CXCL12/CXCR4 Axis Regulates Aggrecanase Activation and Cartilage Degradation in a Post-Traumatic Osteoarthritis Rat Model
Abstract
:1. Introduction
2. Results
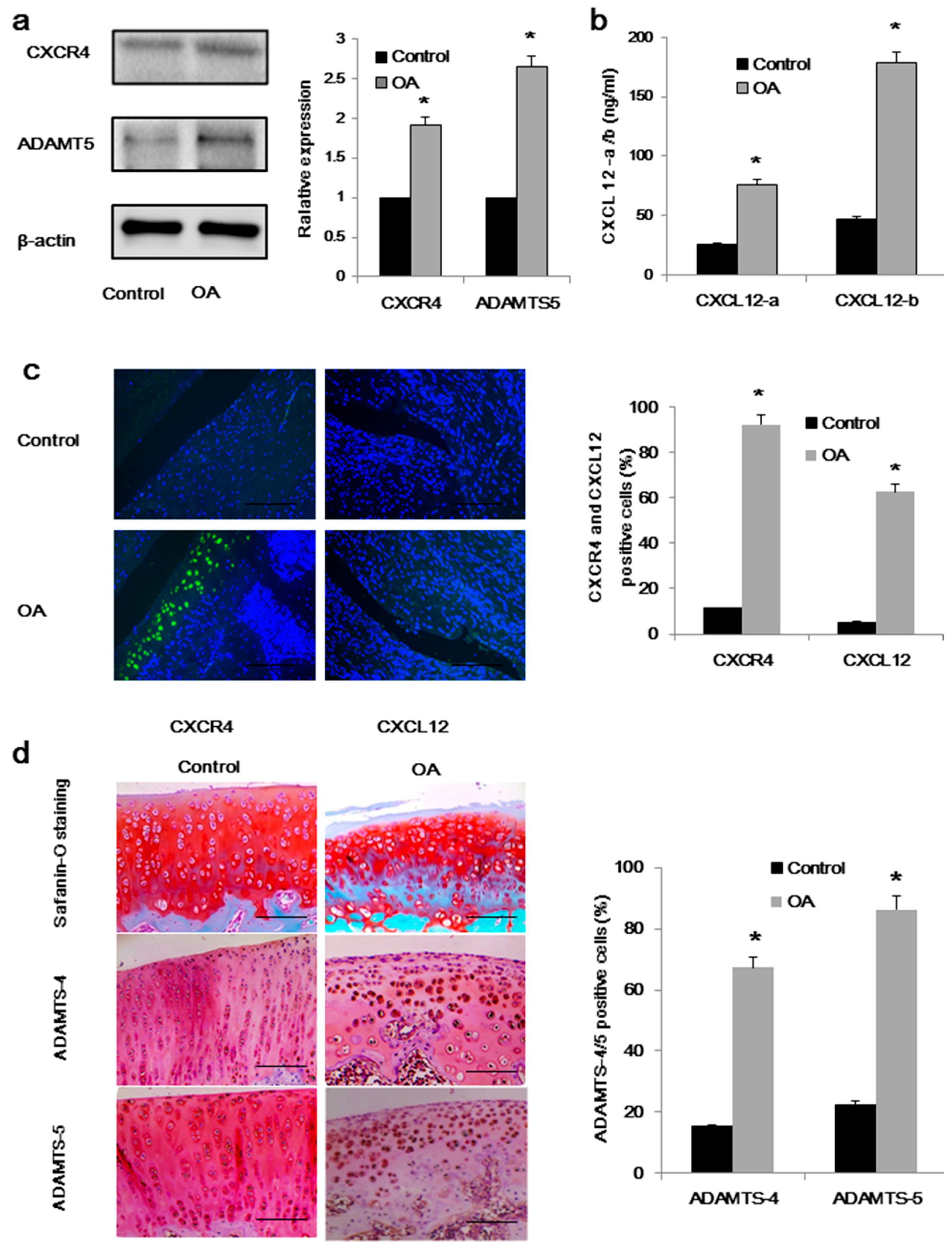
2.1. High Expression of CXCL12/CXCR4 and ADAMTS-5 in Osteoarthritis (OA) Rats
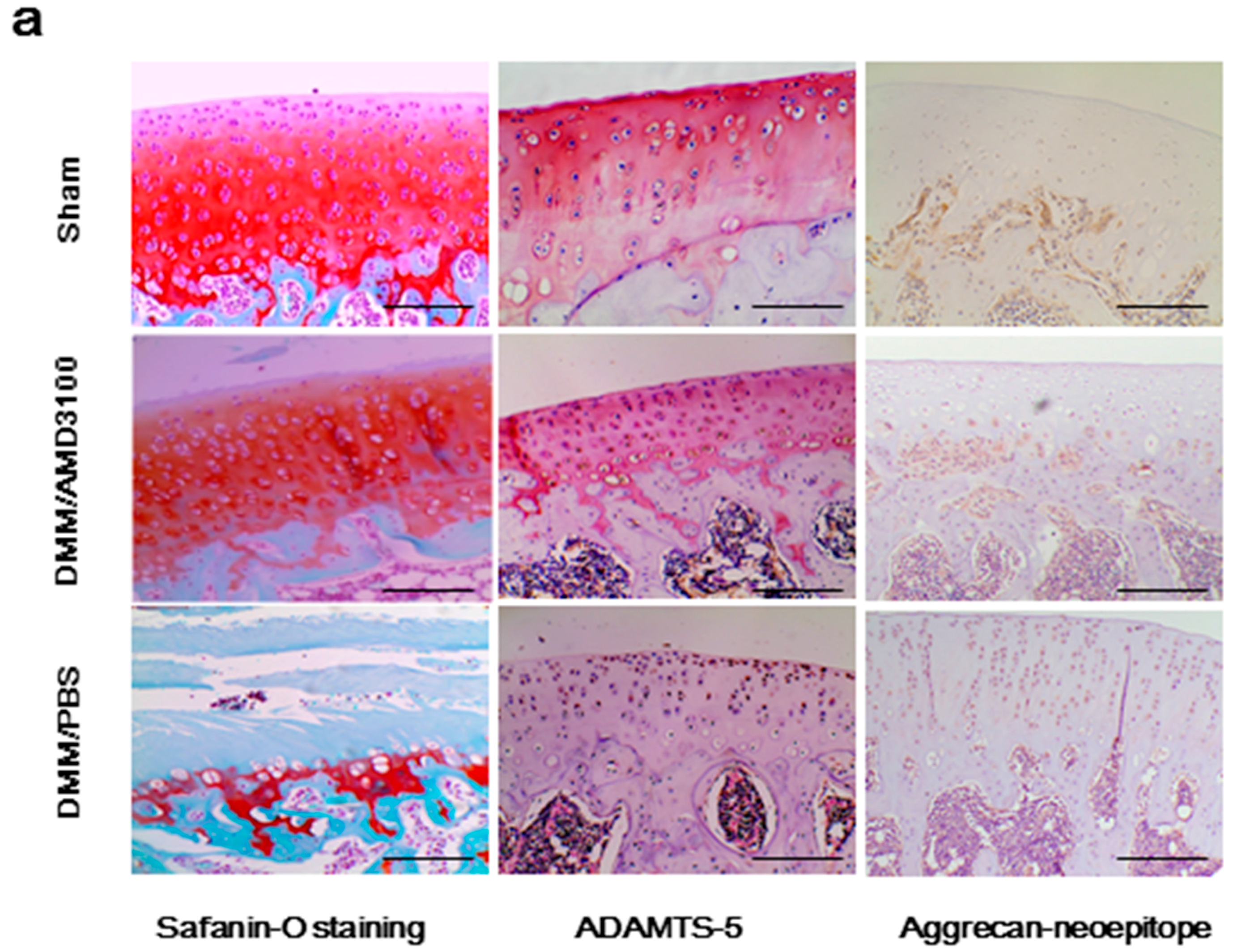
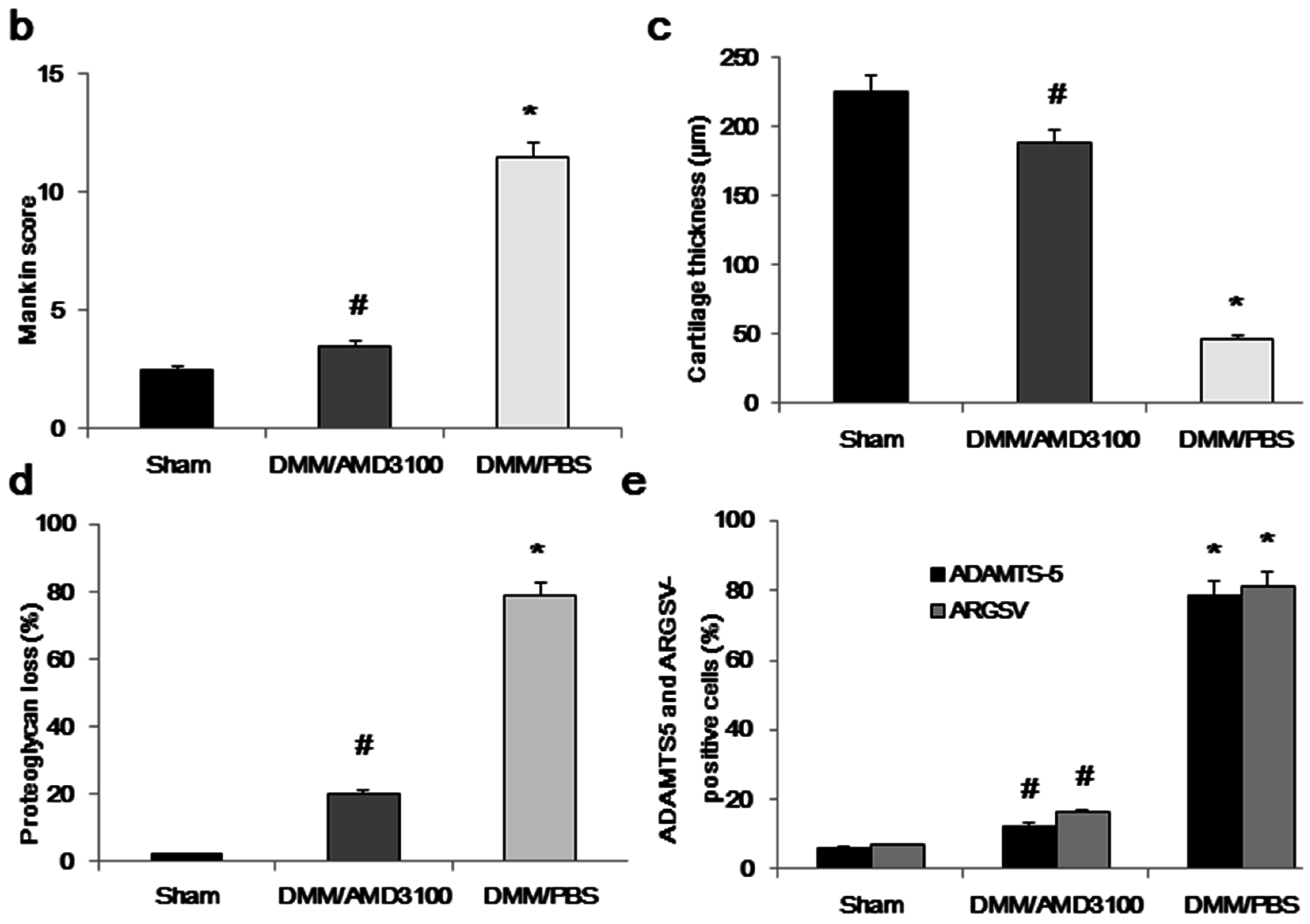
2.2. Inhibition of the CXCL12/CXCR4 Axis Results in Reduced Cartilage Destruction and Alleviates OA Severity
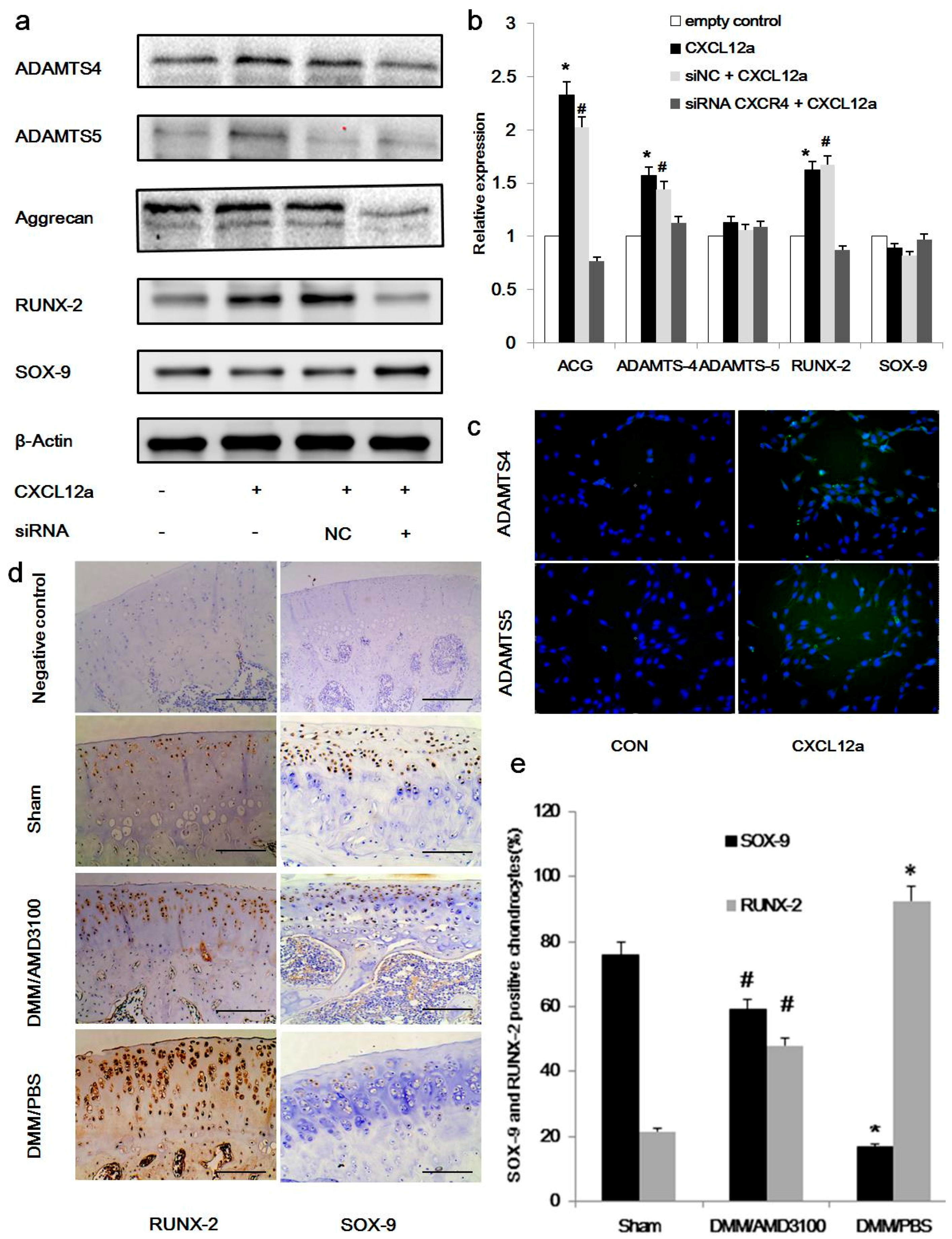
2.3. CXCR4 Knockdown Inhibits IL-Induced Expression of ACAN, RUNX-2, and ADAMTS-4/5, and Enhances the Expression of SOX-9
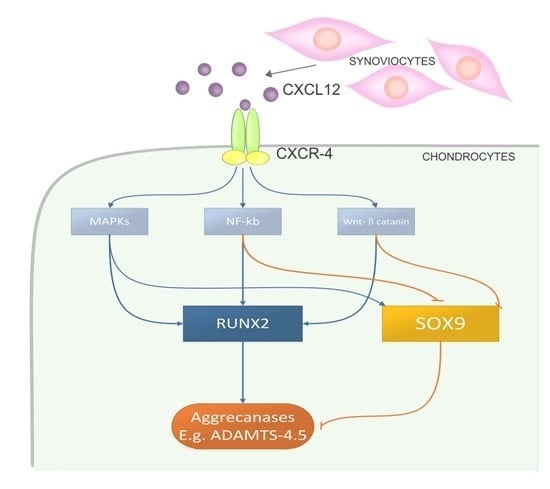
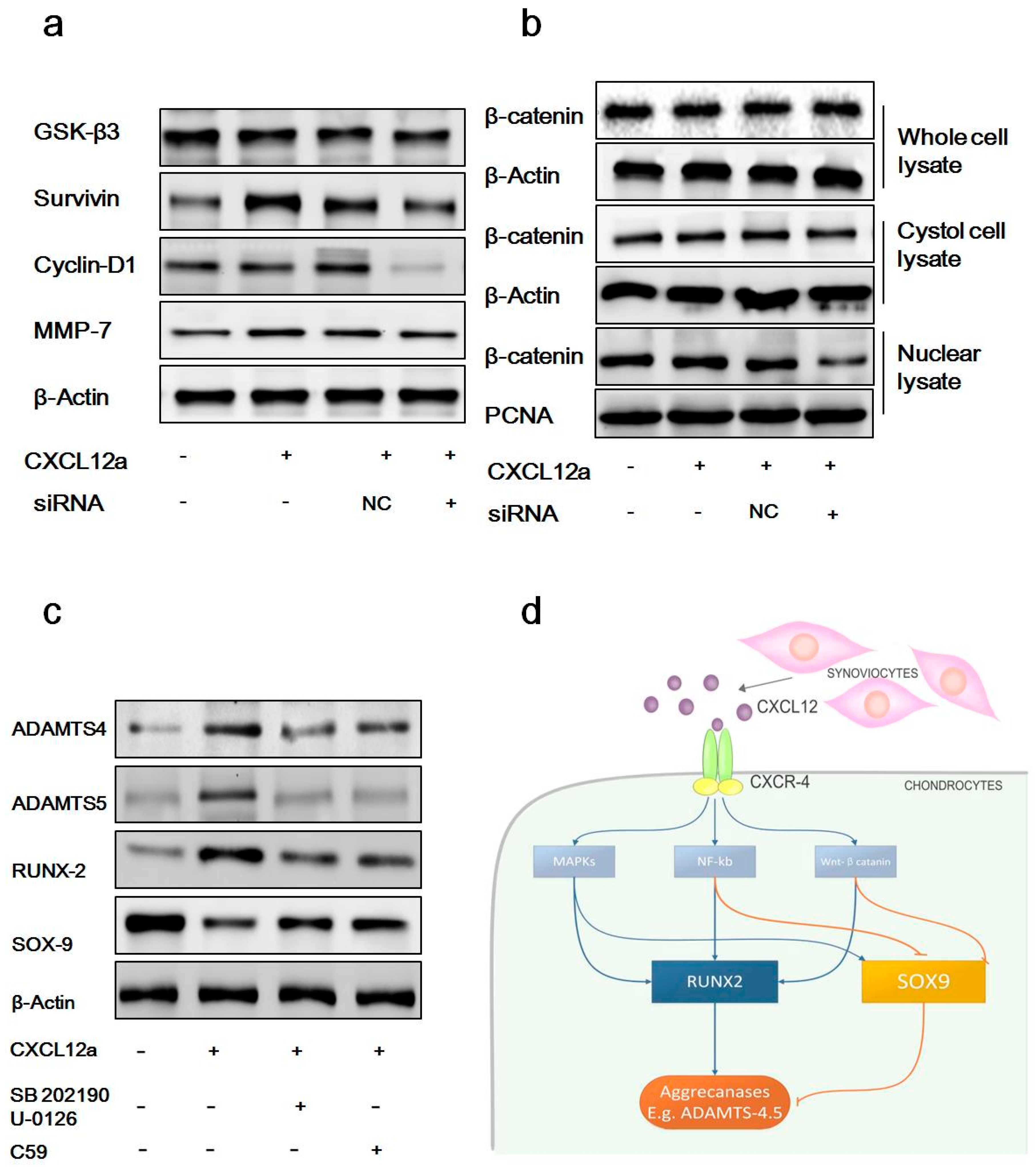
2.4. CXCL12a-Mediated Aggrecanase Activation of Chondrocytes Is Dependent on the NF-κB, MAPK, and Wnt/β-Catenin Pathways
3. Discussion
4. Materials and Methods
4.1. Reagents and Antibodies
4.2. Animals and Experimental Design
4.3. Cell Culture and Treatment
4.4. Western Blot
4.5. Real-Time Reverse Transcription-Polymerase Chain Reaction Analysis
4.6. Enzyme-Linked Immunosorbent Assay for Synovial Fluid
4.7. RNA Interference Assay
4.8. Histological Assessment
4.9. Immunohistochemistry and Immunofluorescence
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brooks, P.M. The burden of musculoskeletal disease—A global perspective. Clin. Rheumatol. 2006, 25, 778–781. [Google Scholar] [CrossRef] [PubMed]
- Goldring, S.R.; Goldring, M.B. Bone and cartilage in osteoarthritis: Is what’s best for one good or bad for the other? Arthritis Res. Ther. 2010, 12, 143. [Google Scholar] [CrossRef] [PubMed]
- Scanzello, C.R.; McKeon, B.; Swaim, B.H.; DiCarlo, E.; Asomugha, E.U.; Kanda, V.; Nair, A.; Lee, D.M.; Richmond, J.C.; Katz, J.N.; et al. Synovial inflammation in patients undergoing arthroscopic meniscectomy: Molecular characterization and relationship to symptoms. Arthritis Rheum. 2011, 63, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Lohmander, L.S.; Englund, P.M.; Dahl, L.L.; Roos, E.M. The long-term consequence of anterior cruciate ligament and meniscus injuries: Osteoarthritis. Am. J. Sports Med. 2007, 35, 1756–1769. [Google Scholar] [CrossRef] [PubMed]
