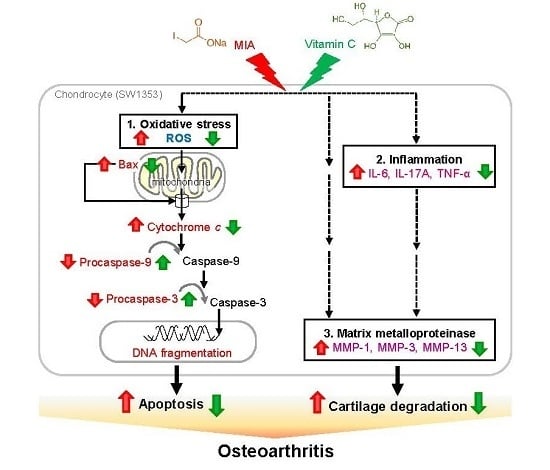
Vitamin C Protects Chondrocytes against Monosodium Iodoacetate-Induced Osteoarthritis by Multiple Pathways
Abstract
:1. Introduction
2. Results
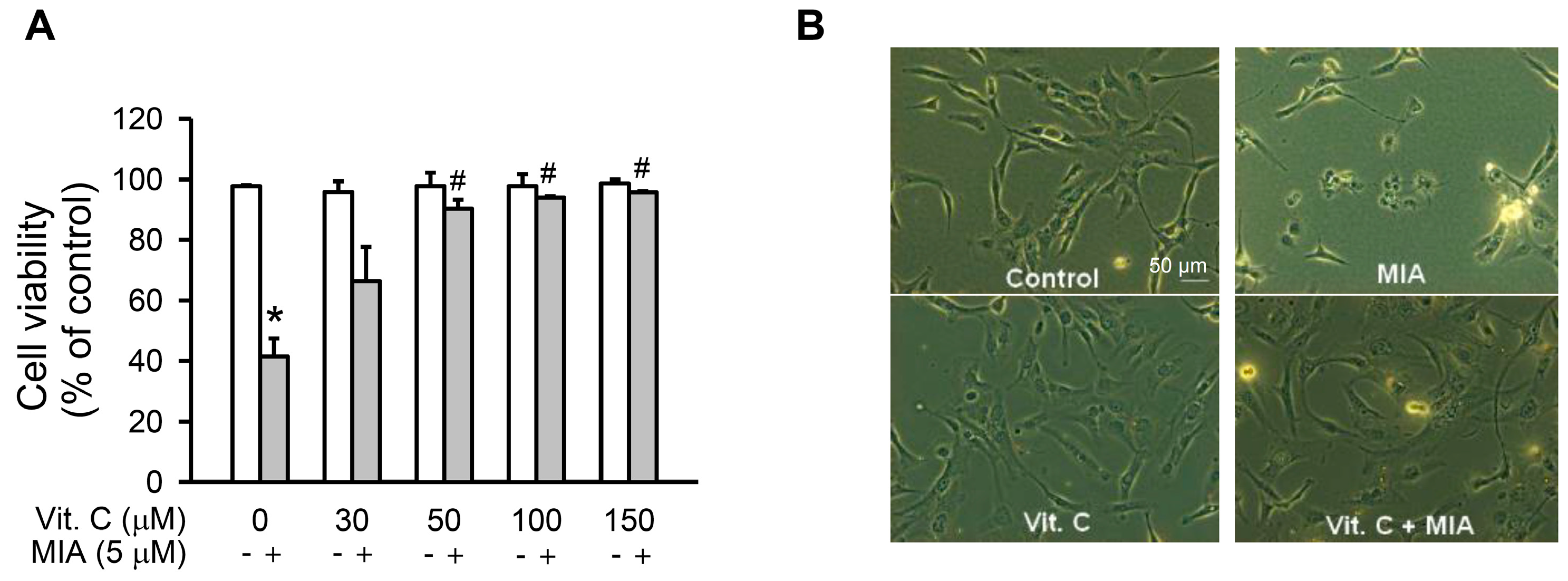
2.1. Cell Growth Inhibition
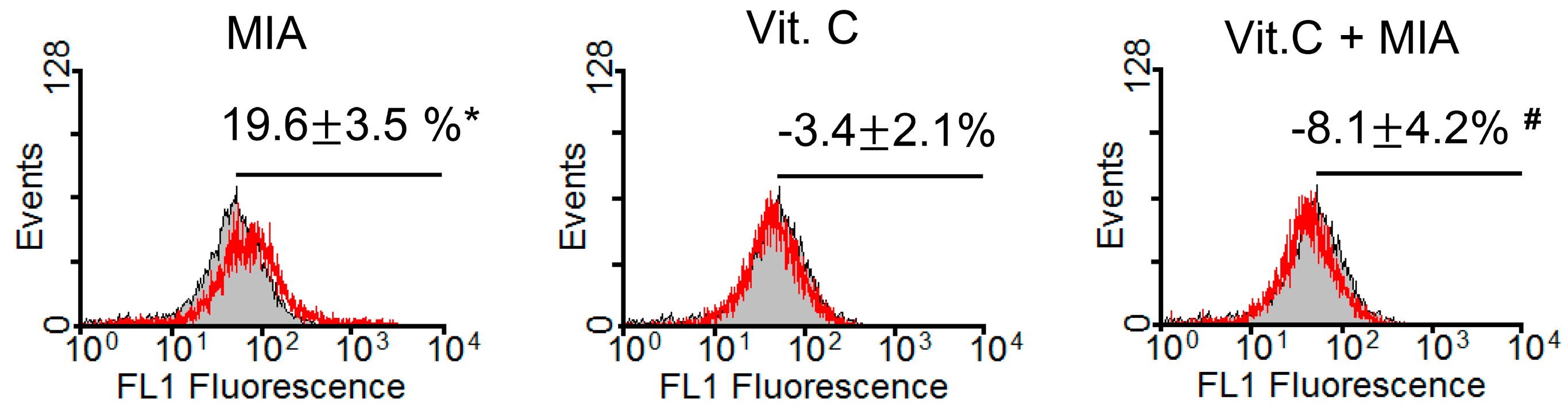
2.2. Oxidative Stress
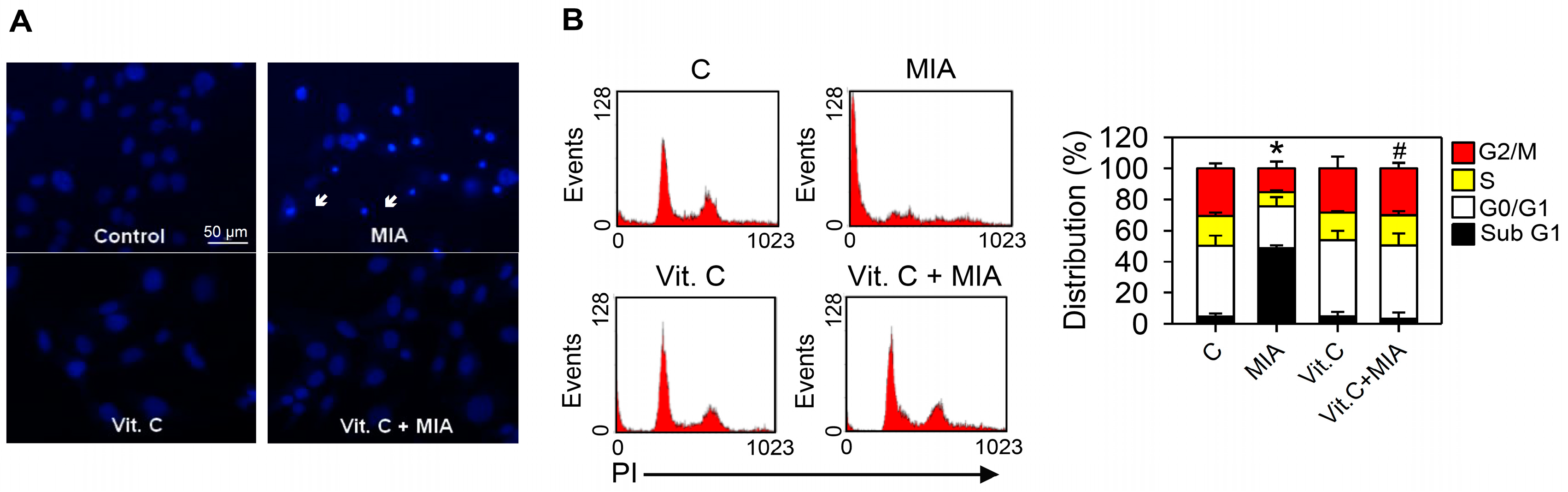
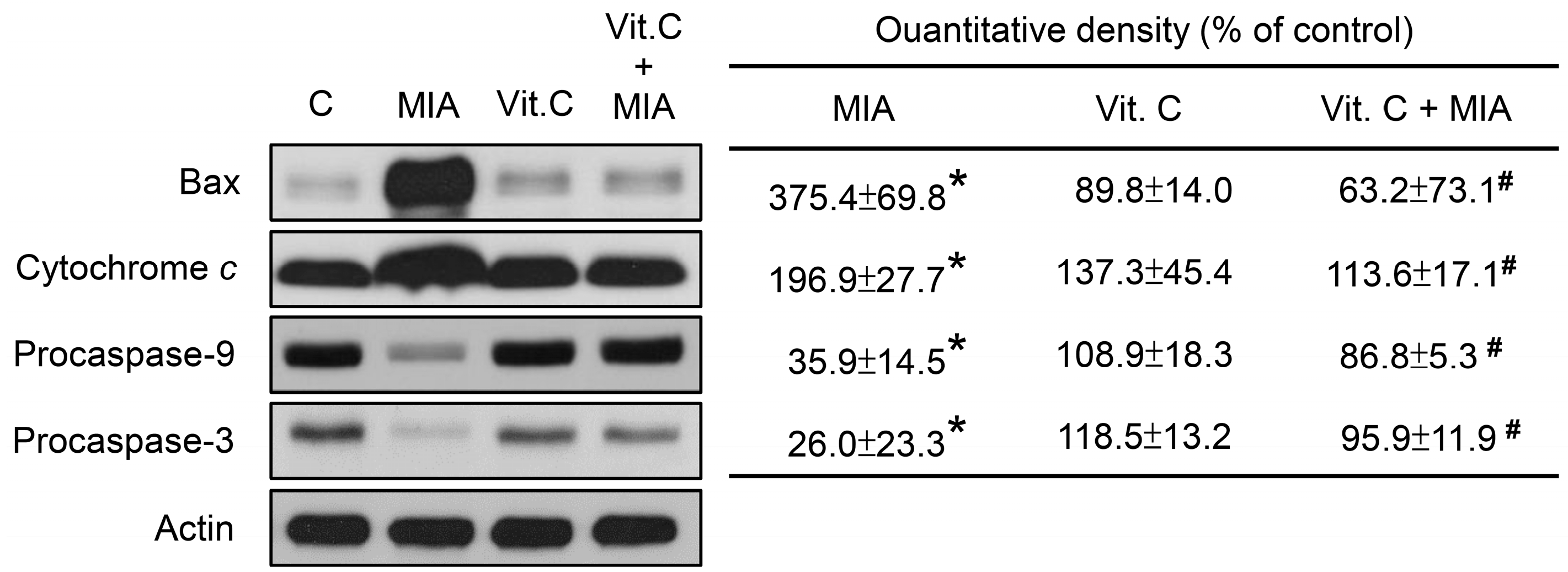
2.3. Apoptosis, Cell Cycle Progress, and Apoptosis-Related Proteins
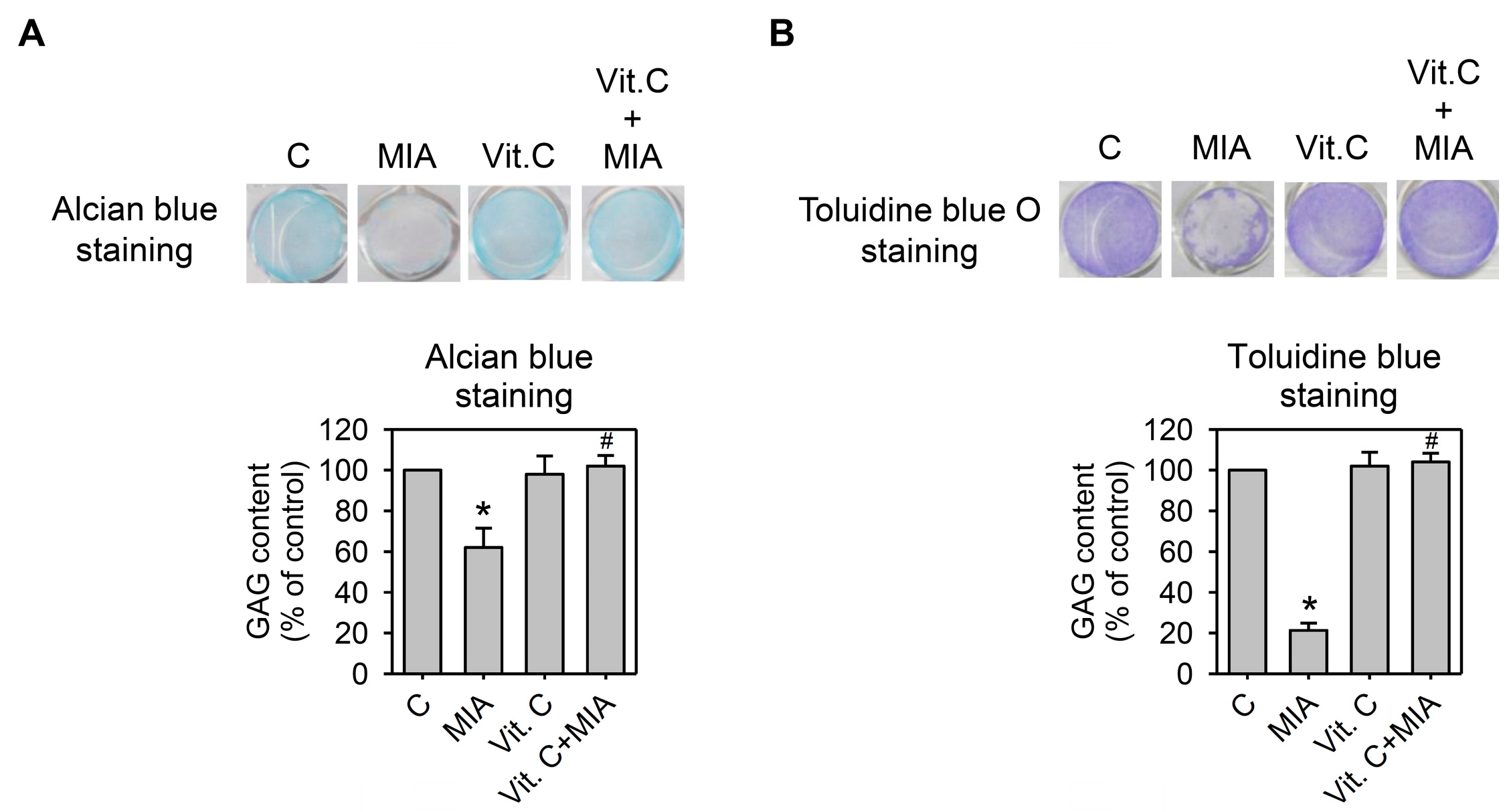
2.4. Proteoglycan Loss
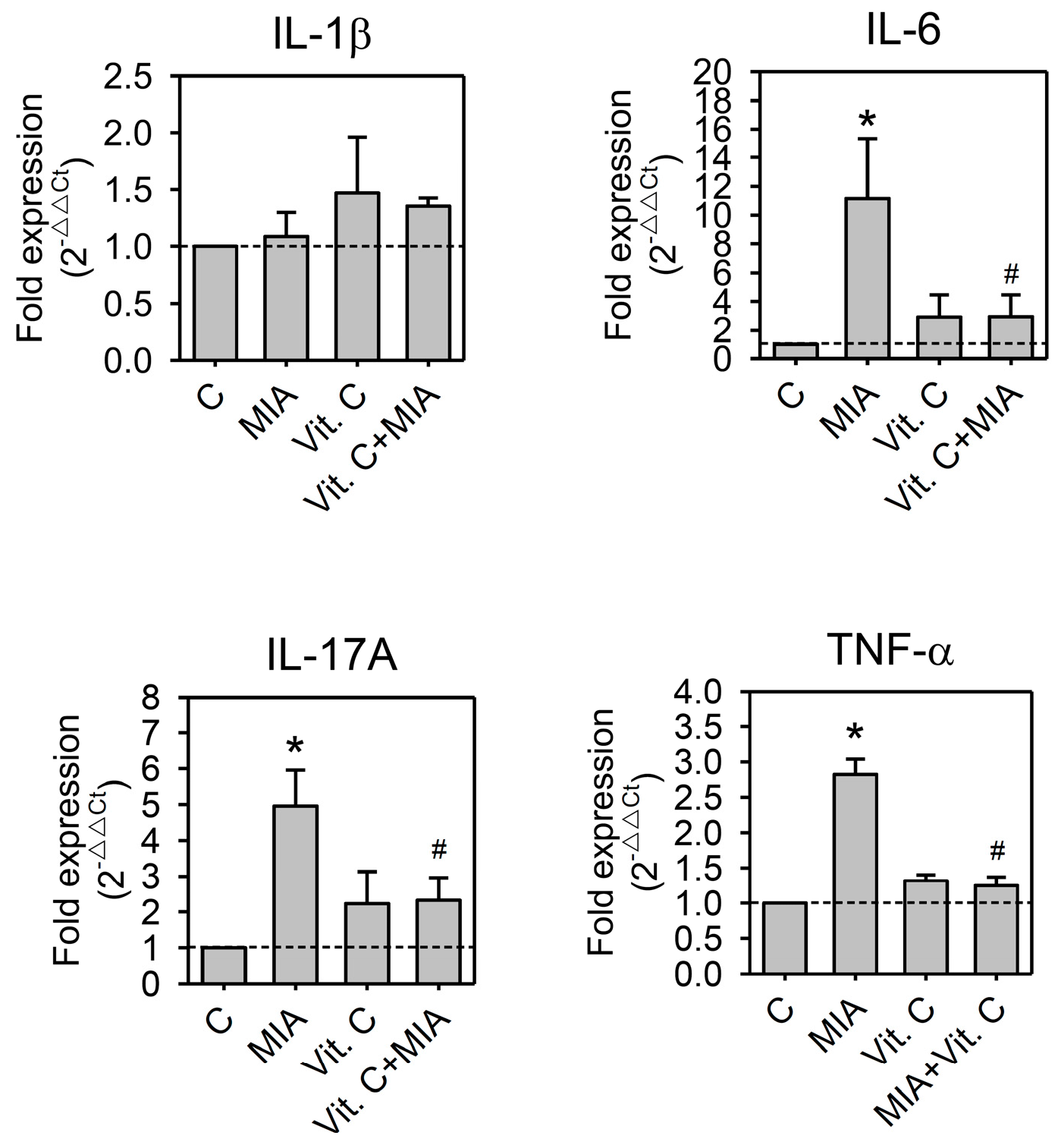
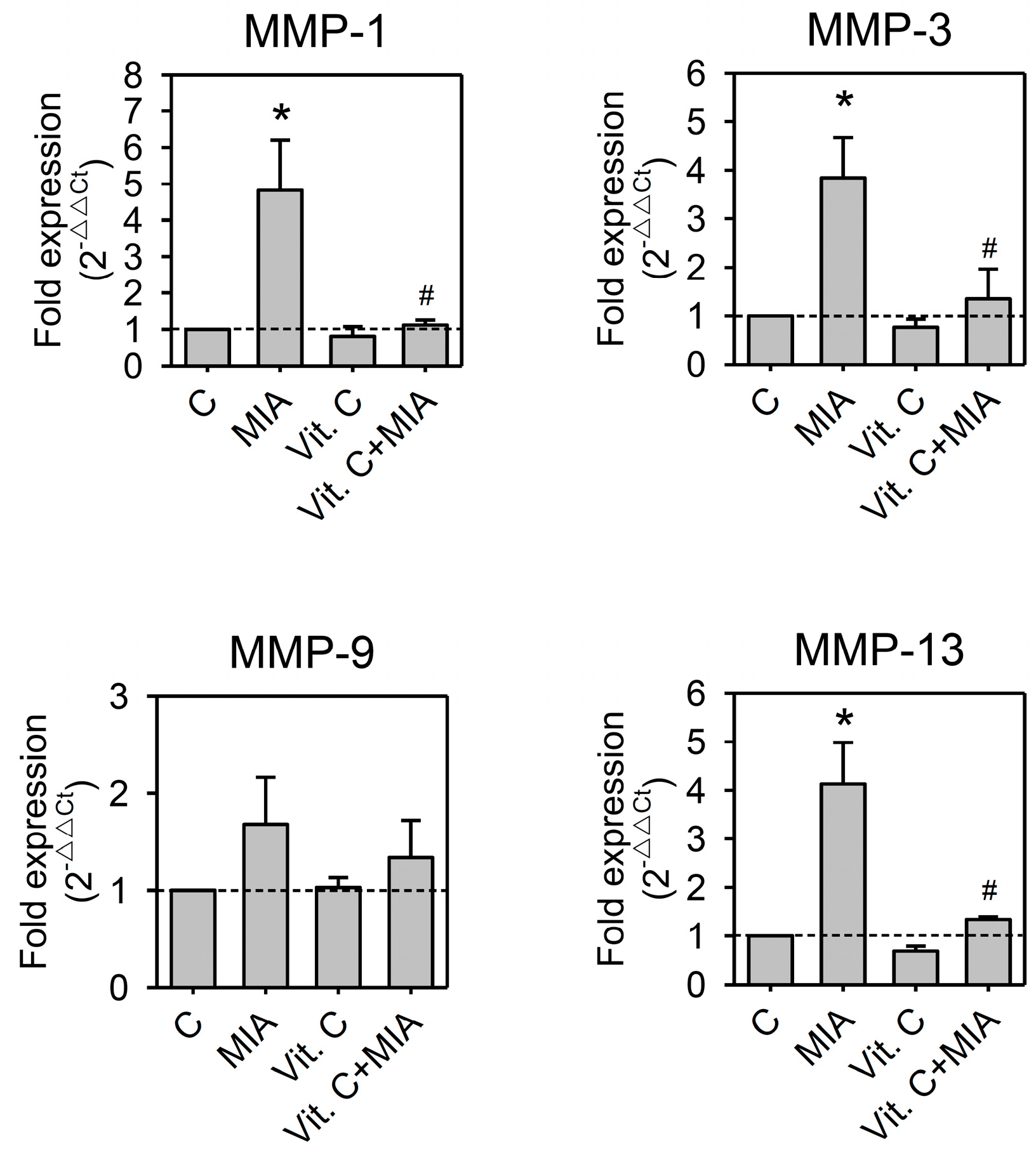
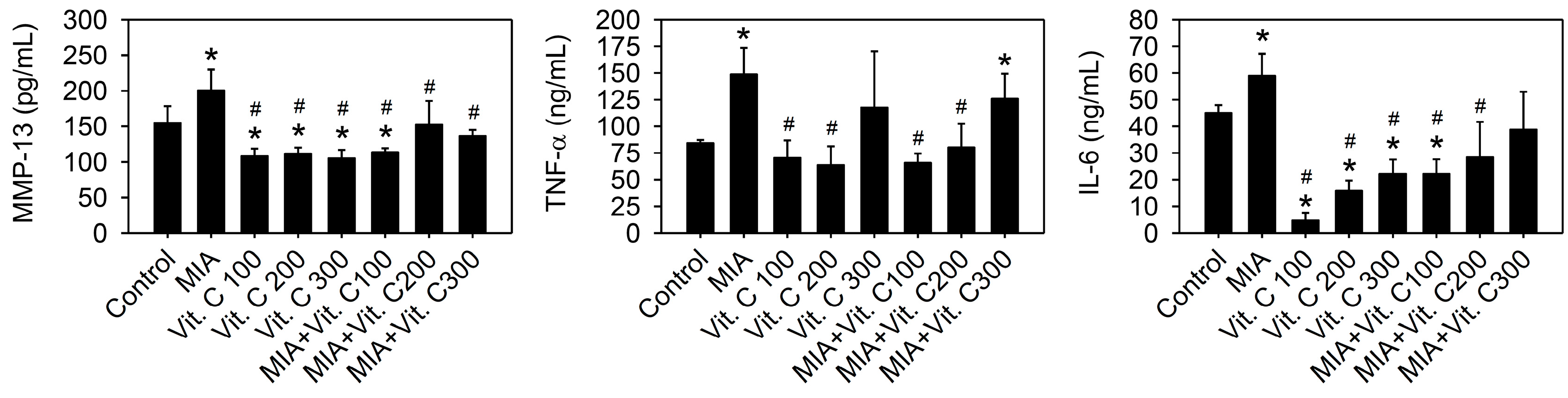
2.5. Expression of Pro-Inflammation Cytokine and MMP
2.6. Articular Cartilage Loss of the MIA-Induced OA in Rats
3. Discussion
4. Materials and Methods
4.1. Reagents and Antibodies
4.2. Cell Culture
4.3. Cell Viability Assay and Morphology
4.4. Measurement of Reactive Oxygen Species (ROS)
4.5. Detection of Apoptosis and Cell Cycle Progress
4.6. Western Blot Analysis
4.7. Glycosaminoglycans Staining
4.8. Quantitative Real-Time PCR Analysis
4.9. Animals and Treatments
4.10. Histopathology of Joint Tissues: Safranin O and Fast Green Staining
4.11. Serum Biomarker Measurements
4.12. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| OA | Osteoarthritis |
| MIA | Monosodium Iodoacetate |
| MMPs | Matrix Metalloproteinases |
| IL | Interleukin |
| TNF-α | Tumor Necrosis Factor-alpha |
| Vit. C | Vitamin C |
| ROS | Reactive Oxygen Species |
| ADAMTS | A Disintegrin And Metalloproteinase with Thrombospondin Motifs |
| MAPKs | Mitogen-Activated Protein Kinases |
