Influence of a Virgin Olive Oil versus Butter Plus Cholesterol-Enriched Diet on Testicular Enzymatic Activities in Adult Male Rats
Abstract
1. Introduction
2. Results
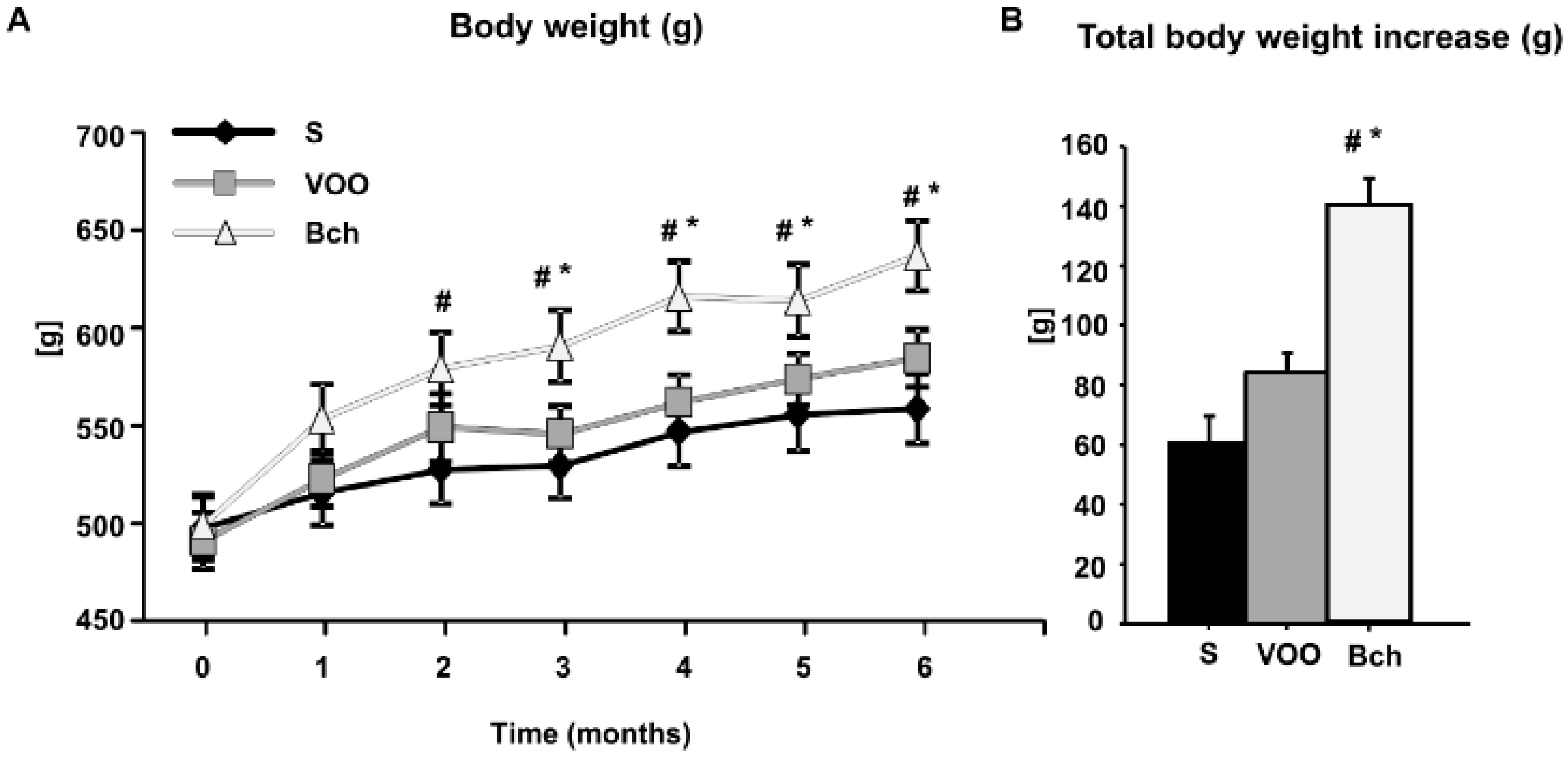
2.1. Body Weight
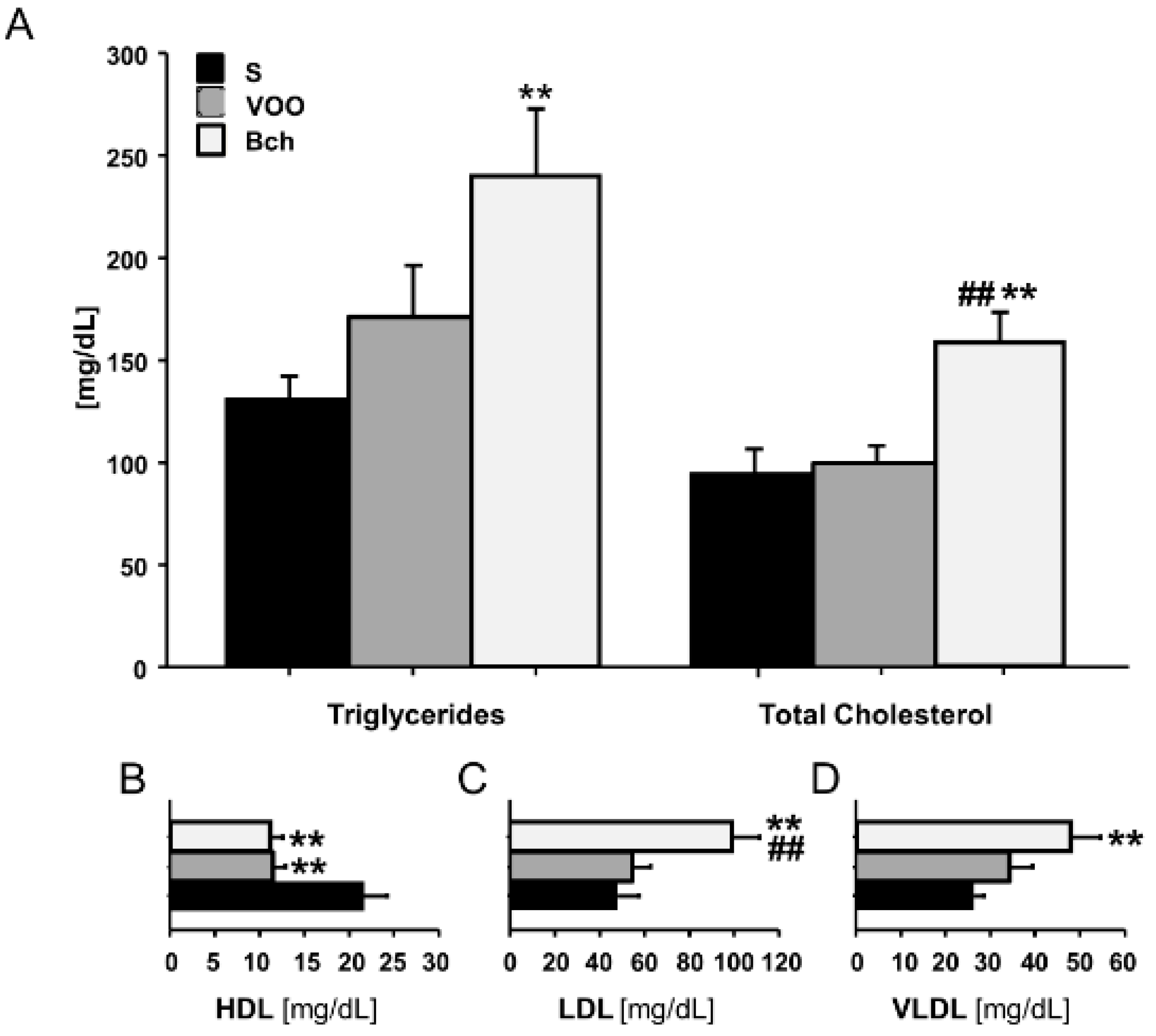
2.2. Lipid Plasma Profile
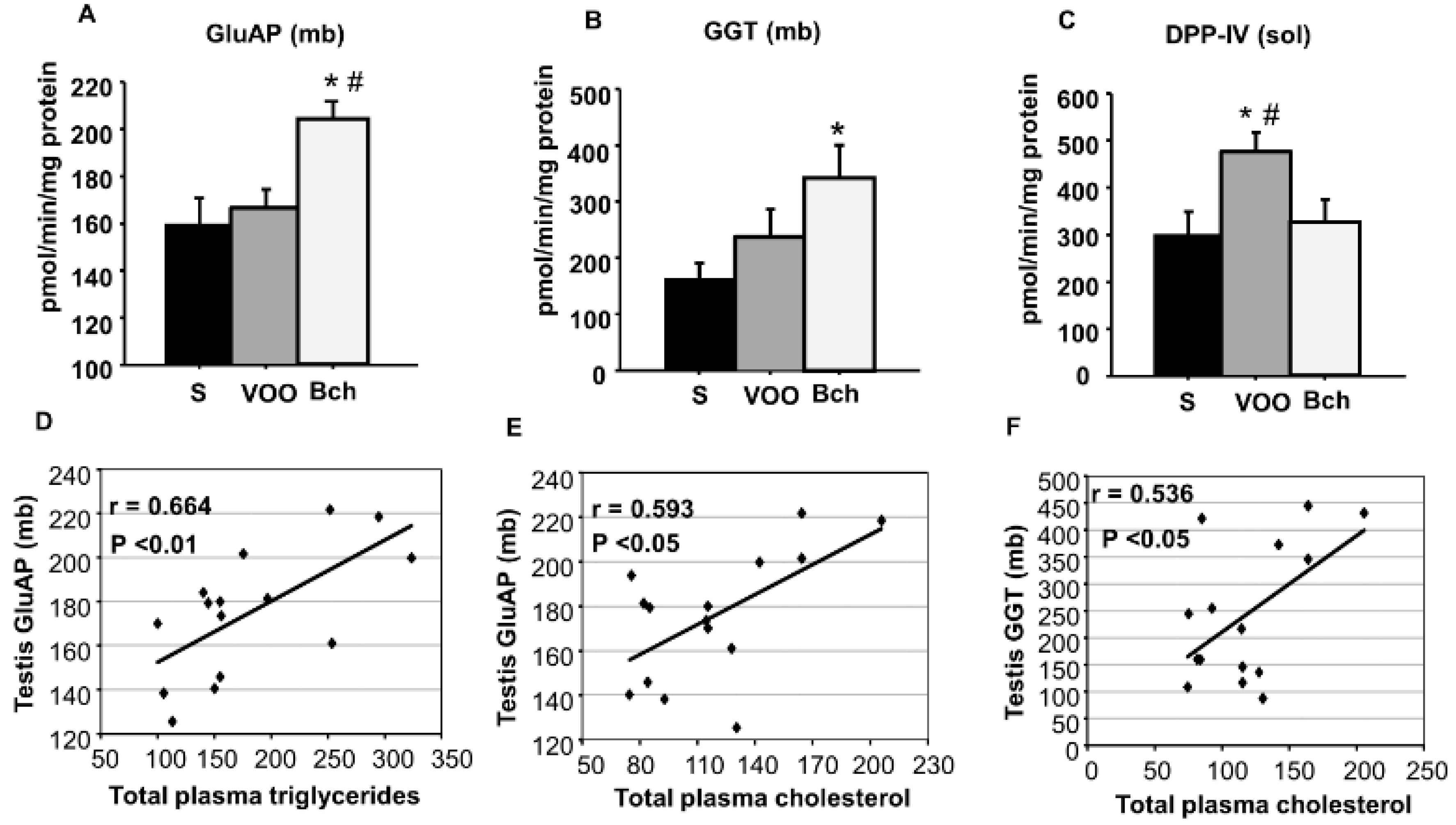
2.3. GluAP, GGT and DPP-IV Activities
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Experimental Design and Diets
4.3. Samples
4.4. Biochemical Assessment
4.4.1. Triglycerides and Cholesterol Assay
4.4.2. Enzymatic Assays
4.4.3. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| DPP-IV | Dipeptidyl Peptidase IV |
| GGT | Gamma Glutamyl Transpeptidase |
| HDL | High Density Lipoprotein |
| LDL | Low Density Lipoprotein |
| VDL | Very Low Density Lipoprotein |
| BTB | Blood Testis Barrier |
| RAS | Renin Angiotensin System |
| AngII | Angiotensin II |
| Bch | Butter plus cholesterol Diet |
| S | Standard Diet |
| VOO | Virgin Olive Oil Diet |
| GluAP | Glutamyl Aminopeptidase |
| Sol | Soluble fraction |
| Mb | Membrane Bound fraction |
| AngIII | Angiotensin III |
| NFE | Nitrogen Free Extract |
| SFA | Saturated Fatty Acids |
| MUFA | Monounsaturated Fatty Acids |
| PUFA | Polyunsaturated Fatty Acids |
| GluNNap | Glutamyl-β-naphthylamine |
| Gly-Pro-NNap | Glycine-Proline-β-naphthylamine |
| γ-GluNNap | γ-Glutamyl-β-naphthylamine |
| BSA | Bovine Serum Albumin |
| DTT | Dithiothreitol |
References
- Shalaby, M.A.; el-Zorba, H.Y.; Kamel, G.M. Effect of alpha-tocopherol and simvastatin on male fertility in hypercholesterolemic rats. Pharmacol. Res. 2004, 50, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Hurtado de Catalfo, G.E.; de Alaniz, M.J.; Marra, C.A. Influence of commercial dietary oils on lipid composition and testosterone production in interstitial cells isolated from rat testis. Lipids 2009, 44, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Padron, R.S.; Mas, J.; Zamora, R.; Riverol, F.; Licea, M.; Mallea, M.; Rodríguez, J. Lipids and testicular function. Int. Urol. Nephrol. 