Melatonin as a Novel Interventional Candidate for Fragile X Syndrome with Autism Spectrum Disorder in Humans
Abstract
1. Introduction
2. Autism Spectrum Disorder (ASD)
2.1. Classification of ASD
2.2. Causes of ASD
2.2.1. Genetic Risk Factors
2.2.2. Environmental Conditions: Pre-, Peri-, and Neonatal Risk Factors of ASD
3. Fragile X Syndrome (FXS)
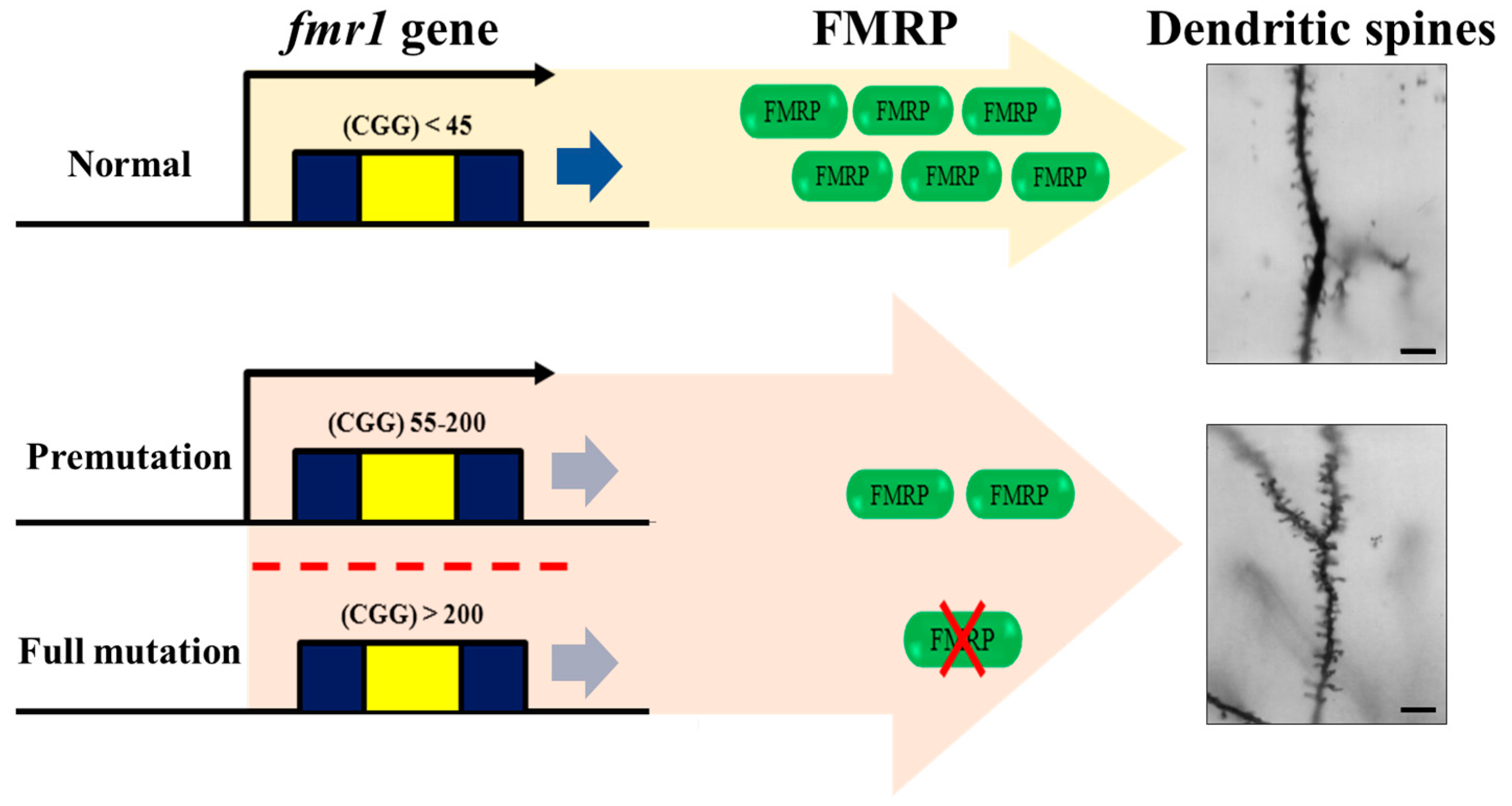
3.1. Mechanism Underlying the Incidence of FXS
3.2. Sleep Problems in Individuals with FXS with ASD
3.3. Correlation between FXS with ASD and Circadian Rhythms
3.4. Neurodevelopmental Abnormalities in FXS with ASD
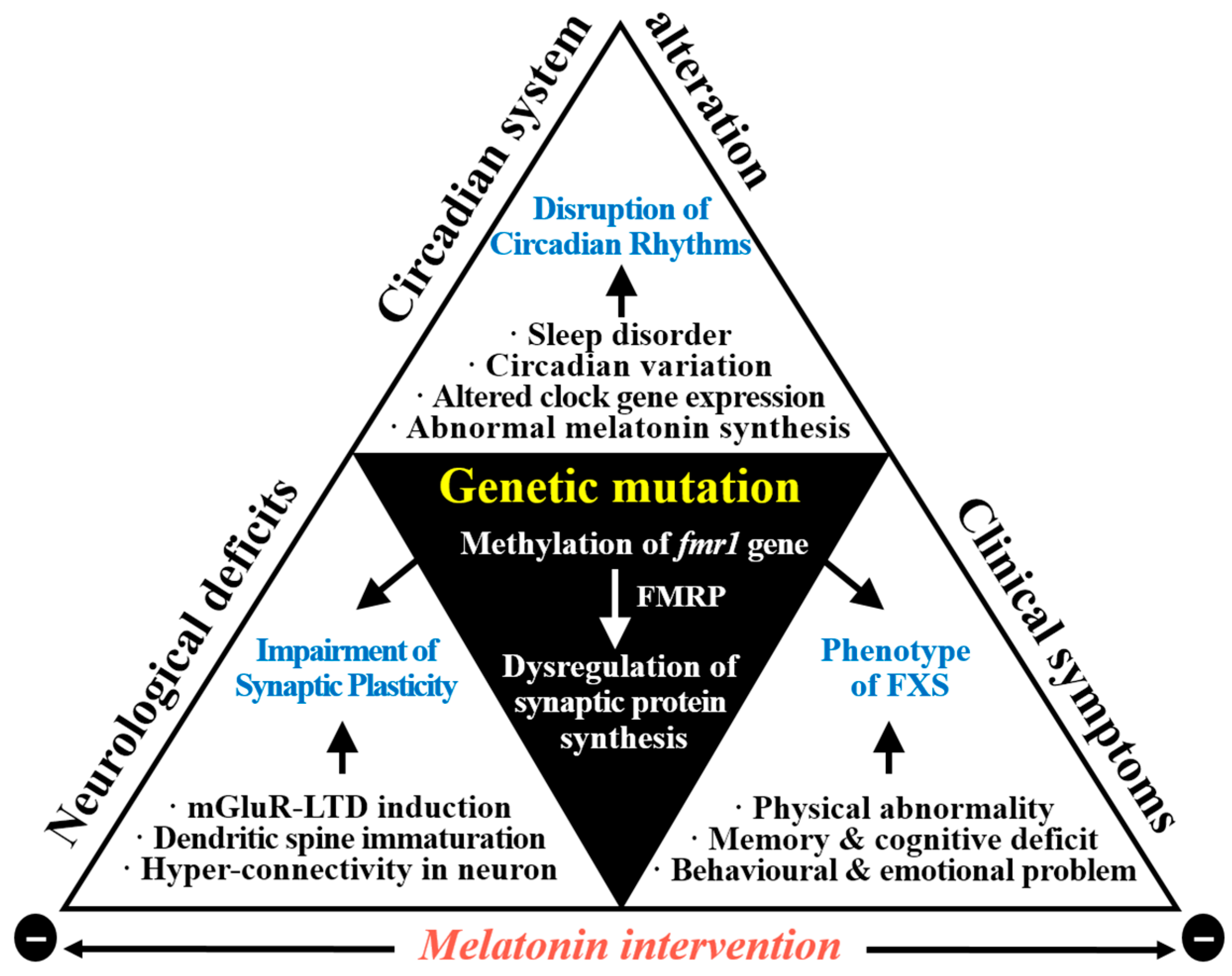
4. Melatonin in FXS with ASD
4.1. Melatonin Signaling Pathways under Normal Condition
4.2. Dysregulation of the Melatonin Pathway in FXS with ASD
4.3. Correlation between Melatonin with Neurodevelopmental Abnormalities in FXS with ASD
5. Melatonin as an Interventional Therapeutic Approach for FXS with ASD: Clinical Assessments
5.1. Effect of Melatonin as a Treatment for Sleep Disorder in FXS with ASD
5.2. Effects of Melatonin on Cognitive and Learning Disabilities in FXS with ASD
5.3. Neuroprotective Effects of Melatonin on Seizures in FXS with ASD
5.4. Synergistic Effects of Melatonin on Synaptic Plasticity in FXS with ASD
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Willemsen, R.; Oostra, B.A.; Bassell, G.J.; Dictenberg, J. The fragile X syndrome: From molecular genetics to neurobiology. Ment. Retard. Dev. Disabil. Res. Rev. 2004, 10, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, L.M.; Phillips, M.A.; Forster-Gibson, C.J.; Beckett, J.; Partington, M.W.; Simpson, N.E.; Holden, J.J.; White, B.N. Genetic mapping of DNA segments relative to the locus for the fragile-X syndrome at Xq27.3. Am. J. Hum. Genet. 1985, 37, 463–472. [Google Scholar] [PubMed]
- Brown, T.C.; Tarleton, J.C.; Go, R.C.; Longshore, J.W.; Descartes, M. Instability of the FMR2 trinucleotide repeat region associated with expanded FMR1 alleles. Am. J. Med. Genet. 1997, 73, 447–455. [Google Scholar] [CrossRef]
- Gecz, J. The FMR2 gene, FRAXE and non-specific X-linked mental retardation: Clinical and molecular aspects. Ann. Hum. Genet. 2000, 64, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Smalley, S.L. Genetic influences in childhood-onset psychiatric disorders: Autism and attention-deficit/hyperactivity disorder. Am. J. Hum. Genet. 1997, 60, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- McLennan, Y.; Polussa, J.; Tassone, F.; Hagerman, R. Fragile X syndrome. Curr. Genom. 2011, 12, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Musumeci, S.A.; Ferri, R.; Elia, M.; Colognola, R.M.; Bergonzi, P.; Tassinari, C.A. Epilepsy and fragile X syndrome: A follow-up study. Am. J. Med. Genet. 1991, 38, 511–513. [Google Scholar] [CrossRef] [PubMed]
- Gould, E.L.; Loesch, D.Z.; Martin, M.J.; Hagerman, R.J.; Armstrong, S.M.; Huggins, R.M. Melatonin profiles and sleep characteristics in boys with fragile X syndrome: A preliminary study. Am. J. Med. Genet. 2000, 95, 307–315. [Google Scholar] [CrossRef]
- Zhang, J.; Fang, Z.; Jud, C.; Vansteensel, M.J.; Kaasik, K.; Lee, C.C.; Albrecht, U.; Tamanini, F.; Meijer, J.H.; Oostra, B.A.; et al. Fragile X-related proteins regulate mammalian circadian behavioral rhythms. Am. J. Hum. Genet. 2008, 83, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Poidevin, M.; Han, E.; Bi, J.; Jin, P. Circadian rhythm-dependent alterations of gene expression in Drosophila brain lacking fragile X mental retardation protein. PLoS ONE 2012, 7, e37937. [Google Scholar] [CrossRef] [PubMed]
