The Dark Side of Cell Fusion
Abstract
1. Introduction
2. Cancer Cell-Cell Fusion
2.1. Cancer Cell-Stromal Cell Fusion
2.1.1. Novel Hybrids
2.1.2. Metastasis
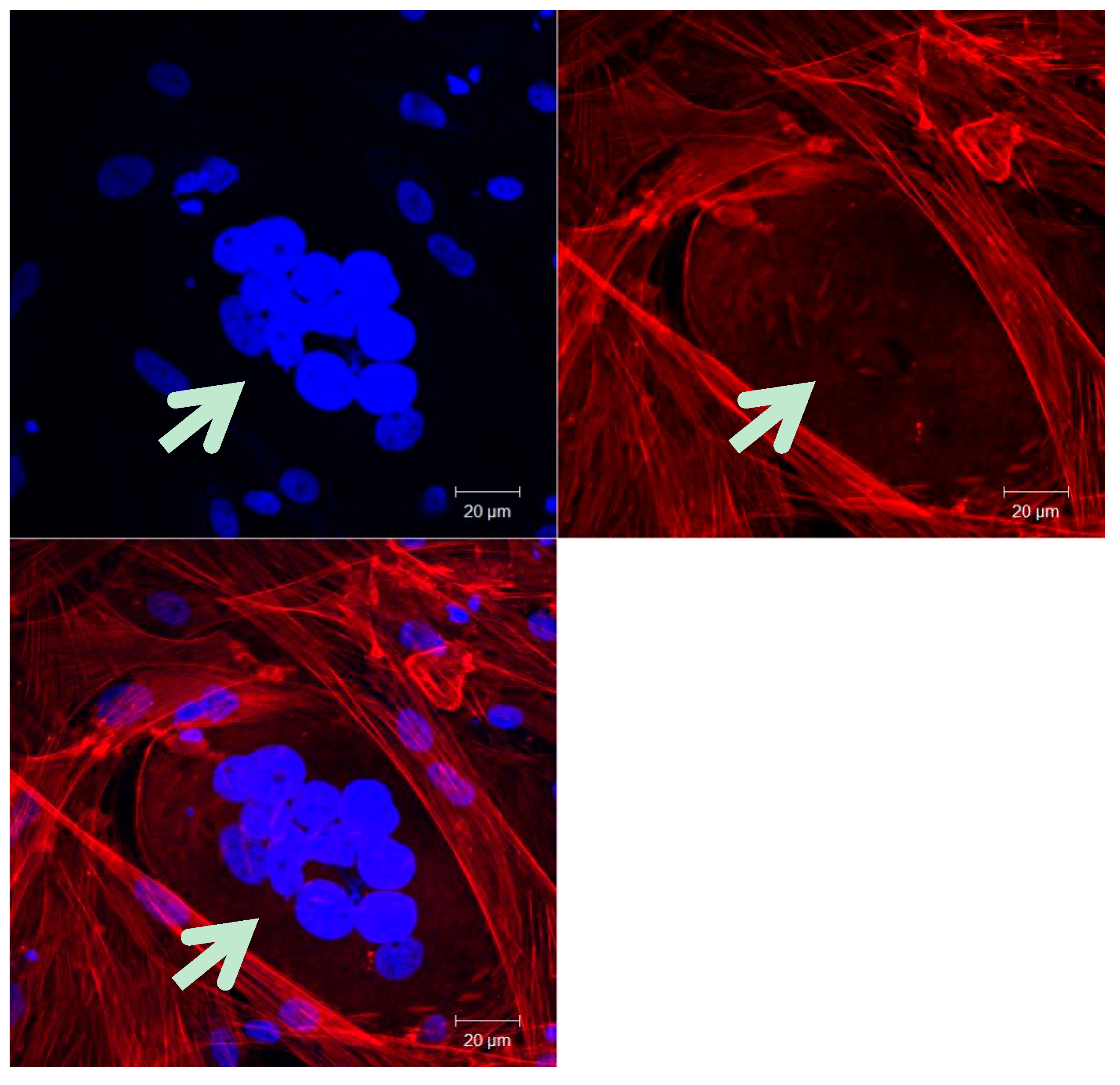
2.2. Cancer Cell-Cancer Cell Fusion
2.3. Cancer Stem Cells
3. Mechanisms of Cell Fusion
3.1. Proteins Associated with Cell-Cell Fusion
3.2. Syncytin-Dependent Fusion Mechanism
3.3. Model of Membrane Fusion
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| Abcb1a | ATP-binding cassette b1a multidrug transporter. |
| Abcb1b | ATP-binding cassette b1b multidrug transporter. |
| ADAM | A disintegrin and metalloproteinase |
| ASCT1 | Alanine, Serine and Cysteine selective transporter 1 |
| ASCT2 | Alanine, serine and cysteine selective transporter 2 |
| BMDCs | Bone marrow-derived cells |
| CD | Cluster of Differentiation |
| CSCs | Cancer stem cells |
| Cyt | Cytosolic tail |
| DC | Dendritic cells |
| DC-STAMP | Dendritic cell-specific transmembrane protein |
| DNA | Deoxyribonucleic acid |
| EMT | Epithelial to mesenchymal transition |
| Env | Envelope |
| FP | Fusion peptide |
| GnT-V | β 1,6 N-acetylglucosaminyltransferase V |
| GRP78 | 78 kDa glucose-regulated protein |
| HERV | Human endogenous retrovirus |
| HR1 | Heptad repeats |
| HR2 | Heptad repeats |
| LD | Linear dichroism |
| MC1R | Melanocortin 1 receptor |
| mCSCs | metastatic cancer stem cells |
| OC-STAMP | Osteoclast-stimulatory transmembrane protein |
| pCSCs | Primary cancer stem cells |
| PGCCs | Polyploid giant cancer cells |
| RBD | Receptor binding domain |
| SPARC | Secreted protein acidic and rich in cysteine |
| SU | Surface unit |
| t-SNARE | target-soluble N-ethylmaleimide-sensitive factor attachment protein receptor |
| TAM | Tumor-associated macrophages |
| TM | Transmembrane unit |
| Tm | Transmembrane anchorage domain |
| v-SNARE | vesicular-soluble N-ethylmaleimide-sensitive factor attachment protein receptor |
| ZO-1 | Zona ocludens-1 |
References
- Yokota, J. Tumor progression and metastasis. Carcinogenesis 2000, 21, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, H.; Nowak, M.A.; Vogelstein, B.; Lengauer, C. The significance of unstable chromosomes in colorectal cancer. Nat. Rev. Cancer 2003, 3, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Kang, Y. Cell fusion as a hidden force in tumor progression. Cancer Res. 2009, 69, 8536–8539. [Google Scholar] [CrossRef] [PubMed]
- Seyfried, T.N.; Huysentruyt, L.C. On the origin of cancer metastasis. Crit. Rev. Oncog. 2013, 18, 43–73. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, P.S.; Baylies, M.K.; Fleissner, A.; Helming, L.; Inoue, N.; Podbilewicz, B.; Wang, H.; Wong, M. Genetic basis of cell-cell fusion mechanisms. Trends Genet. 2013, 29, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Zanker, K.S.; Dittmar, T. Cancer (stem) cell differentiation: An inherent or acquired property? Med. Hypotheses 2015, 85, 1012–1018. [Google Scholar] [CrossRef] [PubMed]
- Coward, J.; Harding, A. Size does matter: Why polyploid tumor cells are critical drug targets in the war on cancer. Front. Oncol. 2014, 4, 123. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Mercado-Uribe, I.; Xing, Z.; Sun, B.; Kuang, J.; Liu, J. Generation of cancer stem-like cells through the formation of polyploid giant cancer cells. Oncogene 2014, 33, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Fox, D.T.; Duronio, R.J. Endoreplication and polyploidy: Insights into development and disease. Development 2013, 140, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Gentric, G.; Desdouets, C. Polyploidization in liver tissue. Am. J. Pathol. 2014, 184, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Davoli, T.; de Lange, T. The causes and consequences of polyploidy in normal development and cancer. Annu. Rev. Cell Dev. Biol. 2011, 27, 585–610. [Google Scholar] [CrossRef] [PubMed]
- Storchova, Z.; Pellman, D. From polyploidy to aneuploidy, genome instability and cancer. Nat. Rev. Mol. Cell Biol. 2004, 5, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Ganem, N.J.; Storchova, Z.; Pellman, D. Tetraploidy, aneuploidy and cancer. Curr. Opin. Genet. Dev. 2007, 17, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Storchova, Z.; Kuffer, C. The consequences of tetraploidy and aneuploidy. J. Cell Sci. 2008, 121, 3859–3866. [Google Scholar] [CrossRef] [PubMed]
- Dewhurst, S.M.; McGranahan, N.; Burrell, R.A.; Rowan, A.J.; Gronroos, E.; Endesfelder, D.; Joshi, T.; Mouradov, D.; Gibbs, P.; Ward, R.L.; et al. Tolerance of whole-genome doubling propagates chromosomal instability and accelerates cancer genome evolution. Cancer Discov. 2014, 4, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Ganem, N.J.; Pellman, D. Limiting the proliferation of polyploid cells. Cell 2007, 131, 437–440. [Google Scholar] [CrossRef] [PubMed]