- Pattoli, M.A.; MacMaster, J.F.; Gregor, K.R.; Burke, J.R. Collagen and aggrecan degradation is blocked in interleukin-1-treated cartilage explants by an inhibitor of IκB kinase through suppression of metalloproteinase expression. J. Pharmacol. Exp. Ther. 2005, 315, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Pratta, M.A.; Yao, W.; Decicco, C.; Tortorella, M.D.; Liu, R.Q.; Copeland, R.A.; Magolda, R.; Newton, R.C.; Trzaskos, J.M.; Arner, E.C. Aggrecan protects cartilage collagen from proteolytic cleavage. J. Biol. Chem. 2003, 278, 45539–45545. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, M.K.; Askew, R.; Schelling, S.; Stedman, N.; Blanchet, T.; Hopkins, B.; Morris, E.A.; Glasson, S.S. Double-knockout of ADAMTS-4 and ADAMTS-5 in mice results in physiologically normal animals and prevents the progression of osteoarthritis. Arthritis Rheum. 2007, 56, 3670–3674. [Google Scholar] [CrossRef] [PubMed]
- Song, R.H.; Tortorella, M.D.; Malfait, A.M.; Alston, J.T.; Yang, Z.; Arner, E.C.; Griggs, D.W. Aggrecan degradation in human articular cartilage explants is mediated by both ADAMTS-4 and ADAMTS-5. Arthritis Rheum. 2007, 56, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Stanton, H.; Rogerson, F.M.; East, C.J.; Golub, S.B.; Lawlor, K.E.; Meeker, C.T.; Little, C.B.; Last, K.; Farmer, P.J.; Campbell, I.K.; et al. ADAMTS-5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 2005, 434, 648–652. [Google Scholar] [CrossRef] [PubMed]
- Verma, P.; Dalal, K. ADAMTS-4 and ADAMTS-5: Key enzymes in osteoarthritis. J. Cell. Biochem. 2011, 112, 3507–3514. [Google Scholar] [CrossRef] [PubMed]
- Glasson, S.S.; Askew, R.; Sheppard, B.; Carito, B.; Blanchet, T.; Ma, H.L.; Flannery, C.R.; Peluso, D.; Kanki, K.; Yang, Z.; et al. Deletion of active ADAMTS-5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 2005, 434, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Kanbe, K.; Takagishi, K.; Chen, Q. Stimulation of matrix metalloprotease 3 release from human chondrocytes by the interaction of stromal cell-derived factor 1 and CXC chemokine receptor 4. Arthritis Rheum. 2002, 46, 130–137. [Google Scholar] [CrossRef]
