Impact of Chronic Hepatitis C Virus Genotype 1b Infection on Triglyceride Concentration in Serum Lipoprotein Fractions
Abstract
:1. Introduction
2. Results
2.1. Features of Patients with Chronic Active HCV G1b Infection (Active HCV Group) and Those with Cleared HCV Infection Due to Anti-Viral Therapy (Sustained Virological Response Group; SVR Group)
| Discrete Traits | Active HCV (n = 103); n (%) | SVR (n = 82); n (%) | p Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Sex | 0.08 | ||||||||
| Male | 36 (35) | 40 (49) | |||||||
| Female | 67 (65) | 42 (51) | |||||||
| Quantitative Traits | Median | (Q1–Q3) | Median | (Q1–Q3) | p Value | ||||
| Age (years) | 71.0 | 63.0–78.0 | 64.0 | 56.0–72.8 | 0.001 | ||||
| BMI (kg/m2) | 21.9 | 20.1–24.2 | 22.6 | 20.9–24.5 | 0.1 | ||||
| HbA1c (%) | 5.5 | 5.2–5.8 | 5.6 | 5.3–6.0 | 0.1 | ||||
| AST (IU/L) | 41.0 | 29.5–63.5 | 22.0 | 19.0–28.0 | <0.001 | ||||
| ALT (IU/L) | 33.0 | 23.0–53.5 | 16.0 | 13.0–22.8 | <0.001 | ||||
| Total bilirubin (mg/dL) | 0.7 | 0.6–0.9 | 0.6 | 0.5–0.9 | 0.050 | ||||
| γ-GTP (IU/L) | 30.0 | 20.5–49.0 | 20.0 | 15.0–30.0 | <0.001 | ||||
| Albumin (g/dL) | 4.1 | 3.8–4.3 | 4.4 | 4.2–4.6 | <0.001 | ||||
| Platelet (104/μL) | 15.0 | 11.2–20.4 | 18.0 | 14.8–21.8 | 0.002 | ||||
| Lipid Profiles | |||||||||
| Total cholesterol (mg/dL) | 174.0 | 151.5–192.0 | 196.0 | 176.3–217.0 | <0.001 | ||||
| Triglyceride (mg/dL) | 88.0 | 67.0–112.0 | 89.5 | 70.3–135.0 | 0.4 | ||||
| LDL cholesterol (mg/dL) | 90.0 | 74.0–111.0 | 112.0 | 93.0–132.0 | <0.001 | ||||
| HDL cholesterol (mg/dL) | 61.2 | 48.0–72.4 | 63.0 | 53.3–77.9 | 0.2 | ||||
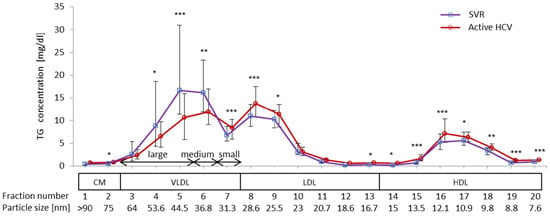
2.2. Difference in TG Profiles in Serum Lipoprotein Fractions between Active HCV and SVR Groups
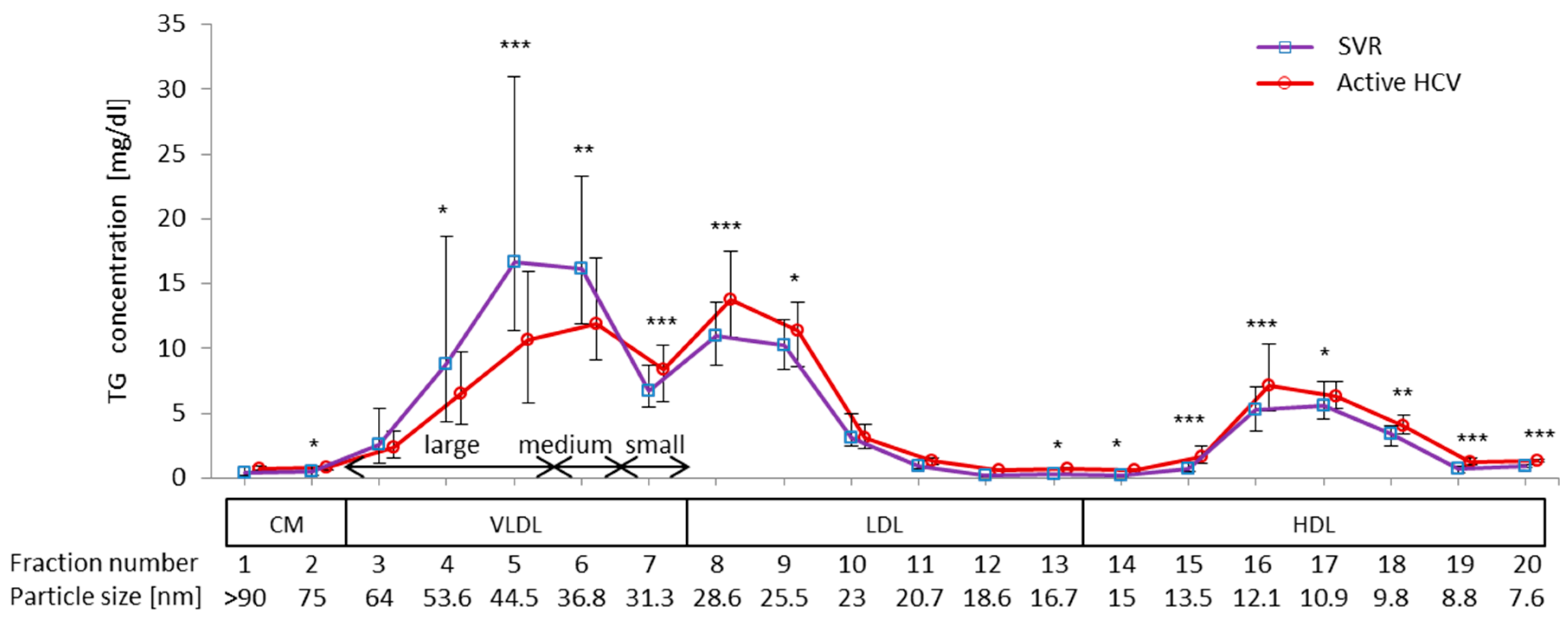
2.3. Impact of Chronic Active HCV G1b Infection on the Concentration of TG in the Four Major Classes of Lipoproteins
| VLDL-TG | LDL-TG | HDL-TG | |||||||
|---|---|---|---|---|---|---|---|---|---|
| B | SE | p Value | B | SE | p Value | B | SE | p Value | |
| Constant | 2.4 | 44.4 | 1.0 | 9.2 | 16.2 | 0.6 | 22.7 | 8.6 | 0.01 |
| HCV infection (Active HCV vs. SVR) | −13.8 | 6.9 | 0.047 | 1.6 | 2.5 | 0.52 | 3.1 | 1.3 | 0.02 |
| Age | −0.01 | 0.2 | 0.96 | 0.2 | 0.08 | 0.01 | 0.005 | 0.04 | 0.9 |
| Female gender | −12.7 | 5.8 | 0.03 | −0.3 | 2.1 | 0.9 | 2.6 | 1.1 | 0.02 |
| BMI | 0.99 | 0.8 | 0.2 | 0.4 | 0.3 | 0.2 | 0.08 | 0.2 | 0.6 |
| ALT | −0.02 | 0.1 | 0.8 | 0.1 | 0.04 | 0.03 | −0.01 | 0.02 | 0.7 |
| γ-GTP | 0.2 | 0.08 | 0.01 | 0.06 | 0.03 | 0.04 | 0.03 | 0.01 | 0.09 |
| Albumin | 10.6 | 7.6 | 0.2 | −2.7 | 2.8 | 0.3 | −1.1 | 1.5 | 0.4 |
| HbA1c | 0.06 | 4.9 | 0.99 | 1.2 | 1.8 | 0.5 | −0.6 | 1.0 | 0.5 |
| Platelet | −0.3 | 0.2 | 0.3 | −0.08 | 0.09 | 0.4 | −0.01 | 0.05 | 0.8 |