- Richdale, A.L. Sleep problems in autism: Prevalence, cause, and intervention. Dev. Med. Child Neurol. 1999, 41, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Wiggs, L.; Stores, G. Sleep patterns and sleep disorders in children with autistic spectrum disorders: Insights using parent report and actigraphy. Dev. Med. Child Neurol. 2004, 46, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Hodge, D.; Carollo, T.M.; Lewin, M.; Hoffman, C.D.; Sweeney, D.P. Sleep patterns in children with and without autism spectrum disorders: Developmental comparisons. Res. Dev. Disabil. 2014, 35, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Mazurek, M.O.; Sohl, K. Sleep and behavioral problems in children with autism spectrum disorder. J. Autism Dev. Disord. 2016, 46, 1906–1915. [Google Scholar] [CrossRef] [PubMed]
- Wiggs, L.; Stores, G. Severe sleep disturbance and daytime challenging behaviour in children with severe learning disabilities. J. Intellect. Disabil. Res. 1996, 40, 518–528. [Google Scholar] [CrossRef] [PubMed]
- Malow, B.A.; Marzec, M.L.; McGrew, S.G.; Wang, L.; Henderson, L.M.; Stone, W.L. Characterizing sleep in children with autism spectrum disorders: A multidimensional approach. Sleep 2006, 29, 1563–1571. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, D.A.; Frye, R.E. Melatonin in autism spectrum disorders. Curr. Clin. Pharmacol. 2014, 9, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Melke, J.; Goubran Botros, H.; Chaste, P.; Betancur, C.; Nygren, G.; Anckarsäter, H.; Rastam, M.; Ståhlberg, O.; Gillberg, I.C.; Delorme, R.; et al. Abnormal melatonin synthesis in autism spectrum disorders. Mol. Psychiatry 2008, 13, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Kulman, G.; Lissoni, P.; Rovelli, F.; Roselli, M.G.; Brivio, F.; Sequeri, P. Evidence of pineal endocrine hypofunction in autistic children. Neuro Endocrinol. Lett. 2000, 21, 31–34. [Google Scholar] [PubMed]
- O’Hare, J.P.; O’Brien, I.A.; Arendt, J.; Astley, P.; Ratcliffe, W.; Andrews, H.; Walters, R.; Corrall, R.J. Does melatonin deficiency cause the enlarged genitalia of the fragile-X syndrome? Clin. Endocrinol. 1986, 24, 327–333. [Google Scholar] [CrossRef]
- Reiter, R.J.; Calvo, J.R.; Karbownik, M.; Qi, W.; Tan, D.X. Melatonin and its relation to the immune system and inflammation. Ann. N. Y. Acad. Sci. 2000, 917, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Foulkes, N.S.; Whitmore, D.; Sassone-Corsi, P. Rhythmic transcription: The molecular basis of circadian melatonin synthesis. Biol. Cell 1997, 89, 487–494. [Google Scholar] [CrossRef]
- Reiter, R.J.; Maestroni, G.J. Melatonin in relation to the antioxidative defense and immune systems: Possible implications for cell and organ transplantation. J. Mol. Med. 1999, 77, 36–39. [Google Scholar] [CrossRef] [PubMed]
- El-Sherif, Y.; Tesoriero, J.; Hogan, M.V.; Wieraszko, A. Melatonin regulates neuronal plasticity in the hippocampus. J. Neurosci. Res. 2003, 72, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Boutin, J.A.; Audinot, V.; Ferry, G.; Delagrange, P. Molecular tools to study melatonin pathways and actions. Trends Pharmacol. Sci. 2005, 26, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Pilar Terron, M.; Flores, L.J.; Koppisepi, S. Medical implications of melatonin: Receptor-mediated and receptor-independent actions. Adv. Med. Sci. 2007, 52, 11–28. [Google Scholar] [PubMed]
- Gupta, Y.K.; Gupta, M.; Kohli, K. Neuroprotective role of melatonin in oxidative stress vulnerable brain. Indian J. Physiol. Pharmacol. 2003, 47, 373–386. [Google Scholar] [PubMed]
