Bajijiasu Abrogates Osteoclast Differentiation via the Suppression of RANKL Signaling Pathways through NF-κB and NFAT
Abstract
:1. Introduction
2. Results
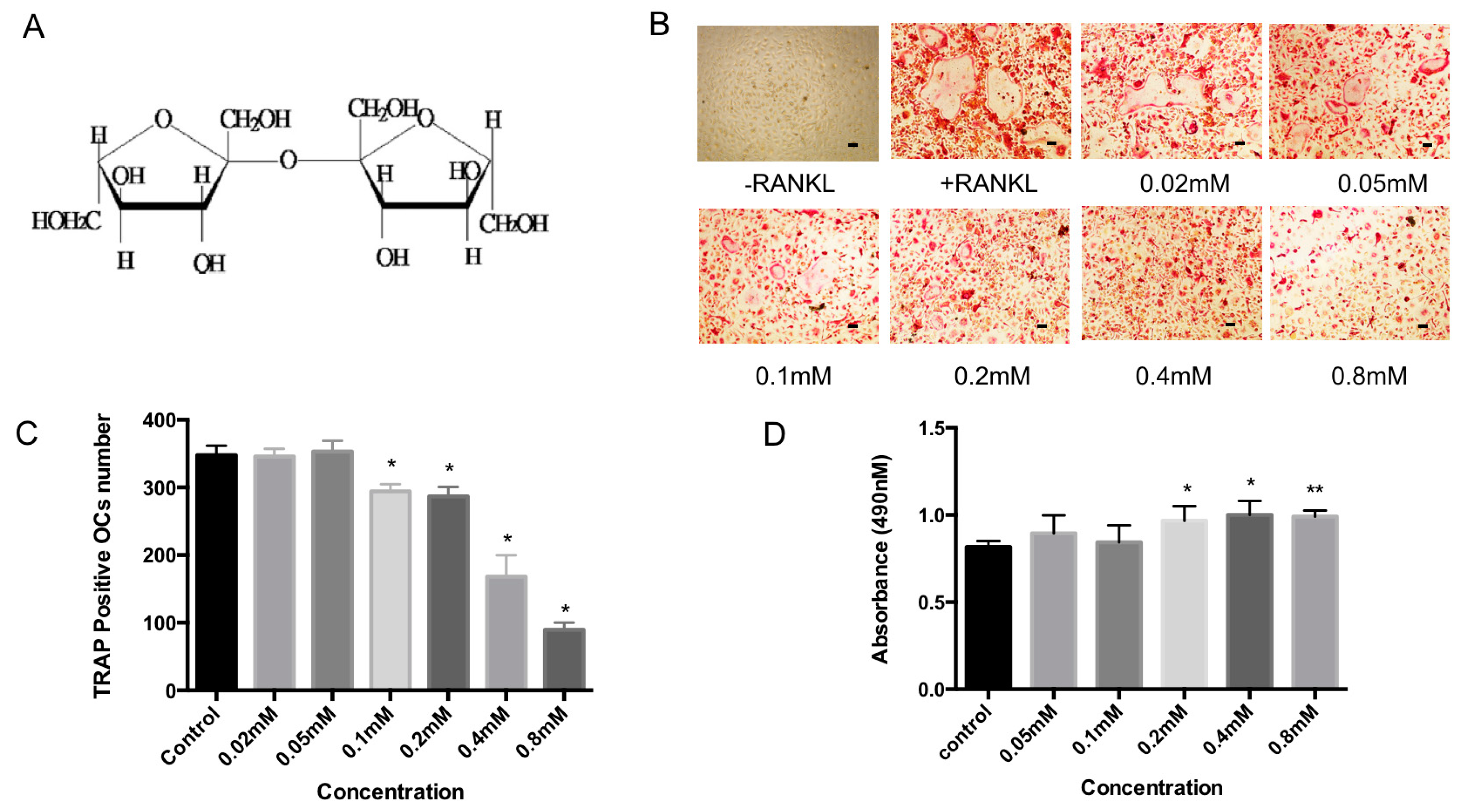
2.1. Bajijiasu Inhibits RANKL-Induced Osteoclastogenesis
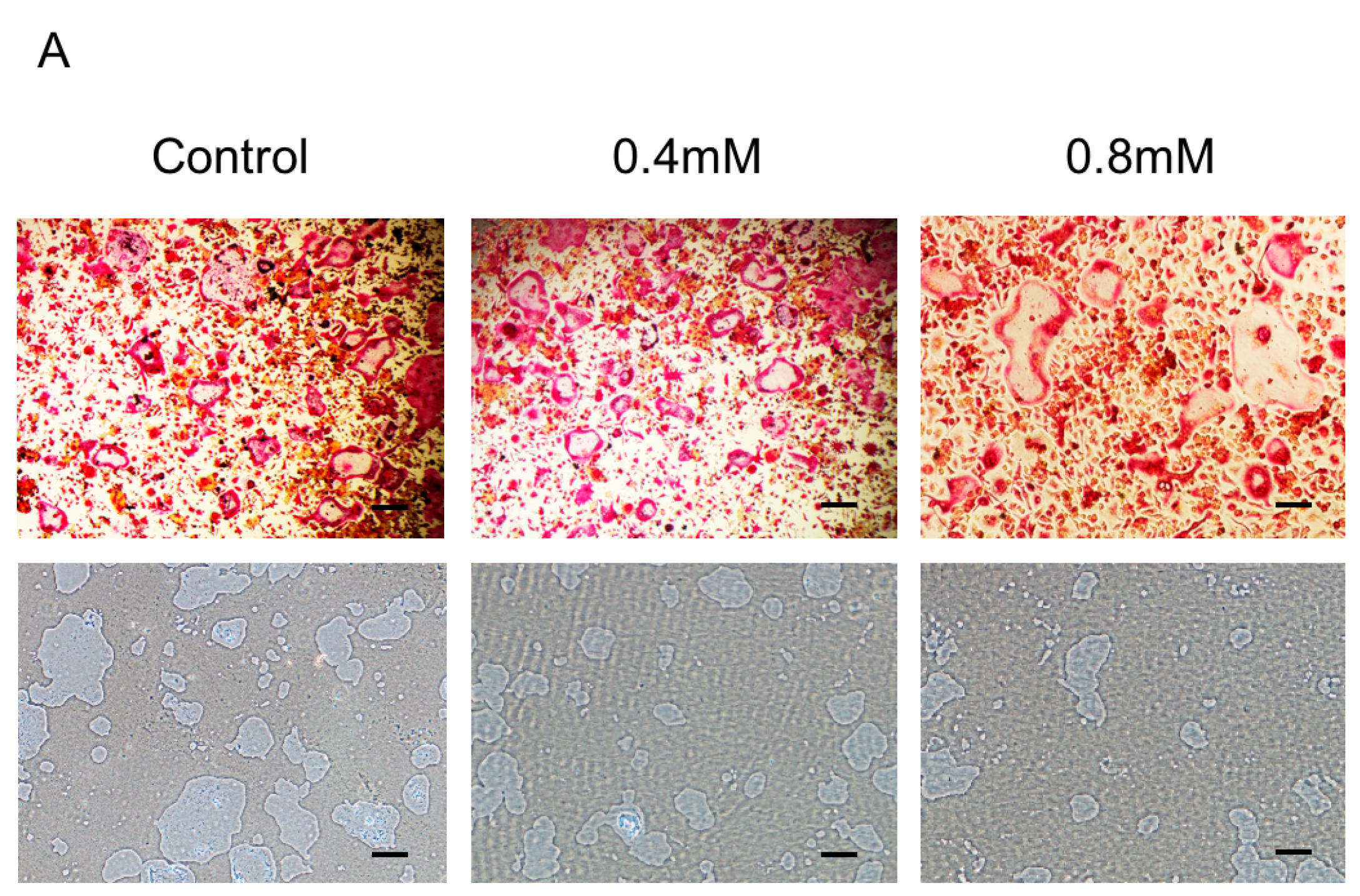
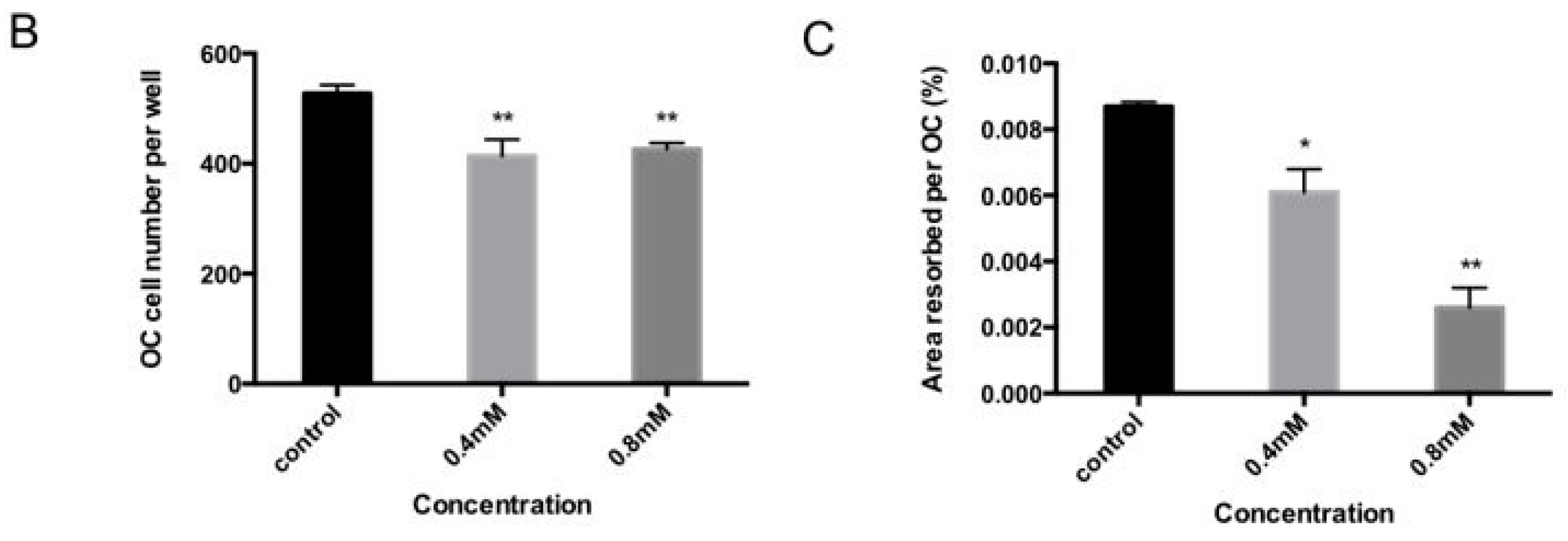
2.2. Bajijiasu Suppresses RANKL-Induced Osteoclast Function
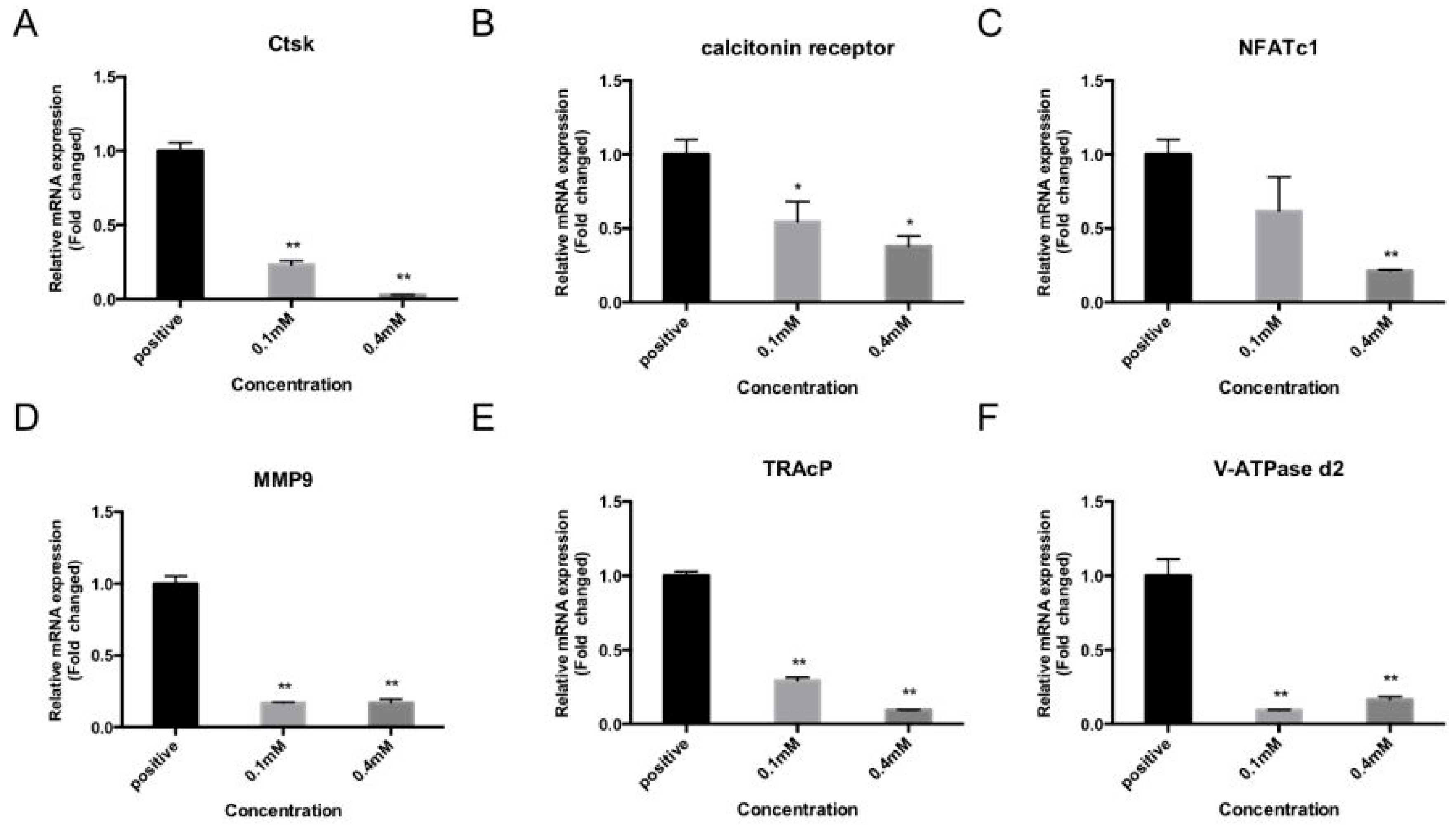
2.3. Bajijiasu Suppresses RANKL-Induced Osteoclast-Associated Gene Expression
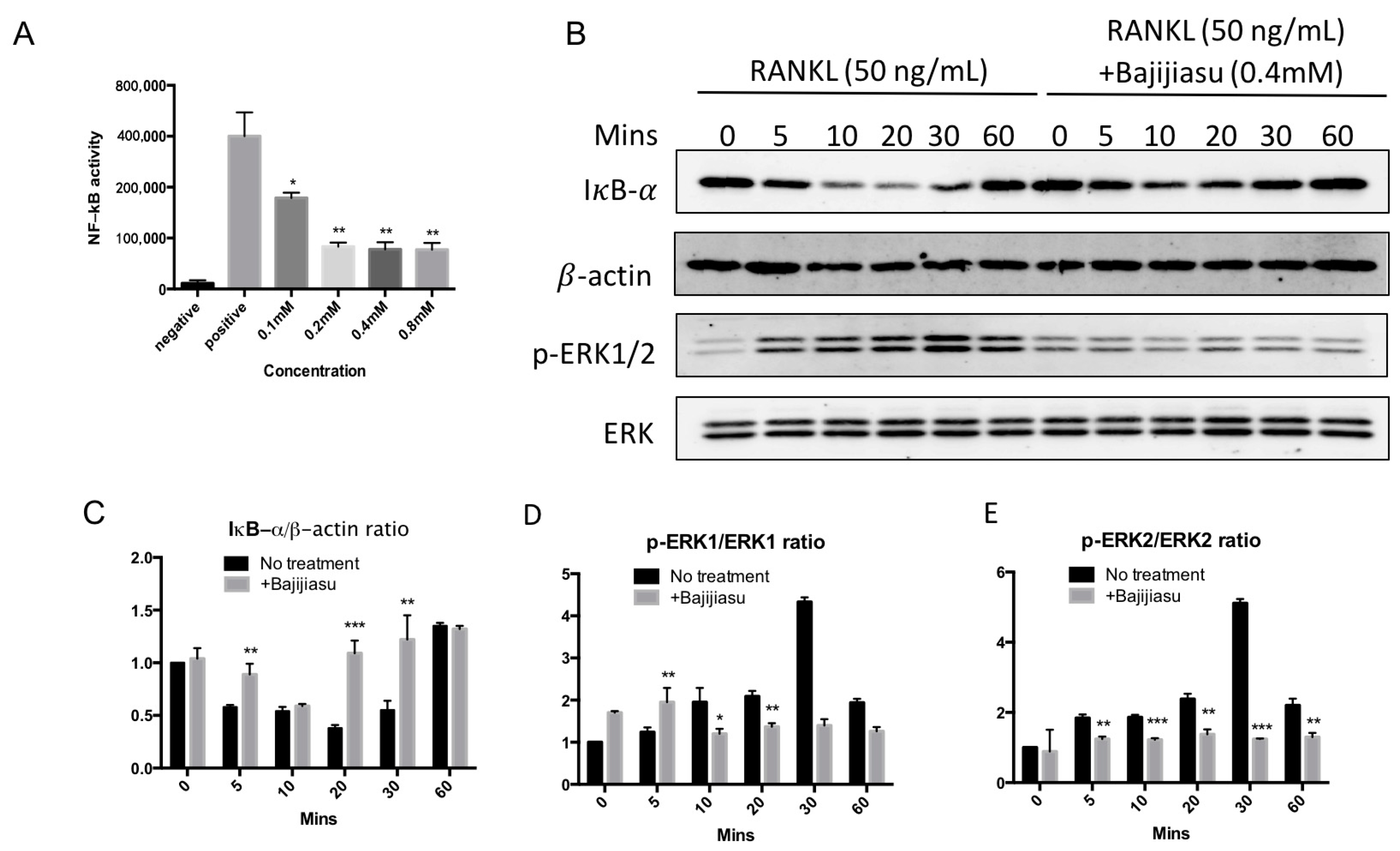
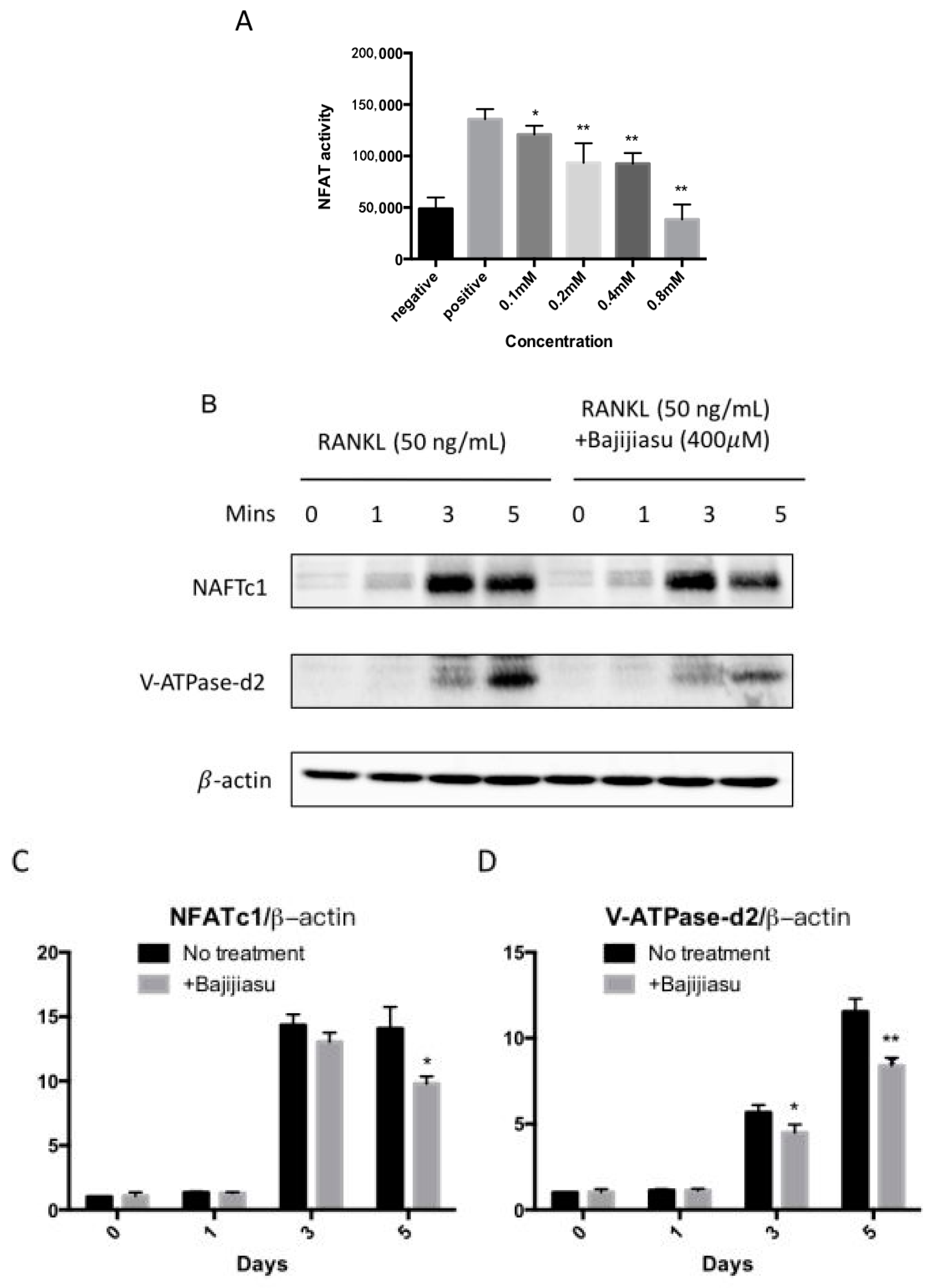
2.4. Bajijiasu Suppresses RANKL-Induced NF-κB and NFAT Transcriptional Activation
2.5. Bajijiasu Inhibits RANKL-Induced IκB-α, P-ERK, NFATc1 and V-ATPase d2 Protein Expression
3. Discussion
4. Materials and Methods
4.1. Materials
4.1.1. Reagents and Antibodies
4.1.2. Cell Culture
4.2. Compound Screening Assay and In Vitro Osteoclastogenesis Assay
4.3. Cytotoxicity Assay
4.4. Hydroxyapatite Resorption Assay
4.5. Luciferase Reporter Gene Assay for NF-κB and NFAT
4.6. Real Time Polymerase Chain Reaction (Real-Time PCR)
4.7. Western Blot Assay
4.8. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kurihara, N. The origin of osteoclasts. Bull. Kanagawa Dent. Coll. 1990, 18, 161–164. [Google Scholar] [PubMed]