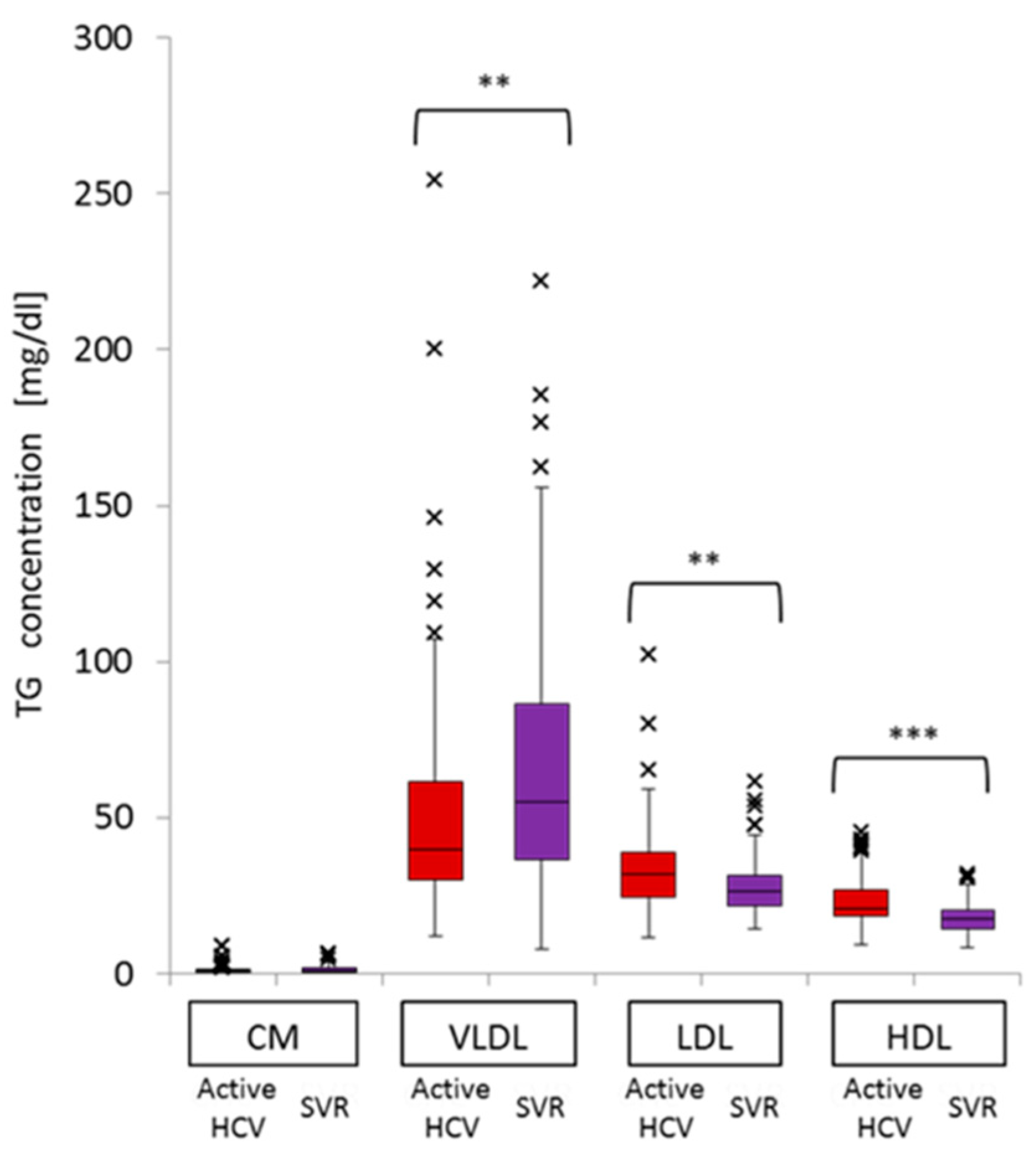
2.4. Impact of Active HCV G1b Infection on TG Concentrations in the Three Sub-Fractions of Very Low-Density Lipoprotein (VLDL)
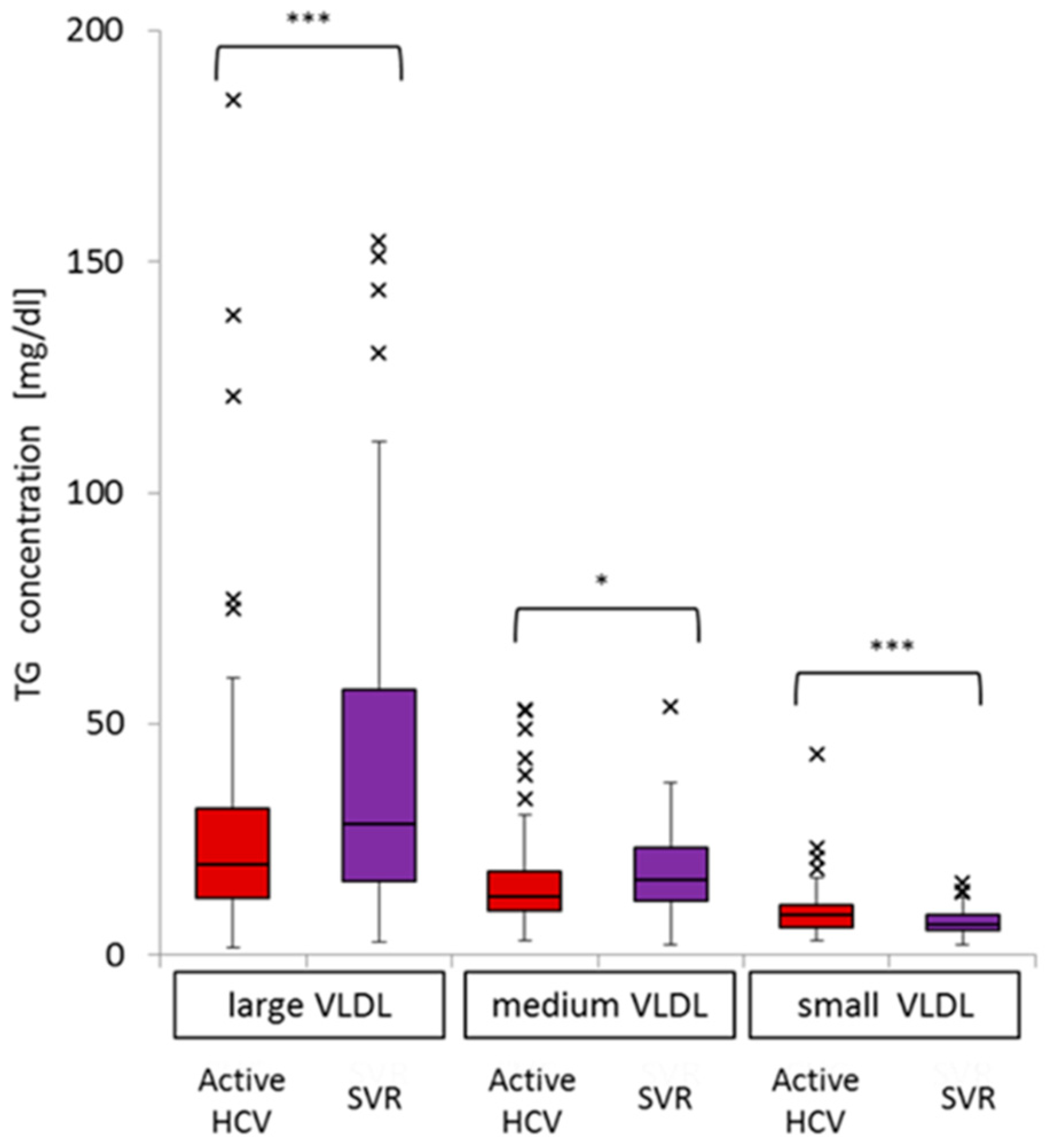
| Large VLDL-TG | Medium VLDL-TG | Small VLDL-TG | |||||||
|---|---|---|---|---|---|---|---|---|---|
| B | SE | p Value | B | SE | p Value | B | SE | p Value | |
| Constant | −28.2 | 33.2 | 0.4 | 12.1 | 11.7 | 0.3 | 18.5 | 5.6 | 0.001 |
| HCV infection (Active HCV vs. SVR) | −10.6 | 5.1 | 0.04 | −3.2 | 1.8 | 0.08 | −0.04 | 0.9 | 0.97 |
| Age | −0.06 | 0.2 | 0.7 | 0.003 | 0.06 | 0.96 | 0.04 | 0.03 | 0.1 |
| Female gender | −10.2 | 4.3 | 0.02 | −2.9 | 1.5 | 0.06 | 0.3 | 0.7 | 0.7 |
| BMI | 0.7 | 0.6 | 0.2 | 0.2 | 0.2 | 0.4 | 0.1 | 0.1 | 0.3 |
| ALT | −0.04 | 0.07 | 0.6 | −0.002 | 0.03 | 0.93 | 0.02 | 0.01 | 0.1 |
| γ-GTP | 0.1 | 0.06 | 0.03 | 0.05 | 0.02 | 0.02 | 0.02 | 0.01 | 0.049 |
| Albumin | 14.3 | 5.7 | 0.01 | −0.3 | 2.00 | 0.9 | −3.4 | 0.9 | <0.001 |
| HbA1c | −0.4 | 3.7 | 0.9 | 0.8 | 1.3 | 0.5 | −0.3 | 0.6 | 0.6 |
| Platelet | −0.2 | 0.2 | 0.4 | −0.09 | 0.06 | 0.2 | −0.02 | 0.03 | 0.5 |
2.5. Significance of Advanced Liver Fibrosis on Serum Levels of VLDL-TG, LDL-TG, and High-Density Lipoprotein (HDL)-TG in Patients with Chronic Active HCV G1b Infection
| Discrete Traits | Advanced Fibrosis (n = 51); n (%) | Mild-Moderate Fibrosis (n = 52); n (%) | p Value | ||
|---|---|---|---|---|---|
| Sex | 0.07 | ||||
| Male | 13 (25) | 23 (44) | |||
| Female | 38 (75) | 29 (56) | |||
| IFNL3 genotype (rs8099917) | 0.06 | ||||
| TT | 40 (78) | 33 (63) | |||
| Non-TT | 9 (18) | 18 (35) | |||
| Not done | 2 (4) | 1 (2) | |||
| Quantitative Traits | Median | (Q1–Q3) | Median | (Q1–Q3) | p Value |
| Age (years) | 75.0 | 68.5–80.5 | 66.0 | 52.8–73.0 | <0.001 |
| BMI (kg/m2) | 22.1 | 20.1–24.1 | 21.5 | 20.3–24.4 | 0.97 |
| HbA1c (%) | 5.5 | 5.0–5.8 | 5.5 | 5.3–5.8 | 0.4 |
| HCV-RNA (log IU/mL) | 6.5 | 6.0–6.8 | 6.7 | 5.8–7.1 | 0.2 |
| AST (IU/L) | 58.0 | 38.5–82.0 | 30.5 | 25.0–42.0 | <0.001 |
| ALT (IU/L) | 44.0 | 27.0–62.5 | 30.0 | 19.8–47.3 | 0.01 |
| Total bilirubin (mg/dL) | 0.7 | 0.6–0.9 | 0.8 | 0.6–0.8 | 0.7 |
| γ-GTP (IU/L) | 33.0 | 22.0–48.5 | 30.0 | 18.0–49.5 | 0.5 |
| Albumin (g/dL) | 4.0 | 3.6–4.3 | 4.2 | 4.0–4.3 | 0.01 |
| Platelet (104/μL) | 11.3 | 8.5–14.3 | 19.6 | 15.6–23.0 | <0.001 |
| Lipid Profiles | |||||
| Total cholesterol (mg/dL) | 168.0 | 142.0–187.5 | 176.5 | 160.8–193.3 | 0.04 |
| Triglyceride (mg/dL) | 89.0 | 68.5–113.5 | 86.5 | 63.0–105.3 | 0.6 |
| LDL cholesterol (mg/dL) | 84.0 | 69.0–99.5 | 96.0 | 79.8–112.5 | 0.04 |
| HDL cholesterol (mg/dL) | 58.4 | 46.9–68.0 | 63.1 | 50.3–73.2 | 0.2 |
2.6. Difference in VLDL-TG, LDL-TG, and HDL-TG Serum Levels between Patients with Advanced Liver Fibrosis and Those with Mild-Moderate Fibrosis