- Kotagal, S.; Broomall, E. Sleep in children with autism spectrum disorder. Pediatr. Neurol. 2012, 47, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Kostoglou-Athanassiou, I. Therapeutic applications of melatonin. Ther. Adv. Endocrinol. Metab. 2013, 4, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, D.A.; Frye, R.E. Evidence linking oxidative stress, mitochondrial dysfunction, and inflammation in the brain of individuals with autism. Front. Physiol. 2014, 5, 150. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Rossignol, D.A. Mitochondrial dysfunction can connect the diverse medical symptoms associated with autism spectrum disorders. Pediatr. Res. 2011, 69, 41R–47R. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, A.; Audhya, T.; Chauhan, V. Brain region-specific glutathione redox imbalance in autism. Neurochem. Res. 2012, 37, 1681–1689. [Google Scholar] [CrossRef] [PubMed]
- Rose, S.; Melnyk, S.; Pavliv, O.; Bai, S.; Nick, T.G.; Frye, R.E.; James, S.J. Evidence of oxidative damage and inflammation associated with low glutathione redox status in the autism brain. Transl. Psychiatry 2012, 2, e134. [Google Scholar] [CrossRef] [PubMed]
- Romero-Zerbo, Y.; Decara, J.; el Bekay, R.; Sanchez-Salido, L.; del Arco-Herrera, I.; de Fonseca, F.R.; de Diego-Otero, Y. Protective effects of melatonin against oxidative stress in FMR1 knockout mice: A therapeutic research model for the fragile X syndrome. J. Pineal Res. 2009, 46, 224–234. [Google Scholar] [CrossRef] [PubMed]
- El Bekay, R.; Romero-Zerbo, Y.; Decara, J.; Sanchez-Salido, L.; del Arco-Herrera, I.; Rodríguez-de Fonseca, F.; de Diego-Otero, Y. Enhanced markers of oxidative stress, altered antioxidants and NADPH-oxidase activation in brains from Fragile X mental retardation 1-deficient mice, a pathological model for Fragile X syndrome. Eur. J. Neurosci. 2007, 26, 3169–3180. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P. Radical-free biology of oxidative stress. Am. J. Physiol. Cell Physiol. 2008, 295, C849–C868. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Role of free radicals in the neurodegenerative diseases: Therapeutic implications for antioxidant treatment. Drugs Aging 2001, 18, 685–716. [Google Scholar] [CrossRef] [PubMed]
- Circu, M.L.; Aw, T.Y. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic. Biol. Med. 2010, 48, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Avery, S.V. Molecular targets of oxidative stress. Biochem. J. 2011, 434, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Barnham, K.J.; Masters, C.L.; Bush, A.I. Neurodegenerative diseases and oxidative stress. Nat. Rev. Drug Discov. 2004, 3, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Emerit, J.; Edeas, M.; Bricaire, F. Neurodegenerative diseases and oxidative stress. Biomed. Pharmacother. 2004, 58, 39–46. [Google Scholar] [CrossRef] [PubMed]
- McGinnis, W.R. Oxidative stress in autism. Altern. Ther. Health Med. 2004, 10, 22–36. [Google Scholar] [PubMed]
- Reiter, R.J.; Tan, D.X.; Qi, W.; Manchester, L.C.; Karbownik, M.; Calvo, J.R. Pharmacology and physiology of melatonin in the reduction of oxidative stress in vivo. Biol. Signals Recept. 2000, 9, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Reiter, R.J.; Manchester, L.C.; Yan, M.T.; El-Sawi, M.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Allegra, M.; Hardeland, R.; et al. Chemical and physical properties and potential mechanisms: Melatonin as a broad spectrum antioxidant and free radical scavenger. Curr. Top. Med. Chem. 2002, 2, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.; Pang, S.F.; Cheung, R.T. Administration of melatonin after onset of ischemia reduces the volume of cerebral infarction in a rat middle cerebral artery occlusion stroke model. Stroke 2003, 34, 770–775. [Google Scholar] [CrossRef] [PubMed]
