A Review of Polyphenolics in Oak Woods
Abstract
:1. Introduction
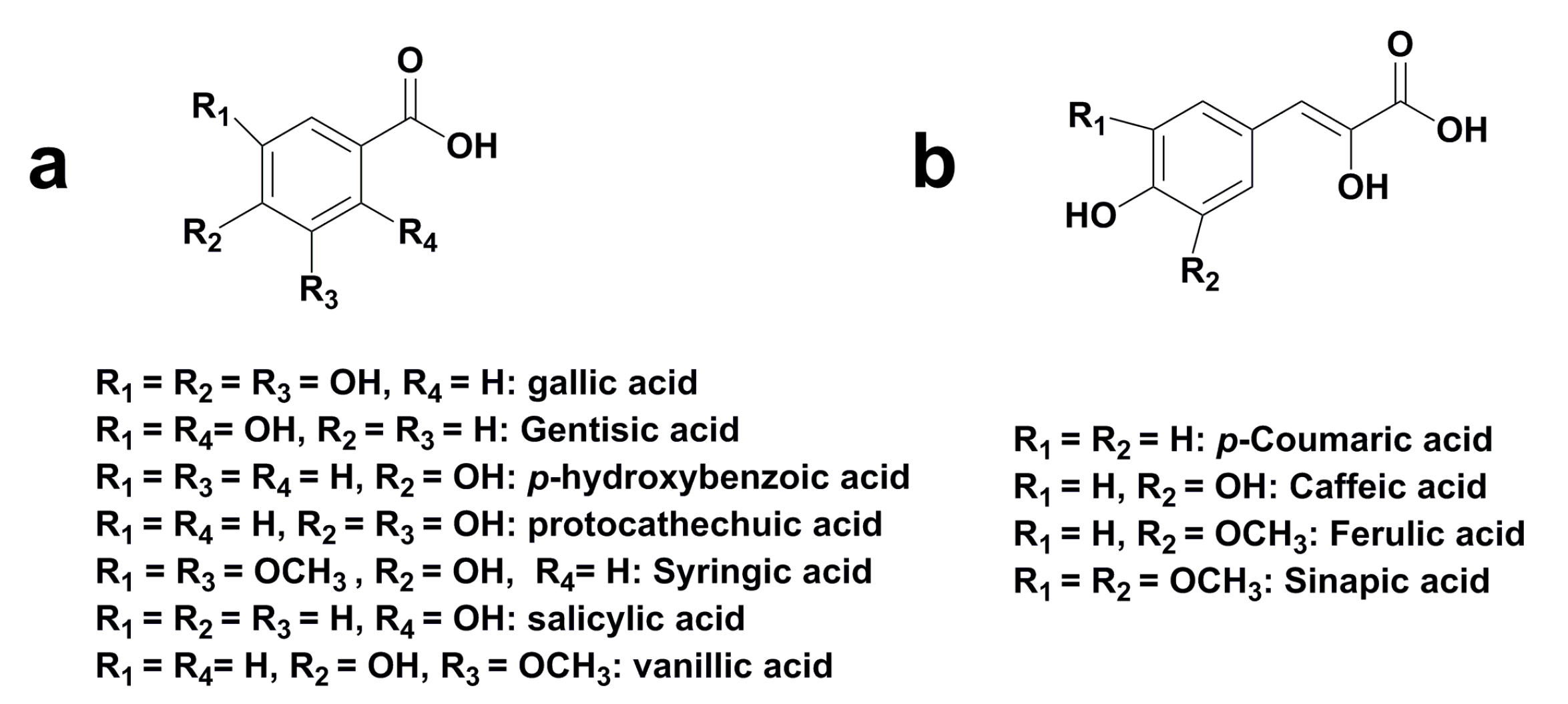
2. Classification of Plant Polyphenols
| Structure | Phenolic Class |
|---|---|
| C6 | Simple phenolics |
| C6-C1 | Phenolic acids and related compounds |
| C6-C2 | Acetophenones and phenylacetic acids |
| C6-C3 | Cinnamic acids, cinnamyl aldehydes/alcohols |
| C6-C3 | Coumarins, isocoumarins, chromones |
| C6-C1-C6 | Benzophenones, xanthones |
| C6-C2-C6 | Stilbenes |
| C6-C3-C6 | Chalcones, aurones, dihydrochalcones |
| C6-C3-C6 | Flavones |
| C6-C3-C6 | Flavonols |
| C6-C3-C6 | Flavanones |
| C6-C3-C6 | Flavanonols |
| C6-C3-C6 | Flavan-3-ols |
| C6-C3-C6 | Isoflavonoids |
| C6-C3-C6 | Anthocyanidins/Anthocyanins |
| (C6-C3-C6)2 | Biflavonoids |
| C6,C10,C14 | Benzoquinones, naphthaquinones, anthraquinones |
| C18 | Betacyanins |
| Lignans, neolignans | Dimers or oligomers |
| Lignin | Polymers |
| Phlobaphenes | Polymers |

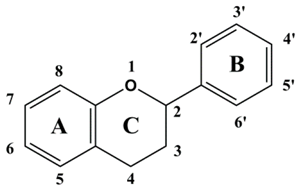
| Flavonoids | Basic Structure |