References
- Rahmati, M.; Mobasheri, A.; Mozafari, M. Inflammatory mediators in osteoarthritis: A critical review of the state-of-the-art, current prospects, and future challenges. Bone 2016, 85, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Li, L.; Geng, C.; Gong, D.; Jiang, L.; Ishikawa, N.; Kajima, K.; Zhong, L. Monosodium iodoacetate induces apoptosis via the mitochondrial pathway involving ROS production and caspase activation in rat chondrocytes in vitro. J. Orthop. Res. 2013, 31, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Goldring, S.R. Osteoarthritis. J. Cell. Physiol. 2007, 213, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Farahat, M.N.; Yanni, G.; Poston, R.; Panayi, G.S. Cytokine expression in synovial membranes of patients with rheumatoid arthritis and osteoarthritis. Ann. Rheum. Dis. 1993, 52, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Thalhamer, T.; McGrath, M.A.; Harnett, M.M. MAPKs and their relevance to arthritis and inflammation. Rheumatology 2008, 47, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Zamli, Z.; Sharif, M. Chondrocyte apoptosis: A cause or consequence of osteoarthritis? Int. J. Rheum. Dis. 2011, 14, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Musumeci, G.; Castrogiovanni, P.; Trovato, F.M.; Weinberg, A.M.; Al-Wasiyah, M.K.; Alqahtani, M.H.; Mobasheri, A. Biomarkers of Chondrocyte Apoptosis and Autophagy in Osteoarthritis. Int. J. Mol. Sci. 2015, 16, 20560–20575. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.S.; Kim, H.A. Chondrocyte Apoptosis in the Pathogenesis of Osteoarthritis. Int. J. Mol. Sci. 2015, 16, 26035–26054. [Google Scholar] [CrossRef] [PubMed]
