Role of Free Radicals and Biotransformation in Trichloronitrobenzene-Induced Nephrotoxicity In Vitro
Abstract
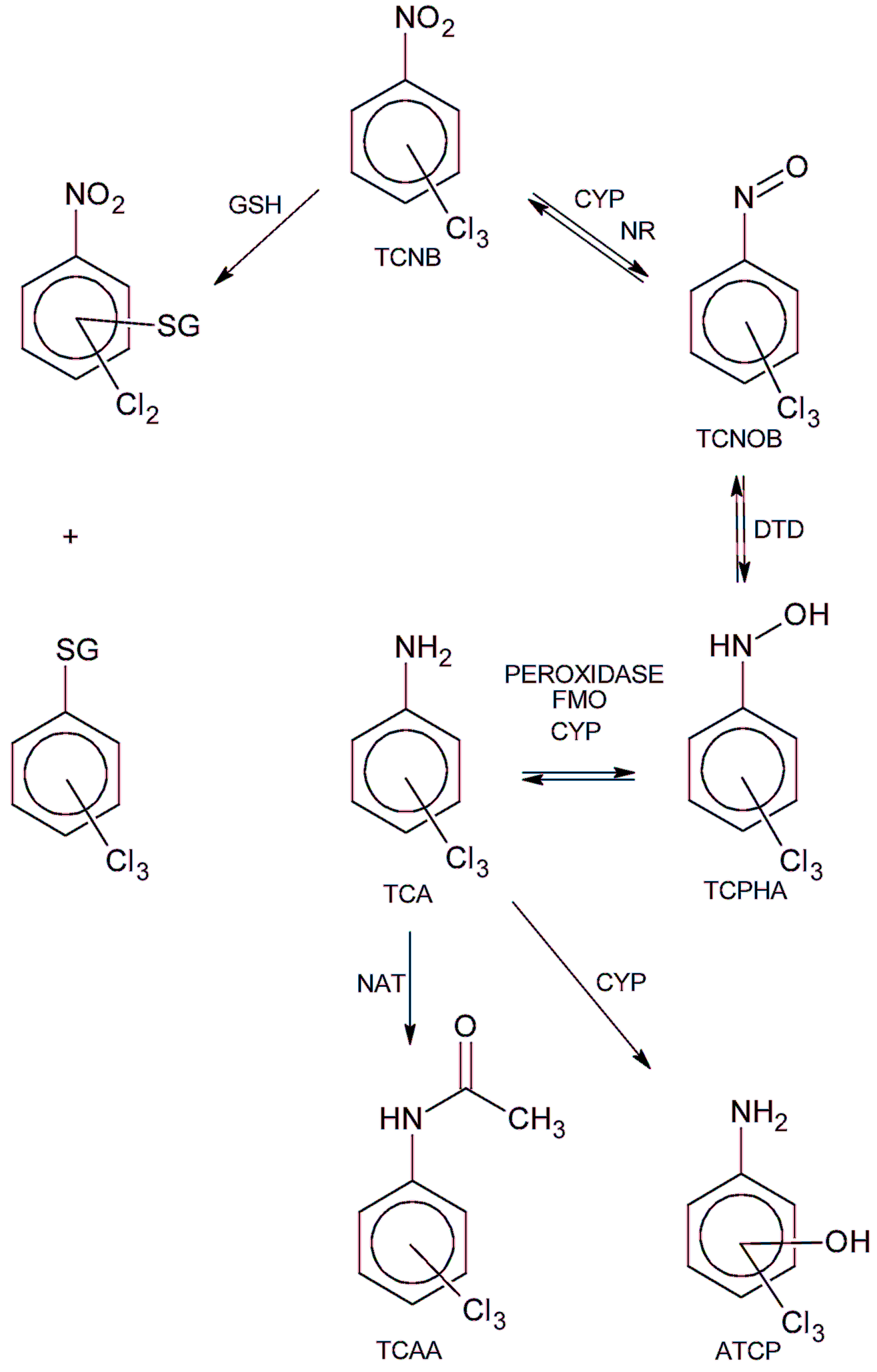
:1. Introduction
2. Results
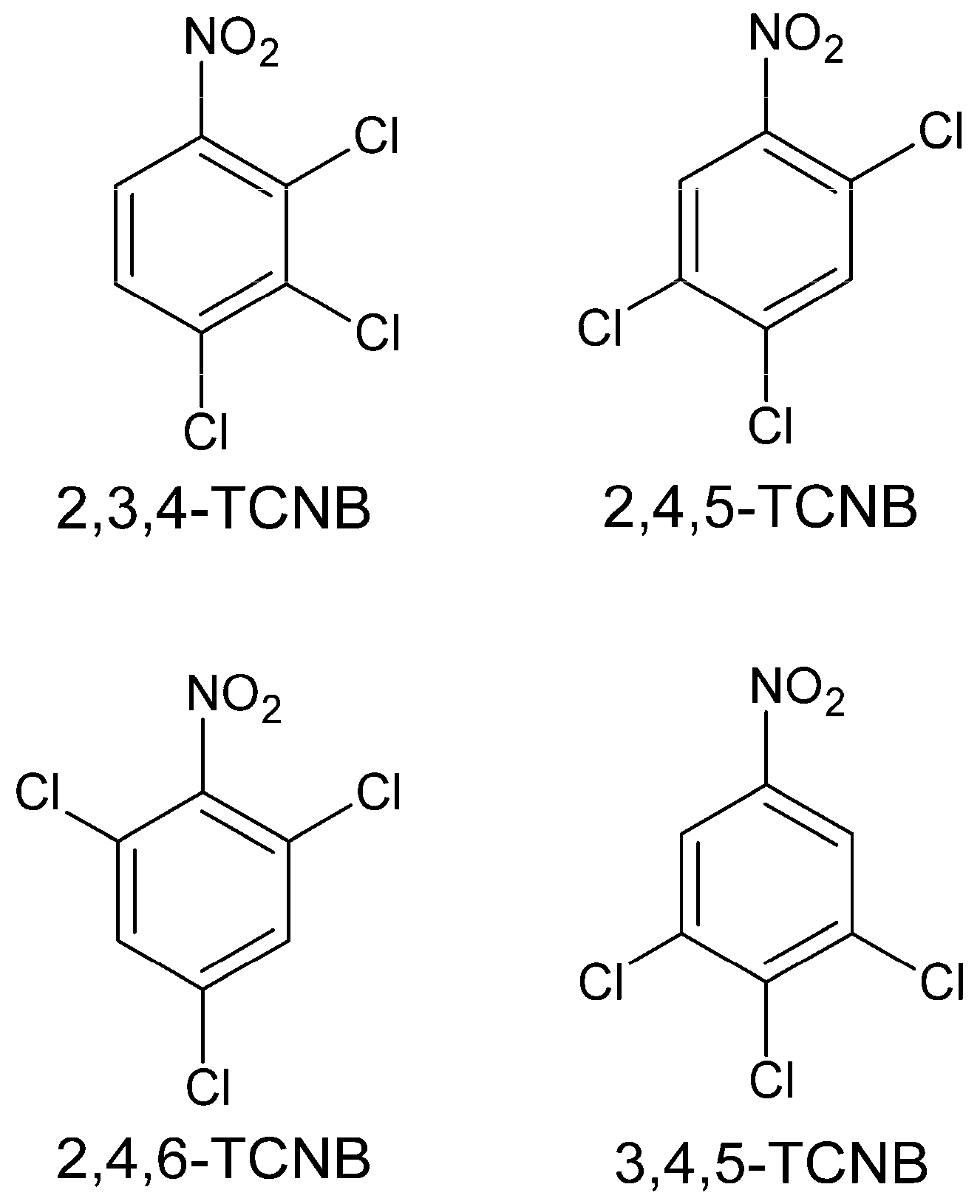
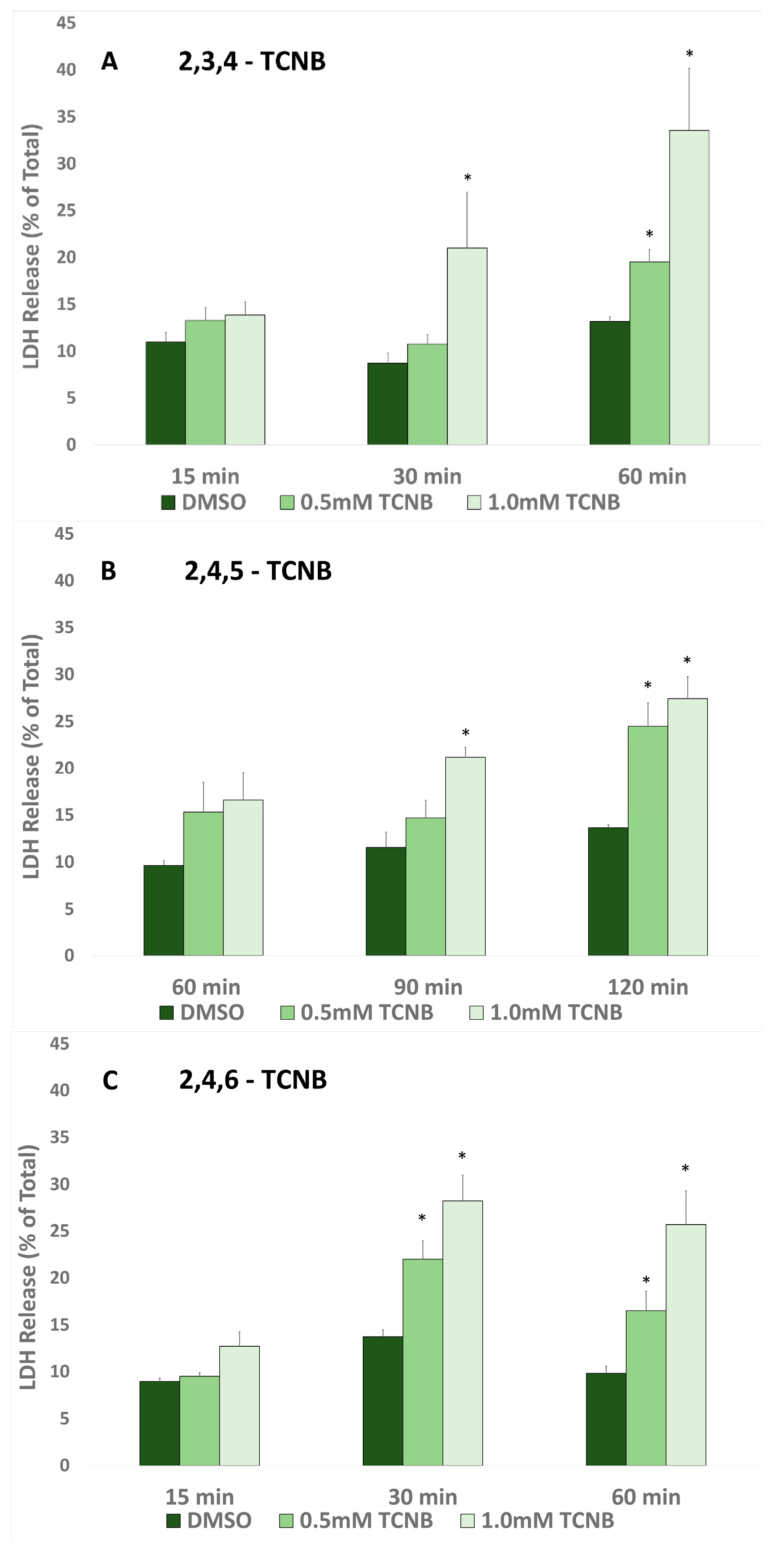
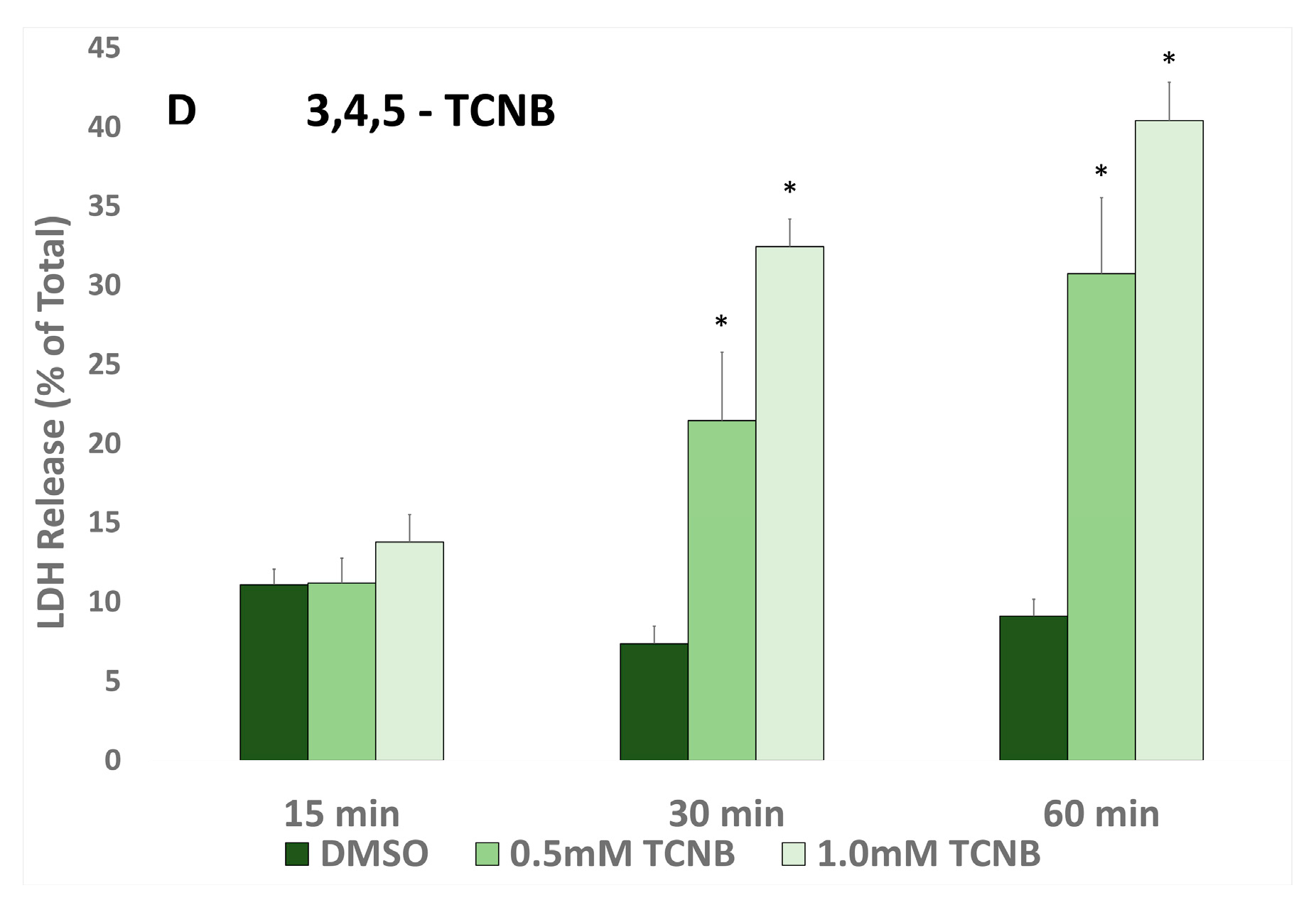
2.1. Time and Concentration Cytotoxicity Studies
2.2. Effects of Antioxidants and Biotransformation Inhibitors on 2,3,4-Trichloronitrobenzene (2,3,4-TCNB) Cytotoxicity
2.3. Effects of Antioxidants and Biotransformation Inhibitors on 2,4,5-TCNB Cytotoxicity
2.4. Effects of Antioxidants and Biotransformation Inhibitors on 2,4,6-TCNB Cytotoxicity
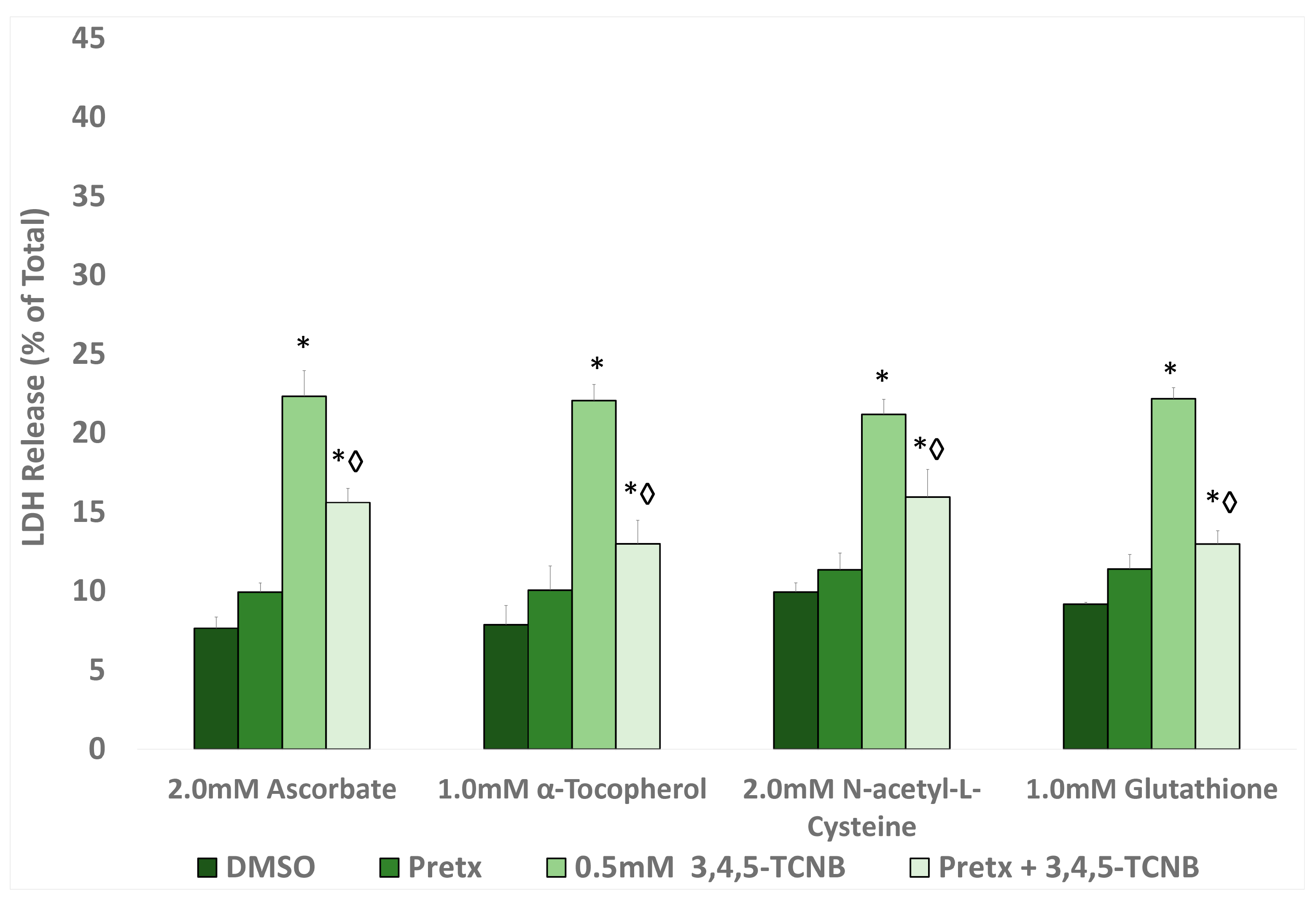
2.5. Effects of Antioxidants and Biotransformation Inhibitors on 3,4,5-TCNB Cytotoxicity
3. Discussion
4. Materials and Methods
4.1. Experimental Animals
4.2. Chemicals
4.3. Isolated Renal Cortical Cell (IRCC) Preparation and Treatment
4.4. Adenine Nucleotides
4.5. Western Blot, Protein Carbonylation (OxyBlot™), and 4-HNE
4.6. Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ATCP | Aminotrichlorophenol |
| CYP | Cytochrome P450 |
| DCNB | Dichloronitrobenzene |
| DTD | DT-diaphorase |
| FMO | Flavin monooxygenase |
| IRCC | Isolated renal cortical cells |
| LDH | Lactate dehydrogenase |
| MCNB | Monchloronitrobenzene |
| NAC | N-acetyl-l-cysteine |
| NR | Nitroreductase |
| PiBx | Piperonyl butoxide |
| ROS | Reactive oxygen species |
| TCA | Trichloroanilines |
| TCAA | Trichloroacetanilide |
| TCNB | Trichloronitrobenzene |
| TCNOP | Trichloronitrosobenzene |
| TCPHA | Trichlorophenylhydroxylamine |
References
- Aizawa, H. Metabolic Maps of Pesticides; Academic Press Inc.: London, UK, 1989; Volume 2. [Google Scholar]
- Laini, A.; Bartoli, M.; Lamastra, L.; Capri, E.; Balderacchi, M.; Trevisan, M. Herbicide contamination and dispersion pattern in lowland springs. Sci. Total Environ. 2012, 438, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Gandar, A.; Jean, S.; Canal, J.; Marty-Gasset, N.; Gilbert, F.; Laffaille, P. Multistress effects on goldfish (Carassius auratus) behavior and metabolism. Environ. Sci. Pollut. Res. Int. 2016, 23, 3184–3194. [Google Scholar] [CrossRef] [PubMed]
- Taygerly, J.P.; McGee, L.R.; Rubenstein, S.M.; Houze, J.B.; Cushing, T.D.; Li, Y.; Montai, A.; Chen, J.L.; Frankmoelle, W.; Ye, G.; et al. Discovery of INT131: A selective PPARγ modulator that enhances insulin sensitivity. Bioorg. Med. Chem. 2013, 21, 979–992. [Google Scholar] [CrossRef] [PubMed]