2.7. Impact of Advanced Liver Fibrosis on TG Concentration in the Four Serum Lipoprotein Classes in Patients with Chronic Active HCV G1b Infection
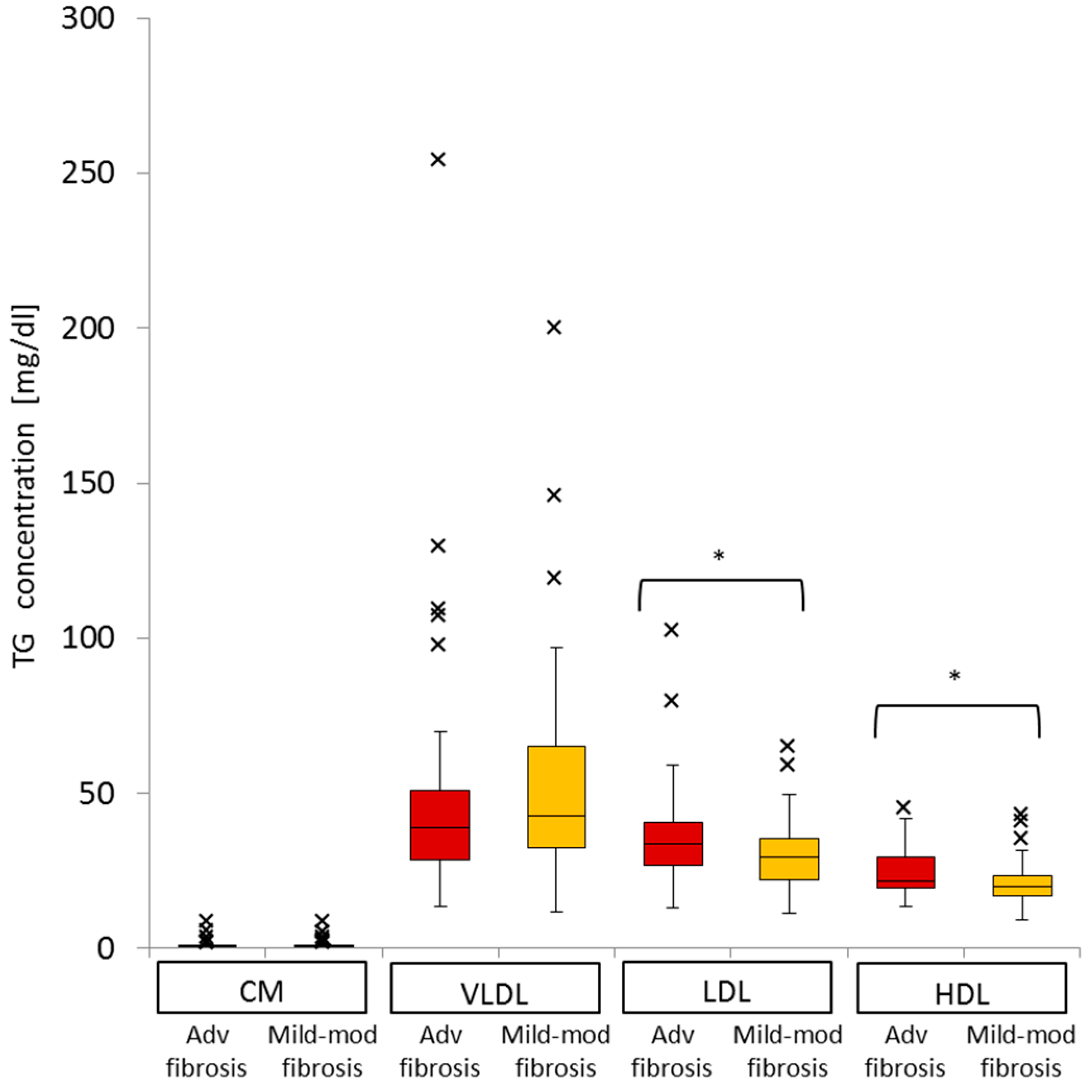
| VLDL-TG | LDL-TG | HDL-TG | |||||||
|---|---|---|---|---|---|---|---|---|---|
| B | SE | p Value | B | SE | p Value | B | SE | p Value | |
| Constant | 10.8 | 42.6 | 0.8 | −23.9 | 21.4 | 0.3 | 15.4 | 10.8 | 0.2 |
| Advanced fibrosis | −2.8 | 6.5 | 0.7 | 10.4 | 3.3 | 0.002 | 4.2 | 1.7 | 0.01 |
| IFNL3 (Non-TT) | 2.9 | 7.2 | 0.7 | −2.1 | 3.6 | 0.6 | 4.3 | 1.8 | 0.02 |
| Female gender | −13.1 | 6.7 | 0.055 | −4.0 | 3.4 | 0.2 | 0.2 | 1.7 | 0.9 |
| HCV-RNA | 7.7 | 3.8 | 0.04 | 4.8 | 1.9 | 0.01 | 0.8 | 1.0 | 0.4 |
| BMI | 0.6 | 1.0 | 0.6 | 0.4 | 0.5 | 0.4 | 0.2 | 0.2 | 0.4 |
| HbA1c | −2.8 | 5.0 | 0.6 | 2.9 | 2.5 | 0.25 | −1.1 | 1.3 | 0.4 |
2.8. Significance of Advanced Fibrosis for Serum TG Concentrations in the Three Subclasses of VLDL
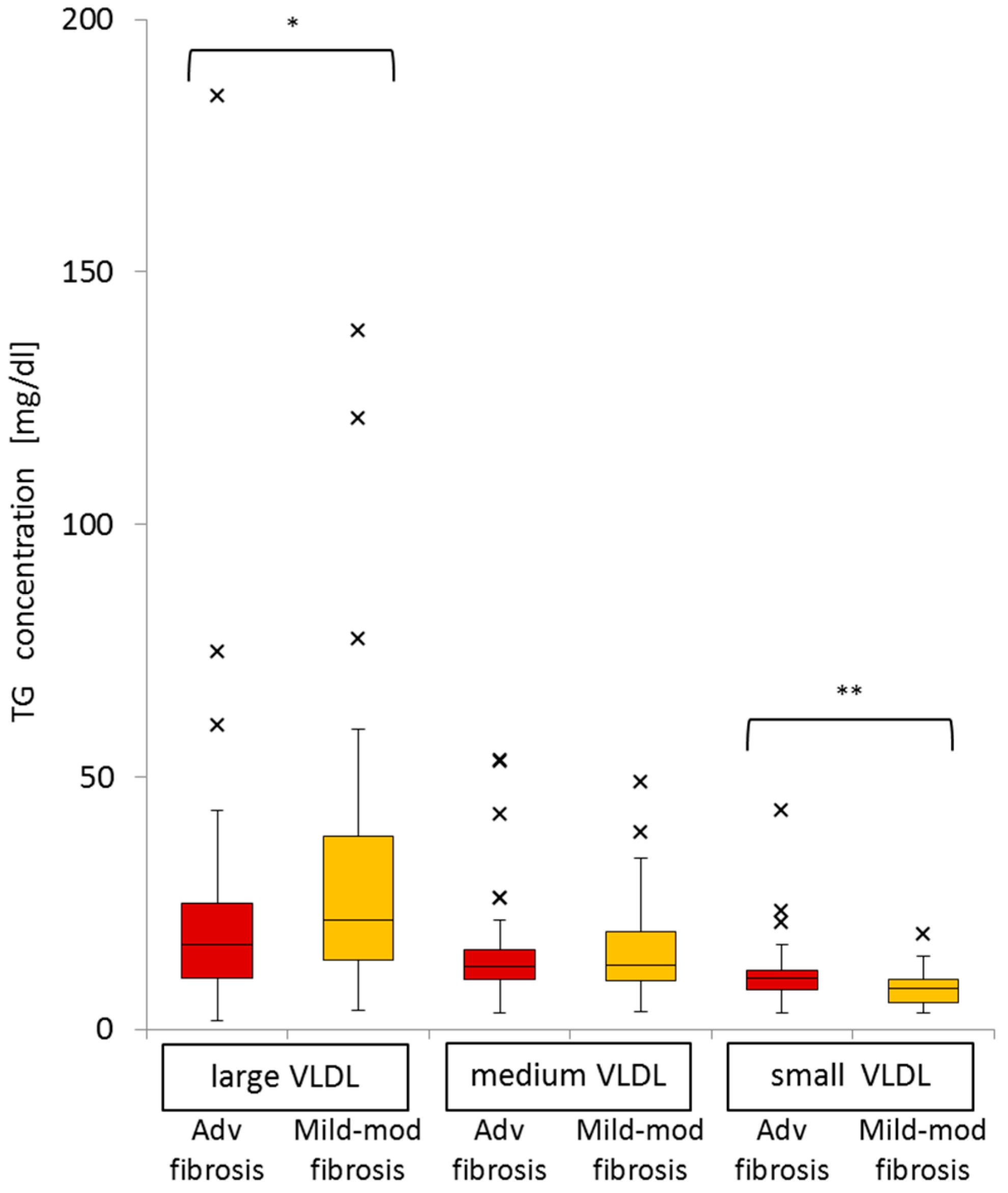
| Large VLDL-TG | Medium VLDL-TG | Small VLDL-TG | |||||||
|---|---|---|---|---|---|---|---|---|---|
| B | SE | p Value | B | SE | p Value | B | SE | p Value | |
| Constant | 7.3 | 27.9 | 0.8 | 0.0 | 14.2 | 1.0 | 3.5 | 8.0 | 0.7 |
| Advanced fibrosis | −6.1 | 4.3 | 0.16 | 0.1 | 2.2 | 0.98 | 3.2 | 1.2 | 0.01 |
| IFNL3 (Non-TT) | 6.0 | 4.7 | 0.2 | −1.3 | 2.4 | 0.6 | −1.7 | 1.4 | 0.2 |
| Female gender | −9.2 | 4.4 | 0.04 | −3.6 | 2.2 | 0.1 | −0.3 | 1.3 | 0.8 |
| HCV-RNA | 4.3 | 2.5 | 0.1 | 2.3 | 1.3 | 0.1 | 1.2 | 0.7 | 0.1 |
| BMI | 0.4 | 0.6 | 0.5 | 0.08 | 0.3 | 0.8 | 0.04 | 0.2 | 0.8 |
| HbA1c | −2.4 | 3.3 | 0.5 | 0.2 | 1.7 | 0.91 | −0.6 | 0.9 | 0.5 |
3. Discussion
4. Experimental Section
4.1. Patients
4.2. Study Design
4.3. Analysis of Serum TG Profiles in Lipoprotein Subclasses
4.4. Demographic and Basic Laboratory Tests
4.5. Evaluation of Serum Lipid Profiles
4.6. Measurement of Serum HCV-RNA Level and Genotyping of Interferon Lambda (IFNL) 3
4.7. Evaluation of Liver Fibrosis in Patients with Chronic Active HCV G1b Infection
4.8. Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Moriya, K.; Yotsuyanagi, H.; Shintani, Y.; Fujie, H.; Ishibashi, K.; Matsuura, Y.; Miyamura, T.; Koike, K. Hepatitis C virus core protein induces hepatic steatosis in transgenic mice. J. Gen. Virol. 1997, 78, 1527–1531. [Google Scholar] [CrossRef] [PubMed]
- Aizawa, Y.; Shimada, N.; Abe, H.; Seki, N.; Aida, Y.; Ishiguro, H.; Ika, M.; Kato, K.; Tsubota, A. Serum lipoprotein profiles and response to pegylated interferon plus ribavirin combination therapy in patients with chronic HCV genotype 1b infection. Hepat. Mon. 2013, 13, e8988. [Google Scholar] [CrossRef] [PubMed]