- Kerachian, M.A.; Seguin, C.; Harvey, E.J. Glucocorticoids in osteonecrosis of the femoral head: A new understanding of the mechanisms of action. J. Steroid Biochem. Mol. Biol. 2009, 114, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Manolagas, S.C.; Jilka, R.L. Bone marrow, cytokines, and bone remodeling. Emerging insights into the pathophysiology of osteoporosis. N. Engl. J. Med. 1995, 332, 305–311. [Google Scholar] [PubMed]
- Boyle, W.J.; Simonet, W.S.; Lacey, D.L. Osteoclast differentiation and activation. Nature 2003, 423, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Baud’huin, M.; Duplomb, L.; Ruiz Velasco, C.; Fortun, Y.; Heymann, D.; Padrines, M. Key roles of the OPG-RANK-RANKL system in bone oncology. Expert Rev. Anticancer Ther. 2007, 7, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H. The role of NFAT in osteoclast formation. Ann. N. Y. Acad. Sci. 2007, 1116, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Whelan, A.M.; Jurgens, T.M.; Bowles, S.K. Natural health products in the prevention and treatment of osteoporosis: Systematic review of randomized controlled trials. Ann. Pharmacother. 2006, 40, 836–849. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Cheng, J.; Liu, Q.; Zhou, L.; Kenny, J.; Wang, T.; Lin, X.; Yuan, J.; Quinn, J.M.; Tickner, J.; et al. Neohesperidin suppresses osteoclast differentiation, bone resorption and ovariectomised-induced osteoporosis in mice. Mol. Cell. Endocrinol. 2017, 439, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Zhou, L.; Zhao, J.; Liu, Q.; Yang, M.; Tan, R.; Xu, J.; Zhang, G.; Quinn, J.M.; Tickner, J.; Huang, Y.; Xu, J. Eriodictyol Inhibits RANKL-Induced Osteoclast Formation and Function Via Inhibition of NFATc1 Activity. J. Cell. Physiol. 