- Karatas, H.; Alp, M.; Yildiz, S.; Goker, H. Synthesis and potent in vitro activity of novel 1H-benzimidazoles as anti-MRSA agents. Chem. Biol. Drug Des. 2012, 80, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Alcock, R.E.; Sweetman, A.; Jones, K.C. Assesment of organic contamination fate in waste water treatment plants. I. Selected compounds and physiochemical properties. Chemosphere 1999, 38, 2247–2262. [Google Scholar] [CrossRef]
- Heudorf, U.; Neumann, H.G.; Peters, M. Accident in the Hoechst AG company 22 February 1993. Public health evaluation. Gesundheitswesen 1994, 56, 405–410. [Google Scholar] [PubMed]
- Brown, T.; Wania, F. Screening chemicals for the potential to be persistent organic pollutants: A case study of artic contaminants. Environ. Sci. Technol. 2008, 42, 5202–5209. [Google Scholar] [CrossRef] [PubMed]
- Travlos, G.S.; Mahler, J.; Ragan, H.A.; Chou, B.J.; Bucher, J.R. Thirteen-week inhalation toxicity of 2-and 4-chloronitrobenzene in F344/N rats and B6C3F1 mice. Fundam. Appl. Toxicol. 1996, 30, 75–92. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Aiso, S.; Senoh, H.; Yamazaki, K.; Arito, H.; Nagano, K.; Yamamoto, S.; Matsushima, T. Carcinogenicity and chronic toxicity of para-chloronitrobenzene in rats and mice by two-year feeding. J. Environ. Pathol. Toxicol. Oncol. 2006, 25, 571–584. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Minami, M.; Hanaoka, T.; Yamamura, Y. Acute immunotoxicity of p-chloronitrobenzene in mice. II. Effect of p-chloronitrobenzene on the immunophenotype of murine splenocytes determined by flow cytometry. Toxicology 1999, 137, 35–45. [Google Scholar] [CrossRef]
- Hong, S.; Anestis, D.K.; Ball, J.G.; Valentovic, M.A.; Rankin, G.O. In vitro nephrotoxicity induced by chloronitrobenzenes in renal cortical slices from Fisher 344 rats. Toxicol. Lett. 2002, 129, 133–141. [Google Scholar] [CrossRef]
- Matsumoto, M.; Umeda, Y.; Senoh, H.; Suzuki, M.; Kano, H.; Katagiri, T.; Aiso, S.; Yamazaki, K.; Arito, H.; Nagano, K.; et al. Two-year feed study of carcinogenicity and chronic toxicity of ortho-chloronitrobenzene in rats and mice. J. Toxicol. Sci. 2006, 31, 247–264. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Yoshikawa, H.; Goto, H.; Hara, I. Evaluation of the nephrotoxicity of aromatic nitro-amino compounds by urinary enzyme activities. J. Toxicol. Sci. 1989, 14, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, X.; Ni, L.; Jiang, R.; Liu, L.; Ye, C.; Han, C. Management of a patient with thermal burns and para-chloronitrobenzene poisoning. Int. J. Occup. Med. Environ. Health 2014, 27, 882–887. [Google Scholar] [CrossRef] [PubMed]