- Frei, B.; England, L.; Ames, B.N. Ascorbate is an outstanding antioxidant in human blood plasma. Proc. Natl. Acad. Sci. USA 1989, 86, 6377–6381. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Katz, A.; Wang, Y.; Eck, P.; Kwon, O.; Lee, J.H.; Chen, S.; Corpe, C.; Dutta, A.; Dutta, S.K.; et al. Vitamin C as an antioxidant: Evaluation of its role in disease prevention. J. Am. Coll. Nutr. 2003, 22, 18–35. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zeng, C.; Wei, J.; Yang, T.; Gao, S.G.; Li, Y.S.; Lei, G.H. Associations between dietary antioxidants intake and radiographic knee osteoarthritis. Clin. Rheumatol. 2016, 35, 1585–1592. [Google Scholar] [CrossRef] [PubMed]
- McAlindon, T.E.; Jacques, P.; Zhang, Y.; Hannan, M.T.; Aliabadi, P.; Weissman, B.; Rush, D.; Levy, D.; Felson, D.T. Do antioxidant micronutrients protect against the development and progression of knee osteoarthritis? Arthritis Rheum. 1996, 39, 648–656. [Google Scholar] [CrossRef] [PubMed]
- Chang, Z.; Huo, L.; Li, P.; Wu, Y.; Zhang, P. Ascorbic acid provides protection for human chondrocytes against oxidative stress. Mol. Med. Rep. 2015, 12, 7086–7092. [Google Scholar] [CrossRef] [PubMed]
- Gebauer, M.; Saas, J.; Sohler, F.; Haag, J.; Soder, S.; Pieper, M.; Bartnik, E.; Beninga, J.; Zimmer, R.; Aigner, T. Comparison of the chondrosarcoma cell line SW1353 with primary human adult articular chondrocytes with regard to their gene expression profile and reactivity to IL-1β. Osteoarthr. Cartil. OARS Osteoarthr. Res. Soc. 2005, 13, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Guzman, R.E.; Evans, M.G.; Bove, S.; Morenko, B.; Kilgore, K. Mono-iodoacetate-induced histologic changes in subchondral bone and articular cartilage of rat femorotibial joints: An animal model of osteoarthritis. Toxicol. Pathol. 2003, 31, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Liu, S.; Huang, J.; Guo, W.; Chen, J.; Zhang, L.; Zhao, B.; Peng, J.; Wang, A.; Wang, Y.; et al. The ECM-cell interaction of cartilage extracellular matrix on chondrocytes. BioMed Res. Int. 2014, 2014, 648459. [Google Scholar] [CrossRef] [PubMed]
- Wojdasiewicz, P.; Poniatowski, L.A.; Szukiewicz, D. The role of inflammatory and anti-inflammatory cytokines in the pathogenesis of osteoarthritis. Mediat. Inflamm. 2014, 2014, 561459. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; Pelletier, J.P.; Fahmi, H. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Vincenti, M.P.; Brinckerhoff, C.E. Transcriptional regulation of collagenase (MMP-1, MMP-13) genes in arthritis: Integration of complex signaling pathways for the recruitment of gene-specific transcription factors. Arthritis Res. 2002, 4, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Meszaros, E.; Malemud, C.J. Prospects for treating osteoarthritis: Enzyme-protein interactions regulating matrix metalloproteinase activity. Ther. Adv. Chronic Dis. 2012, 3, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Poole, A.R.; Rizkalla, G.; Ionescu, M.; Reiner, A.; Brooks, E.; Rorabeck, C.; Bourne, R.; Bogoch, E. Osteoarthritis in the human knee: A dynamic process of cartilage matrix degradation, synthesis and reorganization. Agents Actions Suppl. 1993, 39, 3–13. [Google Scholar] [PubMed]
