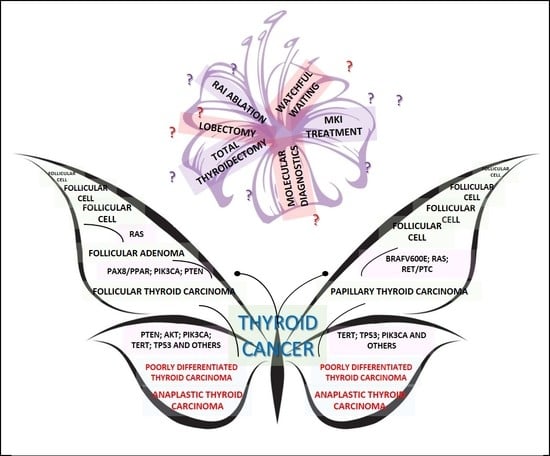
Current Advances in Thyroid Cancer Management. Are We Ready for the Epidemic Rise of Diagnoses?
Abstract
1. Introduction
2. Follicular-Cell Derived Thyroid Cancers
2.1. Is the Increase in TC Incidence a Real Phenomenon or an Artifact Overdiagnosis?
2.2. Is the Diagnosis of Thyroid Cancer an Unquestionable Statement (NIFTP, FTC vs. FA)?
2.3. What Is the Role of Gene Expression/Mutation Classifier in the Diagnosis of TC?
2.4. What Is the Tumor Size That Justifies Total Thyroidectomy?
2.5. When Is Adjuvant Radioiodine Therapy Necessary?
2.6. Are There Any Molecular Factors That Determine TC Aggressiveness?
2.7. Could Small Thyroid Carcinomas Be Managed without Surgery by Watchful Waiting Strategy?
2.8. Is There a Molecular Phenotype of Indolent Thyroid Cancer?
3. A Place for Multi Kinase Inhibitor (MKI) Therapy in RAI-refractory DTC (RR-DTC)
3.1. Role of Tumor Status in Prediction of MKI Therapy Success
3.2. Unsuccessful Attempts of ATC Treatment—Are There Any Chances for Improvement?
4. Medullary Thyroid Carcinoma
4.1. Genotype-Driven Management of Healthy Individuals and Patients with Cancer in MTC
4.2. The Role of Calcitonin in MTC Monitoring
4.3. A Place for MKI Treatment in Advanced MTC
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ATA | American Thyroid Association |
| ATC | Anaplastic thyroid carcinoma |
| AUS | Atypia of undetermined significance |
| BRAF | B-Raf proto-oncogene, serine/threonine kinase |
| CEA | Carcinoembryonic antigen |
| Ct | Calcitonin |
| CVPTC | Classical variant of papillary thyroid carcinoma |
| DTC | Differentiated thyroid carcinoma |
| EANM | European Association of Nuclear Medicine |
| EFC | Encapsulated follicular thyroid carcinoma |
| EFVPTC | Encapsulated follicular variant of papillary thyroid carcinoma |
| ERK | Extracellular signal-regulated kinase |
| FA | Follicular adenomas |
| FLUS | Follicular lesion of undetermined significance |
| FN | Follicular neoplasm |
| FNAB | Fine-needle aspiration biopsy |
| FTC | Follicular thyroid carcinoma |
| FVPTC | Follicular variant of papillary thyroid carcinoma |
| GEC | Afirma Gene Expression Classifier |
| HT | Hashimoto’s thyroiditis |
| ITN | Incidental thyroid nodule |
| MAPK | Mitogen-activated protein kinase |
| MEK | Mitogen-activated protein kinase kinase |
| MEN2 | Multiple endocrine neoplasia type 2 |
| MKIs | Multi kinase inhibitors |
| MTC | Medullary thyroid carcinoma |
| NCCN | National Comprehensive Cancer Network |
| NIFTP | Noninvasive follicular thyroid neoplasm with papillary-like nuclear features |
| NPV | Negative predictive value |
| OS | Overall survival |
| QTc | Corrected QT interval (electrocardiogram) |
| PBDEs | Polybrominated diphenyl etherflame retardants |
| PDTC | Poorly differentiated thyroid carcinoma |
| PFS | Progression-free survival |
| PHEO | Pheochromocytoma |
| PHP | Primary hyperparathyroidism |
| PI3K | Phosphatidylinositol 3-kinase |
| PPV | Positive predictive value |
| PTC | Papillary thyroid carcinoma |
| PTMC | Papillary microcarcinoma |
| RAI | Radioactive iodine |
| RAS | Ras proto-oncogene, GTPase |
| RET | Ret proto-oncogene |
| RR-DTC | Radioiodine-refractory differentiated thyroid carcinoma |
| SEER | Surveillance, Epidemiology, and End Results Program |
| SFN | Suspicious for follicular neoplasm |
| SUSP | Suspicious for malignancy |
| TC | Thyroid cancer |
| TCGA | The Cancer Genome Atlas |
| TKIs | Tyrosine kinase inhibitors |
| TKs | Tyrosine kinases |
| TNM | Tumor, Node, and Metastasis staging |
| TSVPTC | Papillary thyroid carcinoma with trabecular-solid pattern of growth |
| US | United States of America |
References
- National Cancer Institute. Surveilance, Epidemiology, and End Results Program. Available online: https://seer.cancer.gov/statfacts/html/thyro.html (accessed on 25 April 2017).
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Heller, K.S. Do all cancers need to be treated? The role of thyroglobulin in the management of thyroid cancer: The 2006 Hayes Martin lecture. Arch. Otolaryngol. Head Neck Surg. 2007, 133, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M.; et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef] [PubMed]
- Verburg, F.A.; Aktolun, C.; Chiti, A.; Frangos, S.; Giovanella, L.; Hoffmann, M.; Iakovou, I.; Mihailovic, J.; Krause, B.J.; Langsteger, W.; et al. Why the European Association of Nuclear Medicine has declined to endorse the 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1001–1005. [Google Scholar] [CrossRef] [PubMed]
- Verburg, F.A.; Luster, M.; Giovanella, L. Adjuvant post-operative I-131 therapy in differentiated thyroid carcinoma: Are the 2015 ATA guidelines an exact science or a dark art? Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 183–184. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.; Welch, H.G. Current thyroid cancer trends in the United States. JAMA Otolaryngol. Head Neck Surg. 2014, 140, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Elisei, R. Molecular profiles of papillary thyroid tumors have been changing in the last decades: How could we explain it? J. Clin. Endocrinol. Metab. 2014, 99, 412–414. [Google Scholar] [CrossRef] [PubMed]
- Vigneri, R.; Malandrino, P.; Vigneri, P. The changing epidemiology of thyroid cancer: Why is incidence increasing? Curr. Opin. Oncol. 2015, 27, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Udelsman, R.; Zhang, Y. The epidemic of thyroid cancer in the United States: The role of endocrinologists and ultrasounds. Thyroid 2014, 24, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Su, L.; Xiao, H. Review of Factors Related to the Thyroid Cancer Epidemic. Int. J. Endocrinol. 2017, 2017, 5308635. [Google Scholar] [CrossRef] [PubMed]
- Corso, C.; Gomez, X.; Sanabria, A.; Vega, V.; Dominguez, L.C.; Osorio, C. Total thyroidectomy versus hemithyroidectomy for patients with follicular neoplasm. A cost-utility analysis. Int. J. Surg. 2014, 12, 837–842. [Google Scholar] [CrossRef] [PubMed]
- Hoang, J.K.; Langer, J.E.; Middleton, W.D.; Wu, C.C.; Hammers, L.W.; Cronan, J.J.; Tessler, F.N.; Grant, E.G.; Berland, L.L. Managing incidental thyroid nodules detected on imaging: White paper of the ACR Incidental Thyroid Findings Committee. J. Am. Coll. Radiol. 2015, 12, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Harach, H.R.; Franssila, K.O.; Wasenius, V.M. Occult papillary carcinoma of the thyroid. A “normal”; finding in Finland. A systematic autopsy study. Cancer 1985, 56, 531–538. [Google Scholar] [CrossRef]