1989, 21, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Campos-Silva, P.; Furriel, A.; Costa, W.S.; Sampaio, F.J.; Gregorio, B.M. Metabolic and testicular effects of the long-term administration of different high-fat diets in adult rats. Int. Braz. J. Urol. 2015, 41, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Reame, V.; Pytlowanciv, E.Z.; Ribeiro, D.L.; Pissolato, T.F.; Taboga, S.R.; Góes, R.M.; Pinto-Fochi, M.E. Obesogenic environment by excess of dietary fats in different phases of development reduces spermatic efficiency of wistar rats at adulthood: Correlations with metabolic status. Biol. Reprod. 2014, 91, 151. [Google Scholar] [CrossRef] [PubMed]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Mah, P.M.; Wittert, G.A. Obesity and testicular function. Mol. Cell. Endocrinol. 2010, 316, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.H.; Ghribi, O.; Hui, L.; Geiger, J.D.; Chen, X. Cholesterol-enriched diet disrupts the blood-testis barrier in rabbits. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E1125–E1130. [Google Scholar] [CrossRef] [PubMed]
- Prieto, I.; Arechaga, G.; Ramírez-Expósito, M.J.; de Gasparo, M.; Martínez-Martos, J.M.; Ramírez, M. Aminopeptidases in the gonads of male and female rats. Fertil. Steril. 2002, 77, 802–804. [Google Scholar] [CrossRef]
- Segarra, A.B.; Prieto, I.; Villarejo, A.B.; Banegas, I.; Wangensteen, R.; de Gasparo, M.; Vives, F.; Ramírez-Sánchez, M. Effects of antihypertensive drugs on angiotensinase activities in the testis of spontaneously hypertensive rats. Horm. Metab. Res. 2013, 45, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Alves-Pereira, J.L.; Frantz, E.D.; da Fonte Ramos, C. Beneficial effects of renin-angiotensin system blockers on testicular steroidogenesis. J. Urol. 2014, 192, 1878–1883. [Google Scholar] [CrossRef] [PubMed]