- Betts, J.J.; Bray, H.G.; James, S.P.; Thorpe, W.V. The metabolism of the trichloronitrobenzenes in the rabbit. Biochem. J. 1957, 66, 610–621. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Tabuchi, T.; Andoh, K. Identification of urinary metabolites of human subjects acutely poisoned by p-chloronitrobenzene. Xenobiotica 1992, 22, 1459–1470. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Tabuchi, T.; Andoh, K. Pharmacokinetic study of p-chloronitrobenzene in humans suffering from acute poisoning. Drug Metab. Dispos. 1993, 21, 1142–1146. [Google Scholar] [PubMed]
- Rickert, D.E. Metabolism of nitroaromatic compounds. Drug Metab. Rev. 1987, 18, 23–53. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.R.; Sepai, O.; Liu, Y.Y.; Yan, H.; Sabbioni, G. Urinary metabolites and health effects in workers exposed chronically to chloronitrobenzene. Biomarkers 2007, 12, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Rankin, G.O.; Yang, D.J.; Cressey-Veneziano, K.; Casto, S.; Wang, R.T.; Brown, P.I. In vivo and in vitro nephrotoxicity of aniline and its monochlorophenyl derivatives in the Fischer 344 rat. Toxicology 1986, 38, 269–283. [Google Scholar] [CrossRef]
- Lo, H.H.; Brown, P.I.; Rankin, G.O. Trichloroaniline effects on renal function in vivo and in vitro. Toxicol. Lett. 1991, 57, 319–328. [Google Scholar] [CrossRef]
- Valentovic, M.A.; Ball, J.G.; Anestis, D.K.; Rankin, G.O. Comparison of the in vitro toxicity of dichloroaniline structural isomers. Toxicol. In Vitro 1995, 9, 75–81. [Google Scholar] [CrossRef]
- Hong, S.K.; Anestis, D.K.; Henderson, T.T.; Rankin, G.O. Haloaniline-induced in vitro nephrotoxicity: Effects of 4-haloanilines and 3,5-dihaloanilines. Toxicol. Lett. 2000, 14, 125–133. [Google Scholar] [CrossRef]
- Racine, C.; Ward, D.; Anestis, D.K.; Ferguson, T.; Preston, D.; Rankin, G.O. 3,4,5-Trichloroaniline nephrotoxicity in vitro: Potential role of free radicals and renal biotransformation. Int. J. Mol. Sci. 2014, 15, 20900–20912. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.J.; Teets, V.J.; Bolton, B.; Brown, P.I.; Rankin, G.O. Role of glutathione in acute N-(3,5-dichlorophenyl) succinimide-induced nephrotoxicity in Sprague-Dawley and Fischer 344 rats. Toxicology 1987, 45, 25–44. [Google Scholar] [CrossRef]
- Rickert, D.E.; Held, S.D. Metabolism of chloronitrobenzenes by isolated rat hepatocytes. Drug Metab. Dispos. 1990, 18, 5–9. [Google Scholar] [PubMed]
- Yoshida, T.; Andoh, K.; Tabuchi, T. Identification of urinary metabolites in rats treated with p-chloronitrobenzene. Arch. Toxicol. 1991, 65, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T. Pharmacokinetic study of p-chloronitrobenzene in rat. Drug Metab. Dispos. 1994, 22, 275–280. [Google Scholar] [PubMed]
- Bray, H.G.; James, S.P.; Thorpe, W.V. The metabolism of the monochloronitrobenzenes in the rabbit. Biochem. J. 1956, 64, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Dekant, W. Chemical-induced nephrotoxicity mediated by glutathione S-conjugate formation. Toxicol. Lett. 2001, 124, 21–36. [Google Scholar] [CrossRef]