- Wang, T.S.; Goffredo, P.; Sosa, J.A.; Roman, S.A. Papillary thyroid microcarcinoma: An over-treated malignancy? World J. Surg. 2014, 38, 2297–2303. [Google Scholar] [CrossRef] [PubMed]
- Nikiforov, Y.E.; Seethala, R.R.; Tallini, G.; Baloch, Z.W.; Basolo, F.; Thompson, L.D.R.; Barletta, J.A.; Wenig, B.M.; Al Ghuzlan, A.; Kakudo, K.; et al. Nomenclature Revision for Encapsulated Follicular Variant of Papillary Thyroid Carcinoma: A Paradigm Shift to Reduce Overtreatment of Indolent Tumors. JAMA Oncol. 2016, 2, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Y.; Jemal, A.; Ward, E.M. Increasing incidence of differentiated thyroid cancer in the United States, 1988–2005. Cancer 2009, 115, 3801–3807. [Google Scholar] [CrossRef] [PubMed]
- Vergamini, L.B.; Frazier, A.L.; Abrantes, F.L.; Ribeiro, K.B.; Rodriguez-Galindo, C. Increase in the incidence of differentiated thyroid carcinoma in children, adolescents, and young adults: A population-based study. J. Pediatr. 2014, 164, 1481–1485. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.K.; Little, M.P.; Lubin, J.H.; Brenner, A.V.; Wells, S.A.J.; Sigurdson, A.J.; Nikiforov, Y.E. The increase in thyroid cancer incidence during the last four decades is accompanied by a high frequency of BRAF mutations and a sharp increase in RAS mutations. J. Clin. Endocrinol. Metab. 2014, 99, E276–E285. [Google Scholar] [CrossRef] [PubMed]
- Smyth, P.; Finn, S.; Cahill, S.; O’Regan, E.; Flavin, R.; O’Leary, J.J.; Sheils, O. ret/PTC and BRAF act as distinct molecular, time-dependant triggers in a sporadic Irish cohort of papillary thyroid carcinoma. Int. J. Surg. Pathol. 2005, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mathur, A.; Moses, W.; Rahbari, R.; Khanafshar, E.; Duh, Q.-Y.; Clark, O.; Kebebew, E. Higher rate of BRAF mutation in papillary thyroid cancer over time: A single-institution study. Cancer 2011, 117, 4390–4395. [Google Scholar] [CrossRef] [PubMed]
- Romei, C.; Fugazzola, L.; Puxeddu, E.; Frasca, F.; Viola, D.; Muzza, M.; Moretti, S.; Nicolosi, M.L.; Giani, C.; Cirello, V.; et al. Modifications in the papillary thyroid cancer gene profile over the last 15 years. J. Clin. Endocrinol. Metab. 2012, 97, E1758–E1765. [Google Scholar] [CrossRef] [PubMed]
- Guan, H.; Ji, M.; Bao, R.; Yu, H.; Wang, Y.; Hou, P.; Zhang, Y.; Shan, Z.; Teng, W.; Xing, M. Association of high iodine intake with the T1799A BRAF mutation in papillary thyroid cancer. J. Clin. Endocrinol. Metab. 2009, 94, 1612–1617. [Google Scholar] [CrossRef] [PubMed]
- Weihrauch, M.; Benick, M.; Lehner, G.; Wittekind, M.; Bader, M.; Wrbitzk, R.; Tannapfel, A. High prevalence of K-ras-2 mutations in hepatocellular carcinomas in workers exposed to vinyl chloride. Int. Arch. Occup. Environ. Health 2001, 74, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Porta, M.; Crous-Bou, M.; Wark, P.A.; Vineis, P.; Real, F.X.; Malats, N.; Kampman, E. Cigarette smoking and K-ras mutations in pancreas, lung and colorectal adenocarcinomas: Etiopathogenic similarities, differences and paradoxes. Mutat. Res. 2009, 682, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Guo, G.L.; Han, X.; Zhu, C.; Kilfoy, B.A.; Zhu, Y.; Boyle, P.; Zheng, T. Do Polybrominated Diphenyl Ethers (PBDEs) Increase the Risk of Thyroid Cancer? Biosci. Hypotheses 2008, 1, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Leitzmann, M.F.; Brenner, A.; Moore, S.C.; Koebnick, C.; Park, Y.; Hollenbeck, A.; Schatzkin, A.; Ron, E. Prospective study of body mass index, physical activity and thyroid cancer. Int. J. Cancer 2010, 126, 2947–2956. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, S.; Lise, M.; Clavel-Chapelon, F.; Boutron-Ruault, M.-C.; Guillas, G.; Overvad, K.; Tjonneland, A.; Halkjaer, J.; Lukanova, A.; Kaaks, R.; et al. Body size and risk of differentiated thyroid carcinomas: Findings from the EPIC study. Int. J. Cancer 2012, 131, E1004–E1014. [Google Scholar] [CrossRef] [PubMed]
- Larson, S.D.; Jackson, L.N.; Riall, T.S.; Uchida, T.; Thomas, R.P.; Qiu, S.; Evers, B.M. Increased incidence of well-differentiated thyroid cancer associated with Hashimoto thyroiditis and the role of the PI3k/Akt pathway. J. Am. Coll. Surg. 2007, 204. [Google Scholar] [CrossRef] [PubMed]
- Rajoria, S.; Suriano, R.; Shanmugam, A.; Wilson, Y.L.; Schantz, S.P.; Geliebter, J.; Tiwari, R.K. Metastatic phenotype is regulated by estrogen in thyroid cells. Thyroid 2010, 20, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Derwahl, M.; Nicula, D. Estrogen and its role in thyroid cancer. Endocr. Relat. Cancer 2014, 21, T273–T283. [Google Scholar] [CrossRef] [PubMed]
- Zahid, M.; Goldner, W.; Beseler, C.L.; Rogan, E.G.; Cavalieri, E.L. Unbalanced estrogen metabolism in thyroid cancer. Int. J. Cancer 2013, 133, 2642–2649. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Hendryx, M.; Manson, J.E.; Liang, X.; Margolis, K.L. Hysterectomy, Oophorectomy, and Risk of Thyroid Cancer. J. Clin. Endocrinol. Metab. 2016, 101, 3812–3819. [Google Scholar] [CrossRef] [PubMed]
- Wartofsky, L. A rose by any name surely does smell just as sweetly: The controversy over revised nomenclature for encapsulated follicular variant papillary carcinoma. J. Transl. Intern. Med. 2016, 4, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.D. Ninety-four cases of encapsulated follicular variant of papillary thyroid carcinoma: A name change to Noninvasive Follicular Thyroid Neoplasm with Papillary-like Nuclear Features would help prevent overtreatment. Mod. Pathol. 2016, 29, 698–707. [Google Scholar] [CrossRef] [PubMed]
- Seethala, R.R.; Asa, S.L.; Bullock, M.J.; Carty, S.E.; Hodak, S.P.; McHugh, J.B.; Nikiforov, Y.E.; Pettus, J.; Richardson, M.S.; Shah, J. Protocol for the Examination of Specimens From Patients With Carcinomas of the Thyroid Gland. Arch. Pathol. Lab. Med. 1999, 123, 45–49. [Google Scholar]
- Johnson, S.J.; Stephenson, T.J. NIFTP Addendum to the RCPath Dataset for Thyroid Cancer Histopathology Reports. Available online: http://www.niftp.org/NIFTP.pdf (accessed on 20 July 2017).