- Han, Y.; He, T.; Huang, D.R.; Pardo, C.A.; Ransohoff, R.M. TNF-α mediates SDF-1α-induced NF-κB activation and cytotoxic effects in primary astrocytes. J. Clin. Investig. 2001, 108, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Cho, M.L.; Kim, H.R.; Ju, J.H.; Park, M.K.; Oh, H.J.; Kim, J.S.; Park, S.H.; Lee, S.H.; Kim, H.Y. Up-regulation of stromal cell-derived factor 1 (CXCL12) production in rheumatoid synovial fibroblasts through interactions with T lymphocytes: Role of interleukin-17 and CD40L-CD40 interaction. Arthritis Rheum. 2007, 56, 1076–1086. [Google Scholar] [CrossRef] [PubMed]
- Hitchon, C.; Wong, K.; Ma, G.; Reed, J.; Lyttle, D.; El-Gabalawy, H. Hypoxia-induced production of stromal cell-derived factor 1 (CXCL12) and vascular endothelial growth factor by synovial fibroblasts. Arthritis Rheum. 2002, 46, 2587–2597. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Tao, J.; Zhu, S.; Cai, Y.; Mao, Q.; Yu, D.; Dai, J.; Ouyang, H. Radially oriented collagen scaffold with SDF-1 promotes osteochondral repair by facilitating cell homing. Biomaterials 2015, 39, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Park, M.S.; Kim, Y.H.; Jung, Y.; Kim, S.H.; Park, J.C.; Yoon, D.S.; Kim, S.H.; Lee, J.W. In situ recruitment of human bone marrow-derived mesenchymal stem cells using chemokines for articular cartilage regeneration. Cell Transpl. 2015, 24, 1067–1083. [Google Scholar] [CrossRef] [PubMed]
- Thomas, N.P.; Li, P.; Fleming, B.C.; Chen, Q.; Wei, X.; Pan, X.-H.; Li, G.; Wei, L. Attenuation of cartilage pathogenesis in post-traumatic osteoarthritis (PTOA) in mice by blocking the stromal derived factor 1 receptor (CXCR4) with the specific inhibitor, AMD3100. J. Orthop. Res. 2015, 33, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Y.; Chen, Y.; Tang, X.J.; Jiang, L.H.; Ji, P. AMD3100 attenuates matrix metalloprotease-3 and -9 expressions and prevents cartilage degradation in a monosodium iodo-acetate-induced rat model of temporomandibular osteoarthritis. J. Oral Maxillofac. Surg. 2016, 74, 927.e1–927.e13. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Chen, J.; Tao, J.; Jiang, Y.; Hu, C.; Huang, L.; Ji, J.; Ouyang, H.W. The use of type 1 collagen scaffold containing stromal cell-derived factor-1 to create a matrix environment conducive to partial-thickness cartilage defects repair. Biomaterials 2013, 34, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.W.; Han, M.S.; Park, H.R.; Lee, E.J.; Jung, Y.K.; Usmani, S.E.; Ulici, V.; Han, S.W.; Beier, F. CXC chemokine ligand 12a enhances chondrocyte proliferation and maturation during endochondral bone formation. Osteoarthr. Cartil. 2015, 23, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Fleming, B.C.; Sun, X.; Teeple, E.; Wu, W.; Jay, G.D.; Elsaid, K.A.; Luo, J.; Machan, J.T.; Chen, Q. Comparison of differential biomarkers of osteoarthritis with and without posttraumatic injury in the Hartley guinea pig model. J. Orthop. Res. 2010, 28, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Appleton, C.T.; Pitelka, V.; Henry, J.; Beier, F. Global analyses of gene expression in early experimental osteoarthritis. Arthritis Rheum. 2007, 56, 1854–1868. [Google Scholar] [CrossRef] [PubMed]
- Blom, A.B.; Brockbank, S.M.; van Lent, P.L.; van Beuningen, H.M.; Geurts, J.; Takahashi, N.; van der Kraan, P.M.; van de Loo, F.A.; Schreurs, B.W.; Clements, K.; et al. Involvement of the Wnt signaling pathway in experimental and human osteoarthritis: Prominent role of Wnt-induced signaling protein 1. Arthritis Rheum. 2009, 60, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Yaykasli, K.O.; Hatipoglu, O.F.; Yaykasli, E.; Yildirim, K.; Kaya, E.; Ozsahin, M.; Uslu, M.; Gunduz, E. Leptin induces ADAMTS-4, ADAMTS-5, and ADAMTS-9 genes expression by mitogen-activated protein kinases and NF-κB signaling pathways in human chondrocytes. Cell Biol. Int. 2015, 39, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Prasadam, I.; Crawford, R.; Xiao, Y. Aggravation of ADAMTS and matrix metalloproteinase production and role of Erk1/2 pathway in the interaction of osteoarthritic subchondral bone osteoblasts and articular cartilage chondrocytes—Possible pathogenic role in osteoarthritis. J. Rheumatol. 