- Rankin, G.O.; Valentovic, M.A.; Beers, K.W.; Nicoll, D.W.; Ball, J.G.; Anestis, D.K.; Brown, P.I.; Hubbard, J.L. Renal and hepatic toxicity of monochloroacetanilides in the Fischer 344 rat. Toxicology 1993, 79, 181–193. [Google Scholar] [CrossRef]
- Rankin, G.O.; Valentovic, M.A.; Hong, S.K.; Anestis, D.K.; Ball, J.G.; Dial, L.D.; Brown, P.I. In vivo and in vitro renal effects induced by 2-chloro-4-hydroxyacetanilide and 4- chloro-2-hydroxyacetanilide in the Fischer 344 rat. Toxic Sbstance Mech. 1995, 14, 93–109. [Google Scholar]
- Valentovic, M.A.; Rogers, B.A.; Meadows, M.K.; Conner, J.T.; Williams, E.; Hong, S.K.; Rankin, G.O. Characterization of methemoglobin formation induced by 3,5-dichloroaniline, 4-amino-2,6-dichlorophenol and 3,5-dichlorophenylhydroxylamine. Toxicology 1997, 118, 23–36. [Google Scholar] [CrossRef]
- McMillan, D.C.; Leakey, J.E.; Arlotto, M.P.; McMillan, J.M.; Hinson, J.A. Metabolism of the arylamide herbicide propanil. II. Effects of propanil and its derivatives on hepatic microsomal drug metabolizing enzymes in the rat. Toxicol. Appl. Pharmacol. 1990, 103, 102–112. [Google Scholar] [CrossRef]
- Ochiai, Y.; Sakurai, E.; Nomura, A.; Itoh, K.; Tanaka, Y. Metabolism of nicotine in rat lung microvascular endothelial cells. J. Pharm. Pharmacol. 2006, 58, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Ehlhardt, W.J.; Kulanthaivel, P.; Lanza, D.L.; Reilly, A.; Yost, G.S. Dehydrogenation of indoline by cytochrome P450 enzymes: A novel “aromatase” process. J. Pharmacol. Exp. Ther. 2007, 322, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Yanni, S.B.; Annaert, P.P.; Augustijns, P.; Ibrahim, J.G.; Benjamin, D.K., Jr.; Thakker, D.R. In vitro hepatic metabolism explains higher clearance of voriconazole in children versus adults: Role of CYP2C19 and flavin-containing monooxygenase 3. Drug Metab. Dispos. 2010, 38, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Golly, I.; Hlavica, P. N-Oxidation of 4-chloroaniline by prostaglandin synthase. Biochem. J. 1985, 260, 803–809. [Google Scholar] [CrossRef]
- Corbett, M.D.; Chipko, B.R.; Batchelor, A.O. The action of chloride peroxidase on 4-chloroaniline. Biochem. J. 1980, 187, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Bakkenist, A.R.J.; Plat, H.; Wever, R. Oxidation of 4-chloroaniline catalyzed by human myeloperoxidase. Bioorg. Chem. 1981, 10, 324–328. [Google Scholar] [CrossRef]