- Arechaga, G.; Prieto, I.; Segarra, A.B.; Alba, F.; Ruiz-Larrea, M.B.; Ruiz-Sanz, J.I.; de Gasparo, M.; Ramirez, M. Dietary fatty acid composition affects aminopeptidase activities in the testes of mice. Int. J. Androl. 2002, 25, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Arechaga, G.; Martínez, J.M.; Prieto, I.; Ramírez, M.J.; Sánchez, M.J.; Alba, F.; De Gasparo, M.; Ramírez, M. Serum aminopeptidase A activity of mice is related to dietary fat saturation. J. Nutr. 2001, 131, 1177–1179. [Google Scholar] [PubMed]
- Martinez, J.M.; Ramirez, M.J.; Prieto, I.; Alba, F.; Ramirez, M. Sex differences and in vitro effects of steroids on serum aminopeptidase activities. Peptides 1998, 19, 1637–1640. [Google Scholar] [CrossRef]
- Ramírez-Expósito, M.J.; Martínez-Martos, J.M.; Prieto, I.; Alba, F.; Ramírez, M. Angiotensinase activity in mice fed an olive oil-supplemented diet. Peptides 2001, 22, 945–952. [Google Scholar] [CrossRef]
- Kumar, T.R.; Wiseman, A.L.; Kala, G.; Kala, S.V.; Matzuk, M.M.; Lieberman, M.W. Reproductive defects in gamma-glutamyl transpeptidase-deficient mice. Endocrinology 2000, 141, 4270–4277. [Google Scholar] [CrossRef] [PubMed]
- Dubois, V.; Van Ginneken, C.; De Cock, H.; Lambeir, A.M.; Van der Veken, P.; Augustyns, K.; Chen, X.; Scharpé, S.; De Meester, I. Enzyme activity and immunohistochemical localization of dipeptidyl peptidase 8 and 9 in male reproductive tissues. J. Histochem. Cytochem. 2009, 57, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, T.M.A.; Carrera, A.; Zambrana, M. High incidence of hyperestrogenemia and dyslipidemia in a group of infertile men. Gynecol. Obstet. Mex. 2000, 68, 224–229. [Google Scholar]