- Sinha, K.; Degaonkar, M.N.; Jagannathan, N.R.; Gupta, Y.K. Effect of melatonin on ischemia reperfusion injury induced by middle cerebral artery occlusion in rats. Eur. J. Pharmacol. 2001, 428, 185–192. [Google Scholar] [CrossRef]
- Lee, E.J.; Lee, M.Y.; Chen, H.Y.; Hsu, Y.S.; Wu, T.S.; Chen, S.T.; Chang, G.L. Melatonin attenuates gray and white matter damage in a mouse model of transient focal cerebral ischemia. J. Pineal Res. 2005, 38, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Deng, P.; Xu, Z.C.; Chen, J. Moderate traumatic brain injury causes acute dendritic and synaptic degeneration in the hippocampal dentate gyrus. PLoS ONE 2011, 6, e24566. [Google Scholar] [CrossRef] [PubMed]
- Juan, W.S.; Huang, S.Y.; Chang, C.C.; Hung, Y.C.; Lin, Y.W.; Chen, T.Y.; Lee, A.H.; Lee, A.C.; Wu, T.S.; Lee, E.J.; et al. Melatonin improves neuroplasticity by upregulating the growth-associated protein-43 (GAP-43) and NMDAR postsynaptic density-95 (PSD-95) proteins in cultured neurons exposed to glutamate excitotoxicity and in rats subjected to transient focal cerebral ischemia even during a long-term recovery period. J. Pineal Res. 2014, 56, 213–223. [Google Scholar] [PubMed]
- Argüelles, S.; Munoz, M.F.; Cano, M.; Machado, A.; Ayala, A. In vitro and in vivo protection by melatonin against the decline of elongation factor-2 caused by lipid peroxidation: Preservation of protein synthesis. J. Pineal Res. 2012, 53, 1–10. [Google Scholar] [CrossRef] [PubMed]
- McPartland, J.C.; Reichow, B.; Volkmar, F.R. Sensitivity and specificity of proposed DSM-5 diagnostic criteria for autism spectrum disorder. J. Am. Acad. Child Adolesc. Psychiatry 2012, 51, 368–383. [Google Scholar] [CrossRef] [PubMed]
- Fryns, J.P.; Kleczkowska, A.; Kubień, E.; van den Berghe, H. Cytogenetic findings in moderate and severe mental retardation. A study of an institutionalized population of 1991 patients. Acta Paediatr. 1984, 313, 1–23. [Google Scholar] [CrossRef]
- Verkerk, A.J.; Pieretti, M.; Sutcliffe, J.S.; Fu, Y.H.; Kuhl, D.P.; Pizzuti, A.; Reiner, O.; Richards, S.; Victoria, M.F.; Zhang, F.P.; et al. Identification of a gene (FMR1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell 1991, 65, 905–914. [Google Scholar] [CrossRef]
- Macpherson, J.N.; Murray, A. Development of genetic testing for fragile X syndrome and associated disorders, and estimates of the prevalence of FMR1 expansion mutations. Genes 2016, 7, 110. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, R.J.; Berry-Kravis, E.; Kaufmann, W.E.; Ono, M.Y.; Tartaglia, N.; Lachiewicz, A.; Kronk, R.; Delahunty, C.; Hessl, D.; Visootsak, J.; et al. Advances in the treatment of fragile X syndrome. Pediatrics 2009, 123, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Comery, T.A.; Harris, J.B.; Willems, P.J.; Oostra, B.A.; Irwin, S.A.; Weiler, I.J.; Greenough, W.T. Abnormal dendritic spines in fragile X knockout mice: Maturation and pruning deficits. Proc. Natl. Acad. Sci. USA 1997, 94, 5401–5404. [Google Scholar] [CrossRef] [PubMed]
- Bakker, C.E.; Oostra, B.A. Understanding fragile X syndrome: Insights from animal models. Cytogenet. Genome Res. 2003, 100, 111–123. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Martín, A.; Crespo, M.; Portera-Cailliau, C. Delayed stabilization of dendritic spines in fragile X mice. J. Neurosci. 2010, 30, 7793–7803. [Google Scholar] [CrossRef] [PubMed]
- Irwin, S.A.; Patel, B.; Idupulapati, M.; Harris, J.B.; Crisostomo, R.A.; Larsen, B.P.; Kooy, F.; Willems, P.J.; Cras, P.; Kozlowski, P.B.; et al. Abnormal dendritic spine characteristics in the temporal and visual cortices of patients with fragile-X syndrome: A quantitative examination. Am. J. Med. Genet. 2001, 98, 161–167. [Google Scholar] [CrossRef]
