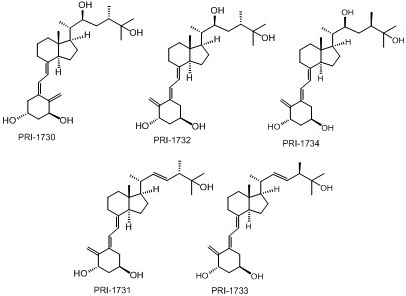
Biological Evaluation of Double Point Modified Analogues of 1,25-Dihydroxyvitamin D2 as Potential Anti-Leukemic Agents
Abstract
:1. Introduction
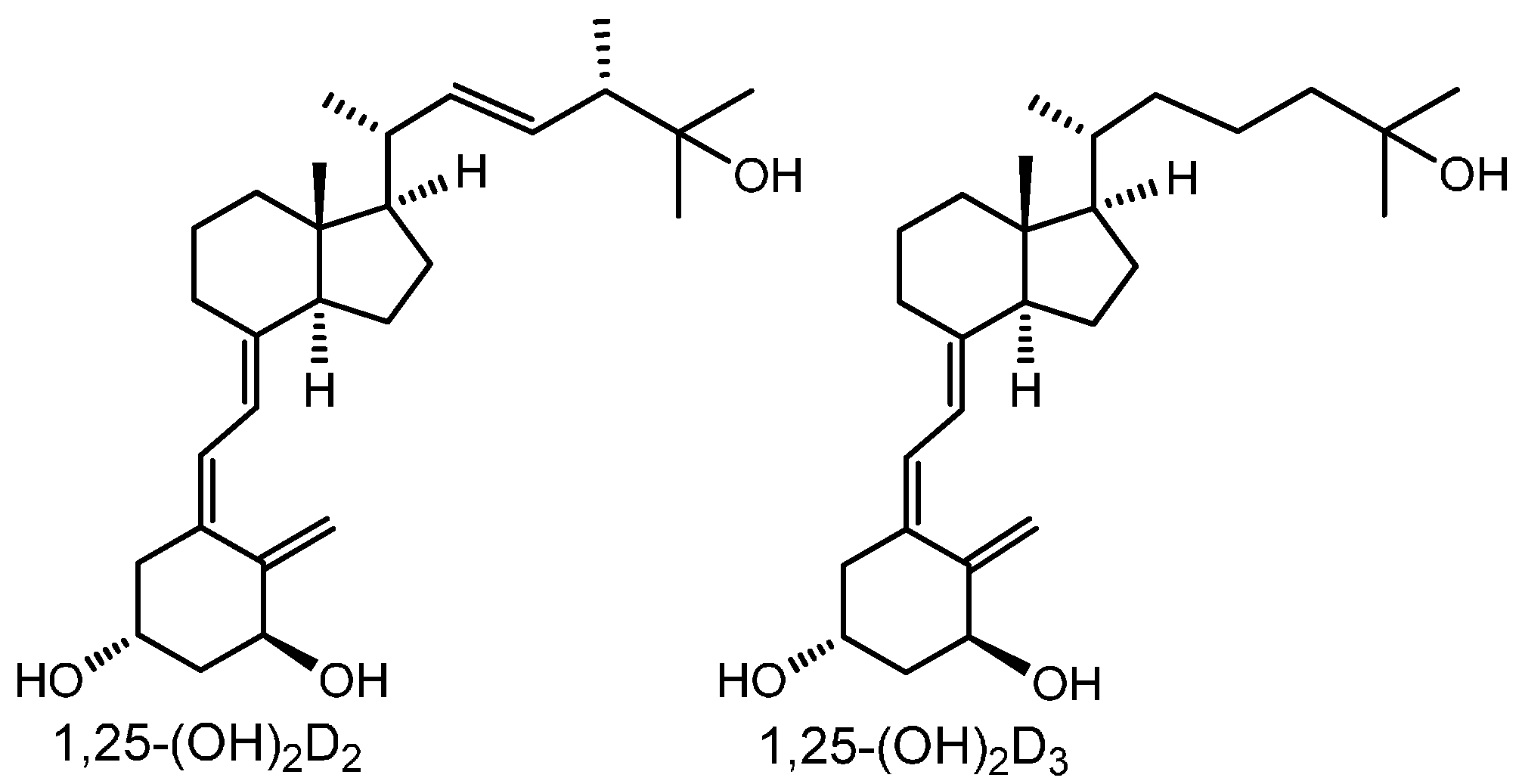
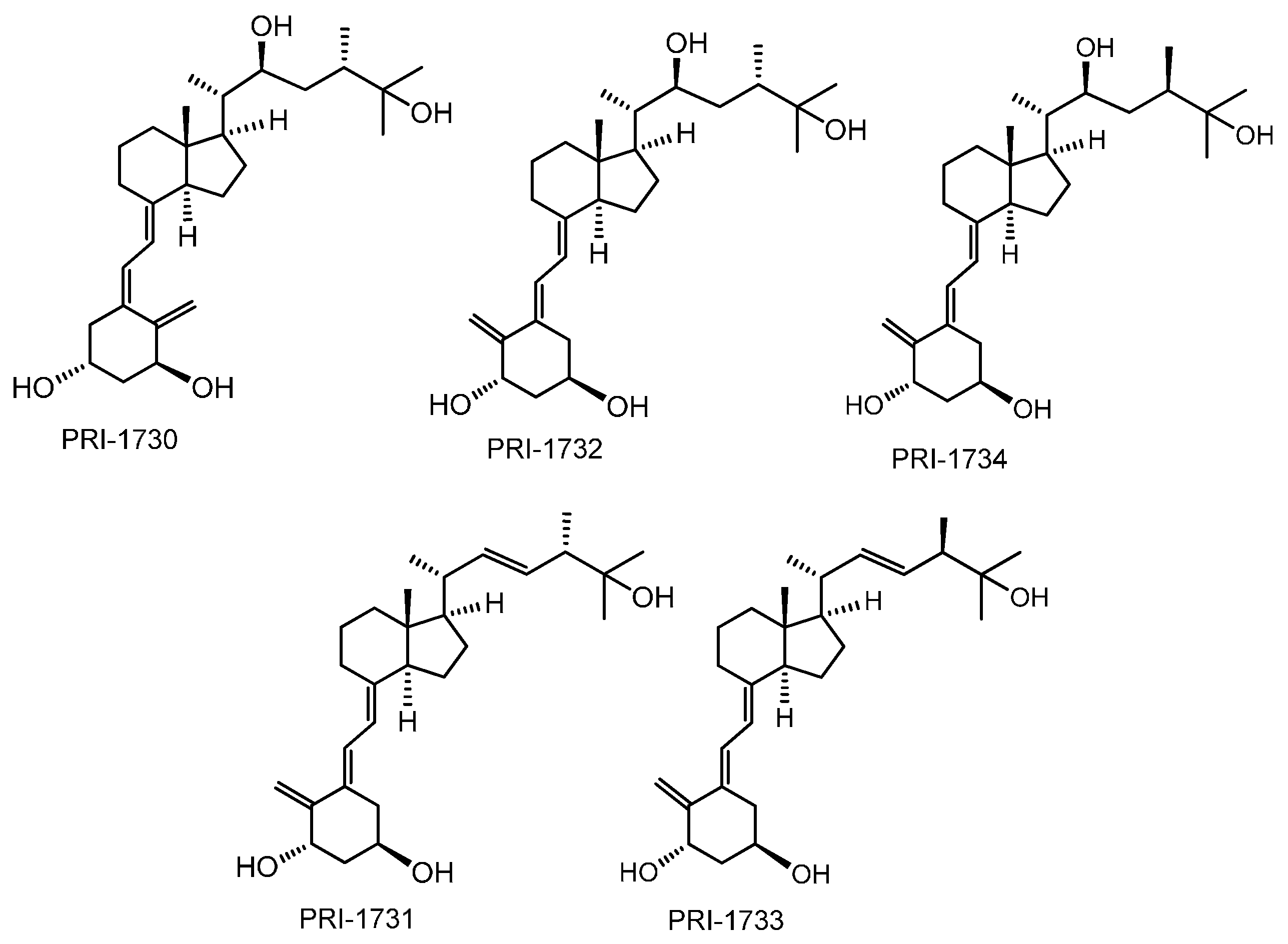
2. Results
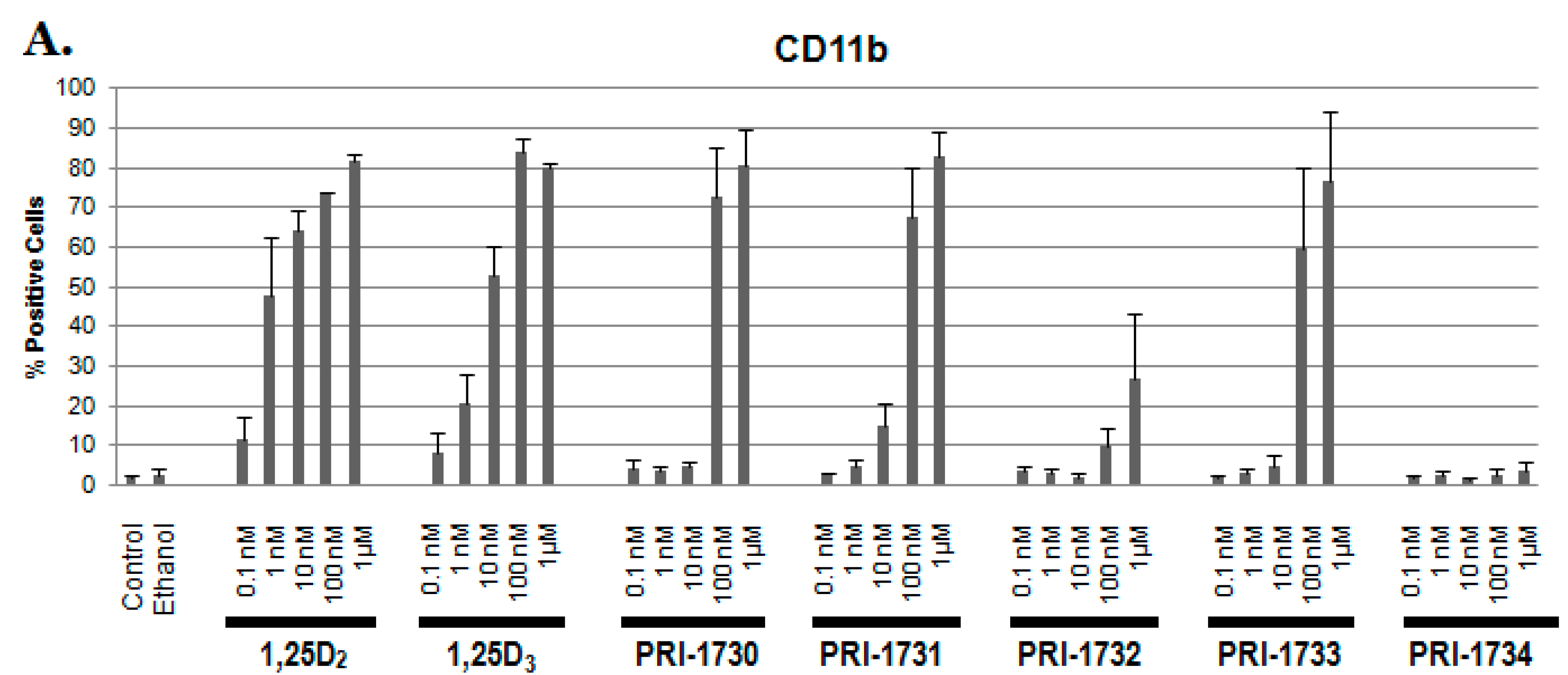
2.1. Differentiation of HL60 Cells
2.2. Nuclear Translocation of VDR
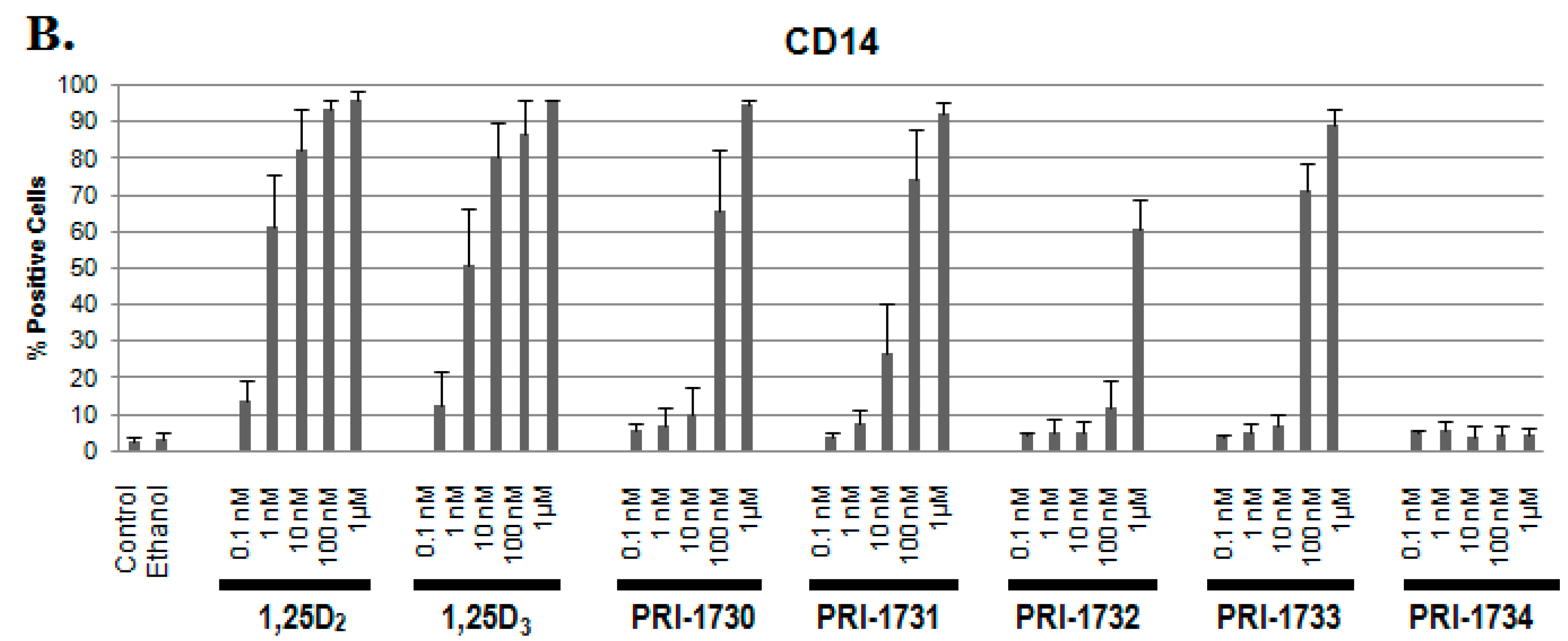

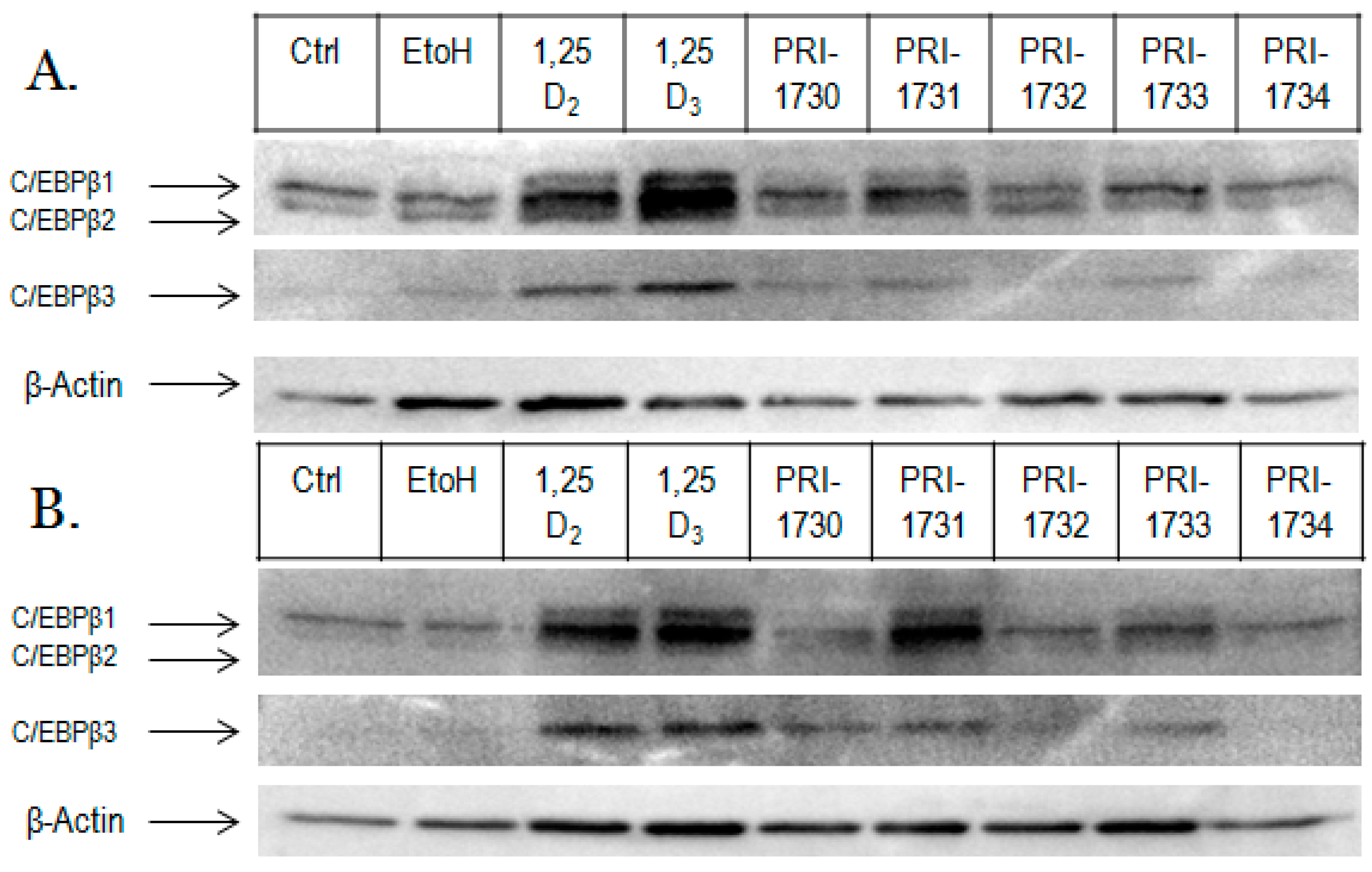
2.3. Activation of C/EBPβ by 1,25D2, 1,25D3 and Analogues
2.4. Binding Affinity of Analogues to VDR
| 1,25D3 | 1,25D2 | PRI-1730 | PRI-1731 | PRI-1732 | PRI-1733 | PRI-1734 | |
|---|---|---|---|---|---|---|---|
| IC50 | 4.494 × 10−9 | 1.466 × 10−8 | 2.006 × 10−7 | 2.218 × 10−8 | 2.138 × 10−7 | 4.386 × 10−6 | 2.641 × 10−6 |
| RBA a | 100 | 30.66 | 2.175 | 20.26 | 2.03 | 0.102 | 0.170 |
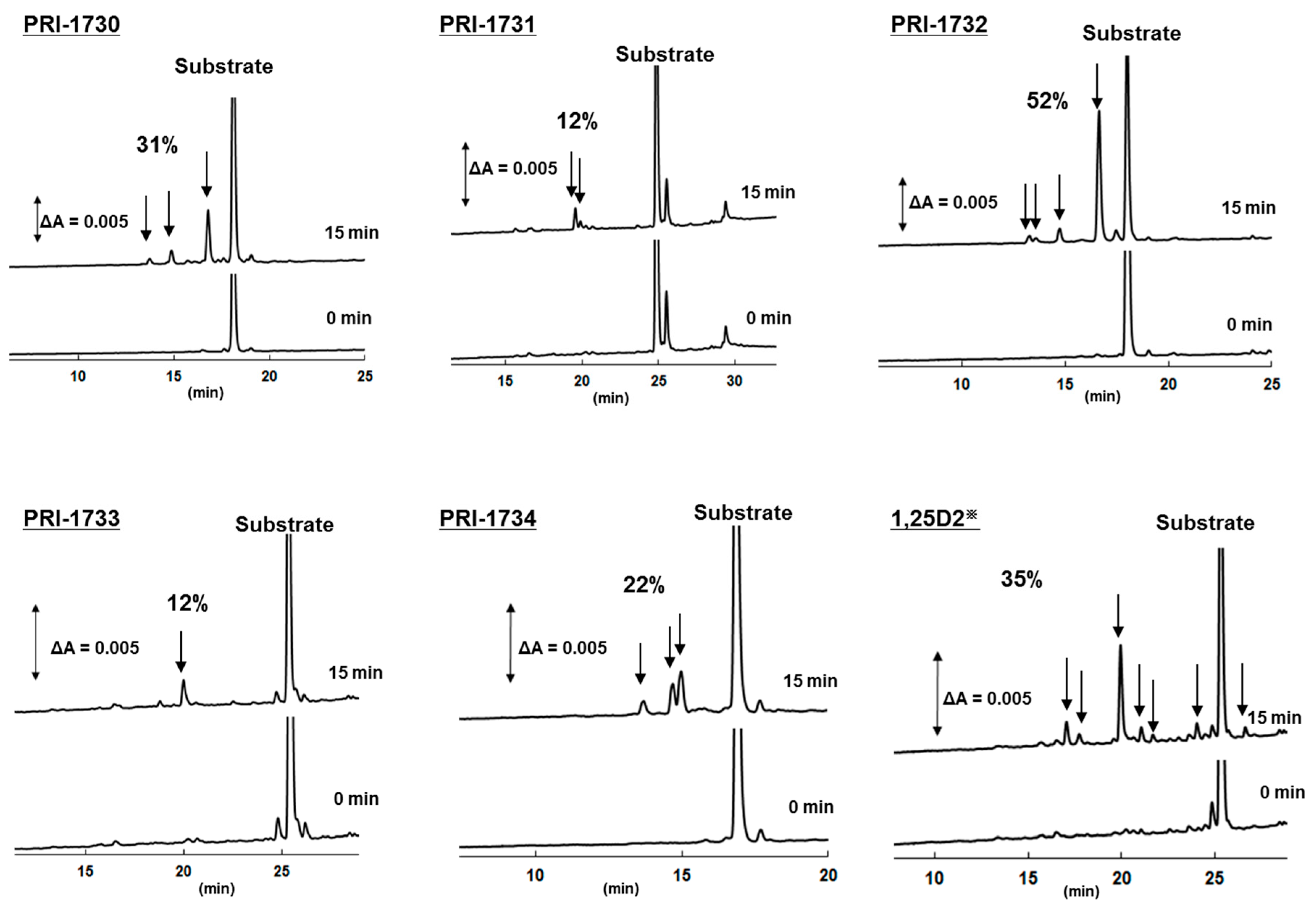
2.5. Metabolic Resistance of Analogs to CYP24A1