2016, 231, 1983–1993. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Yuan, L.; Xu, Y.K.; Yang, M.; Zhao, Y.M.; Luo, Z.P. Antistress effect of oligosaccharides extracted from Morinda officinalis in mice and rats. Acta Pharmacol. Sin. 2001, 22, 1084–1088. [Google Scholar] [PubMed]
- Li, Y.F.; Liu, Y.Q.; Yang, M.; Wang, H.L.; Huang, W.C.; Zhao, Y.M.; Luo, Z.P. The cytoprotective effect of inulin-type hexasaccharide extracted from Morinda officinalis on PC12 cells against the lesion induced by corticosterone. Life Sci. 2004, 75, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- Bao, L.; Qin, L.; Liu, L.; Wu, Y.; Han, T.; Xue, L.; Zhang, Q. Anthraquinone compounds from Morinda officinalis inhibit osteoclastic bone resorption in vitro. Chem. Biol. Interact. 2011, 194, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Qin, L.P.; Han, T.; Wu, Y.B.; Zhang, Q.Y.; Zhang, H. Inhibitory effects of morinda officinalis extract on bone loss in ovariectomized rats. Molecules 2009, 14, 2049–2061. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.B.; Zheng, C.J.; Qin, L.P.; Sun, L.N.; Han, T.; Jiao, L.; Zhang, Q.Y.; Wu, J.Z. Antiosteoporotic activity of anthraquinones from Morinda officinalis on osteoblasts and osteoclasts. Molecules 2009, 14, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, Q.; Yang, H.; Liu, W.; Zhang, N.; Qin, L.; Xin, H. Monotropein