- Cortesi, F.; Giannotti, F.; Ivanenko, A.; Johnson, K. Sleep in children with autistic spectrum disorder. Sleep Med. 2010, 11, 659–664. [Google Scholar] [CrossRef] [PubMed]
- DeVincent, C.J.; Gadow, K.D.; Delosh, D.; Geller, L. Sleep disturbance and its relation to DSM-IV psychiatric symptoms in preschool-age children with pervasive developmental disorder and community controls. J. Child Neurol. 2007, 22, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Budimirovic, D.B.; Kaufmann, W.E. What can we learn about autism from studying fragile X syndrome? Dev. Neurosci. 2011, 33, 379–394. [Google Scholar] [CrossRef] [PubMed]
- Wirojanan, J.; Jacquemont, S.; Diaz, R.; Bacalman, S.; Anders, T.F.; Hagerman, R.J.; Goodlin-Jones, B.L. The efficacy of melatonin for sleep problems in children with autism, fragile X syndrome, or autism and fragile X syndrome. J. Clin. Sleep Med. 2009, 5, 145–150. [Google Scholar] [PubMed]
- Lumaban, J.G.; Nelson, D.L. The fragile X proteins FMRP and FXR2P cooperate to regulate glucose metabolism in mice. Hum. Mol. Genet. 2015, 24, 2175–2184. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.F.; Soares, C.; Béïque, J.C. Examining form and function of dendritic spines. Neural Plast. 2012, 2012, 704103. [Google Scholar] [CrossRef] [PubMed]
- Von Bohlen Und Halbach, O. Dendritic spine abnormalities in mental retardation. Cell Tissue Res. 2010, 342, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Purpura, D.P. Dendritic spine “dysgenesis” and mental retardation. Science 1974, 186, 1126–1128. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, D.R.; Torrey, E.F.; Neophytides, A.N.; Wyatt, R.J. Structural abnormalities in the cerebral cortex of chronic schizophrenic patients. Arch. Gen. Psychiatry 1979, 36, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Irwin, S.A.; Idupulapati, M.; Gilbert, M.E.; Harris, J.B.; Chakravarti, A.B.; Rogers, E.J.; Crisostomo, R.A.; Larsen, B.P.; Mehta, A.; Alcantara, C.J.; et al. Dendritic spine and dendritic field characteristics of layer V pyramidal neurons in the visual cortex of fragile-X knockout mice. Am. J. Med. Genet. 2002, 111, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Antar, L.N.; Li, C.; Zhang, H.; Carroll, R.C.; Bassell, G.J. Local functions for FMRP in axon growth cone motility and activity-dependent regulation of filopodia and spine synapses. Mol. Cell. Neurosci. 2006, 32, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, K.; Stehle, J.H. Melatonin synthesis in the human pineal gland: Advantages, implications, and difficulties. Chronobiol. Int. 2006, 23, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.J.; McIntosh, D.N.; McGrath, J.; Shyu, V.; Lampe, M.; Taylor, A.K.; Tassone, F.; Neitzel, K.; Stackhouse, T.; Hagerman, R.J. Electrodermal responses to sensory stimuli in individuals with fragile X syndrome: A preliminary report. Am. J. Med. Genet. 1999, 83, 268–279. [Google Scholar] [CrossRef]
- Veatch, O.J.; Goldman, S.E.; Adkins, K.W.; Malow, B.A. Melatonin in children with autism spectrum disorders: How does the evidence fit together? J. Nat. Sci. 2015, 1, e125. [Google Scholar] [PubMed]
- Pagan, C.; Delorme, R.; Callebert, J.; Goubran-Botros, H.; Amsellem, F.; Drouot, X.; Boudebesse, C.; Le Dudal, K.; Ngo-Nguyen, N.; Laouamri, H.; et al. The serotonin-N-acetylserotonin-melatonin pathway as a biomarker for autism spectrum disorders. Transl. Psychiatry 2014, 4, e479. [Google Scholar] [CrossRef] [PubMed]
- Hou, L.; Antion, M.D.; Hu, D.; Spencer, C.M.; Paylor, R.; Klann, E. Dynamic translational and proteasomal regulation of fragile X mental retardation protein controls mGluR-dependent long-term depression. Neuron 2006, 51, 441–454. [Google Scholar] [CrossRef] [PubMed]