2012, 39, 621–634. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, G.; Cao, B.; Gao, G.; Ma, K.; Chen, W.; Xu, P.; Yang, G. Influence on matrix metalloproteinases 3, 9, and 13 levels after blocking stromal cell derived factor 1/chemokine receptor 4 signaling pathway with AMD3100. Chin. J. Repar. Reconstr. Surg. 2012, 26, 652–656. [Google Scholar]
- Wei, F.; Moore, D.C.; Wei, L.; Li, Y.; Zhang, G.; Wei, X.; Lee, J.K.; Chen, Q. Attenuation of osteoarthritis via blockade of the SDF-1/CXCR4 signaling pathway. Arthritis Res. Ther. 2012, 14, R177. [Google Scholar] [CrossRef] [PubMed]
- Thirunavukkarasu, K.; Pei, Y.; Wei, T. Characterization of the human ADAMTS-5 (aggrecanase-2) gene promoter. Mol. Biol. Rep. 2007, 34, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Thirunavukkarasu, K.; Pei, Y.; Moore, T.L.; Wang, H.; Yu, X.P.; Geiser, A.G.; Chandrasekhar, S. Regulation of the human ADAMTS-4 promoter by transcription factors and cytokines. Biochem. Biophys. Res. Commun. 2006, 345, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Miyaki, S.; Sato, T.; Inoue, A.; Otsuki, S.; Ito, Y.; Yokoyama, S.; Kato, Y.; Takemoto, F.; Nakasa, T.; Yamashita, S.; et al. MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev. 2010, 24, 1173–1185. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Ji, Q.; Wang, X.; Kang, L.; Fu, Y.; Yin, Y.; Li, Z.; Liu, Y.; Xu, X.; Wang, Y. SOX-9 is a regulator of ADAMTSs-induced cartilage degeneration at the early stage of human osteoarthritis. Osteoarthr. Cartil. 2015, 23, 2259–2268. [Google Scholar] [CrossRef] [PubMed]