- Kobayashi, K.; Imaizumi, R.; Sumichika, H.; Tanaka, H.; Goda, M.; Fukunari, A.; Komatsu, H. Sodium iodoacetate-induced experimental osteoarthritis and associated pain model in rats. J. Vet. Med. Sci. Jpn. Soc. Vet. Sci. 2003, 65, 1195–1199. [Google Scholar] [CrossRef]
- Bowles, R.D.; Mata, B.A.; Bell, R.D.; Mwangi, T.K.; Huebner, J.L.; Kraus, V.B.; Setton, L.A. In vivo luminescence imaging of NF-κB activity and serum cytokine levels predict pain sensitivities in a rodent model of osteoarthritis. Arthritis Rheumatol. 2014, 66, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Marcu, K.B. Cartilage homeostasis in health and rheumatic diseases. Arthritis Res. Ther. 2009, 11, 224. [Google Scholar] [CrossRef] [PubMed]
- Poole, A.R.; Kojima, T.; Yasuda, T.; Mwale, F.; Kobayashi, M.; Laverty, S. Composition and structure of articular cartilage: A template for tissue repair. Clin. Orthop. Relat. Res. 2001, 391, S26–S33. [Google Scholar] [CrossRef]
- Mariani, E.; Pulsatelli, L.; Facchini, A. Signaling pathways in cartilage repair. Int. J. Mol. Sci. 2014, 15, 8667–8698. [Google Scholar] [CrossRef] [PubMed]
- Loeser, R.F. Aging and osteoarthritis: The role of chondrocyte senescence and aging changes in the cartilage matrix. Osteoarthr. Cartil. OARS Osteoarthr. Res. Soc. 2009, 17, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Otero, M. Inflammation in osteoarthritis. Curr. Opin. Rheumatol. 2011, 23, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Abramson, S.B.; Attur, M. Developments in the scientific understanding of osteoarthritis. Arthritis Res. Ther. 2009, 11, 227. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B. Osteoarthritis and cartilage: The role of cytokines. Curr. Rheumatol. Rep. 2000, 2, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Food and Nutrition Board of the Institute of Medicine. Dietary Reference Intakes (DRIs): Recommended Dietary Allowances; United States National Academy of Sciences: Washington, DC, USA, 2013. [Google Scholar]
- Lindblad, M.; Tveden-Nyborg, P.; Lykkesfeldt, J. Regulation of vitamin C homeostasis during deficiency. Nutrients 2013, 5, 2860–2879. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.; Conry-Cantilena, C.; Wang, Y.; Welch, R.W.; Washko, P.W.; Dhariwal, K.R.; Park, J.B.; Lazarev, A.; Graumlich, J.F.; King, J.; et al. Vitamin C pharmacokinetics in healthy volunteers: Evidence for a recommended dietary allowance. Proc. Natl. Acad. Sci. USA 1996, 93, 3704–3709. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Chaganti, R.K.; Tolstykh, I.; Javaid, M.K.; Neogi, T.; Torner, J.; Curtis, J.; Jacques, P.; Felson, D.; Lane, N.E.; Nevitt, M.C.; et al. High plasma levels of vitamin C and E are associated with incident radiographic knee osteoarthritis. Osteoarthr. Cartil. OARS Osteoarthr. Res. Soc. 2014, 22, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, B.S.; Huang, L.W.; Su, S.J.; Cheng, H.L.; Hu, Y.C.; Hung, T.C.; Chang, K.L. Combined arginine and ascorbic acid treatment induces apoptosis in the hepatoma cell line HA22T/VGH and changes in redox status involving the pentose phosphate pathway and reactive oxygen and nitrogen species. J. Nutr. Biochem. 