- Bear, M.F.; Huber, K.M.; Warren, S.T. The mGluR theory of fragile X mental retardation. Trends Neurosci. 2004, 27, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Jacquemont, S.; Hagerman, R.J.; Hagerman, P.J.; Leehey, M.A. Fragile-X syndrome and fragile X-associated tremor/ataxia syndrome: Two faces of FMR1. Lancet Neurol. 2007, 6, 45–55. [Google Scholar] [CrossRef]
- Dölen, G.; Osterweil, E.; Rao, B.S.; Smith, G.B.; Auerbach, B.D.; Chattarji, S.; Bear, M.F. Correction of fragile X syndrome in mice. Neuron 2007, 56, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Johnston, M.V.; Ishida, A.; Ishida, W.N.; Matsushita, H.B.; Nishimura, A.; Tsuji, M. Plasticity and injury in the developing brain. Brain Dev. 2009, 31, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Shan, G.; Guo, W.; Smrt, R.D.; Johnson, E.B.; Li, X.; Pfeiffer, R.L.; Szulwach, K.E.; Duan, R.; Barkho, B.Z.; et al. Fragile X mental retardation protein regulates proliferation and differentiation of adult neural stem/progenitor cells. PLoS Genet. 2010, 6, e1000898. [Google Scholar] [CrossRef] [PubMed]
- Ceman, S.; O’Donnell, W.T.; Reed, M.; Patton, S.; Pohl, J.; Warren, S.T. Phosphorylation influences the translation state of FMRP-associated polyribosomes. Hum. Mol. Genet. 2003, 12, 3295–3305. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, M.; Nalavadi, V.; Epstein, M.P.; Narayanan, U.; Bassell, G.J.; Warren, S.T. Phosphorylation al. Fragile X mental retardation protein deficiency leads to excessive mGluR5-dependent internalization of AMPA receptors. Proc. Natl. Acad. Sci. USA 2007, 104, 15537–15542. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Yatsushiro, S.; Ishio, S.; Hayashi, M.; Nishi, T.; Yamamoto, A.; Futai, M.; Yamaguchi, A.; Moriyama, Y. Metabotropic glutamate receptors negatively regulate melatonin synthesis in rat pinealocytes. J. Neurosci. 1998, 18, 2056–2062. [Google Scholar] [PubMed]
- Bonsi, P.; Cuomo, D.; de Persis, C.; Centonze, D.; Bernardi, G.; Calabresi, P.; Pisani, A. Modulatory action of metabotropic glutamate receptor (mGluR) 5 on mGluR1 function in striatal cholinergic interneurons. Neuropharmacology 2005, 1, 104–113. [Google Scholar] [CrossRef] [PubMed]
- De Blasi, A.; Conn, P.J.; Pin, J.; Nicoletti, F. Molecular determinants of metabotropic glutamate receptor signaling. Trends Pharmacol. Sci. 2001, 22, 114–120. [Google Scholar] [CrossRef]
- Praninskiene, R.; Dumalakiene, I.; Kemezys, R.; Mauricas, M.; Jucaite, A. Melatonin secretion in children with epilepsy. Epilepsy Behav. 2012, 25, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Byrnes, K.R.; Loane, D.J.; Faden, A.I. Metabotropic glutamate receptors as targets for multipotential treatment of neurological disorders. Neurotherapeutics 2009, 6, 94–107. [Google Scholar] [CrossRef] [PubMed]
- Yatsushiro, S.; Yamada, H.; Hayashi, M.; Tsuboi, S.; Moriyama, Y. Functional expression of metabotropic glutamate receptor type 5 in rat pinealocytes. Neuroreport 1999, 10, 1599–1603. [Google Scholar] [CrossRef] [PubMed]
- Deschenes, C.L.; McCurry, S.M. Current treatments for sleep disturbances in individuals with dementia. Curr. Psychiatry Rep. 2009, 11, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Galli-Carminati, G.; Deriaz, N.; Bertschy, G. Melatonin in treatment of chronic sleep disorders in adults with autism: A retrospective study. Swiss Med. Wkly. 2009, 139, 293–296. [Google Scholar] [PubMed]