isolated from the roots of Morinda officinalis increases osteoblastic bone formation and prevents bone loss in ovariectomized mice. Fitoterapia 2016, 110, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.Q.; Chen, D.L.; Lin, F.H.; Lin, L.; Shuai, O.; Wang, J.Y.; Qi, L.K.; Zhang, P. Effect of bajijiasu isolated from Morinda officinalis F. C. how on sexual function in male mice and its antioxidant protection of human sperm. J. Ethnopharmacol. 2015, 164, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.L.; Zhang, P.; Lin, L.; Zhang, H.M.; Deng, S.D.; Wu, Z.Q.; Ou, S.; Liu, S.H.; Wang, J.Y. Protective effects of bajijiasu in a rat model of Aβ25–35-induced neurotoxicity. J. Ethnopharmacol. 2014, 154, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.L.; Ning, L.; Li, L.; Deng, S.D.; Zhang, H.M.; Liu, S.H. Method to detect the variants of the erythrocyte in a rat model of Aβ25–35-induced neurotoxicity based on micro-Raman spectroscopy. J. Biomed. Opt. 2013, 18, 118003. [Google Scholar] [PubMed]
- Shinohara, M.; Takayanagi, H. Novel osteoclast signaling mechanisms. Curr. Osteoporos. Rep. 2007, 5, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.Y.; Yoshida, H.; Sarosi, I.; Tan, H.L.; Timms, E.; Capparelli, C.; Morony, S.; Oliveira-dos-Santos, A.J.; Van, G.; Itie, A.; et al. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 1999, 397, 315–323. [Google Scholar] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Rachner, T.D.; Khosla, S.; Hofbauer, L.C. Osteoporosis: Now and the future. Lancet 2011, 377, 1276–1287. [Google Scholar] [CrossRef]