- Akintonwa, D.A.A. Theoretical mechanistic basis of oxidants of methaemoglobin formation. Med. Hypotheses 2000, 54, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Purnell, E.; Smith, C. Mechanistic study on aniline-induced erythrocyte toxicity. Arh. Hig. Rada Toksikol. 2007, 58, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Eyer, P.; Ascheri, M. Reactions of para-substituted nitrosobenzenes with human hemoglobin. Biol. Chem. Hoppe Seyler 1987, 368, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Kiese, M.; Taeger, K. The fate of phenylhydroxylamine in human red cells. Naunyn Schmiedebergs Arch. Pharmacol. 1976, 292, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Stiborova, M.; Frei, E.; Schmeiser, H.H.; Wiessler, M.; Anzenbacher, P. peroxidase oxidizes N-nitrosomethylaniline to ultimate carcinogens: Binding to DNA and transfer RNA in vitro. Cancer Lett. 1992, 63, 53–59. [Google Scholar] [CrossRef]
- Valentovic, M.; Ball, J.G.; Stoll, S.; Rankin, G.O. 3,4-Dichlorophenylhydroxylamine cytotoxicity in renal cortical slices from Fischer 344 rats. Toxicology 2001, 162, 149–156. [Google Scholar] [CrossRef]
- Hong, S.K.; Valentovic, M.A.; Anestis, D.K.; Ball, J.G.; Brown, P.I.; Rankin, G.O. Nephrotoxicity of 4-amino-2-chlorophenol and 2-amino-4-chlorophenol in the Fischer 344 rat. Toxicology 1996, 110, 47–58. [Google Scholar] [CrossRef]
- Hong, S.K.; Anestis, D.K.; Ball, J.G.; Valentovic, M.A.; Brown, P.I.; Rankin, G.O. 4-Amino-2,6-dichlorophenol nephrotoxicity in the Fischer 344 rat: Protection by ascorbic acid, AT-125, and aminooxyacetic acid. Toxicol. Appl. Pharmacol. 1997, 147, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Rankin, G.O.; Hong, S.K.; Anestis, D.K.; Ball, J.G.; Valentovic, M.A. Mechanistic aspects of 4-amino-2,6-dichlorophenol-induced in vitro nephrotoxicity. Toxicology 2008, 245, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Valentovic, M.A.; Ball, J.G.; Sun, H.; Rankin, G.O. Characterization of 2-amino-4,5-dichlorophenol (2A45CP) in vitro toxicity in renal cortical slices from male Fischer 344 rats. Toxicology 2002, 172, 113–123. [Google Scholar] [CrossRef]
- Racine, C.R.; Ferguson, T.L.; Preston, D.; Ward, D.; Anestis, D.K.; Ball, J.; Valentovic, M.A.; Rankin, G.O. The role of biotransformation and oxidative stress in 3,5-dichloroaniline induced nephrotoxicity in isolated renal cortical cells from Fischer 344 rats. Toxicology 2016, 341, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Loew, G.H.; Goldblum, A. Metabolic activation and toxicity of acetaminophen and related analogs: A theoretical study. Mol. Pharmacol. 1985, 27, 375–386. [Google Scholar] [PubMed]
- Umbreit, J. Methemoglobin—It’s not just blue: A concise review. Am. J. Hematol. 2007, 82, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Harmon, R.C.; Kiningham, K.K.; Valentovic, M.A. Pyruvate reduces 4-aminophenol in vitro toxicity. Toxicol. Appl. Pharmacol. 2006, 213, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P.; Sundby, G.-B.; Ormsted, K.; Orrenius, S. Use of isolated kidney cells for study of drug metabolism. Biochem. Pharmacol. 1979, 28, 929–935. [Google Scholar] [CrossRef]