- Saez Lancellotti, T.E.; Boarelli, P.V.; Monclus, M.A.; Cabrillana, M.E.; Clementi, M.A.; Espínola, L.S.; Cid Barría, J.L.; Vincenti, A.E.; Santi, A.G.; Fornés, M.W. Hypercholesterolemia impaired sperm functionality in rabbits. PLoS ONE 2010, 5, e13457. [Google Scholar] [CrossRef] [PubMed]
- Oi-Kano, Y.; Kawada, T.; Watanabe, T.; Koyama, F.; Watanabe, K.; Senbongi, R.; Iwai, K. Oleuropein supplementation increases urinary noradrenaline and testicular testosterone levels and decreases plasma corticosterone level in rats fed high-protein diet. J. Nutr. Biochem. 2013, 24, 887–893. [Google Scholar] [CrossRef] [PubMed]
- Saez Lancellotti, T.E.; Boarelli, P.V.; Romero, A.A.; Funes, A.K.; Cid-Barria, M.; Cabrillana, M.E.; Monclus, M.A.; Simón, L.; Vicenti, A.E.; Fornés, M.W. Semen quality and sperm function loss by hypercholesterolemic diet was recovered by addition of olive oil to diet in rabbit. PLoS ONE 2013, 8, e52386. [Google Scholar] [CrossRef] [PubMed]
- Segarra, A.B.; Ramirez, M.; Banegas, I.; Alba, F.; Vives, F.; Gasparo, M.; Ortega, E.; Ruiz, E.; Prieto, I. Dietary fat influences testosterone; cholesterol; aminopeptidase A.; and blood pressure in male rats. Horm. Metab. Res. 2008, 40, 289–291. [Google Scholar] [CrossRef] [PubMed]
- El-Kholy, T.A.; Al-Abbadi, H.A.; Qahwaji, D.; Al-Ghamdi, A.K.; Shelat, V.G.; Sobhy, H.M.; Abu Hilal, M. Ameliorating effect of olive oil on fertility of male rats fed on genetically modified soya bean. Food Nutr. Res. 2015, 59, 27758. [Google Scholar] [CrossRef] [PubMed]
- Hurtado de Catalfo, G.E.; de Alaniz, M.J.; Marra, C.A. Dietary lipids modify redox homeostasis and steroidogenic status in rat testis. Nutrition 2008, 24, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Zhu, Y.; Chen, X.; Sha, J.; Fan, L.; Chen, Q. Effects of diet-induced hypercholesterolemia on testosterone-regulated protein expression in mice liver. J. Nanosci. Nanotechnol. 2005, 5, 1273–1276. [Google Scholar] [CrossRef] [PubMed]