2011, 22, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.L.; Hung, T.C.; Hsieh, B.S.; Chen, Y.H.; Chen, T.F.; Cheng, H.L. Zinc at pharmacologic concentrations affects cytokine expression and induces apoptosis of human peripheral blood mononuclear cells. Nutrition 2006, 22, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Hung, T.C.; Huang, L.W.; Su, S.J.; Hsieh, B.S.; Cheng, H.L.; Hu, Y.C.; Chen, Y.H.; Hwang, C.C.; Chang, K.L. Hemeoxygenase-1 expression in response to arecoline-induced oxidative stress in human umbilical vein endothelial cells. Int. J. Cardiol. 2011, 151, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.L.; Su, S.J.; Huang, L.W.; Hsieh, B.S.; Hu, Y.C.; Hung, T.C.; Chang, K.L. Arecoline induces HA22T/VGH hepatoma cells to undergo anoikis—Involvement of STAT3 and RhoA activation. Mol. Cancer 2010, 9, 126. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Gerwin, N.; Bendele, A.M.; Glasson, S.; Carlson, C.S. The OARSI histopathology initiative—Recommendations for histological assessments of osteoarthritis in the rat. Osteoarthr. Cartil. OARS Osteoarthr. Res. Soc. 2010, 18, 24–34. [Google Scholar] [CrossRef] [PubMed]
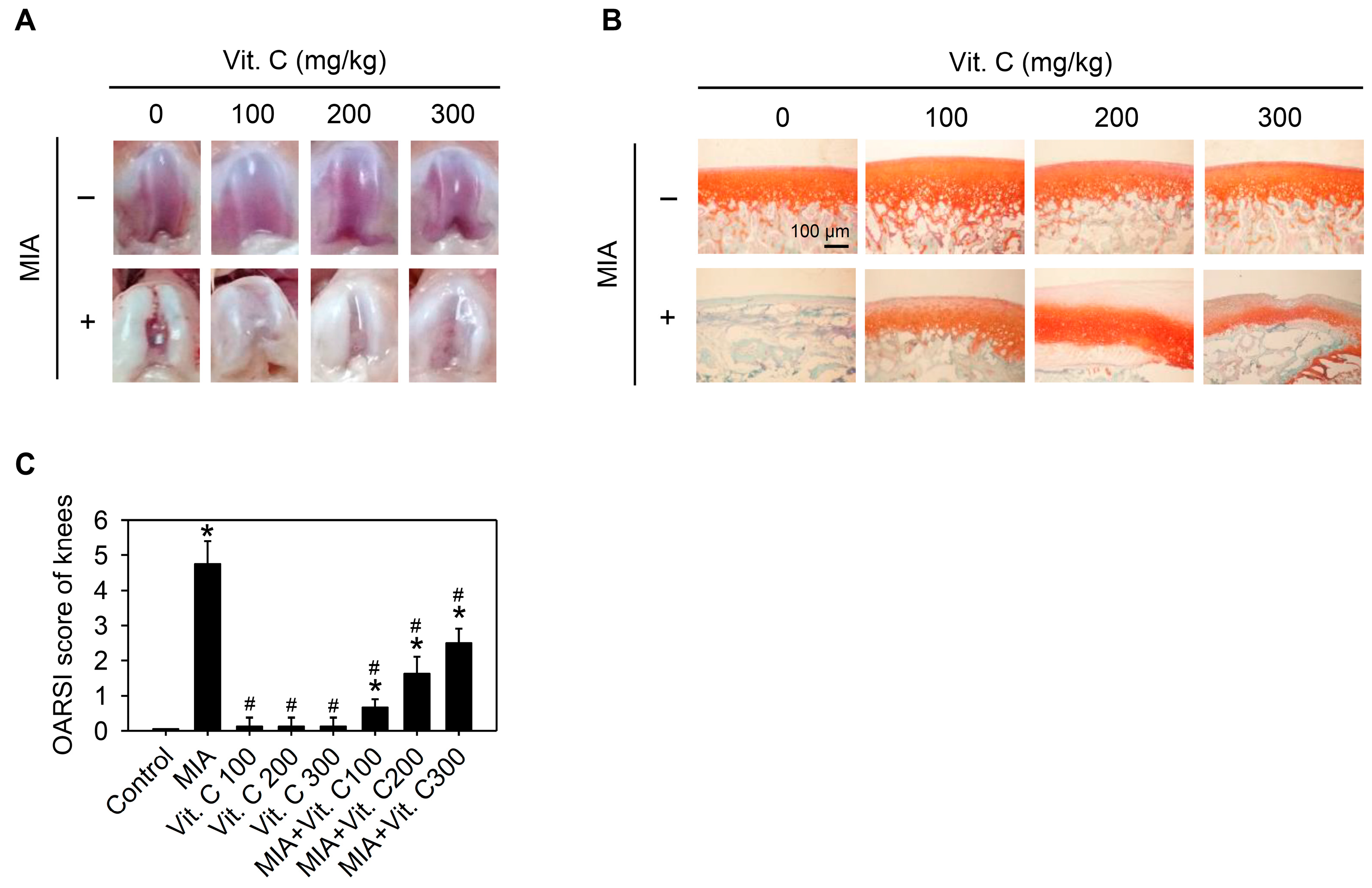
 : enhanced by MIA; Red
: enhanced by MIA; Red  : decreased by MIA; Green
: decreased by MIA; Green  : enhanced by Vitamin C; Green
: enhanced by Vitamin C; Green  : decreased by Vitamin C.
: decreased by Vitamin C.
 : enhanced by MIA; Red
: enhanced by MIA; Red  : decreased by MIA; Green
: decreased by MIA; Green  : enhanced by Vitamin C; Green
: enhanced by Vitamin C; Green  : decreased by Vitamin C.