- McClendon, A.K.; Dean, J.L.; Ertel, A.; Fu, Z.; Rivadeneira, D.B.; Reed, C.A.; Bourgo, R.J.; Witkiewicz, A.; Addya, S.; Mayhew, C.N.; et al. Rb and p53 cooperate to prevent liver tumorigenesis in response to tissue damage. Gastroenterology 2011, 141, 1439–1450. [Google Scholar] [CrossRef] [PubMed]
- Martincorena, I.; Campbell, P.J. Somatic mutation in cancer and normal cells. Science 2015, 349, 1483–1489. [Google Scholar] [CrossRef] [PubMed]
- Duelli, D.; Lazebnik, Y. Cell fusion: A hidden enemy? Cancer Cell 2003, 3, 445–448. [Google Scholar] [CrossRef]
- Wakeling, W.F.; Greetham, J.; Bennett, D.C. Efficient spontaneous fusion between some co-cultured cells, especially murine melanoma cells. Cell Biol. Int. 1994, 18, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Fortuna, M.B.; Dewey, M.J.; Furmanski, P. Cell fusion in tumor development and progression: Occurrence of cell fusion in primary methylcholanthrene-induced tumorigenesis. Int. J. Cancer 1989, 44, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Miller, F.R.; McInerney, D.; Rogers, C.; Miller, B.E. Spontaneous fusion between metastatic mammary tumor subpopulations. J. Cell. Biochem. 1988, 36, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Dittmar, T.; Zanker, K.S. Tissue regeneration in the chronically inflamed tumor environment: Implications for cell fusion driven tumor progression and therapy resistant tumor hybrid cells. Int. J. Mol. Sci. 2015, 16, 30362–30381. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Wang, J.; Liu, L. Chemotherapy promotes tumour cell hybridization in vivo. Tumour Biol. 2016, 37, 5025–5030. [Google Scholar] [CrossRef] [PubMed]
- Clawson, G.A.; Matters, G.L.; Xin, P.; Imamura-Kawasawa, Y.; Du, Z.; Thiboutot, D.M.; Helm, K.F.; Neves, R.I.; Abraham, T. Macrophage-tumor cell fusions from peripheral blood of melanoma patients. PLoS ONE 2015, 10, e0134320. [Google Scholar]
- Goldenberg, D.M.; Zagzag, D.; Heselmeyer-Haddad, K.M.; Berroa Garcia, L.Y.; Ried, T.; Loo, M.; Chang, C.H.; Gold, D.V. Horizontal transmission and retention of malignancy, as well as functional human genes, after spontaneous fusion of human glioblastoma and hamster host cells in vivo. Int. J. Cancer 2012, 131, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Tlsty, T.D. Stromal cells can contribute oncogenic signals. Semin. Cancer Biol. 2001, 11, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Mueller, M.M.; Fusenig, N.E. Friends or foes—Bipolar effects of the tumour stroma in cancer. Nat. Rev. Cancer 2004, 4, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Proia, D.A.; Kuperwasser, C. Stroma: Tumor agonist or antagonist. Cell Cycle 2005, 4, 1022–1025. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.A.; Pollard, J.W. Microenvironmental regulation of metastasis. Nat. Rev. Cancer 2009, 9, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, A.K.; Sodi, S.; Rachkovsky, M.; Kolesnikova, N.; Platt, J.T.; Bolognia, J.L.; Pawelek, J.M. A spontaneous murine melanoma lung metastasis comprised of host x tumor hybrids. Cancer Res. 2000, 60, 2512–2519. [Google Scholar] [PubMed]
- Miller, F.R.; Mohamed, A.N.; McEachern, D. Production of a more aggressive tumor cell variant by spontaneous fusion of two mouse tumor subpopulations. Cancer Res. 1989, 49, 4316–4321. [Google Scholar] [PubMed]
- Jacobsen, B.M.; Harrell, J.C.; Jedlicka, P.; Borges, V.F.; Varella-Garcia, M.; Horwitz, K.B. Spontaneous fusion with, and transformation of mouse stroma by, malignant human breast cancer epithelium. Cancer Res. 2006, 66, 8274–8279. [Google Scholar] [CrossRef] [PubMed]
- Ogle, B.M.; Cascalho, M.; Platt, J.L. Biological implications of cell fusion. Nat. Rev. Mol. Cell Biol. 2005, 6, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, D.M.; Rooney, R.J.; Loo, M.; Liu, D.; Chang, C.H. In-vivo fusion of human cancer and hamster stromal cells permanently transduces and transcribes human DNA. PLoS ONE 2014, 9, e107927. [Google Scholar] [CrossRef] [PubMed]
- Powell, A.E.; Anderson, E.C.; Davies, P.S.; Silk, A.D.; Pelz, C.; Impey, S.; Wong, M.H. Fusion between intestinal epithelial cells and macrophages in a cancer context results in nuclear reprogramming. Cancer Res. 2011, 71, 1497–1505. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.M. Tumor suppressor genes. In The Cell: A Molecular Approach, 2nd ed.; Sinauer Associates: Sunderland, MA, USA, 2000. [Google Scholar]
- Chaffer, C.L.; Weinberg, R.A. A perspective on cancer cell metastasis. Science 2011, 331, 1559–1564. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Neilson, E.G. Epithelial-mesenchymal transition and its implications for fibrosis. J. Clin. Investig. 2003, 112, 1776–1784. [Google Scholar] [CrossRef] [PubMed]