- Lange, C.M.; von Wagner, M.; Bojunga, J.; Berg, T.; Farnik, H.; Hassler, A.; Sarrazin, C.; Herrmann, E.; Zeuzem, S. Serum lipids in European chronic HCV genotype 1 patients during and after treatment with pegylated interferon-α-2a and ribavirin. Eur. J. Gastroenterol. Hepatol. 2010, 22, 1303–1307. [Google Scholar] [CrossRef] [PubMed]
- Maggi, G.; Bottelli, R.; Gola, D.; Perricone, G.; Posca, M.; Zavaglia, C.; Ideo, G. Serum cholesterol and chronic hepatitis C. Ital. J. Gastroenterol. 1996, 28, 436–440. [Google Scholar] [PubMed]
- Petit, J.M.; Benichou, M.; Duvillard, L.; Jooste, V.; Bour, J.B.; Minello, A.; Verges, B.; Brun, J.M.; Gambert, P.; Hillon, P. Hepatitis C virus-associated hypobetalipoproteinemia is correlated with plasma viral load, steatosis, and liver fibrosis. Am. J. Gastroenterol. 2003, 98, 1150–1154. [Google Scholar] [PubMed]
- Brown, M.S.; Goldstein, J.L. A receptor-mediated pathway for cholesterol homeostasis. Science 1986, 232, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Barth, H.; Liang, T.J.; Baumert, T.F. Hepatitis C virus entry: Molecular biology and clinical implications. Hepatology 2006, 44, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Sun, F.; Owen, D.M.; Li, W.; Chen, Y.; Gale, M., Jr.; Ye, J. Hepatitis C virus production by human hepatocytes dependent on assembly and secretion of very low-density lipoproteins. Proc. Natl. Acad. Sci. USA 2007, 104, 5848–5853. [Google Scholar] [CrossRef] [PubMed]
- Perlemuter, G.; Sabile, A.; Letteron, P.; Vona, G.; Topilco, A.; Chrétien, Y.; Koike, K.; Pessayre, D.; Chapman, J.; Barba, G.; et al. Hepatitis C virus core protein inhibits microsomal triglyceride transfer protein activity and very low density lipoprotein secretion: A model of viral-related steatosis. FASEB J. 2002, 16, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, A.; Tazuma, S.; Nishioka, T.; Ohishi, W.; Hyogo, H.; Nomura, S.; Chayama, K. Hepatitis C virus core protein modulates fatty acid metabolism and thereby causes lipid accumulation in the liver. Dig. Dis. Sci. 2005, 50, 1361–1371. [Google Scholar] [CrossRef] [PubMed]
- Serfaty, L.; Andreani, T.; Giral, P.; Carbonell, N.; Chazouillères, O.; Poupon, R. Hepatitis C virus induced hypobetalipoproteinemia: A possible mechanism for steatosis in chronic hepatitis C. J. Hepatol. 2001, 34, 428–434. [Google Scholar] [CrossRef]
- Moriya, K.; Shintani, Y.; Fujie, H.; Miyoshi, H.; Tsutsumi, T.; Yotsuyanagi, H.; Iino, S.; Kimura, S.; Koike, K. Serum lipid profile of patients with genotype 1b hepatitis C viral infection in Japan. Hepatol. Res. 2003, 25, 371–376. [Google Scholar] [CrossRef]