- Xu, B.; Tallini, G.; Ghossein, R.A. Noninvasive Follicular Thyroid Neoplasm with Papillary-Like Nuclear Features: Historical Context, Diagnosis, and Future Challenges. Endocr. Pathol. 2017, 28, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Farat, N.; Xu, B.; Onernek, A.; Tuttle, R.; Roman, B.; Katabi, N.; Nose, V.; Sadow, P.; Tallini, G.; Faqiun, W.; et al. Should subcentimeter non-invasive encapsulated, follicular variant of papillary thyroid carcinoma (NI-EFV PTC) be included as noninvasive follicular thyroid neoplasms with papillary-like nuclear features (NIFTP)? In Proceedings of the USCAP 106th Annual Meeting, San Antonio, TX, USA, 4–10 March 2017; p. 138. [Google Scholar]
- Xu, B.; Tallini, G.; Scognamiglio, T.; Roman, B.R.; Tuttle, R.M.; Ghossein, R.A. Outcome of Large Noninvasive Follicular Thyroid Neoplasm with Papillary-Like Nuclear Features. Thyroid 2017, 27, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Poller, D.N.; Nikiforov, Y.E. Non-invasive follicular thyroid neoplasm with papillary-like nuclei: Reducing overtreatment by reclassifying an indolent variant of papillary thyroid cancer. J. Clin. Pathol. 2016, 69, 947–948. [Google Scholar] [CrossRef] [PubMed]
- Ganly, I.; Wang, L.; Tuttle, R.M.; Katabi, N.; Ceballos, G.A.; Harach, H.R.; Ghossein, R. Invasion rather than nuclear features correlates with outcome in encapsulated follicular tumors: Further evidence for the reclassification of the encapsulated papillary thyroid carcinoma follicular variant. Hum. Pathol. 2015, 46, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Lastra, R.R.; Pramick, M.R.; Crammer, C.J.; LiVolsi, V.A.; Baloch, Z.W. Implications of a suspicious afirma test result in thyroid fine-needle aspiration cytology: An institutional experience. Cancer Cytopathol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-C.C.; Friedman, L.; Kennedy, G.C.; Wang, H.; Kebebew, E.; Steward, D.L.; Zeiger, M.A.; Westra, W.H.; Wang, Y.; Khanafshar, E.; et al. A large multicenter correlation study of thyroid nodule cytopathology and histopathology. Thyroid 2011, 21, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Xing, M.; Haugen, B.R.; Schlumberger, M. Progress in molecular-based management of differentiated thyroid cancer. Lancet 2013, 381, 1058–1069. [Google Scholar] [CrossRef]
- Chudova, D.; Wilde, J.I.; Wang, E.T.; Wang, H.; Rabbee, N.; Egidio, C.M.; Reynolds, J.; Tom, E.; Pagan, M.; Rigl, C.T.; et al. Molecular classification of thyroid nodules using high-dimensionality genomic data. J. Clin. Endocrinol. Metab. 2010, 95, 5296–5304. [Google Scholar] [CrossRef] [PubMed]
- Alexander, E.K.; Kennedy, G.C.; Baloch, Z.W.; Cibas, E.S.; Chudova, D.; Diggans, J.; Friedman, L.; Kloos, R.T.; LiVolsi, V.A.; Mandel, S.J.; et al. Preoperative diagnosis of benign thyroid nodules with indeterminate cytology. N. Engl. J. Med. 2012, 367, 705–715. [Google Scholar] [CrossRef] [PubMed]
- Alexander, E.K.; Schorr, M.; Klopper, J.; Kim, C.; Sipos, J.; Nabhan, F.; Parker, C.; Steward, D.L.; Mandel, S.J.; Haugen, B.R. Multicenter clinical experience with the Afirma gene expression classifier. J. Clin. Endocrinol. Metab. 2014, 99, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Nishino, M. Molecular cytopathology for thyroid nodules: A review of methodology and test performance. Cancer Cytopathol. 2016, 124, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Lin, O. Molecular Testing of Thyroid Nodules: A Review of Current Available Tests for Fine-Needle Aspiration Specimens. Arch. Pathol. Lab. Med. 2016, 140, 1338–1344. [Google Scholar] [CrossRef] [PubMed]
- Brauner, E.; Holmes, B.J.; Krane, J.F.; Nishino, M.; Zurakowski, D.; Hennessey, J.V.; Faquin, W.C.; Parangi, S. Performance of the Afirma Gene Expression Classifier in Hurthle Cell Thyroid Nodules Differs from Other Indeterminate Thyroid Nodules. Thyroid 2015, 25, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Nikiforova, M.N.; Wald, A.I.; Roy, S.; Durso, M.B.; Nikiforov, Y.E. Targeted Next-Generation Sequencing Panel (ThyroSeq) for Detection of Mutations in Thyroid Cancer. J. Clin. Endocrinol. Metab. 2013, 98, E1852–E1860. [Google Scholar] [CrossRef] [PubMed]
- Nikiforov, Y.E.; Carty, S.E.; Chiosea, S.I.; Coyne, C.; Duvvuri, U.; Ferris, R.L.; Gooding, W.E.; Hodak, S.P.; LeBeau, S.O.; Ohori, N.P.; et al. Highly accurate diagnosis of cancer in thyroid nodules with follicular neoplasm/suspicious for a follicular neoplasm cytology by ThyroSeq v2 next-generation sequencing assay. Cancer 2014, 120, 3627–3634. [Google Scholar] [CrossRef] [PubMed]
- Nixon, I.J. Wysoko zróżnicowany rak tarczycy—Czy nie leczysz swoich pacjentów zbyt intensywnie? Endokrynol. Pol. 2016, 67, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Allan, E.; Owens, S.E.; Waller, M.L. Differentiated thyroid cancer: Lobectomy and radioiodine, a treatment suitable for all cases? Nucl. Med. Commun. 1999, 20, 983–989. [Google Scholar] [CrossRef] [PubMed]
- Bramley, M.D.; Harrison, B.J. Papillary microcarcinoma of the thyroid gland. Br. J. Surg. 1996, 83, 1674–1683. [Google Scholar] [CrossRef] [PubMed]
- Cohn, K.H.; Backdahl, M.; Forsslund, G.; Auer, G.; Zetterberg, A.; Lundell, G.; Granberg, P.O.; Lowhagen, T.; Willems, J.S.; Cady, B. Biologic considerations and operative strategy in papillary thyroid carcinoma: Arguments against the routine performance of total thyroidectomy. Surgery 1984, 96, 957–971. [Google Scholar] [PubMed]
- De Montes, F.B.G.; Huerta, H.D.; Ortiz, M.G.; Camacho, G.V.; Solano, F.R. Comparison of three prognostic indexes in differentiated thyroid cancer. Eur. J. Surg. Oncol. 1999, 51, 285–288. [Google Scholar]
- DeGroot, L.J.; Kaplan, E.L.; McCormick, M.; Straus, F.H. Natural history, treatment, and course of papillary thyroid carcinoma. J. Clin. Endocrinol. Metab. 1990, 71, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferri, E.L. An overview of the management of papillary and follicular thyroid carcinoma. Thyroid 1999, 9, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Czarniecka, A.; Jarzab, M.; Krajewska, J.; Sacher, A.; Poltorak, S.; Wloch, J. The impact of the extent and time of operation on the survival in patients with differentiated thyroid carcinoma (DTC). Endokrynol. Pol. 2006, 57, 347–355. [Google Scholar] [PubMed]
- Cooper, D.S.; Doherty, G.M.; Haugen, B.R.; Hauger, B.R.; Kloos, R.T.; Lee, S.L.; Mandel, S.J.; Mazzaferri, E.L.; McIver, B.; Pacini, F.; et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 2009, 19, 1167–1214. [Google Scholar] [CrossRef] [PubMed]
- Adam, M.A.; Goffredo, P.; Youngwirth, L.; Scheri, R.P.; Roman, S.A.; Sosa, J.A. Same thyroid cancer, different national practice guidelines: When discordant American Thyroid Association and National Comprehensive Cancer Network surgery recommendations are associated with compromised patient outcome. Surgery 2016, 159, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Adam, M.A.; Pura, J.; Gu, L.; Dinan, M.A.; Tyler, D.S.; Reed, S.D.; Scheri, R.; Roman, S.A.; Sosa, J.A. Extent of surgery for papillary thyroid cancer is not associated with survival: An analysis of 61,775 patients. Ann. Surg. 2014, 260. [Google Scholar] [CrossRef] [PubMed]