- Qian, B.Z.; Pollard, J.W. Macrophage diversity enhances tumor progression and metastasis. Cell 2010, 141, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Coussens, L.M. Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Peinado, H.; Rafii, S.; Lyden, D. Inflammation joins the "Niche". Cancer Cell 2008, 14, 347–349. [Google Scholar] [CrossRef] [PubMed]
- Pawelek, J.M.; Chakraborty, A.K. Fusion of tumour cells with bone marrow-derived cells: A unifying explanation for metastasis. Nat. Rev. Cancer 2008, 8, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.K.; Sica, A.; Lewis, C.E. Plasticity of macrophage function during tumor progression: Regulation by distinct molecular mechanisms. J. Immunol. 2008, 180, 2011–2017. [Google Scholar] [CrossRef] [PubMed]
- Pawelek, J.M. Tumour cell hybridization and metastasis revisited. Melanoma Res. 2000, 10, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Friedl, P. Prespecification and plasticity: Shifting mechanisms of cell migration. Curr. Opin. Cell Biol. 2004, 16, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Massague, J. Epithelial-mesenchymal transitions: Twist in development and metastasis. Cell 2004, 118, 277–279. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Mani, S.A.; Donaher, J.L.; Ramaswamy, S.; Itzykson, R.A.; Come, C.; Savagner, P.; Gitelman, I.; Richardson, A.; Weinberg, R.A. Twist, a master regulator of morphogenesis, plays an essential role in tumor metastasis. Cell 2004, 117, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Fidler, I.J. The pathogenesis of cancer metastasis: The “seed and soil” hypothesis revisited. Nat. Rev. Cancer 2003, 3, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P. Epithelial-mesenchymal transitions in tumour progression. Nat. Rev. Cancer 2002, 2, 442–454. [Google Scholar] [CrossRef] [PubMed]
- Huysentruyt, L.C.; Seyfried, T.N. Perspectives on the mesenchymal origin of metastatic cancer. Cancer Metastasis Rev. 2010, 29, 695–707. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, A.K.; de Freitas Sousa, J.; Espreafico, E.M.; Pawelek, J.M. Human monocyte x mouse melanoma fusion hybrids express human gene. Gene 2001, 275, 103–106. [Google Scholar] [CrossRef]
- Mekler, L.B.; Drize, O.B.; Osechinskii, I.V.; Shliankevich, M.A. Transformation of a normal differentiated cell of an adult organism, induced by the fusion of this cell with another normal cell of the same organism but with different organ or tissue specificity. Vestn. Akad. Med. Nauk. SSSR 1971, 26, 75–80. [Google Scholar] [PubMed]
- Warner, T.F. Cell hybridizaiton: An explanation for the phenotypic diversity of certain tumours. Med. Hypotheses 1975, 1, 51–57. [Google Scholar] [CrossRef]
- Shabo, I.; Olsson, H.; Sun, X.F.; Svanvik, J. Expression of the macrophage antigen CD163 in rectal cancer cells is associated with early local recurrence and reduced survival time. Int. J. Cancer 2009, 125, 1826–1831. [Google Scholar] [CrossRef] [PubMed]
- Shabo, I.; Midtbo, K.; Andersson, H.; Akerlund, E.; Olsson, H.; Wegman, P.; Gunnarsson, C.; Lindstrom, A. Macrophage traits in cancer cells are induced by macrophage-cancer cell fusion and cannot be explained by cellular interaction. BMC Cancer 2015, 15, 922. [Google Scholar] [PubMed]
- Chambers, A.F.; Groom, A.C.; MacDonald, I.C. Dissemination and growth of cancer cells in metastatic sites. Nat. Rev. Cancer 2002, 2, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Paget, S. The distribution of secondary growths in cancer of the breast. 1889. Cancer Metastasis Rev. 1989, 8, 98–101. [Google Scholar] [PubMed]
- Seyfried, T. Cancer as a Metabolic Disease: On the Origin, Management, and Prevention of Cancer, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Hofer, S.O.; Molema, G.; Hermens, R.A.; Wanebo, H.J.; Reichner, J.S.; Hoekstra, H.J. The effect of surgical wounding on tumour development. Eur. J. Surg. Oncol. 1999, 25, 231–243. [Google Scholar] [CrossRef] [PubMed]
- De Baetselier, P.; Roos, E.; Brys, L.; Remels, L.; Gobert, M.; Dekegel, D.; Segal, S.; Feldman, M. Nonmetastatic tumor cells acquire metastatic properties following somatic hybridization with normal cells. Cancer Metastasis Rev. 1984, 3, 5–24. [Google Scholar] [CrossRef] [PubMed]
- Podsypanina, K.; Du, Y.C.; Jechlinger, M.; Beverly, L.J.; Hambardzumyan, D.; Varmus, H. Seeding and propagation of untransformed mouse mammary cells in the lung. Science 2008, 321, 1841–1844. [Google Scholar] [CrossRef] [PubMed]
- Levin, T.G.; Powell, A.E.; Davies, P.S.; Silk, A.D.; Dismuke, A.D.; Anderson, E.C.; Swain, J.R.; Wong, M.H. Characterization of the intestinal cancer stem cell marker CD166 in the human and mouse gastrointestinal tract. Gastroenterology 2010, 139, 2072–2082. [Google Scholar] [CrossRef] [PubMed]