- Lippuner, K. The future of osteoporosis treatment—A research update. Swiss Med. Wkly. 2012, 142, w13624. [Google Scholar] [CrossRef] [PubMed]
- Baek, K.H.; Oh, K.W.; Lee, W.Y.; Lee, S.S.; Kim, M.K.; Kwon, H.S.; Rhee, E.J.; Han, J.H.; Song, K.H.; Cha, B.Y.; et al. Association of oxidative stress with postmenopausal osteoporosis and the effects of hydrogen peroxide on osteoclast formation in human bone marrow cell cultures. Calcif. Tissue Int. 2010, 87, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Yip, K.H.; Zheng, M.H.; Steer, J.H.; Giardina, T.M.; Han, R.; Lo, S.Z.; Bakker, A.J.; Cassady, A.I.; Joyce, D.A.; Xu, J. Thapsigargin modulates osteoclastogenesis through the regulation of RANKL-induced signaling pathways and reactive oxygen species production. J. Bone Miner. Res. 2005, 20, 1462–1471. [Google Scholar] [CrossRef] [PubMed]
- Lacey, D.L.; Timms, E.; Tan, H.L.; Kelley, M.J.; Dunstan, C.R.; Burgess, T.; Elliott, R.; Colombero, A.; Elliott, G.; Scully, S.; et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 1998, 93, 165–176. [Google Scholar] [CrossRef]
- Boyce, B.F.; Xing, L.; Franzoso, G.; Siebenlist, U. Required and nonessential functions of nuclear factor-κB in bone cells. Bone 1999, 25, 137–139. [Google Scholar] [CrossRef]