- Czarniecka, A.; Poltorak, S.; Sacher, A.; Maciejewski, A.; Krajewska, J.; Jarzab, M.; Stobiecka, E.; Zembala-Nozynska, E.; Chmielik, E.; Rusinek, D.; et al. Surgical strategy in low advanced differentiated thyroid cancer staged T1N0M0-results of a pilot feasibility study. 38th Annual Meeting of the European Thyroid Association, Santiago de Compostela, Spain, September 6–10, 2014; OP93. Eur. Thyroid J. 2014, 3, 73–226. [Google Scholar]
- Tuttle, R.M.; Haddad, R.I.; Ball, D.W.; Byrd, D.; Dickson, P.; Duh, Q.-Y.; Ehya, H.; Haymart, M.; Hoh, C.; Hunt, J.P.; et al. Thyroid carcinoma, version 2.2014. J. Natl. Compr. Cancer Netw. 2014, 12, 1671–1680. [Google Scholar] [CrossRef]
- Perros, P.; Boelaert, K.; Colley, S.; Evans, C.; Evans, R.M.; Gerrard Ba, G.; Gilbert, J.; Harrison, B.; Johnson, S.J.; Giles, T.E.; et al. Guidelines for the management of thyroid cancer. Clin. Endocrinol. 2014, 81, 1–122. [Google Scholar] [CrossRef] [PubMed]
- Barney, B.M.; Hitchcock, Y.J.; Sharma, P.; Shrieve, D.C.; Tward, J.D. Overall and cause-specific survival for patients undergoing lobectomy, near-total, or total thyroidectomy for differentiated thyroid cancer. Head Neck 2011, 33, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Mendelsohn, A.H.; Elashoff, D.A.; Abemayor, E.; St John, M.A. Surgery for papillary thyroid carcinoma: Is lobectomy enough? Arch. Otolaryngol. Head Neck Surg. 2010, 136, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Bilimoria, K.Y.; Bentrem, D.J.; Ko, C.Y.; Stewart, A.K.; Winchester, D.P.; Talamonti, M.S.; Sturgeon, C. Extent of surgery affects survival for papillary thyroid cancer. Ann. Surg. 2007, 246, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Miyauchi, A.; Oda, H. Low-risk papillary microcarcinoma of the thyroid: A review of active surveillance trials. Eur. J. Surg. Oncol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Schlumberger, M.; Pacini, F.; Tuttle, R. Initial treatment. In Thyroid Tumors, 4th ed.; Estimprim: Besançon, France, 2016; pp. 141–148. [Google Scholar]
- Schmidbauer, B.; Menhart, K.; Hellwig, D.; Grosse, J. Differentiated Thyroid Cancer-Treatment: State of the Art. Int. J. Mol. Sci. 2017, 18, 1292. [Google Scholar] [CrossRef] [PubMed]
- Jarząb, B.; Dedecjus, M.; Handkiewicz-Junak, D.; Lange, D.; Lewiński, A.; Nasierowska-Guttmejer, A.; Ruchała, M.; Słowińska-Klencka, D.; Nauman, J.; Adamczewski, Z.; et al. Diagnostics and Treatment of Thyroid Carcinoma. Endokrynol. Pol. 2016, 67, 74–107. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, N.; Akbani, R.; Aksoy, B.A.; Ally, A.; Arachchi, H.; Asa, S.L.; Auman, J.T.; Balasundaram, M.; Balu, S.; Baylin, S.B.; et al. Integrated Genomic Characterization of Papillary Thyroid Carcinoma. Cell 2014, 159, 676–690. [Google Scholar] [CrossRef] [PubMed]
- Tavares, C.; Melo, M.; Cameselle-Teijeiro, J.M.; Soares, P.; Sobrinho-Simoes, M. Endocrine Tumours: Genetic predictors of thyroid cancer outcome. Eur. J. Endocrinol. 2016, 174, R117–R126. [Google Scholar] [CrossRef] [PubMed]
- Kebebew, E.; Weng, J.; Bauer, J.; Ranvier, G.; Clark, O.H.; Duh, Q.-Y.; Shibru, D.; Bastian, B.; Griffin, A. The prevalence and prognostic value of BRAF mutation in thyroid cancer. Ann. Surg. 2007, 246, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.Y.; Kim, W.B.; Rhee, Y.S.; Song, J.Y.; Kim, J.M.; Gong, G.; Lee, S.; Kim, S.Y.; Kim, S.C.; Hong, S.J.; et al. The BRAF mutation is useful for prediction of clinical recurrence in low-risk patients with conventional papillary thyroid carcinoma. Clin. Endocrinol. 2006, 65, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Namba, H.; Nakashima, M.; Hayashi, T.; Hayashida, N.; Maeda, S.; Rogounovitch, T.I.; Ohtsuru, A.; Saenko, V.A.; Kanematsu, T.; Yamashita, S. Clinical implication of hot spot BRAF mutation, V599E, in papillary thyroid cancers. J. Clin. Endocrinol. Metab. 2003, 88, 4393–4397. [Google Scholar] [CrossRef] [PubMed]
- Oler, G.; Cerutti, J.M. High prevalence of BRAF mutation in a Brazilian cohort of patients with sporadic papillary thyroid carcinomas: Correlation with more aggressive phenotype and decreased expression of iodide-metabolizing genes. Cancer 2009, 115, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Elisei, R.; Ugolini, C.; Viola, D.; Lupi, C.; Biagini, A.; Giannini, R.; Romei, C.; Miccoli, P.; Pinchera, A.; Basolo, F. BRAF(V600E) mutation and outcome of patients with papillary thyroid carcinoma: A 15-year median follow-up study. J. Clin. Endocrinol. Metab. 2008, 93, 3943–3949. [Google Scholar] [CrossRef] [PubMed]
- Xing, M.; Westra, W.H.; Tufano, R.P.; Cohen, Y.; Rosenbaum, E.; Rhoden, K.J.; Carson, K.A.; Vasko, V.; Larin, A.; Tallini, G.; et al. BRAF mutation predicts a poorer clinical prognosis for papillary thyroid cancer. J. Clin. Endocrinol. Metab. 2005, 90, 6373–6379. [Google Scholar] [CrossRef] [PubMed]
- Fugazzola, L.; Puxeddu, E.; Avenia, N.; Romei, C.; Cirello, V.; Cavaliere, A.; Faviana, P.; Mannavola, D.; Moretti, S.; Rossi, S.; et al. Correlation between B-RAFV600E mutation and clinico-pathologic parameters in papillary thyroid carcinoma: Data from a multicentric Italian study and review of the literature. Endocr. Relat. Cancer 2006, 13, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Riesco-Eizaguirre, G.; Gutierrez-Martinez, P.; Garcia-Cabezas, M.A.; Nistal, M.; Santisteban, P. The oncogene BRAF V600E is associated with a high risk of recurrence and less differentiated papillary thyroid carcinoma due to the impairment of Na+/I− targeting to the membrane. Endocr. Relat. Cancer 2006, 13, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Hardee, S.; Prasad, M.L.; Hui, P.; Dinauer, C.A.; Morotti, R.A. Pathologic Characteristics, Natural History, and Prognostic Implications of BRAFV600E Mutation in Pediatric Papillary Thyroid Carcinoma. Pediatr. Dev. Pathol. 2017, 20, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Geng, J.; Wang, H.; Liu, Y.; Tai, J.; Jin, Y.; Zhang, J.; He, L.; Fu, L.; Qin, H.; Song, Y.; et al. Correlation between BRAF V600E mutation and clinicopathological features in pediatric papillary thyroid carcinoma. Sci. China Life Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Xing, M.; Alzahrani, A.S.; Carson, K.A.; Viola, D.; Elisei, R.; Bendlova, B.; Yip, L.; Mian, C.; Vianello, F.; Tuttle, R.M.; et al. Association between BRAF V600E mutation and mortality in patients with papillary thyroid cancer. JAMA 2013, 309, 1493–1501. [Google Scholar] [CrossRef] [PubMed]
- Xing, M. Molecular pathogenesis and mechanisms of thyroid cancer. Nat. Rev. Cancer 2013, 13, 184–199. [Google Scholar] [CrossRef] [PubMed]
- Xing, M.; Alzahrani, A.S.; Carson, K.A.; Shong, Y.K.; Kim, T.Y.; Viola, D.; Elisei, R.; Bendlová, B.; Yip, L.; Mian, C.; et al. Association Between BRAF V600E Mutation and Recurrence of Papillary Thyroid Cancer. J. Clin. Oncol. 2015, 33, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Xing, M. BRAF Mutation and Thyroid Cancer Recurrence. J. Clin. Oncol. 2015, 33, 2482–2483. [Google Scholar] [CrossRef] [PubMed]
- Abrosimov, A.; Saenko, V.; Rogounovitch, T.; Namba, H.; Lushnikov, E.; Mitsutake, N.; Yamashita, S. Different structural components of conventional papillary thyroid carcinoma display mostly identical BRAF status. Int. J. Cancer 2007, 120, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Ghossein, R.A.; Katabi, N.; Fagin, J.A. Immunohistochemical detection of mutated BRAF V600E supports the clonal origin of BRAF-induced thyroid cancers along the spectrum of disease progression. J. Clin. Endocrinol. Metab. 2013, 98, E1414–E1421. [Google Scholar] [CrossRef] [PubMed]