- Seki, N.; Sugita, T.; Aida, Y.; Itagaki, M.; Ishiguro, H.; Sutoh, S.; Abe, H.; Tsubota, A.; Matsushima, M.; Aizawa, Y. Assessment of the features of serum apolipoprotein profiles in chronic HCV infection: Difference between HCV genotypes 1b and 2. Hepatol. Int. 2014, 8, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Furusyo, N.; Ai, M.; Okazaki, M.; Ikezaki, H.; Ihara, T.; Hayashi, T.; Hiramine, S.; Ura, K.; Kohzuma, T.; Schaefer, E.J.; et al. Serum cholesterol and triglyceride reference ranges of twenty lipoprotein subclasses for healthy Japanese men and women. Atherosclerosis 2013, 231, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, G.; Conca, P.; Ariello, M.; Mastrolia, M. Does a lower insulin resistance affect antiviral therapy response in patients suffering from HCV related chronic hepatitis? Gut 2006, 55, 585. [Google Scholar] [PubMed]
- Tarantino, G.; Conca, P.; Sorrentino, P.; Ariello, M. Metabolic factors involved in the therapeutic response of patients with hepatitis C virus-related chronic hepatitis. J. Gastroenterol. Hepatol. 2006, 21, 1266–1268. [Google Scholar] [CrossRef] [PubMed]
- Siagris, D.; Christofidou, M.; Theocharis, G.J.; Pagoni, N.; Papadimitriou, C.; Lekkou, A.; Thomopoulos, K.; Starakis, I.; Tsamandas, A.C.; Labropoulou-Karatza, C. Serum lipid pattern in chronic hepatitis C: Histological and virological correlations. J. Viral Hepat. 2006, 13, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Rowell, J.; Thompson, A.J.; Guyton, J.R.; Lao, X.Q.; McHutchison, J.G.; McCarthy, J.J.; Patel, K. Serum apolipoprotein C-III is independently associated with chronic hepatitis C infection and advanced fibrosis. Hepatol. Int. 2012, 6, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Mawatari, H.; Yoneda, M.; Fujita, K.; Nozaki, Y.; Shinohara, Y.; Sasaki, H.; Iida, H.; Takahashi, H.; Inamori, M.; Abe, Y.; et al. Association between lipoprotein subfraction profile and the response to hepatitis C treatment in Japanese patients with genotype 1b. J. Viral Hepat. 2010, 17, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Khattab, M.A.; Eslam, M.; Aly, M.M.; Shatat, M.; Mousa, Y.I.; Abd-Aalhalim, H.; Aly, H.; Shaker, Y. Serum lipids and chronic hepatitis C genotype 4: Interaction and significance. Ann. Hepatol. 2012, 11, 37–46. [Google Scholar] [PubMed]
- Hsu, C.S.; Liu, C.J.; Liu, C.H.; Chen, C.L.; Lai, M.Y.; Chen, P.J.; Chen, D.S.; Kao, J.H. Metabolic profiles in patients with chronic hepatitis C: A case-control study. Hepatol. Int. 2008, 2, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, M.; Yamamoto, H.; Yoshida, T.; Seimiya, M.; Sawabe, Y.; Matsushita, K.; Umemura, H.; Sogawa, K.; Takizawa, H.; Yokosuka, O.; et al. Decreases in the serum VLDL-TG/non-VLDL-TG ratio from early stages of chronic hepatitis C: Alterations in TG-rich lipoprotein levels. PLoS ONE 2011, 25, e17309. [Google Scholar] [CrossRef] [PubMed]