- Nakagomi, D.; Suzuki, K.; Nakajima, H. Critical roles of IκB kinase subunits in mast cell degranulation. Int. Arch. Allergy Immunol. 2012, 158 (Suppl. 1), 92–95. [Google Scholar] [CrossRef] [PubMed]
- Asagiri, M.; Takayanagi, H. The molecular understanding of osteoclast differentiation. Bone 2007, 40, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Hirata, A.; Tsuji, T.; Yamamoto, T. Role of osteoclast extracellular signal-regulated kinase (ERK) in cell survival and maintenance of cell polarity. J. Bone Miner. Res. 2003, 18, 1198–1205. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, F.; Nishimura, R.; Matsubara, T.; Tanaka, S.; Inoue, J.; Reddy, S.V.; Hata, K.; Yamashita, K.; Hiraga, T.; Watanabe, T.; et al. Critical roles of c-Jun signaling in regulation of NFAT family and RANKL-regulated osteoclast differentiation. J. Clin. Investig. 2004, 114, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Asagiri, M.; Sato, K.; Usami, T.; Ochi, S.; Nishina, H.; Yoshida, H.; Morita, I.; Wagner, E.F.; Mak, T.W.; Serfling, E.; et al. Autoamplification of NFATc1 expression determines its essential role in bone homeostasis. J. Exp. Med. 2005, 202, 1261–1269. [Google Scholar] [CrossRef] [PubMed]
- Aliprantis, A.O.; Ueki, Y.; Sulyanto, R.; Park, A.; Sigrist, K.S.; Sharma, S.M.; Ostrowski, M.C.; Olsen, B.R.; Glimcher, L.H. NFATc1 in mice represses osteoprotegerin during osteoclastogenesis and dissociates systemic osteopenia from inflammation in cherubism. J. Clin. Investig. 2008, 118, 3775–3789. [Google Scholar] [CrossRef] [PubMed]