- Rankin, G.O.; Racine, C.; Sweeney, A.; Kraynie, A.; Anestis, D.K.; Barnett, J.B. In vitro nephrotoxicity induced by propanil. Environ. Toxicol. 2008, 23, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Katsuda, H.; Yamashita, M.; Katsura, H.; Yu, J.; Waki, Y.; Nagata, N.; Sai, Y.; Miyamoto, K.-I. Protecting cisplatin-induced nephrotoxicity with cimetidine does not affect antitumor activity. Biol. Pharm. Bull. 2010, 33, 1867–1871. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Sudo, J. Lipid peroxidation and generations of oxygen radicals induced by cephaloridine in renal cortical microsomes of rats. Jpn. J. Pharmacol. 1990, 52, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.S.; Monks, T.J. Co-oxidation of 2-bromohydroquinone by renal prostaglandin synthase. Modulation of prostaglandin synthesis by 2-bromohydroquinone and glutathione. Drug Metab. Dispos. 1987, 15, 801–807. [Google Scholar] [PubMed]
- Lock, E.A.; Cross, T.J.; Schnellmann, R.G. Studies on the mechanism of 4-aminophenol-induced toxicity to renal proximal tubules. Hum. Exp. Toxicol. 1993, 12, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Harmon, R.C.; Terneus, M.V.; Kiningham, K.K.; Valentovic, M. Time-dependent effect of p-aminophenol (PAP) toxicity in renal slices and development of oxidative stress. Toxicol. Appl. Pharmacol. 2005, 209, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.J.; Acosta, D. N-deacetyl ketoconazole-induced hepatotoxicity in a primary culture system of rat hepatocytes. Toxicology 1997, 117, 123–131. [Google Scholar] [CrossRef]
- Baliga, R.; Zhang, Z.; Baliga, M.; Ueda, N.; Shah, S.V. Role of cytochrome P-450 as a source of catalytic iron in cisplatin-induced nephrotoxicity. Kidney Int. 1998, 54, 1562–1569. [Google Scholar] [CrossRef] [PubMed]
- Harleton, E.; Webster, M.; Bumpus, N.N.; Kent, U.M.; Rae, J.M.; Hollenberg, P.F. Metabolism of N,N’,N’’-triethylenethiophosphoramide by CYP2B1 and CYP2B6 results in the inactivation of both isoforms by two distinct mechanisms. J. Pharmacol. Exp. Ther. 2004, 310, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Eagling, V.A.; Tjia, J.F.; Back, D.J. Differential selectivity of cytochrome P450 inhibitors against probe substrates in human and rat liver microsomes. Br. J. Clin. Pharmacol. 1998, 45, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Urashima, K.; Shimada, N.; Chiba, K. Selectivities of human cytochrome P450 inhibitors toward rat P450 isoforms: Study with cDNA-expressed systems of the rat. Drug Metab. Dispos. 2003, 31, 833–836. [Google Scholar] [CrossRef] [PubMed]