- Dufau, M.L.; Khanum, A.; Tsai-Morris, C.R. Multistep regulation of leydig cell function. J. Steroid Biochem. 1987, 27, 343–350. [Google Scholar] [CrossRef]
- Khanum, A.; Dufau, M.L. Angiotensin II receptors and inhibitory actions in leydig cells. J. Biol. Chem 1988, 263, 5070–5074. [Google Scholar] [PubMed]
- Banegas, I.; Prieto, I.; Vives, F.; Alba, F.; de Gasparo, M.; Segarra, A.B.; Hermoso, F.; Durán, R.; Ramírez, M. Brain aminopeptidases and hypertension. J. Renin Angiotensin Aldosterone Syst. 2006, 7, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, M.; Prieto, I.; Alba, F.; Vives, F.; Banegas, I.; de Gasparo, M. Role of central and peripheral aminopeptidase activities in the control of blood pressure: A working hypothesis. Heart Fail. Rev. 2008, 13, 339–353. [Google Scholar] [CrossRef] [PubMed]
- Persson, H.; Rehfeld, J.F.; Ericsson, A.; Schalling, M.; Pelto-Huikko, M.; Hökfelt, T. Transient expression of the cholecystokinin gene in male germ cells and accumulation of the peptide in the acrosomal granule: Possible role of cholecystokinin in fertilization. Proc. Natl. Acad. Sci. USA 1989, 86, 6166–6170. [Google Scholar] [CrossRef] [PubMed]
- Hanigan, M.H.; Ricketts, W.A. Extracellular glutathione is a source of cysteine for cells that express gamma-glutamyl transpeptidase. Biochemistry 1993, 32, 6302–6306. [Google Scholar] [CrossRef] [PubMed]
- Meroni, S.B.; Cánepa, D.F.; Pellizzari, E.H.; Schteingart, H.F.; Cigorraga, S.B. Effects of purinergic agonists on aromatase and gamma-glutamyl transpeptidase activities and on transferrin secretion in cultured Sertoli cells. J. Endocrinol. 1998, 157, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, Y.P.; Vanha-Perttula, T. Gamma-glutamyl transpeptidase; glutathione; and L-glutamic acid in the rat epididymis during postnatal development. Biol. Reprod. 1988, 38, 996–1000. [Google Scholar] [CrossRef] [PubMed]