| Compound | 1,25D3 | 1,25D2 | PRI-1730 | PRI-1731 | PRI-1732 | PRI-1733 | PRI-1734 |
|---|---|---|---|---|---|---|---|
| Metabolic conversion (%) | 44 | 35 | 31 | 12 | 52 | 12 | 22 |
2.6. Identifying If PRI-1734 Has Antagonistic Activity

3. Discussion
4. Materials and Methods
4.1. Chemicals and Antibodies
4.2. Cell Lines
4.3. Determination of Cell Differentiation by Flow Cytometry
4.4. Preparation of Cell Lysates
4.5. Western Blotting
4.6. Human VDR Binding Assay
4.7. Metabolic Resistance of Analogues to CYP24A1
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holick, M.F. Vitamin D and bone health. J. Nutr. 1996, 126, 1159–1164. [Google Scholar]
- Trump, D.L.; Deeb, K.; Johnson, C.S. Vitamin D: Considerations in the continued development as an agent for cancer prevention and therapy. Cancer J. 2010, 16, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Van Etten, E.; Mathieu, C. Immunoregulation by 1,25-dihydroxyvitamin D3: Basic concepts. J. Steroid Biochem. Mol. Biol. 2005, 97, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Houghton, L.A.; Vieth, R. The case against ergocalciferol (vitamin D2) as a vitamin supplement. Am. J. Clin. Nutr. 2006, 84, 694–697. [Google Scholar] [PubMed]
- Nuijten, M.; Andress, D.L.; Marx, S.E.; Sterz, R. Chronic kidney disease Markov model comparing paricalcitol to calcitriol for secondary hyperparathyroidism: A US perspective. Curr. Med. Res. Opin. 2009, 25, 1221–1234. [Google Scholar] [CrossRef] [PubMed]
- Koeffler, H.P.; Aslanian, N.; O'Kelly, J. Vitamin D2 analog (Paricalcitol; Zemplar) for treatment of myelodysplastic syndrome. Leuk. Res. 2005, 29, 1259–1262. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.S.; Bershadskiy, A. Clinical experience using vitamin D and analogs in the treatment of myelodysplasia and acute myeloid leukemia: A review of the literature. Leuk. Res. Treat. 2012. [CrossRef] [PubMed]