- Guerra, A.; Sapio, M.R.; Marotta, V.; Campanile, E.; Rossi, S.; Forno, I.; Fugazzola, L.; Budillon, A.; Moccia, T.; Fenzi, G.; et al. The primary occurrence of BRAF(V600E) is a rare clonal event in papillary thyroid carcinoma. J. Clin. Endocrinol. Metab. 2012, 97, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, G.; Sancisi, V.; Torricelli, F.; Ragazzi, M.; Frasoldati, A.; Piana, S.; Ciarrocchi, A. Allele percentage of the BRAF V600E mutation in papillary thyroid carcinomas and corresponding lymph node metastases: No evidence for a role in tumor progression. J. Clin. Endocrinol. Metab. 2013, 98, E934–E942. [Google Scholar] [CrossRef] [PubMed]
- De Biase, D.; Cesari, V.; Visani, M.; Casadei, G.P.; Cremonini, N.; Gandolfi, G.; Sancisi, V.; Ragazzi, M.; Pession, A.; Ciarrocchi, A.; et al. High-sensitivity BRAF mutation analysis: BRAF V600E is acquired early during tumor development but is heterogeneously distributed in a subset of papillary thyroid carcinomas. J. Clin. Endocrinol. Metab. 2014, 99, E1530–E1538. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.W.; Hodis, E.; Xu, M.J.; Kryukov, G.V.; Chin, L.; Garraway, L.A. Highly recurrent TERT promoter mutations in human melanoma. Science 2013, 339, 957–959. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Bishop, J.; Shan, Y.; Pai, S.; Liu, D.; Murugan, A.K.; Sun, H.; El-Naggar, A.K.; Xing, M. Highly prevalent TERT promoter mutations in aggressive thyroid cancers. Endocr. Relat. Cancer 2013, 20, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Melo, M.; da Rocha, A.G.; Vinagre, J.; Batista, R.; Peixoto, J.; Tavares, C.; Celestino, R.; Almeida, A.; Salgado, C.; Eloy, C.; et al. TERT promoter mutations are a major indicator of poor outcome in differentiated thyroid carcinomas. J. Clin. Endocrinol. Metab. 2014, 99, E754–E765. [Google Scholar] [CrossRef] [PubMed]
- Xing, M.; Liu, R.; Liu, X.; Murugan, A.K.; Zhu, G.; Zeiger, M.A.; Pai, S.; Bishop, J. BRAF V600E and TERT promoter mutations cooperatively identify the most aggressive papillary thyroid cancer with highest recurrence. J. Clin. Oncol. 2014, 32, 2718–2726. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Qu, S.; Liu, R.; Sheng, C.; Shi, X.; Zhu, G.; Murugan, A.K.; Guan, H.; Yu, H.; Wang, Y.; et al. TERT promoter mutations and their association with braf v600e mutation and aggressive clinicopathological characteristics of thyroid cancer. J. Clin. Endocrinol. Metab. 2014, 99, E1130–E1136. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.S.; Lim, J.A.; Choi, H.; Won, J.-K.; Moon, J.H.; Cho, S.W.; Lee, K.E.; Park, Y.J.; Yi, K.H.; Park, D.J.; et al. Prognostic effects of TERT promoter mutations are enhanced by coexistence with BRAF or RAS mutations and strengthen the risk prediction by the ATA or TNM staging system in differentiated thyroid cancer patients. Cancer 2016, 122, 1370–1379. [Google Scholar] [CrossRef] [PubMed]
- Vinagre, J.; Almeida, A.; Populo, H.; Batista, R.; Lyra, J.; Pinto, V.; Coelho, R.; Celestino, R.; Prazeres, H.; Lima, L.; et al. Frequency of TERT promoter mutations in human cancers. Nat. Commun. 2013, 4, 2185. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Ghossein, R. Genomic landscape of poorly differentiated and anaplastic thyroid Carcinoma. Endocr. Pathol. 2016, 27, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rostan, G.; Camp, R.L.; Herrero, A.; Carcangiu, M.L.; Rimm, D.L.; Tallini, G. Beta-catenin dysregulation in thyroid neoplasms: Down-regulation, aberrant nuclear expression, and CTNNB1 exon 3 mutations are markers for aggressive tumor phenotypes and poor prognosis. Am. J. Pathol. 2001, 158, 987–996. [Google Scholar] [CrossRef]
- Garcia-Rostan, G.; Tallini, G.; Herrero, A.; D’Aquila, T.G.; Carcangiu, M.L.; Rimm, D.L. Frequent mutation and nuclear localization of beta-catenin in anaplastic thyroid carcinoma. Cancer Res. 1999, 59, 1811–1815. [Google Scholar] [PubMed]
- Kurihara, T.; Ikeda, S.; Ishizaki, Y.; Fujimori, M.; Tokumoto, N.; Hirata, Y.; Ozaki, S.; Okajima, M.; Sugino, K.; Asahara, T. Immunohistochemical and sequencing analyses of the Wnt signaling components in Japanese anaplastic thyroid cancers. Thyroid 2004, 14, 1020–1029. [Google Scholar] [CrossRef] [PubMed]
- Landa, I.; Ibrahimpasic, T.; Boucai, L.; Sinha, R.; Knauf, J.A.; Shah, R.H.; Dogan, S.; Ricarte-Filho, J.C.; Krishnamoorthy, G.P.; Xu, B.; et al. Genomic and transcriptomic hallmarks of poorly differentiated and anaplastic thyroid cancers. J. Clin. Investig. 2016, 126, 1052–1066. [Google Scholar] [CrossRef] [PubMed]
- Kunstman, J.W.; Juhlin, C.C.; Goh, G.; Brown, T.C.; Stenman, A.; Healy, J.M.; Rubinstein, J.C.; Choi, M.; Kiss, N.; Nelson-Williams, C.; et al. Characterization of the mutational landscape of anaplastic thyroid cancer via whole-exome sequencing. Hum. Mol. Genet. 2015, 24, 2318–2329. [Google Scholar] [CrossRef] [PubMed]
- Ringel, M.D. Molecular markers of aggressiveness of thyroid cancer. Curr. Opin. Endocrinol. Diabetes Obes. 2009, 16, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.-K.; Liu, R.-T.; Kang, H.-Y. MicroRNA-146b: A novel biomarker and therapeutic target for human papillary thyroid cancer. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Hardin, H.; Montemayor-Garcia, C.; Asioli, S.; Righi, A.; Maletta, F.; Sapino, A.; Lloyd, R.V. In situ hybridization analysis of mir-146b-5p and mir-21 in thyroid nodules: Diagnostic implications. Endocr. Pathol. 2015, 26, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Maeda, T.; Izumi, K.; Otsuka, H. Occult papillary carcinoma of the thyroid. A study of 408 autopsy cases. Cancer 1990, 65, 1173–1179. [Google Scholar] [CrossRef]
- Davison, B.J.; Oliffe, J.L.; Pickles, T.; Mroz, L. Factors influencing men undertaking active surveillance for the management of low-risk prostate cancer. Oncol. Nurs. Forum 2009, 36, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Gorin, M.A.; Eldefrawy, A.; Ekwenna, O.; Soloway, M.S. Active surveillance for low-risk prostate cancer: Knowledge, acceptance and practice among urologists. Prostate Cancer Prostatic Dis. 2012, 15, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Uruno, T.; Nakano, K.; Takamura, Y.; Miya, A.; Kobayashi, K.; Yokozawa, T.; Matsuzuka, F.; Kuma, S.; Kuma, K.; et al. An observation trial without surgical treatment in patients with papillary microcarcinoma of the thyroid. Thyroid 2003, 13, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Sugitani, I.; Toda, K.; Yamada, K.; Yamamoto, N.; Ikenaga, M.; Fujimoto, Y. Three distinctly different kinds of papillary thyroid microcarcinoma should be recognized: Our treatment strategies and outcomes. World J. Surg. 2010, 34, 1222–1231. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Miyauchi, A.; Inoue, H.; Fukushima, M.; Kihara, M.; Higashiyama, T.; Tomoda, C.; Takamura, Y.; Kobayashi, K.; Miya, A. An observational trial for papillary thyroid microcarcinoma in Japanese patients. World J. Surg. 2010, 34, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Miyauchi, A.; Kihara, M.; Higashiyama, T.; Kobayashi, K.; Miya, A. Patient age is significantly related to the progression of papillary microcarcinoma of the thyroid under observation. Thyroid 2014, 24, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Oda, H.; Miyauchi, A.; Ito, Y.; Yoshioka, K.; Nakayama, A.; Sasai, H.; Masuoka, H.; Yabuta, T.; Fukushima, M.; Higashiyama, T.; et al. Incidences of unfavorable events in the management of low-risk papillary Microcarcinoma of the Thyroid by Active Surveillance Versus Immediate Surgery. Thyroid 2016, 26, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Fukuoka, O.; Sugitani, I.; Ebina, A.; Toda, K.; Kawabata, K.; Yamada, K. Natural history of asymptomatic papillary thyroid microcarcinoma: Time-dependent changes in calcification and vascularity during active surveillance. World J. Surg. 2016, 40, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Hay, I.D. Management of patients with low-risk papillary thyroid carcinoma. Endocr. Pract. 2007, 13, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferri, E.L. Management of low-risk differentiated thyroid cancer. Endocr. Pract. 2007, 13, 498–512. [Google Scholar] [CrossRef] [PubMed]