|---|---|
| Chalcones | 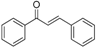 |
| Dihydrochalcones | 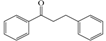 |
| Aurones |  |
| Flavonols | 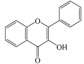 |
| Dihydroflavonols | 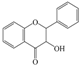 |
| Flavanones | 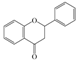 |
| Flavanols | 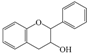 |
| Isoflavonoids | 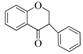 |
| Biflavonoids | 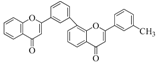 |
| Anthocyanidins |  |
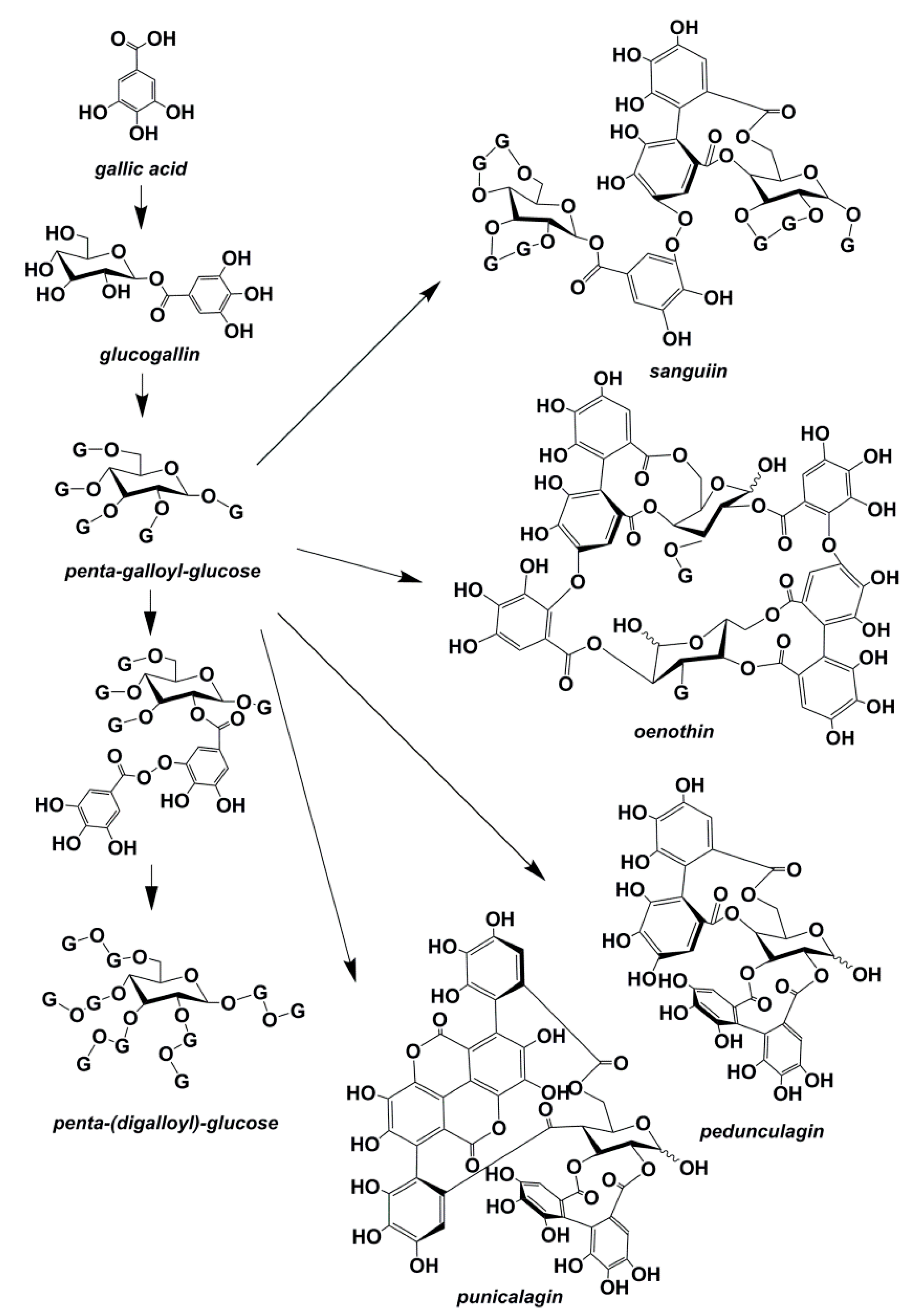
3. Oak Polyphenolics
3.1. Volatile Phenols
3.2. Phenolic Acid
| Compounds | Sample | λ (Max) | MW | ESI-MS/MS Prominent Ions (m/z) | References |
|---|---|---|---|---|---|
| Hydroxybenzoic Acids | |||||
| Gallic acid | A, B, D–I | 270 | 170 | 169 [M-H]−, 154, 125, 81, 79 | [5,40,50,51,52,53,54,55,56] |
| Gentisic acid | B | 340 | 154 | 153 [M-H]−, 109 | [50] |
| p-Hydroxybenzoic acid | H | 255 | 138 | 137 [M-H]−, 93 | [55] |