- Baurska, H.; Kiełbiński, M.; Biecek, P.; Haus, O.; Jaźwiec, B.; Kutner, A.; Marcinkowska, E. Monocytic differentiation induced by side-chain modified analogs of vitamin D in ex vivo ex vivo cells from patients with acute myeloid leukemia. Leuk. Res. 2014, 38, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Baurska, H.; Klopot, A.; Kielbinski, M.; Chrobak, A.; Wijas, E.; Kutner, A.; Marcinkowska, E. Structure-function analysis of vitamin D2 analogs as potential inducers of leukemia differentiation and inhibitors of prostate cancer proliferation. J. Steroid Biochem. Mol. Biol. 2011, 126, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, A.; Bermudez, M.A.; Seoane, S.; Perez-Fernandez, R.; Krupa, M.; Pietraszek, A.; Chodyński, M.; Kutner, A.; Brown, G.; Marcinkowska, E. Biological evaluation of new vitamin D2 analogues. J. Steroid Biochem. Mol. Biol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Pike, J.W.; Meyer, M.B.; Lee, S.M. The vitamin D receptor: Biochemical, molecular, biological, and genomic era investigations. Vitam. D 2011, 1, 97–135. [Google Scholar]
- Pike, J.W.; Meyer, M.B. Fundamentals of vitamin D hormone-regulated gene expression. J. Steroid Biochem. Mol. Biol. 2014, 114, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Horváth, H.C.; Khabir, Z.; Nittke, T.; Gruber, S.; Speer, G.; Manhardt, T.; Bonner, E.; Kallay, E. CYP24A1 splice variants--implications for the antitumorigenic actions of 1,25-(OH)2D3 in colorectal cancer. J. Steroid Biochem Mol. Biol. 2010, 121, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Nadkarni, S.; Chodyński, M.; Krajewski, K.; Cmoch, P.; Marcinkowska, E.; Brown, G.; Kutner, A. Convergent synthesis of double point modified analogs of 1α,25-dihydroxyvitamin D2 for biological evaluation. J. Steroid Biochem. Mol. Biol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Kusudo, T.; Sakaki, T.; Abe, D.; Fujishima, T.; Kittaka, A.; Takayama, H.; Hatakeyama, S.; Ohta, M.; Inouye, K. Metabolism