- Seyfried, T.N.; Kiebish, M.A.; Marsh, J.; Shelton, L.M.; Huysentruyt, L.C.; Mukherjee, P. Metabolic management of brain cancer. Biochim. Biophys. Acta 2011, 1807, 577–594. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lou, X.; Zellmer, L.; Liu, S.; Xu, N.; Liao, D.J. Just like the rest of evolution in mother nature, the evolution of cancers may be driven by natural selection, and not by haphazard mutations. Oncoscience 2014, 1, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Nagler, C.; Hardt, C.; Zanker, K.S.; Dittmar, T. Co-cultivation of murine bmdcs with 67NR mouse mammary carcinoma cells give rise to highly drug resistant cells. Cancer Cell Int. 2011, 11, 21. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Kang, Y. Efficient acquisition of dual metastasis organotropism to bone and lung through stable spontaneous fusion between MDA-MB-231 variants. Proc. Natl. Acad. Sci. USA 2009, 106, 9385–9390. [Google Scholar] [CrossRef] [PubMed]
- Clarke, M.F.; Dick, J.E.; Dirks, P.B.; Eaves, C.J.; Jamieson, C.H.; Jones, D.L.; Visvader, J.; Weissman, I.L.; Wahl, G.M. Cancer stem cells-perspectives on current status and future directions: AACR workshop on cancer stem cells. Cancer Res. 2006, 66, 9339–9344. [Google Scholar] [CrossRef] [PubMed]
- Bjerkvig, R.; Tysnes, B.B.; Aboody, K.S.; Najbauer, J.; Terzis, A.J. Opinion: The origin of the cancer stem cell: Current controversies and new insights. Nat. Rev. Cancer 2005, 5, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Tiede, B.; Massague, J.; Kang, Y. Beyond tumorigenesis: Cancer stem cells in metastasis. Cell Res. 2007, 17, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Dittmar, T.; Nagler, C.; Schwitalla, S.; Reith, G.; Niggemann, B.; Zanker, K.S. Recurrence cancer stem cells—Made by cell fusion? Med. Hypotheses 2009, 73, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Kang, Y. Cell fusion hypothesis of cancer stem cells. In Cell Fusion in Health and Disease. Volume ii: Cell Fusion in Disease, 1st ed.; Dittmar, T., Zänker, K.S., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2011; pp. 129–140. [Google Scholar]
- Houghton, J.M. Bone-marrow-derived cells and cancer—An opportunity for improved therapy. Nat. Rev. Clin. Oncol. 2007, 4, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Quintana, E.; Shackleton, M.; Sabel, M.S.; Fullen, D.R.; Johnson, T.M.; Morrison, S.J. Efficient tumour formation by single human melanoma cells. Nature 2008, 456, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.B.; Zhang, H.; Damelin, M.; Geles, K.G.; Grindley, J.C.; Dirks, P.B. Tumour-initiating cells: Challenges and opportunities for anticancer drug discovery. Nat. Rev. Drug Discov. 2009, 8, 806–823. [Google Scholar] [CrossRef] [PubMed]
- Phillips, T.M.; McBride, W.H.; Pajonk, F. The response of CD24(-/low)/CD44+ breast cancer-initiating cells to radiation. J. Natl. Cancer Inst. 2006, 98, 1777–1785. [Google Scholar] [CrossRef] [PubMed]
- Woodward, W.A.; Chen, M.S.; Behbod, F.; Alfaro, M.P.; Buchholz, T.A.; Rosen, J.M. WNT/β-catenin mediates radiation resistance of mouse mammary progenitor cells. Proc. Natl. Acad. Sci. USA 2007, 104, 618–623. [Google Scholar] [CrossRef] [PubMed]
- Nagler, C.; Zänker, K.S.; Dittmar, T. Cell fusion, drug resistance and recurrence cscs. In Cell Fusion in Health and Disease. Volume II: Cell Fusion in Disease, 1st ed.; Zänker, K.S., Dittmar, T., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2011; pp. 173–182. [Google Scholar]
- Zhang, D.; Wang, Y.; Zhang, S. Asymmetric cell division in polyploid giant cancer cells and low eukaryotic cells. BioMed Res. Int. 2014, 2014, 432652. [Google Scholar] [CrossRef] [PubMed]
- Kawano, N.; Harada, Y.; Yoshida, K.; Miyado, M.; Miyado, K. Role of CD9 in sperm-egg fusion and its general role in fusion phenomena. In Cell Fusions: Regulation and Control, 1st ed.; Larsson, L.-I., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2010; pp. 171–184. [Google Scholar]
- Yi, Y.-J.; Zimmerman, S.W.; Sutovsky, P. Gamete binding and fusion. In Cell Fusions: Regulation and Control, 1st ed.; Larsson, L.-I., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2010; pp. 185–201. [Google Scholar]
- Chen, E.H.; Grote, E.; Mohler, W.; Vignery, A. Cell-cell fusion. FEBS Lett. 2007, 581, 2181–2193. [Google Scholar] [CrossRef] [PubMed]
- Horsley, V.; Pavlath, G.K. Forming a multinucleated cell: Molecules that regulate myoblast fusion. Cells Tissues Organs 2004, 176, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Buckingham, M. Myogenic progenitor cells and skeletal myogenesis in vertebrates. Curr. Opin. Genet. Dev. 2006, 16, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Vignery, A. Osteoclasts and giant cells: Macrophage-macrophage fusion mechanism. Int. J. Exp. Pathol. 2000, 81, 291–304. [Google Scholar] [CrossRef] [PubMed]