- Hu, V.W.; Sarachana, T.; Kim, K.S.; Nguyen, A.; Kulkarni, S.; Steinberg, M.E.; Luu, T.; Lai, Y.; Lee, N.H. Gene expression profiling differentiates autism case-controls and phenotypic variants of autism spectrum disorders: Evidence for circadian rhythm dysfunction in severe autism. Autism Res. 2009, 2, 78–97. [Google Scholar] [CrossRef] [PubMed]
- Bassell, G.J.; Warren, S.T. Fragile X syndrome: Loss of local mRNA regulation alters synaptic development and function. Neuron 2008, 60, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Berry-Kravis, E.; Raspa, M.; Loggin-Hester, L.; Bishop, E.; Holiday, D.; Bailey, D.B. Seizures in fragile X syndrome: Characteristics and comorbid diagnoses. Am. J. Intellect. Dev. Disabil. 2010, 115, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.P.; Malow, B.A. Assessment and pharmacologic treatment of sleep disturbance in autism. Child Adolesc. Psychiatr. Clin. N. Am. 2008, 17, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Andersen, I.M.; Kaczmarska, J.; McGrew, S.G.; Malow, B.A. Melatonin for insomnia in children with autism spectrum disorders. J. Child Neurol. 2008, 23, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Leu, R.M.; Beyderman, L.; Botzolakis, E.J.; Surdyka, K.; Wang, L.; Malow, B.A. Relation of melatonin to sleep architecture in children with autism. Autism Dev. Disord. 2011, 41, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Nimchinsky, E.A.; Oberlander, A.M.; Svoboda, K. Abnormal development of dendritic spines in FMR1 knock-out mice. J. Neurosci. 2001, 21, 5139–5146. [Google Scholar] [PubMed]
- Kasai, H.; Fukuda, M.; Watanabe, S.; Hayashi-Takagi, A.; Noguchi, J. Structural dynamics of dendritic spines in memory and cognition. Trends Neurosci. 2010, 33, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Baydas, G.; Ozer, M.; Yasar, A.; Tuzcu, M.; Koz, S.T. Melatonin improves learning and memory performances impaired by hyperhomocysteinemia in rats. Brain Res. 2005, 1046, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Baydas, G.; Ozveren, F.; Akdemir, I.; Tuzcu, M.; Yasar, A. Learning and memory deficits in rats induced by chronic thinner exposure are reversed by melatonin. J. Pineal Res. 2005, 39, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Bourgeron, T. The possible interplay of synaptic and clock genes in autism spectrum disorders. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Bechara, E.G.; Didiot, M.C.; Melko, M.; Davidovic, L.; Bensaid, M.; Martin, P.; Castets, M.; Pognonec, P.; Khandjian, E.W.; Moine, H.; et al. A novel function for fragile X mental retardation protein in translational activation. PLoS Biol. 2009, 7, e16. [Google Scholar] [CrossRef] [PubMed]
- Berry-Kravis, E. Epilepsy in fragile X syndrome. Dev. Med. Child Neurol. 2002, 44, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Bazil, C.W.; Short, D.; Crispin, D.; Zheng, W. Patients with intractable epilepsy have low melatonin, which increases following seizures. Neurology 2000, 55, 1746–1748. [Google Scholar] [CrossRef] [PubMed]
- Tomycz, N.D.; Richardson, R.M.; Friedlander, R.M. Seizures and free radicals: The antioxidant may become the next anticonvulsant. Neurosurgery 2011, 69, N12–N13. [Google Scholar] [CrossRef] [PubMed]
- Beal, M.F. Mitochondrial dysfunction in neurodegenerative diseases. Biochim. Biophys. Acta 1998, 1366, 211–223. [Google Scholar] [CrossRef]
- Peled, N.; Shorer, Z.; Peled, E.; Pillar, G. Melatonin effect on seizures in children with severe neurologic deficit disorders. Epilepsia 2001, 42, 1208–1210. [Google Scholar] [CrossRef] [PubMed]