- Takayanagi, H.; Kim, S.; Koga, T.; Nishina, H.; Isshiki, M.; Yoshida, H.; Saiura, A.; Isobe, M.; Yokochi, T.; Inoue, J.; et al. Induction and activation of the transcription factor NFATc1 (NFAT2) integrate RANKL signaling in terminal differentiation of osteoclasts. Dev. Cell 2002, 3, 889–901. [Google Scholar] [CrossRef]
- Feng, H.; Cheng, T.; Steer, J.H.; Joyce, D.A.; Pavlos, N.J.; Leong, C.; Kular, J.; Liu, J.; Feng, X.; Zheng, M.H.; Xu, J. Myocyte enhancer factor 2 and microphthalmia-associated transcription factor cooperate with NFATc1 to transactivate the V-ATPase d2 promoter during RANKL-induced osteoclastogenesis. J. Biol. Chem. 2009, 284, 14667–14676. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tan, J.W.; Huang, L.; Gao, X.H.; Laird, R.; Liu, D.; Wysocki, S.; Zheng, M.H. Cloning, sequencing, and functional characterization of the rat homologue of receptor activator of NF-κB ligand. J. Bone Miner. Res. 2000, 15, 2178–2186. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Steer, J.H.; Joyce, D.A.; Yip, K.H.; Zheng, M.H.; Xu, J. 12-O-tetradecanoylphorbol-13-acetate (TPA) inhibits osteoclastogenesis by suppressing RANKL-induced NF-κB activation. J. Bone Miner. Res. 2003, 18, 2159–2168. [Google Scholar] [CrossRef] [PubMed]