- Kajiyama, H.; Kikkawa, F.; Suzuki, T.; Shibata, K.; Ino, K.; Mizutani, S. Prolonged survival and decreased invasive activity attributable to dipeptidyl peptidase IV overexpression in ovarian carcinoma. Cancer Res. 2002, 62, 2753–2757. [Google Scholar] [PubMed]
- Abbot, C.A.; Yu, D.M.T.; Woollatt, E.; Sutherland, G.R.; McCaughan, G.W.; Gorrell, M.D. Cloning; expression and chromosomal localization of a novel human dipeptidyl peptidase (DPP) IV homolog; DPP8. Eur. J. Biochem. 2000, 267, 6140–6150. [Google Scholar] [CrossRef]
- Dutta, S.; Sengupta, P. Men and mice: Relating their ages. Life Sci. 2016, 152, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, P. The Laboratory Rat: Relating Its Age With Human’s. Int. J. Prev. Med. 2013, 4, 624–630. [Google Scholar] [PubMed]
- Ramírez, M.; Prieto, I.; Banegas, I.; Segarra, A.B.; Alba, F. Neuropeptidases. Methods Mol. Biol. 2011, 789, 287–294. [Google Scholar] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
| Activity | Fraction | S | VOD | Bch | p |
|---|---|---|---|---|---|
| GluAP | sol | 81.31 ± 10.396 | 117.88 ± 12.987 | 90.35 ± 11.804 | 0.108 |
| mb | 159.89 ± 10.956 | 166.61 ± 7.936 | 204.18 ± 7.496 | 0.011 | |
| GGT | sol | 45.25 ± 7.671 | 60.52 ± 8.196 | 49.14 ± 6.447 | 0.361 |
| mb | 163.98 ± 27.150 | 236.83 ± 50.370 | 341.62 ± 59.247 | 0.046 | |
| DPP-IV | sol | 302.29 ± 47.355 | 476.63 ± 41.176 | 326.55 ± 48.995 | 0.043 |
| mb | 425.90 ± 56.707 | 453.29 ± 87.556 | 593.78 ± 90.857 | 0.297 |
| Components | S | VOO | Bch | ||||||
|---|---|---|---|---|---|---|---|---|---|
| g/100 g | KJ/100 g | Total E % | g/100 g | KJ/100 g | Total E % | g/100 g | KJ/100 g | Total E % | |
| NFE | 60 | 1003.2 | 72.1 | 48 | 802.6 | 43.4 | 48 | 802.6 | 43.9 |
| Fiber | 4 | 3 | 3 | ||||||
| Protein | 16.5 | 275.9 | 19.8 | 13 | 217.4 | 11.8 | 14 | 234.1 | 12.8 |