- Fauteck, J.; Schmidt, H.; Lerchl, A.; Kurlemann, G.; Wittkowski, W. Melatonin in epilepsy: First results of replacement therapy and first clinical results. Biol. Signals Recept. 1999, 8, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Jan, J.E.; Freeman, R.D. Melatonin therapy for circadian rhythm sleep disorders in children with multiple disabilities: What have we learned in the last decade? Dev. Med. Child Neurol. 2004, 46, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Stewart, L.S.; Leung, L.S. Hippocampal melatonin receptors modulate seizure threshold. Epilepsia 2005, 46, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Lee, S.K.; Park, K.; Lee, Y.; Hong, Y.; Lee, S.; Jeon, J.C.; Kim, J.H.; Lee, S.R.; Chang, K.T.; et al. Beneficial effects of endogenous and exogenous melatonin on neural reconstruction and functional recovery in an animal model of spinal cord injury. J. Pineal Res. 2012, 52, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Palaksha, K.J.; Park, K.; Park, S.; Kim, H.D.; Reiter, R.J.; Chang, K.T. Melatonin plus exercise-based neurorehabilitative therapy for spinal cord injury. J. Pineal Res. 2010, 49, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Park, K.; Lee, Y.; Park, S.; Lee, S.; Hong, Y.; Kil Lee, S.; Hong, Y. Synergistic effect of melatonin on exercise-induced neuronal reconstruction and functional recovery in a spinal cord injury animal model. J. Pineal Res. 2010, 48, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Lee, S.; Lee, S.R.; Park, K.; Hong, Y.; Lee, M.; Park, S.; Jin, Y.; Chang, K.T.; Hong, Y. Beneficial effects of melatonin combined with exercise on endogenous neural stem/progenitor cells proliferation after spinal cord injury. Int. J. Mol. Sci. 2014, 15, 2207–2222. [Google Scholar] [CrossRef] [PubMed]
- Jansen, S.L.; Forbes, D.A.; Duncan, V.; Morgan, D.G. Melatonin for cognitive impairment. Cochrane Database Syst. Rev. 2006, 25, CD003802. [Google Scholar]
- Lee, S.; Shin, J.; Hong, Y.; Lee, M.; Kim, K.; Lee, S.R.; Chang, K.T.; Hong, Y. Beneficial effects of melatonin on stroke-induced muscle atrophy in focal cerebral ischemic rats. Lab. Anim. Res. 2012, 28, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Letechipía-Vallejo, G.; González-Burgos, I.; Cervantes, M. Neuroprotective effect of melatonin on brain damage induced by acute global cerebral ischemia in cats. Arch. Med. Res. 2001, 32, 186–192. [Google Scholar] [CrossRef]
- Patel, A.B.; Loerwald, K.W.; Huber, K.M.; Gibson, J.R. Postsynaptic FMRP promotes the pruning of cell-to-cell connections among pyramidal neurons in the L5A neocortical network. J. Neurosci. 2014, 34, 3413–3418. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Won, J.; Jin, Y.; Choi, J.; Park, S.; Lee, T.H.; Lee, S.-R.; Chang, K.-T.; Hong, Y. Melatonin as a Novel Interventional Candidate for Fragile X Syndrome with Autism Spectrum Disorder in Humans. Int. J. Mol. Sci. 2017, 18, 1314. https://doi.org/10.3390/ijms18061314
Won J, Jin Y, Choi J, Park S, Lee TH, Lee S-R, Chang K-T, Hong Y. Melatonin as a Novel Interventional Candidate for Fragile X Syndrome with Autism Spectrum Disorder in Humans. International Journal of Molecular Sciences. 2017; 18(6):1314. https://doi.org/10.3390/ijms18061314
Chicago/Turabian StyleWon, Jinyoung, Yunho Jin, Jeonghyun Choi, Sookyoung Park, Tae Ho Lee, Sang-Rae Lee, Kyu-Tae Chang, and Yonggeun Hong. 2017. "Melatonin as a Novel Interventional Candidate for Fragile X Syndrome with Autism Spectrum Disorder in Humans" International Journal of Molecular Sciences 18, no. 6: 1314. https://doi.org/10.3390/ijms18061314
APA StyleWon, J., Jin, Y., Choi, J., Park, S., Lee, T. H., Lee, S.-R., Chang, K.-T., & Hong, Y. (2017). Melatonin as a Novel Interventional Candidate for Fragile X Syndrome with Autism Spectrum Disorder in Humans. International Journal of Molecular Sciences, 18(6), 1314. https://doi.org/10.3390/ijms18061314