- Vignery, A. Macrophage fusion: The making of osteoclasts and giant cells. J. Exp. Med. 2005, 202, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Kaji, K.; Oda, S.; Miyazaki, S.; Kudo, A. Infertility of CD9-deficient mouse eggs is reversed by mouse CD9, human CD9, or mouse CD81; polyadenylated mrna injection developed for molecular analysis of sperm-egg fusion. Dev. Biol. 2002, 247, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Kaji, K.; Oda, S.; Shikano, T.; Ohnuki, T.; Uematsu, Y.; Sakagami, J.; Tada, N.; Miyazaki, S.; Kudo, A. The gamete fusion process is defective in eggs of CD9-deficient mice. Nat. Genet. 2000, 24, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Le Naour, F.; Rubinstein, E.; Jasmin, C.; Prenant, M.; Boucheix, C. Severely reduced female fertility in CD9-deficient mice. Science 2000, 287, 319–321. [Google Scholar] [CrossRef] [PubMed]
- Miyado, K.; Yamada, G.; Yamada, S.; Hasuwa, H.; Nakamura, Y.; Ryu, F.; Suzuki, K.; Kosai, K.; Inoue, K.; Ogura, A.; et al. Requirement of CD9 on the egg plasma membrane for fertilization. Science 2000, 287, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Rubinstein, E.; Ziyyat, A.; Prenant, M.; Wrobel, E.; Wolf, J.P.; Levy, S.; Le Naour, F.; Boucheix, C. Reduced fertility of female mice lacking CD81. Dev. Biol. 2006, 290, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Tanigawa, M.; Miyamoto, K.; Kobayashi, S.; Sato, M.; Akutsu, H.; Okabe, M.; Mekada, E.; Sakakibara, K.; Miyado, M.; Umezawa, A.; et al. Possible involvement of CD81 in acrosome reaction of sperm in mice. Mol. Reprod. Dev. 2008, 75, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.P. Fertilin β and other adams as integrin ligands: Insights into cell adhesion and fertilization. Bioessays 2001, 23, 628–639. [Google Scholar] [CrossRef] [PubMed]
- Okabe, M.; Adachi, T.; Takada, K.; Oda, H.; Yagasaki, M.; Kohama, Y.; Mimura, T. Capacitation-related changes in antigen distribution on mouse sperm heads and its relation to fertilization rate in vitro. J. Reprod. Immunol. 1987, 11, 91–100. [Google Scholar] [CrossRef]
- Inoue, N.; Ikawa, M.; Isotani, A.; Okabe, M. The immunoglobulin superfamily protein izumo is required for sperm to fuse with eggs. Nature 2005, 434, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Önel, S.-F.; Dottermusch, C.; Sickmann, A.; Buttgereit, D.; Renkawitz-Pohl, R. Role of the actin cytoskeleton within furmas during drosophila myoblast fusion and first functionally conserved factors in vertebrates. In Cell Fusions: Regulation and Control, 1st ed.; Larsson, L.-I., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2010; pp. 139–170. [Google Scholar]
- Fischbach, K.F.; Linneweber, G.A.; Andlauer, T.F.; Hertenstein, A.; Bonengel, B.; Chaudhary, K. The IRRE cell recognition module (IRM) proteins. J. Neurogenet. 2009, 23, 48–67. [Google Scholar] [CrossRef] [PubMed]
- Uluckan, O.; Becker, S.N.; Deng, H.; Zou, W.; Prior, J.L.; Piwnica-Worms, D.; Frazier, W.A.; Weilbaecher, K.N. CD47 regulates bone mass and tumor metastasis to bone. Cancer Res. 2009, 69, 3196–3204. [Google Scholar] [CrossRef] [PubMed]
- Kukreja, A.; Radfar, S.; Sun, B.H.; Insogna, K.; Dhodapkar, M.V. Dominant role of CD47-thrombospondin-1 interactions in myeloma-induced fusion of human dendritic cells: Implications for bone disease. Blood 2009, 114, 3413–3421. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Ke, J.Z.; Zhang, Q.; Ke, H.Z.; Chalouni, C.; Vignery, A. The intracellular domain of CD44 promotes the fusion of macrophages. Blood 2006, 107, 796–805. [Google Scholar] [CrossRef] [PubMed]
- Mensah, K.A.; Ritchlin, C.T.; Schwarz, E.M. RANKl induces heterogeneous DC-STAMPlo and DC-STAMPhi osteoclast precursors of which the DC-STAMPlo precursors are the master fusogens. J. Cell. Physiol. 2010, 223, 76–83. [Google Scholar] [PubMed]
- Yagi, M.; Miyamoto, T.; Sawatani, Y.; Iwamoto, K.; Hosogane, N.; Fujita, N.; Morita, K.; Ninomiya, K.; Suzuki, T.; Miyamoto, K.; et al. DC-STAMP is essential for cell-cell fusion in osteoclasts and foreign body giant cells. J. Exp. Med. 2005, 202, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Witwicka, H.; Hwang, S.Y.; Reyes-Gutierrez, P.; Jia, H.; Odgren, P.E.; Donahue, L.R.; Birnbaum, M.J.; Odgren, P.R. Studies of OC-STAMP in osteoclast fusion: A new knockout mouse model, rescue of cell fusion, and transmembrane topology. PLoS ONE 2015, 10, e0128275. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.G.; Ritchlin, C.T. Dc-stamp: A key regulator in osteoclast differentiation. J. Cell. Physiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Pérot, P.; Montgiraud, C.; Lavillette, D.; Mallet, F. A comparative portrait of retroviral fusogens and syncytins. In Cell Fusions: Regulation and Control, 1st ed.; Larsson, L.-I., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2010; pp. 63–115. [Google Scholar]