- Tetsunaga, T.; Nishida, K.; Furumatsu, T.; Naruse, K.; Hirohata, S.; Yoshida, A.; Saito, T.; Ozaki, T. Regulation of mechanical stress-induced MMP-13 and ADAMTS-5 expression by RUNX-2 transcriptional factor in SW1353 chondrocyte-like cells. Osteoarthr. Cartil. 2011, 19, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Beier, F.; Loeser, R.F. Biology and pathology of Rho GTPase, PI-3 kinase-Akt, and MAP kinase signaling pathways in chondrocytes. J. Cell. Biochem. 2010, 110, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Sondergaard, B.C.; Schultz, N.; Madsen, S.H.; Bay-Jensen, A.C.; Kassem, M.; Karsdal, M.A. MAPKs are essential upstream signaling pathways in proteolytic cartilage degradation—Divergence in pathways leading to aggrecanase and MMP-mediated articular cartilage degradation. Osteoarthr. Cartil. 2010, 18, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Mengshol, J.A.; Vincenti, M.P.; Brinckerhoff, C.E. IL-1 induces collagenase-3 (MMP-13) promoter activity in stably transfected chondrocytic cells: Requirement for RUNX-2 and activation by p38 MAPK and JNK pathways. Nucleic Acids Res. 2001, 29, 4361–4372. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Nishida, K.; Furumatsu, T.; Yoshida, A.; Ozawa, M.; Ozaki, T. Histone deacetylase inhibitors suppress mechanical stress-induced expression of RUNX-2 and ADAMTS-5 through the inhibition of the MAPK signaling pathway in cultured human chondrocytes. Osteoarthr. Cartil. 2013, 21, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Kan, M.; McKeehan, W.L.; de Crombrugghe, B. Up-regulation of the chondrogenic SOX-9 gene by fibroblast growth factors is mediated by the mitogen-activated protein kinase pathway. Proc. Natl. Acad. Sci. USA 2000, 97, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Yuan, W.; Fujita, N.; Wang, J.; Wang, H.; Shapiro, I.M.; Risbud, M.V. Inflammatory cytokines associated with degenerative disc disease control aggrecanase-1 (ADAMTS-4) expression in nucleus pulposus cells through MAPK and NF-κB. Am. J. Pathol. 2013, 182, 2310–2321. [Google Scholar] [CrossRef] [PubMed]