- Sikorski, J.A. Oral cholesteryl ester transfer protein (CETP) inhibitors: A potential new approach for treating coronary artery disease. J. Med. Chem. 2006, 49, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, K.; Abe, H.; Aida, Y.; Ishiguro, H.; Ika, M.; Shimada, N.; Tsubota, A.; Aizawa, Y. Serum apolipoprotein B-100 concentration predicts the virological response to pegylated interferon plus ribavirin combination therapy in patients infected with chronic hepatitis C virus genotype 1b. J. Med. Virol. 2013, 85, 1180–1190. [Google Scholar] [CrossRef] [PubMed]
- Icard, V.; Diaz, O.; Scholtes, C.; Perrin-Cocon, L.; Ramière, C.; Bartenschlager, R.; Penin, F.; Lotteau, V.; André, P. Secretion of hepatitis C virus envelope glycoproteins depends on assembly of apolipoprotein B positive lipoproteins. PLoS ONE 2009, 4, e4233. [Google Scholar] [CrossRef] [PubMed]
- Stillemark-Billton, P.; Beck, C.; Borén, J.; Olofsson, S.O. Relation of the size and intracellular sorting of apoB to the formation of VLDL 1 and VLDL 2. J. Lipid Res. 2005, 46, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, Y.; Imajo, K.; Yoneda, M.; Tomeno, W.; Ogawa, Y.; Fujita, K.; Kirikoshi, H.; Takahashi, J.; Funakoshi, K.; Ikeda, M.; et al. Hepatic triglyceride lipase plays an essential role in changing the lipid metabolism in genotype 1b hepatitis C virus replicon cells and hepatitis C patients. Hepatol. Res. 2013, 43, 1190–1198. [Google Scholar] [CrossRef] [PubMed]
- Clark, P.J.; Thompson, A.J.; Zhu, M.; Vock, D.M.; Zhu, Q.; Ge, D.; Patel, K.; Harrison, S.A.; Urban, T.J.; Naggie, S.; et al. Interleukin 28B polymorphisms are the only common genetic variants associated with low-density lipoprotein cholesterol (LDL-C) in genotype-1 chronic hepatitis C and determine the association between LDL-C and treatment response. J. Viral. Hepat. 2012, 19, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Olubamwo, O.O.; Onyeka, I.N.; Miettola, J.; Kauhanen, J.; Tuomainen, T.P. Hepatitis C as a risk factor for carotid atherosclerosis—A systematic review. Clin. Physiol. Funct. Imaging 2015, 26. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Kang, R.; Zhao, Z. Is hepatitis C associated with atherosclerotic burden? A systematic review and meta-analysis. PLoS ONE 2014, 10, 1371. [Google Scholar] [CrossRef] [PubMed]
- Adinolfi, L.E.; Zampino, R.; Restivo, L.; Lonardo, A.; Guerrera, B.; Marrone, A.; Nascimbeni, F.; Florio, A.; Loria, P. Chronic hepatitis C virus infection and atherosclerosis: Clinical impact and mechanisms. World J. Gastroenterol. 2014, 20, 3410–3417. [Google Scholar] [CrossRef] [PubMed]
- Negro, F. Facts and fictions of HCV and comorbidities: Steatosis, diabetes mellitus, and cardiovascular diseases. J. Hepatol. 2014, 61, S69–S78. [Google Scholar] [CrossRef] [PubMed]