- Gerbaud, P.; Pidoux, G. Review: An overview of molecular events occurring in human trophoblast fusion. Placenta 2015, 36, S35–S42. [Google Scholar] [CrossRef] [PubMed]
- Benirschke, K.; Burton, G.J.; Baergen, R.N. Architecture of normal villous trees. In Pathology of the Human Placenta, 6th ed.; Springer-Verlag: Berlin, Germany; Heidelberg, Germany, 2012; pp. 101–144. [Google Scholar]
- Benirschke, K.; Burton, G.J.; Baergen, R.N. Early development of the human placenta. In Pathology of the Human Placenta, 6th ed.; Springer-Verlag: Berlin, Germany; Heidelberg, Germany, 2012; pp. 41–53. [Google Scholar]
- Eaton, B.; Contractor, S. In vitro assessment of trophoblast receptors and placental transport mechanisms. In The Human Placenta, 1st ed.; Redman, C.W., Sargent, I.L., Starkey, P.M., Eds.; Blackwell Scientific Publication: London, UK, 1993; pp. 471–503. [Google Scholar]
- Ogren, L.; Talamentes, F. The placenta as an endocrine organ: Polypeptides. In The Physiology of Reproduction, 2nd ed.; Knobil, E., Neill, J., Eds.; Raven Press: New York, NY, USA, 1994; pp. 875–945. [Google Scholar]
- Bozzuto, G.; Ruggieri, P.; Molinari, A. Molecular aspects of tumor cell migration and invasion. Ann. Ist. Super Sanita. 2010, 46, 66–80. [Google Scholar] [CrossRef] [PubMed]
- Blond, J.L.; Beseme, F.; Duret, L.; Bouton, O.; Bedin, F.; Perron, H.; Mandrand, B.; Mallet, F. Molecular characterization and placental expression of HERV-W, a new human endogenous retrovirus family. J. Virol. 1999, 73, 1175–1185. [Google Scholar] [PubMed]
- Mi, S.; Lee, X.; Li, X.; Veldman, G.M.; Finnerty, H.; Racie, L.; LaVallie, E.; Tang, X.Y.; Edouard, P.; Howes, S.; et al. Syncytin is a captive retroviral envelope protein involved in human placental morphogenesis. Nature 2000, 403, 785–789. [Google Scholar] [PubMed]
- Blaise, S.; de Parseval, N.; Heidmann, T. Functional characterization of two newly identified human endogenous retrovirus coding envelope genes. Retrovirology 2005, 2, 19. [Google Scholar] [CrossRef] [PubMed]
- Larsson, L.I.; Bjerregaard, B.; Talts, J.F. Cell fusions in mammals. Histochem. Cell Biol. 2008, 129, 551–561. [Google Scholar] [CrossRef] [PubMed]
- Jahn, R.; Lang, T.; Sudhof, T.C. Membrane fusion. Cell 2003, 112, 519–533. [Google Scholar] [CrossRef]
- Chen, E.H.; Olson, E.N. Unveiling the mechanisms of cell-cell fusion. Science 2005, 308, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Ahmed, M.; Melia, T.J.; Sollner, T.H.; Mayer, T.; Rothman, J.E. Fusion of cells by flipped snares. Science 2003, 300, 1745–1749. [Google Scholar] [CrossRef] [PubMed]
- Caceres, M.; Thomas, J.W. The gene of retroviral origin syncytin 1 is specific to hominoids and is inactive in old world monkeys. J. Hered. 2006, 97, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Lander, E.S.; Linton, L.M.; Birren, B.; Nusbaum, C.; Zody, M.C.; Baldwin, J.; Devon, K.; Dewar, K.; Doyle, M.; FitzHugh, W.; et al. Initial sequencing and analysis of the human genome. Nature 2001, 409, 860–921. [Google Scholar] [CrossRef] [PubMed]
- Maliniemi, P.; Vincendeau, M.; Mayer, J.; Frank, O.; Hahtola, S.; Karenko, L.; Carlsson, E.; Mallet, F.; Seifarth, W.; Leib-Mosch, C.; et al. Expression of human endogenous retrovirus-w including syncytin-1 in cutaneous t-cell lymphoma. PLoS ONE 2013, 8, e76281. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Liu, T.; Zhao, Z.; Chen, Y.; Zeng, J.; Liu, S.; Zhu, F. Mutations in 3’-long terminal repeat of HERV-W family in chromosome 7 upregulate syncytin-1 expression in urothelial cell carcinoma of the bladder through interacting with C-MYB. Oncogene 2014, 33, 3947–3958. [Google Scholar] [CrossRef] [PubMed]
- Strissel, P.L.; Ruebner, M.; Thiel, F.; Wachter, D.; Ekici, A.B.; Wolf, F.; Thieme, F.; Ruprecht, K.; Beckmann, M.W.; Strick, R. Reactivation of codogenic endogenous retroviral (ERV) envelope genes in human endometrial carcinoma and prestages: Emergence of new molecular targets. Oncotarget. 2012, 3, 1204–1219. [Google Scholar] [CrossRef] [PubMed]
