.; Tai, M.-H.; Chiang, P.-H. MiR-30a-5p Inhibits Epithelial-to-Mesenchymal Transition and Upregulates Expression of Tight Junction Protein Claudin-5 in Human Upper Tract Urothelial Carcinoma Cells. Int. J. Mol. Sci. 2017, 18, 1826. https://doi.org/10.3390/ijms18081826
Chung Y-H, Li S-C, Kao Y-H, Luo H-L, Cheng Y-T, Lin P-R, Tai M-H, Chiang P-H. MiR-30a-5p Inhibits Epithelial-to-Mesenchymal Transition and Upregulates Expression of Tight Junction Protein Claudin-5 in Human Upper Tract Urothelial Carcinoma Cells. International Journal of Molecular Sciences. 2017; 18(8):1826. https://doi.org/10.3390/ijms18081826
Chicago/Turabian StyleChung, Yueh-Hua, Sung-Chou Li, Ying-Hsien Kao, Hao-Lun Luo, Yuan-Tso Cheng, Pey-Ru Lin, Ming-Hong Tai, and Po-Hui Chiang. 2017. "MiR-30a-5p Inhibits Epithelial-to-Mesenchymal Transition and Upregulates Expression of Tight Junction Protein Claudin-5 in Human Upper Tract Urothelial Carcinoma Cells" International Journal of Molecular Sciences 18, no. 8: 1826. https://doi.org/10.3390/ijms18081826
APA StyleChung, Y.-H., Li, S.-C., Kao, Y.-H., Luo, H.-L., Cheng, Y.-T., Lin, P.-R., Tai, M.-H., & Chiang, P.-H. (2017). MiR-30a-5p Inhibits Epithelial-to-Mesenchymal Transition and Upregulates Expression of Tight Junction Protein Claudin-5 in Human Upper Tract Urothelial Carcinoma Cells. International Journal of Molecular Sciences, 18(8), 1826. https://doi.org/10.3390/ijms18081826