- Chockalingam, P.S.; Varadarajan, U.; Sheldon, R.; Fortier, E.; LaVallie, E.R.; Morris, E.A.; Yaworsky, P.J.; Majumdar, M.K. Involvement of protein kinase Czeta in interleukin-1beta induction of ADAMTS-4 and type 2 nitric oxide synthase via NF-κB signaling in primary human osteoarthritic chondrocytes. Arthritis Rheum. 2007, 56, 4074–4083. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Lefebvre, V.; de Crombrugghe, B. Potent inhibition of the master chondrogenic factor SOX-9 gene by interleukin-1 and tumor necrosis factor-α. J. Biol. Chem. 2000, 275, 3687–3692. [Google Scholar] [CrossRef] [PubMed]
- Corr, M. Wnt-β-catenin signaling in the pathogenesis of osteoarthritis. Nat. Clin. Pract. Rheumatol. 2008, 4, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Papathanasiou, I.; Malizos, K.N.; Tsezou, A. Low-density lipoprotein receptor-related protein 5 (LRP5) expression in human osteoarthritic chondrocytes. J. Orthop. Res. 2010, 28, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Yano, F.; Kugimiya, F.; Ohba, S.; Ikeda, T.; Chikuda, H.; Ogasawara, T.; Ogata, N.; Takato, T.; Nakamura, K.; Kawaguchi, H.; et al. The canonical Wnt signaling pathway promotes chondrocyte differentiation in a SOX-9-dependent manner. Biochem. Biophys. Res. Commun. 2005, 333, 1300–1308. [Google Scholar] [CrossRef] [PubMed]
- Glasson, S.S.; Blanchet, T.J.; Morris, E.A. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthr. Cartil. 2007, 15, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Thirion, S.; Berenbaum, F. Culture and phenotyping of chondrocytes in primary culture. Methods Mol. Med. 2004, 100, 1–14. [Google Scholar] [PubMed]
- Guan, H.; Xie, L.; Klapproth, K.; Weitzer, C.D.; Wirth, T.; Ushmorov, A. Decitabine represses translocated MYC oncogene in Burkitt lymphoma. J. Pathol. 2013, 229, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Liu, H.; Zhang, X.; Xu, F.; Qin, L.; Cheng, P.; Huang, H.; Guo, F.; Yang, Q.; Chen, A. Inhibition of SDF-1α/CXCR4 signalling in subchondral bone attenuates post-traumatic osteoarthritis. Int. J. Mol. Sci. 2016, 17, 943. [Google Scholar] [CrossRef] [PubMed]