- Bradley, N.L.; Wiseman, S.M. Papillary thyroid microcarcinoma: The significance of high risk features. BMC Cancer 2017, 17, 142. [Google Scholar] [CrossRef] [PubMed]
- Brito, J.P.; Ito, Y.; Miyauchi, A.; Tuttle, R.M. A clinical framework to facilitate risk stratification when considering an active surveillance alternative to immediate biopsy and surgery in papillary microcarcinoma. Thyroid 2016, 26, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Leboulleux, S.; Tuttle, R.M.; Pacini, F.; Schlumberger, M. Papillary thyroid microcarcinoma: Time to shift from surgery to active surveillance? Lancet Diabetes Endocrinol. 2016, 4, 933–942. [Google Scholar] [CrossRef]
- Torregrossa, L.; Viola, D.; Sensi, E.; Giordano, M.; Piaggi, P.; Romei, C.; Materazzi, G.; Miccoli, P.; Elisei, R.; Basolo, F. Papillary thyroid carcinoma with rare exon 15 braf mutation has indolent behavior: A single-institution experience. J. Clin. Endocrinol. Metab. 2016, 101, 4413–4420. [Google Scholar] [CrossRef] [PubMed]
- Kakarmath, S.; Heller, H.T.; Alexander, C.A.; Cibas, E.S.; Krane, J.F.; Barletta, J.A.; Lindeman, N.I.; Frates, M.C.; Benson, C.B.; Gawande, A.A.; et al. Clinical, sonographic, and pathological characteristics of ras-positive versus braf-positive thyroid carcinoma. J. Clin. Endocrinol. Metab. 2016, 101, 4938–4944. [Google Scholar] [CrossRef] [PubMed]
- Medici, M.; Kwong, N.; Angell, T.E.; Marqusee, E.; Kim, M.I.; Frates, M.C.; Benson, C.B.; Cibas, E.S.; Barletta, J.A.; Krane, J.F.; et al. The variable phenotype and low-risk nature of RAS-positive thyroid nodules. BMC Med. 2015, 13, 184. [Google Scholar] [CrossRef] [PubMed]
- Krajewska, J.; Gawlik, T.; Jarzab, B. Advances in small molecule therapy for treating metastatic thyroid cancer. Expert Opin. Pharmacother. 2017, 18, 1049–1060. [Google Scholar] [CrossRef] [PubMed]
- Brose, M.S.; Nutting, C.M.; Jarzab, B.; Elisei, R.; Siena, S.; Bastholt, L.; de la Fouchardiere, C.; Pacini, F.; Paschke, R.; Shong, Y.K.; et al. Sorafenib in radioactive iodine-refractory, locally advanced or metastatic differentiated thyroid cancer: A randomised, double-blind, phase 3 trial. Lancet 2014. [Google Scholar] [CrossRef]
- Schlumberger, M.; Tahara, M.; Wirth, L.J.; Robinson, B.; Brose, M.S.; Elisei, R.; Habra, M.A.; Newbold, K.; Shah, M.H.; Hoff, A.O.; et al. Lenvatinib versus Placebo in Radioiodine-Refractory Thyroid Cancer. N. Engl. J. Med. 2015, 372, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Brose, M.; Jarzab, B.; Elisei, R.; Siena, S.; Bastholt, L.; de la Fouchardiere, C.; Pacini, F.; Paschke, R.; Nutting, C.; Shong, Y.; et al. Updated overall survival analysis of patients with locally advanced or metastatic radioiodine-refractory differentiated thyroid cancer treated with sorafenib on phase 3 DECISION trial (ABSTRACT). J. Clin. Oncol. 2014, 32, 6060. [Google Scholar]
- Brose, M.S.; Worden, F.P.; Newbold, K.L.; Guo, M.; Hurria, A. Effect of age on the efficacy and safety of lenvatinib in radioiodine-refractory differentiated thyroid cancer in the phase III select trial. J. Clin. Oncol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Krajewska, J.; Paliczka-Cieslik, E.; Jarzab, B. Managing tyrosine kinase inhibitors side effects in thyroid cancer. Expert Rev. Endocrinol. Metab. 2017. [Google Scholar] [CrossRef]
- Lote, H.; Bhosle, J.; Thway, K.; Newbold, K.; O’Brien, M. Epidermal growth factor mutation as a diagnostic and therapeutic target in metastatic poorly differentiated thyroid carcinoma: A case report and review of the literature. Case Rep. Oncol. 2014, 7, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Duquette, M.; Sadow, P.M.; Husain, A.; Sims, J.N.; Antonello, Z.A.; Fischer, A.H.; Song, C.; Castellanos-Rizaldos, E.; Makrigiorgos, G.M.; Kurebayashi, J.; et al. Metastasis-associated MCL1 and P16 copy number alterations dictate resistance to vemurafenib in a BRAFV600E patient-derived papillary thyroid carcinoma preclinical model. Oncotarget 2015, 6, 42445–42467. [Google Scholar] [CrossRef] [PubMed]
- Danysh, B.P.; Rieger, E.Y.; Sinha, D.K.; Evers, C.V.; Cote, G.J.; Cabanillas, M.E.; Hofmann, M.-C. Long-term vemurafenib treatment drives inhibitor resistance through a spontaneous KRAS G12D mutation in a BRAF V600E papillary thyroid carcinoma model. Oncotarget 2016, 7, 30907–30923. [Google Scholar] [CrossRef] [PubMed]
- Lito, P.; Saborowski, A.; Yue, J.; Solomon, M.; Joseph, E.; Gadal, S.; Saborowski, M.; Kastenhuber, E.; Fellmann, C.; Ohara, K.; et al. Disruption of CRAF-mediated MEK activation is required for effective MEK inhibition in KRAS mutant tumors. Cancer Cell 2014, 25, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Lacouture, M.E.; Duvic, M.; Hauschild, A.; Prieto, V.G.; Robert, C.; Schadendorf, D.; Kim, C.C.; McCormack, C.J.; Myskowski, P.L.; Spleiss, O.; et al. Analysis of dermatologic events in vemurafenib-treated patients with melanoma. Oncologist 2013, 18, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-H.; Zhang, Y.; Van Horn, R.D.; Yin, T.; Buchanan, S.; Yadav, V.; Mochalkin, I.; Wong, S.S.; Yue, Y.G.; Huber, L.; et al. Oncogenic BRAF Deletions That Function as Homodimers and Are Sensitive to Inhibition by RAF Dimer Inhibitor LY3009120. Cancer Discov. 2016, 6, 300–315. [Google Scholar] [CrossRef] [PubMed]
- De la Fouchardiere, C.; Oussaid, N.; Derbel, O.; Decaussin-Petrucci, M.; Fondrevelle, M.-E.; Wang, Q.; Bringuier, P.-P.; Bournaud-Salinas, C.; Peix, J.-L.; Lifante, J.-C.; et al. Does Molecular Genotype Provide Useful Information in the Management of Radioiodine Refractory Thyroid Cancers? Results of a Retrospective Study. Target Oncol. 2016, 11, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Tahara, M.; Schlumberger, M.; Elisei, R.; Habra, M.A.; Kiyota, N.; Paschke, R.; Dutcus, C.E.; Hihara, T.; McGrath, S.; Matijevic, M.; et al. Exploratory analysis of biomarkers associated with clinical outcomes from the study of lenvatinib in differentiated cancer of the thyroid. Eur. J. Cancer 2017, 75, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Pinto, N.; Black, M.; Patel, K.; Yoo, J.; Mymryk, J.S.; Barrett, J.W.; Nichols, A.C. Genomically driven precision medicine to improve outcomes in anaplastic thyroid cancer. J. Oncol. 2014, 2014, 936285. [Google Scholar] [CrossRef] [PubMed]
- Schlumberger, M.; Baudin, E.; Travagli, J.P. Papillary and follicular cancers of the thyroid. Presse Med. 1998, 27, 1479–1481. [Google Scholar] [PubMed]