: decreased by Vitamin C.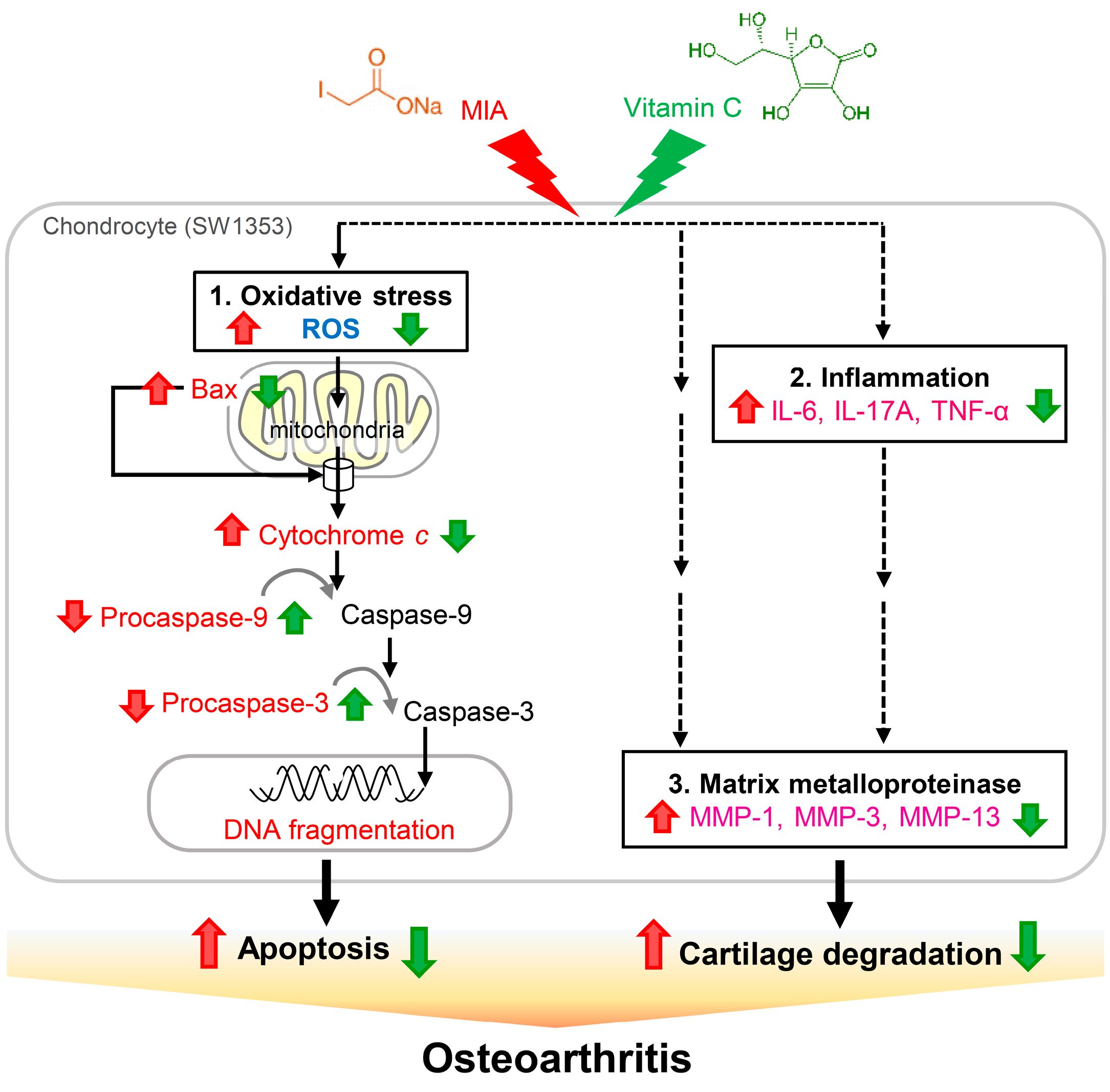
| Primer Name | NCBI Reference Sequence | Primer Sequence (5′→3′) |
|---|---|---|
| β-actin | NM_001101.3 | F: ATCGGCGGCTCCATCCTG |
| R: ACTCGTCATACTCCTGCTTGC | ||
| MMP-1 | NM_002421.3 | F: AGATGTGGAGTGCCTGATGTG |
| R: CTTGACCCTCAGAGACCTTGG | ||
| MMP-3 | NM_002422.3 | F: CCACTCTATCACTCACTCACAG |
| R: GACAGCATCAAAGGACAAAGC | ||
| MMP-9 | NM_004994.2 | F: CTGGTCCTGGTGCTCCTG |
| R: TGCCTGTCGGTGAGATTGG | ||
| MMP-13 | NM_002427.3 | F: GACCCTGGAGCACTCATGTTTC |
| R: TCCTCGGAGACTGGTAATGGC | ||
| IL-1β | NM_000576.2 | F: TGATGGCTTATTACAGTGGCAATG |
| R: GTAGTGGTGGTCGGAGATTCG | ||
| IL-6 | NM_000600.4 | F: ACCCCCAATAAATATAGGACTGGA |
| R: GAGAAGGCAACTGGACCGAA | ||
| TNF-α | NM_000594.3 | F: TCAGCAAGGACAGCAGAGGAC |
| R: GGAGCCGTGGGTCAGTATGTG | ||
| IL-17A | NM_002190.2 | F: GGCTGGAGAAGATACTGGTGTC |
| R: AGGCTGTCTTTGAAGGATGAGG |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiu, P.-R.; Hu, Y.-C.; Huang, T.-C.; Hsieh, B.-S.; Yeh, J.-P.; Cheng, H.-L.; Huang, L.-W.; Chang, K.-L. Vitamin C Protects Chondrocytes against Monosodium Iodoacetate-Induced Osteoarthritis by Multiple Pathways. Int. J. Mol. Sci. 2017, 18, 38. https://doi.org/10.3390/ijms18010038
Chiu P-R, Hu Y-C, Huang T-C, Hsieh B-S, Yeh J-P, Cheng H-L, Huang L-W, Chang K-L. Vitamin C Protects Chondrocytes against Monosodium Iodoacetate-Induced Osteoarthritis by Multiple Pathways. International Journal of Molecular Sciences. 2017; 18(1):38. https://doi.org/10.3390/ijms18010038
Chicago/Turabian StyleChiu, Pu-Rong, Yu-Chen Hu, Tzu-Ching Huang, Bau-Shan Hsieh, Jou-Pei Yeh, Hsiao-Ling Cheng, Li-Wen Huang, and Kee-Lung Chang. 2017. "Vitamin C Protects Chondrocytes against Monosodium Iodoacetate-Induced Osteoarthritis by Multiple Pathways" International Journal of Molecular Sciences 18, no. 1: 38. https://doi.org/10.3390/ijms18010038
APA StyleChiu, P.-R., Hu, Y.-C., Huang, T.-C., Hsieh, B.-S., Yeh, J.-P., Cheng, H.-L., Huang, L.-W., & Chang, K.-L. (2017). Vitamin C Protects Chondrocytes against Monosodium Iodoacetate-Induced Osteoarthritis by Multiple Pathways. International Journal of Molecular Sciences, 18(1), 38. https://doi.org/10.3390/ijms18010038