- Valentovic, M.A.; Minigh, J. Pyruvate attenuates myoglobin in vitro toxicity. Toxicol. Sci. 2003, 74, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Valentovic, M.A.; Ball, J.G.; Brown, J.M.; Terneus, M.V.; McQuade, E.; van Meter, S.; Hedrick, H.M.; Roy, A.A.; Williams, T. Resveratrol attenuates cisplatin renal cortical cytotoxicity by modifying oxidative stress. Toxicol. In Vitro 2014, 28, 248–257. [Google Scholar] [CrossRef] [PubMed]
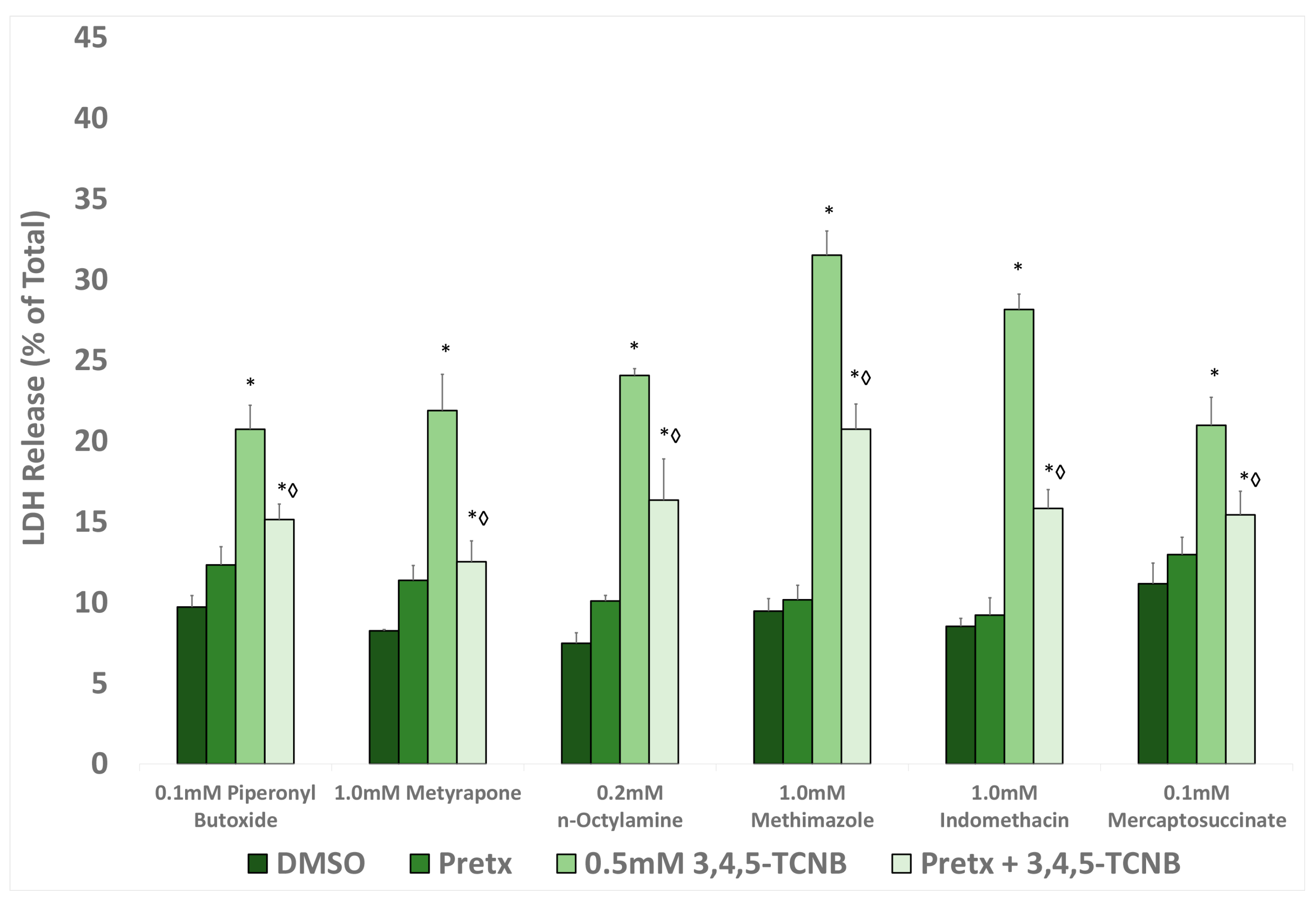
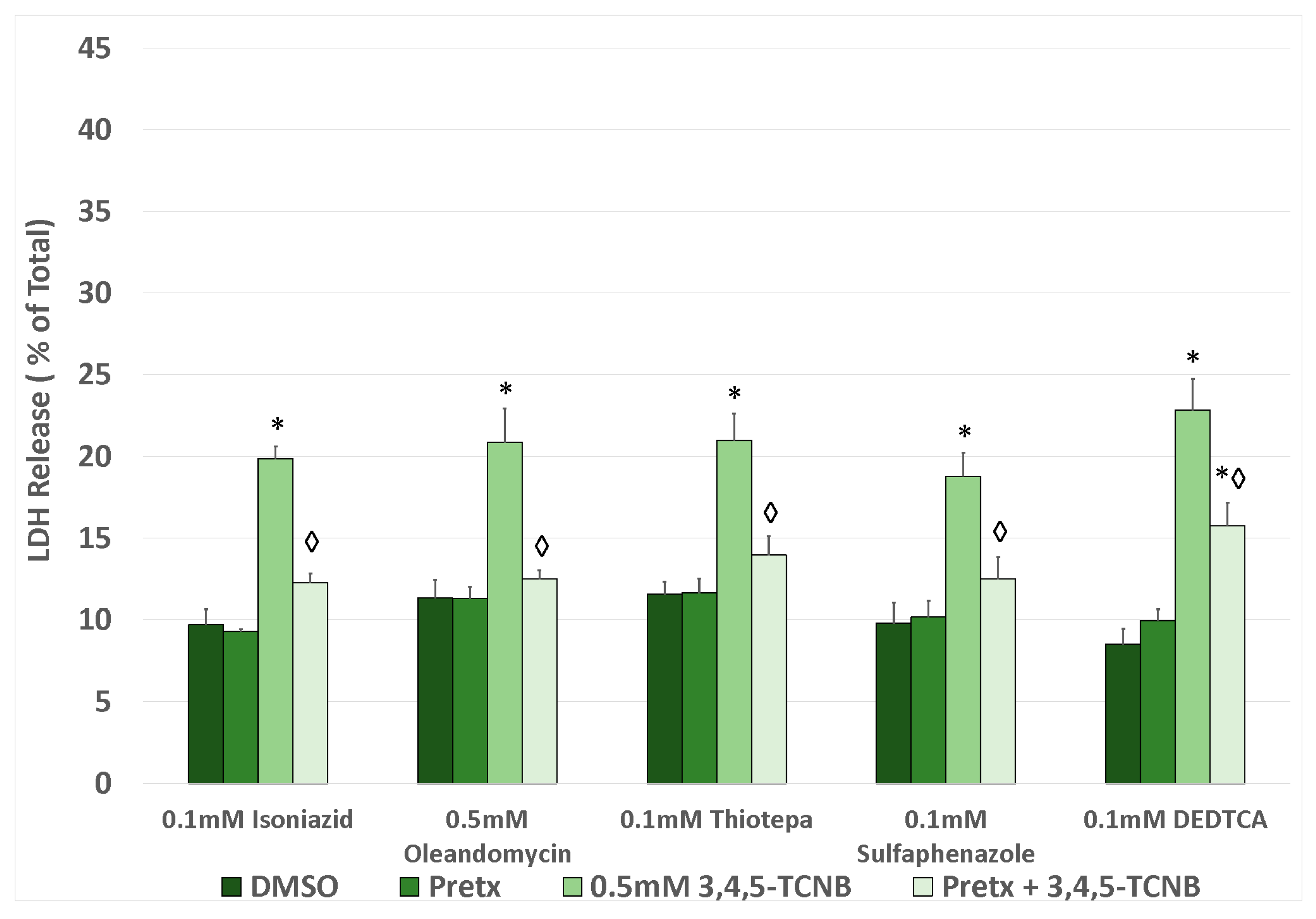
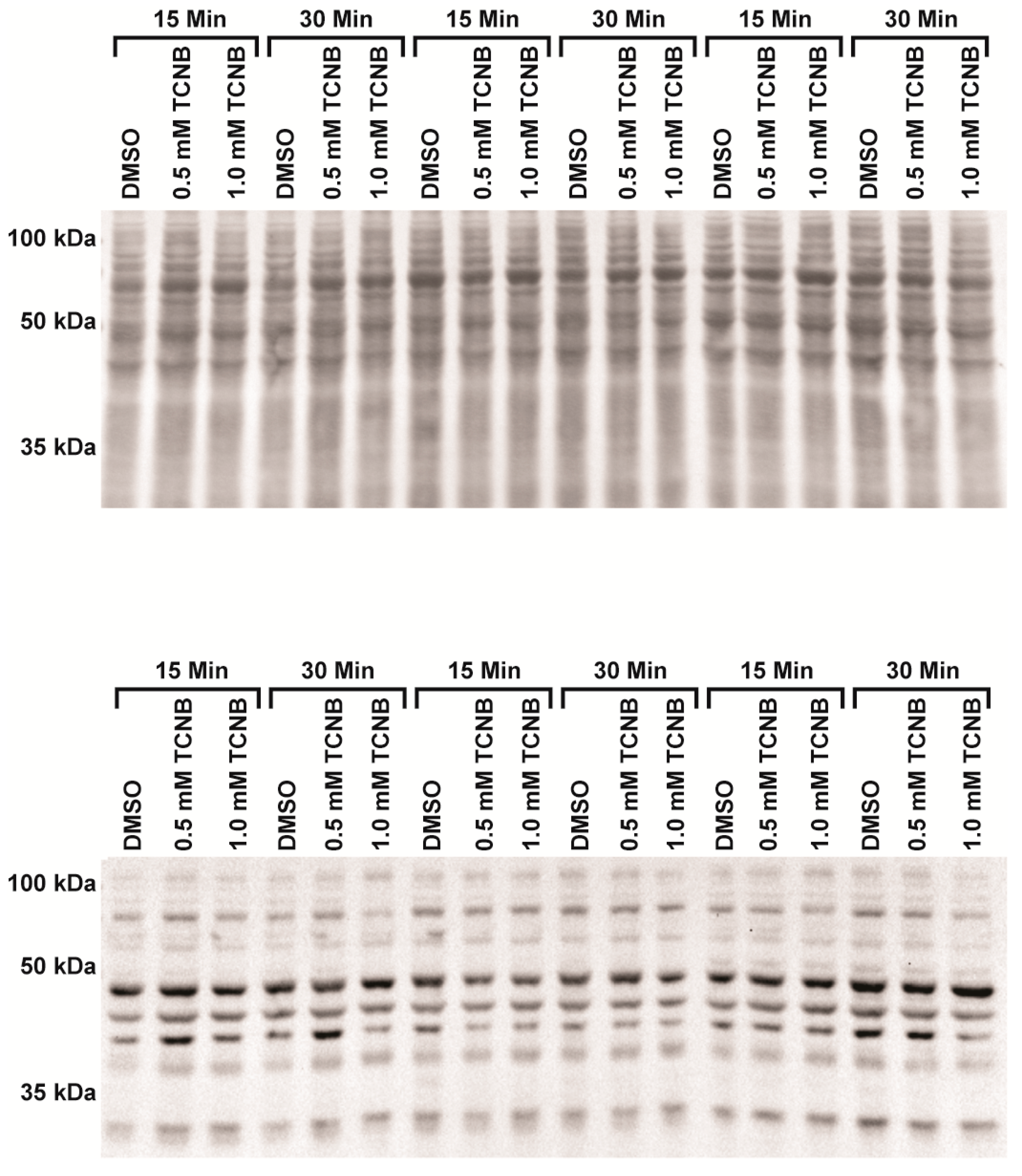
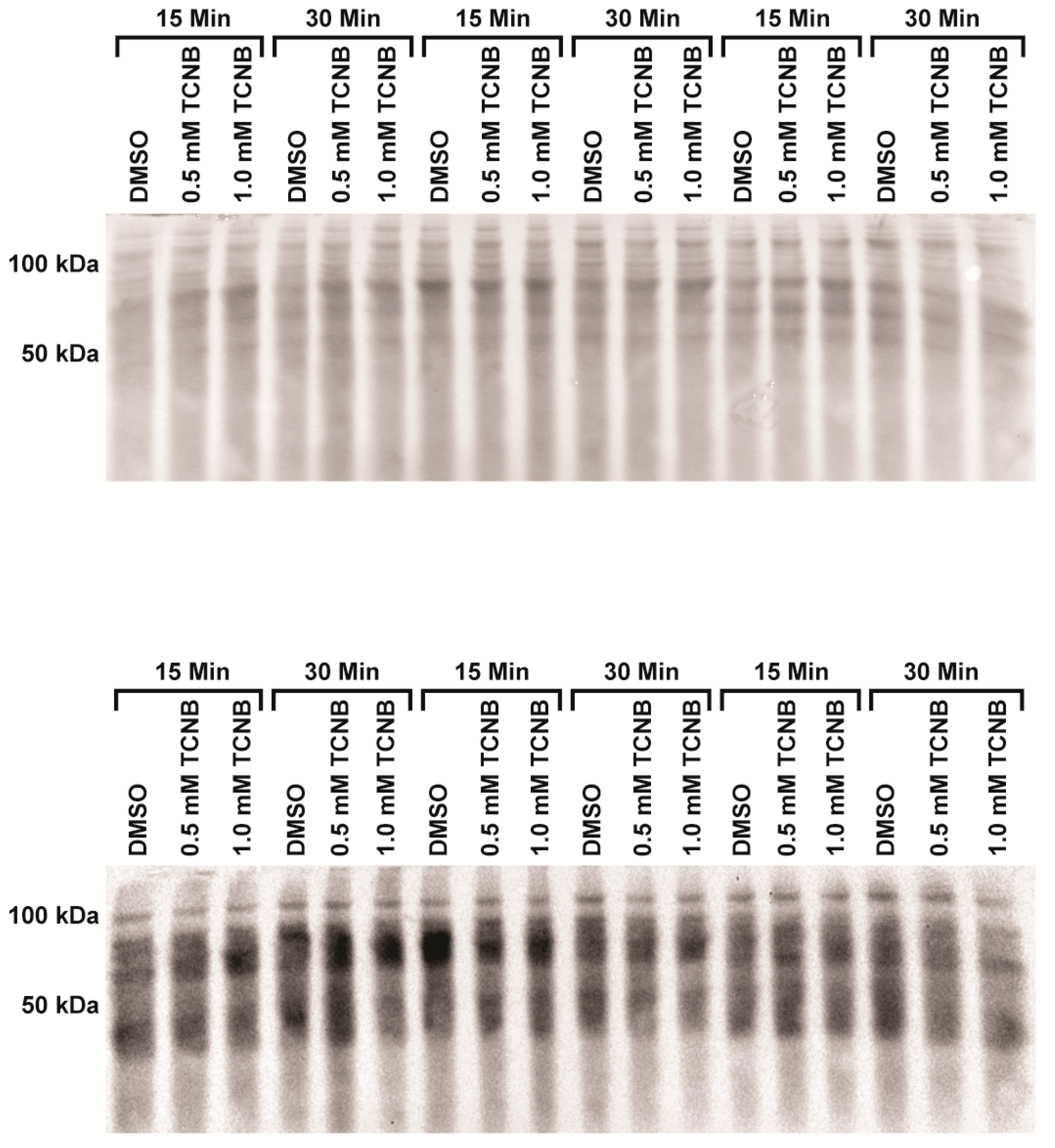
| Pretreatment b | Vehicle | PreTx | 2,3,4-TCNB | PreTx + TCNB |
|---|---|---|---|---|
| Ascorbate | 12 ± 1 | 12 ± 1 | 20 ± 2 c | 14 ± 1 d |
| α–Tocopherol | 11 ± 1 | 11± 1 | 23 ± 2 c | 12 ± 2 d |
| Glutathione | 12 ± 1 | 13 ± 1 | 20 ± 1 c | 14 ± 1 d |
| NAC | 9 ± 2 | 13± 1 | 23 ± 2 c | 16 ± 1 c,d |
| PiBx | 8 ±1 | 10 ± 1 | 21 ± 1 c | 12 ± 1 c,d |
| Metyrapone | 11 ± 1 | 12 ± 1 | 19 ± 1 c | 17 ± 1 c |
| Isoniazid | 10 ± 1 | 11 ± 1 | 23 ± 2 c | 19 ± 1 c,d |
| n-Octylamine | 10 ± 1 | 16 ± 1 c | 28 ± 2 c | 19 ± 1 c,d |
| Methimazole | 8 ± 1 | 9 ± 1 | 21 ± 1 c | 11 ± 1 d |
| Indomethacin | 10 ± 1 | 13 ± 1 | 22 ± 1 c | 16 ± 2 c,d |
| Mercaptosuccinate | 9 ± 2 | 10 ± 1 | 18 ± 1 c | 10 ± 1 d |
| Pretreatment b | Vehicle | PreTx | 2,3,4-TCNB | PreTx + TCNB |
|---|---|---|---|---|
| Ascorbate | 11 ± 1 | 12 ± 1 | 22 ± 1 c | 16 ± 2 d |
| α–Tocopherol | 12 ± 1 | 13 ± 1 | 22 ± 1 c | 14 ± 3 d |
| Glutathione | 14 ± 1 | 15 ± 1 | 26 ± 3 c | 17 ± 2 d |
| NAC | 14 ± 1 | 15 ± 1 | 26 ± 2 c | 17 ± 1 d |
| PiBx | 9 ± 1 | 11 ± 2 | 27 ±2 c | 24 ± 3 c |
| Metyrapone | 9 ± 1 | 16 ± 1 c | 26 ± 2 c | 28 ± 2 c |
| Isoniazid | 9 ± 1 | 10 ± 1 | 28 ± 2 c | 34 ± 1 c,d |
| n-Octylamine | 9 ± 1 | 11 ± 2 | 28 ± 2 c | 32 ± 2 c |
| Methimazole | 12 ± 2 | 14 ± 2 | 28 ± 1 c | 23 ± 2 c |
| Indomethacin | 13 ± 1 | 16 ± 2 | 26 ± 1 c | 21 ± 2 c |
| Mercaptosuccinate | 13 ± 1 | 17 ± 1 c | 26 ± 1 c | 24 ± 1 c |
| Pretreatment b | Vehicle | PreTx | 2,4,6-TCNB | PreTx + TCNB |
|---|---|---|---|---|
| Ascorbate | 12 ± 1 | 12 ± 1 | 24 ± 1 c | 17 ± 2 c,d |
| α–Tocopherol | 13 ± 1 | 14 ± 1 | 30 ± 2 c | 25 ± 2 c |
| Glutathione | 12 ± 1 | 15 ± 1 | 30 ± 1 c | 20 ± 1 c,d |
| NAC | 13 ± 1 | 13 ± 1 | 28 ± 1 c | 23 ± 2 c |
| PiBx | 13 ± 1 | 13 ± 1 | 26 ± 2 c | 22 ± 1 c |
| Metyrapone | 11± 1 | 15 ± 1 | 22 ± 1 c | 27 ± 2 c |
| Isoniazid | 11 ± 1 | 10 ± 1 | 26 ± 3 c | 28 ± 2 c |
| n-Octylamine | 11 ± 1 | 11 ± 1 | 27 ± 3 c | 29 ± 3 c |
| Methimazole | 14 ± 1 | 17 ± 1 | 29 ± 2 c | 31 ± 2 c |
| Indomethacin | 11 ± 1 | 16 ± 1 c | 28 ± 2 c | 21 ± 1 c,d |