- Petta, S.; Torres, D.; Fazio, G.; Cammà, C.; Cabibi, D.; di Marco, V.; Licata, A.; Marchesini, G.; Mazzola, A.; Parrinello, G.; et al. Carotid atherosclerosis and chronic hepatitis C: A prospective study of risk associations. Hepatology 2012, 55, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Perticone, M.; Maio, R.; Tassone, E.J.; Tripepi, G.; di Cello, S.; Miceli, S.; Caroleo, B.; Sciacqua, A.; Licata, A.; Sesti, G.; et al. Insulin-resistance HCV infection-related affects vascular stiffness in normotensives. Atherosclerosis 2015, 238, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, J.R.; Barrie, H.D.; O’Rourke, P.; Clouston, A.D.; Powell, E.E. Obesity and steatosis influence serum and hepatic inflammatory markers in chronic hepatitis C. Hepatology 2008, 48, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Vidali, M.; Tripodi, M.F.; Ivaldi, A.; Zampino, R.; Occhino, G.; Restivo, L.; Sutti, S.; Marrone, A.; Ruggiero, G.; Albano, E.; et al. Interplay between oxidative stress and hepatic steatosis in the progression of chronic hepatitis C. J. Hepatol. 2008, 48, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Munteanu, D.; Negru, A.; Radulescu, M.; Mihailescu, R.; Arama, S.S.; Arama, V. Evaluation of bacterial translocation in patients with chronic HCV infection. Rom. J. Intern. Med. 2014, 52, 91–96. [Google Scholar] [PubMed]
- Minopetrou, M.; Hadziyannis, E.; Deutsch, M.; Tampaki, M.; Georgiadou, A.; Dimopoulou, E.; Vassilopoulos, D.; Koskinas, J. Hepatitis C virus (HCV)-related cryoglobulinemia: Cryoglobulin type and anti-HCV profile. Clin. Vaccine Immunol. 2013, 20, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Nakada, Y.; Kurosawa, H.; Tohyama, J.; Inoue, Y.; Ikewaki, K. Increased remnant lipoprotein in patients with coronary artery disease-evaluation utilizing a newly developed remnant assay, remnant lipoproteins cholesterol homogenous assay (RemL-C). J. Atheroscler. Thromb. 2007, 14, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Takeichi, S.; Yukawa, N.; Nakajima, Y.; Osawa, M.; Saito, T.; Seto, Y.; Nakano, T.; Saniabadi, A.R.; Adachi, M.; Wang, T.; et al. Association of plasma triglyceride-rich lipoprotein remnants with coronary atherosclerosis in cases of sudden cardiac death. Atherosclerosis 1999, 142, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Barone, M.; Viggiani, M.T.; Amoruso, A.; Schiraldi, S.; Zito, A.; Devito, F.; Cortese, F.; Gesualdo, M.; Brunetti, N.; di Leo, A.; et al. Endothelial dysfunction correlates with liver fibrosis in chronic HCV infection. Gastroenterol. Res. Pract. 2015, 2015, 682174. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, P.; Mellor, J.; Sakuldamrongpanich, T. Evolutionary analysis of variants of hepatitis C virus found in South-East Asia: Comparison with classifications based upon sequence similarity. J. Gen. Virol. 1996, 77, 3013–3024. [Google Scholar] [CrossRef] [PubMed]