| Minerals (Ash) | 5 | 5 | 5 | ||||||
| Lipids | 3 | 112.9 | 8.1 | 22 | 827.6 | 44.8 | 21 | 790 | 43.3 |
| SFA | 0.75 | 28.22 | 2.0 | 3.74 | 140.7 | 7.6 | 13.02 | 489.8 | 26.8 |
| MUFA | 0.50 | 18.81 | 1.4 | 17.38 | 653.8 | 35.4 | 7.56 | 284.4 | 15.5 |
| PUFA | 1.75 | 65.84 | 4.7 | 0.88 | 33.1 | 1.8 | 0.42 | 15.8 | 0.9 |
| Cholesterol | 1.50 | ||||||||
| Polyphenols | 0.105 | ||||||||
| α-tocopherol | 0.050 | 0.0004 | |||||||
| Moisture | 11 | 9 | 8 | ||||||
| Total E | 1392 | 1848 | 1827 | ||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domínguez-Vías, G.; Segarra, A.B.; Martínez-Cañamero, M.; Ramírez-Sánchez, M.; Prieto, I. Influence of a Virgin Olive Oil versus Butter Plus Cholesterol-Enriched Diet on Testicular Enzymatic Activities in Adult Male Rats. Int. J. Mol. Sci. 2017, 18, 1701. https://doi.org/10.3390/ijms18081701
Domínguez-Vías G, Segarra AB, Martínez-Cañamero M, Ramírez-Sánchez M, Prieto I. Influence of a Virgin Olive Oil versus Butter Plus Cholesterol-Enriched Diet on Testicular Enzymatic Activities in Adult Male Rats. International Journal of Molecular Sciences. 2017; 18(8):1701. https://doi.org/10.3390/ijms18081701
Chicago/Turabian StyleDomínguez-Vías, Germán, Ana Belén Segarra, Magdalena Martínez-Cañamero, Manuel Ramírez-Sánchez, and Isabel Prieto. 2017. "Influence of a Virgin Olive Oil versus Butter Plus Cholesterol-Enriched Diet on Testicular Enzymatic Activities in Adult Male Rats" International Journal of Molecular Sciences 18, no. 8: 1701. https://doi.org/10.3390/ijms18081701
APA StyleDomínguez-Vías, G., Segarra, A. B., Martínez-Cañamero, M., Ramírez-Sánchez, M., & Prieto, I. (2017). Influence of a Virgin Olive Oil versus Butter Plus Cholesterol-Enriched Diet on Testicular Enzymatic Activities in Adult Male Rats. International Journal of Molecular Sciences, 18(8), 1701. https://doi.org/10.3390/ijms18081701