| Mercaptosuccinate | 14 ± 1 | 16 ± 1 | 28 ± 2 c | 31 ± 1 c |
| Treatment | Time (min) | ATP nmol/4 Million Cells | ADP nmol/4 Million Cells |
|---|---|---|---|
| DMSO | 15 | 13.47 ± 1.65 | 14.21 ± 1.26 |
| DMSO | 30 | 12.74 ± 1.87 | 15.28 ± 1.23 |
| 3,4,5-TCNB 1.0 mM | 15 | 7.93 ± 1.37 b | 13.74 ± 1.51 |
| 3,4,5-TCNB 1.0 mM | 30 | 6.51 ± 0.98 b | 12.62 ± 1.63 |
| Pretreatment (PreTx) Compound | PreTx Conc. | PreTx Time (min) | Mechanism or Enzyme Inhibited | Reference |
|---|---|---|---|---|
| N-acetyl-l-cysteine (NAC) | 2.0 mM | 30 | Antioxidant | [58] |
| α-Tocopherol | 1.0 mM | 5 | Antioxidant | [59] |
| Glutathione | 1.0 mM | 30 | Antioxidant | [60] |
| Ascorbate | 2.0 mM | 5 | Antioxidant | [61] |
| Pyruvate | 1.0 mM | 15 | Antioxidant | [55,62] |
| Methimazole | 1.0 mM | 30 | FMO | [63] |
| N-Octylamine | 2.0 mM | 5 | FMO | [63] |
| Indomethacin | 1.0 mM | 15 | Cyclooxygenase | [60] |
| Mercaptosuccinate | 0.1 mM | 15 | Peroxidase | [25] |
| Piperonyl butoxide (PiBx) | 1.0 mM | 15 | NS CYP | [64] |
| Metyrapone | 1.0 mM | 5 | NS CYP | [61] |
| Oleandomycin triacetate | 0.5 mM | 30 | CYP3A1/2 | [25] |
| Thio-tepa | 0.1 mM | 5 | CYP2B1/2 | [65] |
| Isoniazid | 1.0 mM | 5 | CYP2E1 | [25] |
| Diethyldithiocarbamate (DEDTCA) | 0.1 mM | 30 | CYP2C > CYP2E1 | [66] |
| Sulfaphenazole | 0.1 mM | 30 | CYP2C | [66,67] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rankin, G.O.; Tyree, C.; Pope, D.; Tate, J.; Racine, C.; Anestis, D.K.; Brown, K.C.; Dial, M.; Valentovic, M.A. Role of Free Radicals and Biotransformation in Trichloronitrobenzene-Induced Nephrotoxicity In Vitro. Int. J. Mol. Sci. 2017, 18, 1165. https://doi.org/10.3390/ijms18061165
Rankin GO, Tyree C, Pope D, Tate J, Racine C, Anestis DK, Brown KC, Dial M, Valentovic MA. Role of Free Radicals and Biotransformation in Trichloronitrobenzene-Induced Nephrotoxicity In Vitro. International Journal of Molecular Sciences. 2017; 18(6):1165. https://doi.org/10.3390/ijms18061165
Chicago/Turabian StyleRankin, Gary O., Connor Tyree, Deborah Pope, Jordan Tate, Christopher Racine, Dianne K. Anestis, Kathleen C. Brown, Mason Dial, and Monica A. Valentovic. 2017. "Role of Free Radicals and Biotransformation in Trichloronitrobenzene-Induced Nephrotoxicity In Vitro" International Journal of Molecular Sciences 18, no. 6: 1165. https://doi.org/10.3390/ijms18061165
APA StyleRankin, G. O., Tyree, C., Pope, D., Tate, J., Racine, C., Anestis, D. K., Brown, K. C., Dial, M., & Valentovic, M. A. (2017). Role of Free Radicals and Biotransformation in Trichloronitrobenzene-Induced Nephrotoxicity In Vitro. International Journal of Molecular Sciences, 18(6), 1165. https://doi.org/10.3390/ijms18061165