- Smallridge, R.C.; Ain, K.B.; Asa, S.L.; Bible, K.C.; Brierley, J.D.; Burman, K.D.; Kebebew, E.; Lee, N.Y.; Nikiforov, Y.E.; Rosenthal, M.S.; et al. American Thyroid Association guidelines for management of patients with anaplastic thyroid cancer. Thyroid 2012, 22, 1104–1139. [Google Scholar] [CrossRef] [PubMed]
- Demeter, J.G.; De Jong, S.A.; Lawrence, A.M.; Paloyan, E. Anaplastic thyroid carcinoma: Risk factors and outcome. Surgery 1991, 110, 956–961. [Google Scholar] [PubMed]
- Rosove, M.H.; Peddi, P.F.; Glaspy, J.A. BRAF V600E inhibition in anaplastic thyroid cancer. N. Engl. J. Med. 2013, 368, 684–685. [Google Scholar] [CrossRef] [PubMed]
- Logan, C.Y.; Nusse, R. The Wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol. 2004, 20, 781–810. [Google Scholar] [CrossRef] [PubMed]
- Takemaru, K.-I.; Ohmitsu, M.; Li, F.-Q. An oncogenic hub: Beta-catenin as a molecular target for cancer therapeutics. Handb. Exp. Pharmacol. 2008, 261–284. [Google Scholar] [CrossRef]
- Ha, H.T.; Lee, J.S.; Urba, S.; Koenig, R.J.; Sisson, J.; Giordano, T.; Worden, F.P. A phase II study of imatinib in patients with advanced anaplastic thyroid cancer. Thyroid 2010, 20, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Bible, K.C.; Suman, V.J.; Menefee, M.E.; Smallridge, R.C.; Molina, J.R.; Maples, W.J.; Karlin, N.J.; Traynor, A.M.; Kumar, P.; Goh, B.C.; et al. A multiinstitutional phase 2 trial of pazopanib monotherapy in advanced anaplastic thyroid cancer. J. Clin. Endocrinol. Metab. 2012, 97, 3179–3184. [Google Scholar] [CrossRef] [PubMed]
- Savvides, P.; Nagaiah, G.; Lavertu, P.; Fu, P.; Wright, J.J.; Chapman, R.; Wasman, J.; Dowlati, A.; Remick, S.C. Phase II trial of sorafenib in patients with advanced anaplastic carcinoma of the thyroid. Thyroid 2013, 23, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Hogan, A.R.; Zhuge, Y.; Perez, E.A.; Koniaris, L.G.; Lew, J.I.; Sola, J.E. Pediatric thyroid carcinoma: Incidence and outcomes in 1753 patients. J. Surg. Res. 2009, 156, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Donis-Keller, H.; Dou, S.; Chi, D.; Carlson, K.M.; Toshima, K.; Lairmore, T.C.; Howe, J.R.; Moley, J.F.; Goodfellow, P.; Wells, S.A.J. Mutations in the RET proto-oncogene are associated with MEN 2A and FMTC. Hum. Mol. Genet. 1993, 2, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Frank-Raue, K.; Rondot, S.; Raue, F. Molecular genetics and phenomics of RET mutations: Impact on prognosis of MTC. Mol. Cell. Endocrinol. 2010, 322, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Eng, C.; Clayton, D.; Schuffenecker, I.; Lenoir, G.; Cote, G.; Gagel, R.F.; van Amstel, H.K.; Lips, C.J.; Nishisho, I.; Takai, S.I.; et al. The relationship between specific RET proto-oncogene mutations and disease phenotype in multiple endocrine neoplasia type 2. International RET mutation consortium analysis. JAMA 1996, 276, 1575–1579. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, L.M.; Kwok, J.B.; Healey, C.S.; Elsdon, M.J.; Eng, C.; Gardner, E.; Love, D.R.; Mole, S.E.; Moore, J.K.; Papi, L. Germ-line mutations of the RET proto-oncogene in multiple endocrine neoplasia type 2A. Nature 1993, 363, 458–460. [Google Scholar] [CrossRef] [PubMed]
- Romei, C.; Mariotti, S.; Fugazzola, L.; Taccaliti, A.; Pacini, F.; Opocher, G.; Mian, C.; Castellano, M.; degli Uberti, E.; Ceccherini, I.; et al. Multiple endocrine neoplasia type 2 syndromes (MEN 2): Results from the ItaMEN network analysis on the prevalence of different genotypes and phenotypes. Eur. J. Endocrinol. 2010, 163, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Moline, J.; Eng, C. Multiple endocrine neoplasia type 2: An overview. Genet. Med. 2011, 13, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Moura, M.M.; Cavaco, B.M.; Pinto, A.E.; Domingues, R.; Santos, J.R.; Cid, M.O.; Bugalho, M.J.; Leite, V. Correlation of RET somatic mutations with clinicopathological features in sporadic medullary thyroid carcinomas. Br. J. Cancer 2009, 100, 1777–1783. [Google Scholar] [CrossRef] [PubMed]
- Fugazzola, L.; De Leo, S.; Perrino, M. The optimal range of RET mutations to be tested: European comments to the guidelines of the American Thyroid Association. Thyroid Res. 2013, 6 (Suppl. S1), S8. [Google Scholar] [CrossRef] [PubMed]
- Wells, S.A.; Asa, S.L.; Dralle, H.; Elisei, R.; Evans, D.B.; Gagel, R.F.; Lee, N.; Machens, A.; Moley, J.F.; Pacini, F.; et al. Revised American Thyroid Association guidelines for the management of medullary thyroid carcinoma. Thyroid 2015, 25, 567–610. [Google Scholar] [CrossRef] [PubMed]
- Wiench, M.; Wygoda, Z.; Gubala, E.; Wloch, J.; Lisowska, K.; Krassowski, J.; Scieglinska, D.; Fiszer-Kierzkowska, A.; Lange, D.; Kula, D.; et al. Estimation of risk of inherited medullary thyroid carcinoma in apparent sporadic patients. J. Clin. Oncol. 2001, 19, 1374–1380. [Google Scholar] [CrossRef] [PubMed]
- Oczko-Wojciechowska, M.; Pfeifer, A.; Rusinek, D.; Pawlaczek, A.; Zebracka-Gala, J.; Kowalska, M.; Kowal, M.; Swierniak, M.; Krajewska, J.; Gawlik, T.; et al. The prevalence of somatic RAS mutations in medullary thyroid cancer—A Polish population study. Endokrynol. Pol. 2015, 66, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Elisei, R.; Cosci, B.; Romei, C.; Bottici, V.; Renzini, G.; Molinaro, E.; Agate, L.; Vivaldi, A.; Faviana, P.; Basolo, F.; et al. Prognostic Significance of Somatic RET Oncogene Mutations in Sporadic Medullary Thyroid Cancer: A 10-Year Follow-Up Study. J. Clin. Endocrinol. Metab. 2008, 93, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.; Elisei, R.; Mueller, S.; Schoffski, P.; Brose, M.; Shah, M.; Licitra, L.; Jarzab, B.; Medvedev, V.; Kreissl, M.; et al. The Impact of RET and RAS Mutation Status on Overall Survival in the EXAM Trial, a Phase 3 Study of Cabozantinib in Patients with Progressive, Metastatic Medullary Thyroid Cancer (MTC). In Proceedings of the ITC Annual Meeting, Orlando, FL, USA, 18–23 October 2015. [Google Scholar]
- Schlumberger, M.; Pacini, F.; Tuttle, R. Medullary thyroid carcinoma. In Thyroid Tumors; Estimprim: Besançon, France, 2016; pp. 357–361. [Google Scholar]
- Kloos, R.T.; Eng, C.; Evans, D.B.; Francis, G.L.; Gagel, R.F.; Gharib, H.; Moley, J.F.; Pacini, F.; Ringel, M.D.; Schlumberger, M.; et al. Medullary thyroid cancer: Management guidelines of the American Thyroid Association. Thyroid 2009, 19, 565–612. [Google Scholar] [CrossRef] [PubMed]
- Jarząb, B.; Król, A.; Hasse-Lazar, K.; Jurecka-Lubieniecka, B. Presentation of points of general discussion and voting among the speakers of the European Thyroid Association-Cancer Research Network (ETA-CRN) meeting in Lisbon, 2009, entitled “European comments to ATA medullary thyroid cancer guidelines”. Thyroid Res. 2013, 6, S11. [Google Scholar] [CrossRef] [PubMed]
- Jarzab, B.; Szpak-Ulczok, S.; Wloch, J.; Czarniecka, A.; Krajewska, J. Timing and criteria for prophylactic thyroidectomy in asymptomatic RET carriers—The role of Ct serum level. Thyroid Res. 2013, 6, S9. [Google Scholar] [CrossRef] [PubMed]
- Elisei, R.; Pinchera, A. Advances in the follow-up of differentiated or medullary thyroid cancer. Nat. Rev. Endocrinol. 2012, 8, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Modigliani, E.; Cohen, R.; Campos, J.M.; Conte-Devolx, B.; Maes, B.; Boneu, A.; Schlumberger, M.; Bigorgne, J.C.; Dumontier, P.; Leclerc, L.; et al. Prognostic factors for survival and for biochemical cure in medullary thyroid carcinoma: Results in 899 patients. The GETC Study Group. Groupe d’étude des tumeurs à calcitonine. Clin. Endocrinol. (Oxf.) 1998, 48, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Wygoda, Z.; Oczko-Wojciechowska, M.; Gubała, E.; Pawlaczek, A.; Kula, D.; Wiench, M.; Włoch, J. Medullary thyroid carcinoma: The comparison of the hereditary and sporadic types of cancer. Endokrynol. Pol. 2006, 57, 407–414. [Google Scholar] [PubMed]