- Van der Kraan, A.G.; Chai, R.C.; Singh, P.P.; Lang, B.J.; Xu, J.; Gillespie, M.T.; Price, J.T.; Quinn, J.M. HSP90 inhibitors enhance differentiation and MITF (microphthalmia transcription factor) activity in osteoclast progenitors. Biochem. J. 2013, 451, 235–244. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, G.; Zhou, L.; Shi, X.; He, W.; Wang, H.; Wei, Q.; Chen, P.; Qi, L.; Tickner, J.; Lin, L.; et al. Bajijiasu Abrogates Osteoclast Differentiation via the Suppression of RANKL Signaling Pathways through NF-κB and NFAT. Int. J. Mol. Sci. 2017, 18, 203. https://doi.org/10.3390/ijms18010203
Hong G, Zhou L, Shi X, He W, Wang H, Wei Q, Chen P, Qi L, Tickner J, Lin L, et al. Bajijiasu Abrogates Osteoclast Differentiation via the Suppression of RANKL Signaling Pathways through NF-κB and NFAT. International Journal of Molecular Sciences. 2017; 18(1):203. https://doi.org/10.3390/ijms18010203
Chicago/Turabian StyleHong, Guoju, Lin Zhou, Xuguang Shi, Wei He, Haibin Wang, Qiushi Wei, Peng Chen, Longkai Qi, Jennifer Tickner, Li Lin, and et al. 2017. "Bajijiasu Abrogates Osteoclast Differentiation via the Suppression of RANKL Signaling Pathways through NF-κB and NFAT" International Journal of Molecular Sciences 18, no. 1: 203. https://doi.org/10.3390/ijms18010203
APA StyleHong, G., Zhou, L., Shi, X., He, W., Wang, H., Wei, Q., Chen, P., Qi, L., Tickner, J., Lin, L., & Xu, J. (2017). Bajijiasu Abrogates Osteoclast Differentiation via the Suppression of RANKL Signaling Pathways through NF-κB and NFAT. International Journal of Molecular Sciences, 18(1), 203. https://doi.org/10.3390/ijms18010203