- Strick, R.; Ackermann, S.; Langbein, M.; Swiatek, J.; Schubert, S.W.; Hashemolhosseini, S.; Koscheck, T.; Fasching, P.A.; Schild, R.L.; Beckmann, M.W.; et al. Proliferation and cell-cell fusion of endometrial carcinoma are induced by the human endogenous retroviral syncytin-1 and regulated by TGF-β. J. Mol. Med. 2007, 85, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Ouyang, D.Y.; Pang, W.; Tu, Y.Q.; Li, Y.Y.; Shen, X.M.; Tam, S.C.; Yang, H.Y.; Zheng, Y.T. Expression of syncytin in leukemia and lymphoma cells. Leuk. Res. 2010, 34, 1195–1202. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.M.; Kim, H.M.; Kim, H.S. Expression of the human endogenous retrovirus HERV-W family in various human tissues and cancer cells. J. Gen. Virol. 2004, 85, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Larsen, J.M.; Christensen, I.J.; Nielsen, H.J.; Hansen, U.; Bjerregaard, B.; Talts, J.F.; Larsson, L.I. Syncytin immunoreactivity in colorectal cancer: Potential prognostic impact. Cancer Lett. 2009, 280, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Larsson, L.I.; Holck, S.; Christensen, I.J. Prognostic role of syncytin expression in breast cancer. Hum. Pathol. 2007, 38, 726–731. [Google Scholar] [CrossRef] [PubMed]
- Bjerregaard, B.; Holck, S.; Christensen, I.J.; Larsson, L.I. Syncytin is involved in breast cancer-endothelial cell fusions. Cell. Mol. Life Sci. 2006, 63, 1906–1911. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.M.; Lee, W.H.; Kim, H.M.; Kim, H.S. Identification of new endogenous retroviral sequences belonging to the HERV-W family in human cancer cells. Intervirology 2001, 44, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Kudo, Y.; Boyd, C.A. Changes in expression and function of syncytin and its receptor, amino acid transport system B(0) (ASCT2), in human placental choriocarcinoma bewo cells during syncytialization. Placenta 2002, 23, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Borges, M.; Bose, P.; Frank, H.G.; Kaufmann, P.; Potgens, A.J. A two-colour fluorescence assay for the measurement of syncytial fusion between trophoblast-derived cell lines. Placenta 2003, 24, 959–964. [Google Scholar] [CrossRef]
- Esnault, C.; Priet, S.; Ribet, D.; Vernochet, C.; Bruls, T.; Lavialle, C.; Weissenbach, J.; Heidmann, T. A placenta-specific receptor for the fusogenic, endogenous retrovirus-derived, human syncytin-2. Proc. Natl. Acad. Sci. USA 2008, 105, 17532–17537. [Google Scholar] [CrossRef] [PubMed]
- Strick, R.; Beckmann, M.W.; Strissel, P.L. Cell–cell fusions and human endogenous retroviruses in cancer. In Cell Fusions: Regulation and Control, 1st ed.; Larsson, L.-I., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2010; pp. 395–426. [Google Scholar]
- Li, J.; Lee, A.S. Stress induction of GRP78/BIP and its role in cancer. Curr. Mol. Med. 2006, 6, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Fradet, S.; Pierredon, S.; Ribaux, P.; Epiney, M.; Shin Ya, K.; Irion, O.; Cohen, M. Involvement of membrane GRP78 in trophoblastic cell fusion. PLoS ONE 2012, 7, e40596. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.; Ribaux, P.; Epiney, M.; Irion, O. Role of prostate apoptosis response 4 in translocation of GRP78 from the endoplasmic reticulum to the cell surface of trophoblastic cells. PLoS ONE 2013, 8, e80231. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Chen, P.T.; Chang, G.D.; Huang, C.J.; Chen, H. Functional characterization of the placental fusogenic membrane protein syncytin. Biol. Reprod. 2004, 71, 1956–1962. [Google Scholar] [PubMed]
- Gong, R.; Peng, X.; Kang, S.; Feng, H.; Huang, J.; Zhang, W.; Lin, D.; Tien, P.; Xiao, G. Structural characterization of the fusion core in syncytin, envelope protein of human endogenous retrovirus family W. Biochem. Biophys. Res. Commun. 2005, 331, 1193–1200. [Google Scholar] [PubMed]
- Chernomordik, L.V.; Kozlov, M.M. Mechanics of membrane fusion. Nat. Struct. Mol. Biol. 2008, 15, 675–683. [Google Scholar] [PubMed]
- Riddell, M.R.; Winkler-Lowen, B.; Jiang, Y.; Davidge, S.T.; Guilbert, L.J. Pleiotropic actions of forskolin result in phosphatidylserine exposure in primary trophoblasts. PLoS ONE 2013, 8, e81273. [Google Scholar]
- Takeda, M.; Leser, G.P.; Russell, C.J.; Lamb, R.A. Influenza virus hemagglutinin concentrates in lipid raft microdomains for efficient viral fusion. Proc. Natl. Acad. Sci. USA 2003, 100, 14610–14617. [Google Scholar] [PubMed]
- Suomalainen, M. Lipid rafts and assembly of enveloped viruses. Traffic 2002, 3, 705–709. [Google Scholar] [CrossRef] [PubMed]
- Langbein, M.; Strick, R.; Strissel, P.L.; Vogt, N.; Parsch, H.; Beckmann, M.W.; Schild, R.L. Impaired cytotrophoblast cell-cell fusion is associated with reduced syncytin and increased apoptosis in patients with placental dysfunction. Mol. Reprod. Dev. 2008, 75, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.P.; Wang, K.G.; Chen, C.Y.; Yu, C.; Chuang, H.C.; Chen, H. Altered placental syncytin and its receptor ASCT2 expression in placental development and pre-eclampsia. BJOG 2006, 113, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Lee, X.; Keith, J.C., Jr.; Stumm, N.; Moutsatsos, I.; McCoy, J.M.; Crum, C.P.; Genest, D.; Chin, D.; Ehrenfels, C.; Pijnenborg, R.; et al. Downregulation of placental syncytin expression and abnormal protein localization in pre-eclampsia. Placenta 2001, 22, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Mohan, A.; Moore, D.C.; Lin, L.; Zhou, F.L.; Cao, J.; Wu, Q.; Qin, Y.X.; Reginato, A.M.; Ehrlich, M.G.; et al. SHP2 regulates osteoclastogenesis by promoting preosteoclast fusion. FASEB J. 2015, 29, 1635–1645. [Google Scholar] [CrossRef] [PubMed]