- Gawlik, T.; d’Amico, A.; Szpak-Ulczok, S.; Skoczylas, A.; Gubala, E.; Chorazy, A.; Gorczewski, K.; Wloch, J.; Jarzab, B. The prognostic value of tumor markers doubling time in medullary thyroid carcinoma-preliminary report. Thyroid Res. 2010, 3, 10. [Google Scholar] [CrossRef] [PubMed]
- Meijer, J.A.A.; le Cessie, S.; van den Hout, W.B.; Kievit, J.; Schoones, J.W.; Romijn, J.A.; Smit, J.W.A. Calcitonin and carcinoembryonic antigen doubling times as prognostic factors in medullary thyroid carcinoma: A structured meta-analysis. Clin. Endocrinol. 2010, 72, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Kouvaraki, M.A.; Shapiro, S.E.; Fornage, B.D.; Edeiken-Monro, B.S.; Sherman, S.I.; Vassilopoulou-Sellin, R.; Lee, J.E.; Evans, D.B. Role of preoperative ultrasonography in the surgical management of patients with thyroid cancer. Surgery 2003, 134, 946–954. [Google Scholar] [CrossRef]
- Pellegriti, G.; Leboulleux, S.; Baudin, E.; Bellon, N.; Scollo, C.; Travagli, J.P.; Schlumberger, M. Long-term outcome of medullary thyroid carcinoma in patients with normal postoperative medical imaging. Br. J. Cancer 2003, 88, 1537–1542. [Google Scholar] [CrossRef] [PubMed]
- Wells, S.A.; Robinson, B.G.; Gagel, R.F.; Dralle, H.; Fagin, J.A.; Santoro, M.; Baudin, E.; Elisei, R.; Jarzab, B.; Vasselli, J.R.; et al. Vandetanib in patients with locally advanced or metastatic medullary thyroid cancer: A randomized, double-blind phase III trial. J. Clin. Oncol. 2012, 30, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Elisei, R.; Schlumberger, M.J.; Müller, S.P.; Schöffski, P.; Brose, M.S.; Shah, M.H.; Licitra, L.; Jarzab, B.; Medvedev, V.; Kreissl, M.C.; et al. Cabozantinib in progressive medullary thyroid cancer. J. Clin. Oncol. 2013, 31, 3639–3646. [Google Scholar] [CrossRef] [PubMed]
- Schlumberger, M.; Bastholt, L.; Dralle, H.; Jarzab, B.; Pacini, F.; Smit, J.W.A. European thyroid association guidelines for metastatic medullary thyroid cancer. Eur. Thyroid J. 2012, 1, 5–14. [Google Scholar] [CrossRef] [PubMed]
| Type of the Test | Afirma | ThyroSeq |
|---|---|---|
| “Rule out” | *“Rule in” and “Rule out” | |
| Methodology | mRNA gene expression (microarray analysis) | next-generation sequencing |
| Cytology interpretation | Performed in Veracyte laboratory with exception of few academic centers authorized to carry cytology evaluation and submit samples only for molecular testing | Performed by local cytopathologists or centralized laboratory |
| Sample required | 2 dedicated FNA passes | 1–2 drops from first pass, if sufficiently cellular |
| Test report | benign/suspicious | specific mutation/rearrangement |
| Strength | high NPV; validated in a blinded multicenter prospective trial | high NPV and PPV |
| Limitations | low PPV; problems with correct classification of Hürthle cell lesions; trained before the reclassification of EFVPTC as benign NIFTP; reclassification of EFVPTC as benign tumors leads to decrease of PPV | limited validation data; increased risk of “false positive” cases associated with the extended mutational profile of the test; the knowledge of the test result before the histopathological assessment in validation study; reclassification of EFVPTC as benign tumors leads to decrease of PPV |
| Extent of Thyroid Resection | Extent of Central Lymph Node Dissection | |||
|---|---|---|---|---|
| Total/Near Total Thyroidectomy | Lobectomy | Therapeutic | Prophylactic (ipsi or Bilateral) | None |
| Tumor size >4 cm (cT3) | Unifocal, intrathyroidal PMTC without lymph node evidence and distant metastases (cT1aN0M0) | Clinically involved central lymph nodes | Advanced PTC (T3, T4) without involved lymph nodes (cN0) | Low-advanced (T1 or T2), noninvasive, clinically node-negative papillary thyroid carcinoma (cN0) |
| Gross extrathyroidal extension (cT4) | Optionally: thyroid cancer: >1 cm <4 cm (cT1b, cT2), without lymph nodes (cN0) and distant metastases (M0) | Clinically or biopsy proven metastasis in lateral lymph nodes (N1b) | Most FTCs | |
| Metastatic or suspicious cervical lymph nodes or distant metastases | Information necessary to plan further steps in therapy | |||
| Patient or therapeutic team preference in cT1-T2N0M0 stage | ||||
| Older age (>45 years), contralateral thyroid nodules, personal history of radiation therapy to the head and neck, familial DTC history may constitute criteria for a bilateral procedure | ||||
| Molecular Marker (Signaling Pathway) | Type of Thyroid Cancer | Literature |
|---|---|---|
| Indolent thyroid cancers | ||
| Rare BRAF alterations (like K601E mutation) (MAPK signaling) | Mainly in FVPTCs and PTMCs | [75,126] |
| KRAS mutations (MAPK signaling) | FVPTCs with favorable clinical and sonographical profile | [127,128] |
| Aggressive follicular cell derived thyroid cancers | ||
| BRAF V600E mutation (MAPK signaling) | PTCs | [76,77,78,79,80,81,82,84,87,88,89,90] |
| TERT promoter mutations (C228T and C250T) (MAPK signaling, WNT signaling) | According to the frequency, starting from the highest: ATC > PDTC > FTC > PTC; In most PTC cases coexisting with BRAF V600E mutation | [97,98,99,100,101] |
| TP53 inactivating mutations (TP53 signaling) | Mainly in ATCs and PDTCs; also present in a small fraction of aggressive PTCs and FTCs | [52,103] |
| CTNNB1, APC and AXIN1 mutations (WNT signaling) | Mainly in ATC | [104,105,106] |
| PTEN, AKT1 and PIK3CA mutations (PI3K/AKT signaling) | Advanced TCs, mostly in ATCs | [103] |
| miR-146b | Aggressive BRAF-positive PTCs | [110] |
| * RAS mutations (MAPK signaling) | Present in high fraction of ATCs and PDTCs, but also in FTCs | [76] |
| Other follicular cell derived thyroid cancers | ||
| RET/PTC rearrangements (MAPK pathway) | Molecular hallmark of radiation- induced PTCs | [8,19] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rusinek, D.; Chmielik, E.; Krajewska, J.; Jarzab, M.; Oczko-Wojciechowska, M.; Czarniecka, A.; Jarzab, B. Current Advances in Thyroid Cancer Management. Are We Ready for the Epidemic Rise of Diagnoses? Int. J. Mol. Sci. 2017, 18, 1817. https://doi.org/10.3390/ijms18081817
Rusinek D, Chmielik E, Krajewska J, Jarzab M, Oczko-Wojciechowska M, Czarniecka A, Jarzab B. Current Advances in Thyroid Cancer Management. Are We Ready for the Epidemic Rise of Diagnoses? International Journal of Molecular Sciences. 2017; 18(8):1817. https://doi.org/10.3390/ijms18081817
Chicago/Turabian StyleRusinek, Dagmara, Ewa Chmielik, Jolanta Krajewska, Michal Jarzab, Malgorzata Oczko-Wojciechowska, Agnieszka Czarniecka, and Barbara Jarzab. 2017. "Current Advances in Thyroid Cancer Management. Are We Ready for the Epidemic Rise of Diagnoses?" International Journal of Molecular Sciences 18, no. 8: 1817. https://doi.org/10.3390/ijms18081817
APA StyleRusinek, D., Chmielik, E., Krajewska, J., Jarzab, M., Oczko-Wojciechowska, M., Czarniecka, A., & Jarzab, B. (2017). Current Advances in Thyroid Cancer Management. Are We Ready for the Epidemic Rise of Diagnoses? International Journal of Molecular Sciences, 18(8), 1817. https://doi.org/10.3390/ijms18081817