- Ostergaard, K.; Andersen, C.B.; Petersen, J.; Bendtzen, K.; Salter, D.M. Validity of histopathological grading of articular cartilage from osteoarthritic knee joints. Ann. Rheum. Dis. 1999, 58, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Mankin, H.J.; Dorfman, H.; Lippiello, L.; Zarins, A. Biochemical and metabolic abnormalities in articular cartilage from osteo-arthritic human hips. II. Correlation of morphology with biochemical and metabolic data. J. Bone Jt. Surg. Am. 1971, 53, 523–537. [Google Scholar]

| Target Rat Gene | Sequence |
|---|---|
| GAPDH | (F) 5′-GGCACAGTCAAGGCTGAGAATG-3′ |
| (R) 5′-GGTGGTGAAGACGCCAGTA-3′ | |
| Aggrecan | (F) 5′-CTTCCCAACTATCCAGCCAT-3′ |
| (R) 5′-TCACACCGATAGATCCCAGA-3′ | |
| ADAMTS-4 | (F) 5′-GGAATGGTGGAAAGTATTGTGAGG-3′ |
| (R) 5′-GGTCGGTTCGGTGGTTGTAG-3′ | |
| ADAMTS-5 | (F) 5′-CTCCATGCAGCTTTCACTGT-3′ |
| (R) 5′-CAGAATTTGGAATCGTCGTG-3′ | |
| SOX-9 | (F) 5′-ACATCAAGACGGAGCAAC-3′ |
| (R) 5′-TGAAGGTGGAGTAGAGCC-3′ | |
| RUNX-2 | (F) 5′-CCGCACGACAACCGCACCAT-3′; |
| (R) 5′-CGCTCCGGCCCACA AATCTC-3′ |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, W.; Shi, J.; Zhang, J.; Lv, Z.; Guo, F.; Huang, H.; Zhu, W.; Chen, A. CXCL12/CXCR4 Axis Regulates Aggrecanase Activation and Cartilage Degradation in a Post-Traumatic Osteoarthritis Rat Model. Int. J. Mol. Sci. 2016, 17, 1522. https://doi.org/10.3390/ijms17101522
Lu W, Shi J, Zhang J, Lv Z, Guo F, Huang H, Zhu W, Chen A. CXCL12/CXCR4 Axis Regulates Aggrecanase Activation and Cartilage Degradation in a Post-Traumatic Osteoarthritis Rat Model. International Journal of Molecular Sciences. 2016; 17(10):1522. https://doi.org/10.3390/ijms17101522
Chicago/Turabian StyleLu, Weiwei, Jia Shi, Jinming Zhang, Zhengtao Lv, Fengjing Guo, Hui Huang, Wentao Zhu, and Anmin Chen. 2016. "CXCL12/CXCR4 Axis Regulates Aggrecanase Activation and Cartilage Degradation in a Post-Traumatic Osteoarthritis Rat Model" International Journal of Molecular Sciences 17, no. 10: 1522. https://doi.org/10.3390/ijms17101522
APA StyleLu, W., Shi, J., Zhang, J., Lv, Z., Guo, F., Huang, H., Zhu, W., & Chen, A. (2016). CXCL12/CXCR4 Axis Regulates Aggrecanase Activation and Cartilage Degradation in a Post-Traumatic Osteoarthritis Rat Model. International Journal of Molecular Sciences, 17(10), 1522. https://doi.org/10.3390/ijms17101522