- Usui, S.; Hara, Y.; Hosaki, S.; Okazaki, M. A new on-line dual enzymatic method for simultaneous quantification of cholesterol and triglycerides in lipoproteins by HPLC. J. Lipid Res. 2002, 43, 805–814. [Google Scholar] [PubMed]
- Okazaki, M.; Usui, S.; Ishigami, M.; Sakai, N.; Nakamura, T.; Matsuzawa, Y.; Yamashita, S. Identification of unique lipoprotein subclasses for visceral obesity by component analysis of cholesterol profile in high-performance liquid chromatography. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [PubMed]
- Tanaka, Y.; Nishida, N.; Sugiyama, M.; Kurosaki, M.; Matsuura, K.; Sakamoto, N.; Nakagawa, M.; Korenaga, M.; Hino, K.; Hige, S.; et al. Genome-wide association of IL28B with response to pegylated interferon-α and ribavirin therapy for chronic hepatitis C. Nat. Genet. 2009, 41, 1105–1109. [Google Scholar] [CrossRef] [PubMed]
- Sterling, R.K.; Lissen, E.; Clumeck, N.; Sola, R.; Correa, M.C.; Montaner, J.S.; Sulkowski, M.; Torriani, F.J.; Dieterich, D.T.; Thomas, D.L.; et al. Investigators, APRICOT Clinical. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43, 1317–1325. [Google Scholar] [CrossRef] [PubMed]
- Vallet-Pichard, A.; Mallet, V.; Nalpas, B.; Verkarre, V.; Nalpas, A.; Dhalluin-Venier, V.; Fontaine, H.; Pol, S. FIB-4: An inexpensive and accurate marker of fibrosis in HCV infection. Comparison with liver biopsy and fibrotest. Hepatology 2007, 46, 32–36. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagano, T.; Seki, N.; Tomita, Y.; Sugita, T.; Aida, Y.; Itagaki, M.; Sutoh, S.; Abe, H.; Tsubota, A.; Aizawa, Y. Impact of Chronic Hepatitis C Virus Genotype 1b Infection on Triglyceride Concentration in Serum Lipoprotein Fractions. Int. J. Mol. Sci. 2015, 16, 20576-20594. https://doi.org/10.3390/ijms160920576
Nagano T, Seki N, Tomita Y, Sugita T, Aida Y, Itagaki M, Sutoh S, Abe H, Tsubota A, Aizawa Y. Impact of Chronic Hepatitis C Virus Genotype 1b Infection on Triglyceride Concentration in Serum Lipoprotein Fractions. International Journal of Molecular Sciences. 2015; 16(9):20576-20594. https://doi.org/10.3390/ijms160920576
Chicago/Turabian StyleNagano, Tomohisa, Nobuyoshi Seki, Yoichi Tomita, Tomonori Sugita, Yuta Aida, Munenori Itagaki, Satoshi Sutoh, Hiroshi Abe, Akihito Tsubota, and Yoshio Aizawa. 2015. "Impact of Chronic Hepatitis C Virus Genotype 1b Infection on Triglyceride Concentration in Serum Lipoprotein Fractions" International Journal of Molecular Sciences 16, no. 9: 20576-20594. https://doi.org/10.3390/ijms160920576
APA StyleNagano, T., Seki, N., Tomita, Y., Sugita, T., Aida, Y., Itagaki, M., Sutoh, S., Abe, H., Tsubota, A., & Aizawa, Y. (2015). Impact of Chronic Hepatitis C Virus Genotype 1b Infection on Triglyceride Concentration in Serum Lipoprotein Fractions. International Journal of Molecular Sciences, 16(9), 20576-20594. https://doi.org/10.3390/ijms160920576