- Skeel, R.T.; Khleif, S. Handbook of Cancer Chemotherapy, 8th ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2011. [Google Scholar]
- Gong, J.; Koido, S. Cell fusion and dendritic cell-based vaccines. In Cell Fusions: Regulation and Control, 1st ed.; Larsson, L.-I., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2010; pp. 315–350. [Google Scholar]
- Koido, S.; Hara, E.; Homma, S.; Ohkusa, T.; Gong, J.; Tajiri, H. Cancer immunotherapy by fusions of dendritic cells and tumor cells. Immunotherapy 2009, 1, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M. The dendritic cell system and its role in immunogenicity. Annu. Rev. Immunol. 1991, 9, 271–296. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M. Dendritic cells and the control of immunity: Enhancing the efficiency of antigen presentation. Mt. Sinai J. Med. 2001, 68, 160–166. [Google Scholar] [PubMed]
- Homma, S.; Matai, K.; Irie, M.; Ohno, T.; Kufe, D.; Toda, G. Immunotherapy using fusions of autologous dendritic cells and tumor cells showed effective clinical response in a patient with advanced gastric carcinoma. J. Gastroenterol. 2003, 38, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Kjaergaard, J.; Shimizu, K.; Shu, S. Electrofusion of syngeneic dendritic cells and tumor generates potent therapeutic vaccine. Cell. Immunol. 2003, 225, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Trevor, K.T.; Cover, C.; Ruiz, Y.W.; Akporiaye, E.T.; Hersh, E.M.; Landais, D.; Taylor, R.R.; King, A.D.; Walters, R.E. Generation of dendritic cell-tumor cell hybrids by electrofusion for clinical vaccine application. Cancer Immunol. Immunother. 2004, 53, 705–714. [Google Scholar] [CrossRef] [PubMed]
- Scott-Taylor, T.H.; Pettengell, R.; Clarke, I.; Stuhler, G.; La Barthe, M.C.; Walden, P.; Dalgleish, A.G. Human tumour and dendritic cell hybrids generated by electrofusion: Potential for cancer vaccines. Biochim. Biophys. Acta 2000, 1500, 265–279. [Google Scholar] [CrossRef]
- Hiraoka, K.; Yamamoto, S.; Otsuru, S.; Nakai, S.; Tamai, K.; Morishita, R.; Ogihara, T.; Kaneda, Y. Enhanced tumor-specific long-term immunity of hemagglutinating [correction of hemaggluttinating] virus of Japan-mediated dendritic cell-tumor fused cell vaccination by coadministration with CpG oligodeoxynucleotides. J. Immunol. 2004, 173, 4297–4307. [Google Scholar] [CrossRef] [PubMed]
- Phan, V.; Errington, F.; Cheong, S.C.; Kottke, T.; Gough, M.; Altmann, S.; Brandenburger, A.; Emery, S.; Strome, S.; Bateman, A.; et al. A new genetic method to generate and isolate small, short-lived but highly potent dendritic cell-tumor cell hybrid vaccines. Nat. Med. 2003, 9, 1215–1219. [Google Scholar] [CrossRef] [PubMed]

| Protein | Human Tissue or Cell Line | References |
|---|---|---|
| Syncytin 1 | Cutaneous T-cell lymphoma | [122] |
| Syncytin 1 | Urothelial cell carcinoma | [123] |
| Syncytin 1 | Endometrial carcinoma | [124,125] |
| Syncytin 1 | Leukemia cells | [126,127] |
| Syncytin 1 | Lymphoma cells | [126,127] |
| Syncytin 1 | Colorectal carcinoma | [128] |
| Syncytin 1 | Breast cancer | [127,129,130] |
| Syncytin 1 | Ovarian carcinoma cell line | [127,131] |
| Syncytin 1 | Gastric cell line | [127,131] |
| Syncytin 1 | Lung carcinoma cell line | [127,131] |
| Syncytin 1 | Brain carcinoma cell line | [127] |
| Cervix carcinoma cell line | ||
| Melanoma cell line | ||
| Prostate cancer cell line | ||
| Pancreatic carcinoma cell line | ||
| Hepatocellular carcinoma cell line | ||
| Kidney carcinoma cell line | ||
| Squamous carcinoma cell line | ||
| Colon carcinoma cell line | ||
| Bladder carcinoma cell line | ||
| Syncytin 2 | Endometrial carcinoma | [124] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bastida-Ruiz, D.; Van Hoesen, K.; Cohen, M. The Dark Side of Cell Fusion. Int. J. Mol. Sci. 2016, 17, 638. https://doi.org/10.3390/ijms17050638
Bastida-Ruiz D, Van Hoesen K, Cohen M. The Dark Side of Cell Fusion. International Journal of Molecular Sciences. 2016; 17(5):638. https://doi.org/10.3390/ijms17050638
Chicago/Turabian StyleBastida-Ruiz, Daniel, Kylie Van Hoesen, and Marie Cohen. 2016. "The Dark Side of Cell Fusion" International Journal of Molecular Sciences 17, no. 5: 638. https://doi.org/10.3390/ijms17050638
APA StyleBastida-Ruiz, D., Van Hoesen, K., & Cohen, M. (2016). The Dark Side of Cell Fusion. International Journal of Molecular Sciences, 17(5), 638. https://doi.org/10.3390/ijms17050638