of A-ring diastereomers of 1α,25-dihydroxyvitamin D3 by CYP24A1. Biochem. Biophys. Res. Commun. 2014, 321, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Urushino, N.; Yasuda, K.; Ikushiro, S.; Kamakura, M.; Ohta, M.; Sakaki, T. Metabolism of 1α,25-dihydroxyvitamin D2 by human CYP24A1. Biochem. Biophys. Res. Commun. 2009, 384, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Bolla, N.R.; Corcoran, A.; Yasuda, K.; Chodyński, M.; Krajewski, K.; Cmoch, P.; Marcinkowska, E.; Brown, G.; Sakaki, T.; Kutner, A. Synthesis and evaluation of geometric analogs of 1α,25-dihydroxyvitamin D2 as potential therapeutics. J. Steroid Biochem. Mol. Biol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Nadkarni, S.; Chodynski, M.; Corcoran, A.; Marcinkowska, E.; Brown, G.; Kutner, A. Double point modified analogs of vitamin d as potent activators of vitamin D receptor. Curr. Pharm. Des. 2015, 21, 1741–1763. [Google Scholar] [CrossRef] [PubMed]
- Gocek, E.; Kielbinski, M.; Wylob, P.; Kutner, A.; Marcinkowska, E. Side-chain modified vitamin D analogs induce rapid accumulation of VDR in the cell nuclei proportionately to their differentiation-inducing potential. Steroids 2008, 73, 1359–1366. [Google Scholar] [CrossRef] [PubMed]
- Baurska, H.; Marchwicka, A.; Klopot, A.; Kutner, A.; Marcinkowska, E. Studies on the mechanisms of superagonistic pro-differentiating activities of side-chain modified analogs of vitamin D2. Oncol. Rep. 2012, 28, 1110–1116. [Google Scholar] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corcoran, A.; Nadkarni, S.; Yasuda, K.; Sakaki, T.; Brown, G.; Kutner, A.; Marcinkowska, E. Biological Evaluation of Double Point Modified Analogues of 1,25-Dihydroxyvitamin D2 as Potential Anti-Leukemic Agents. Int. J. Mol. Sci. 2016, 17, 91. https://doi.org/10.3390/ijms17020091
Corcoran A, Nadkarni S, Yasuda K, Sakaki T, Brown G, Kutner A, Marcinkowska E. Biological Evaluation of Double Point Modified Analogues of 1,25-Dihydroxyvitamin D2 as Potential Anti-Leukemic Agents. International Journal of Molecular Sciences. 2016; 17(2):91. https://doi.org/10.3390/ijms17020091
Chicago/Turabian StyleCorcoran, Aoife, Sharmin Nadkarni, Kaori Yasuda, Toshiyuki Sakaki, Geoffrey Brown, Andrzej Kutner, and Ewa Marcinkowska. 2016. "Biological Evaluation of Double Point Modified Analogues of 1,25-Dihydroxyvitamin D2 as Potential Anti-Leukemic Agents" International Journal of Molecular Sciences 17, no. 2: 91. https://doi.org/10.3390/ijms17020091
APA StyleCorcoran, A., Nadkarni, S., Yasuda, K., Sakaki, T., Brown, G., Kutner, A., & Marcinkowska, E. (2016). Biological Evaluation of Double Point Modified Analogues of 1,25-Dihydroxyvitamin D2 as Potential Anti-Leukemic Agents. International Journal of Molecular Sciences, 17(2), 91. https://doi.org/10.3390/ijms17020091