| Protocatechuic acid | A, B, H | 259, 293 | 154 | 153 [M-H]−, 109 | [40,50,55] |
| Syringic acid | B, D–I | 275 | 198 | 197 [M-H]−, 182, 167, 153, 138 | [5,50,51,52,53,54,55] |
| Vanillic acid | A, B, D–G, I | 260, 291 | 168 | 167 [M-H]−, 152, 123, 108 | [40,50] |
| p-Hydroxybenzoic aldehyde | B | 280, 254 | 122 | 121 [M-H]− | [50] |
| Protocatechuic aldehyde | A, B, H | 280, 310 | 138 | 137 [M-H]− | [40,50] |
| Syringaldehyde | A, C–I | 307 | 182 | 181 [M-H]−, 166, 151 | [40,51,52,53,54,55,56,57] |
| Vanillin | A–I | 304 | 152 | 151 [M-H]−, 136, 108 | [5,40,50,51,52,53,54,55,56,57] |
| Hydroxycinnamic Acids | |||||
| p-Coumaric acid | A, B, G | 310 | 164 | 163 [M-H]−, 119, 94 | [40,50,54] |
| Caffeic acid | A, B | 324 | 180 | 179 [M-H]−, 135 | [40,50] |
| Ferulic acid | A, B, E, F | 280, 340 | 194 | 193 [M-H]−, 178, 149, 134 | [5,40,50,53] |
| Sinapic acid | A, B | 340 | 222 | 221 [M-H]−, 164, 149, 121 | [40,50] |
| Sinapaldehyde | A, D–F, I | 254 | 208 | 207 [M-H]−, 192, 189 | [5,40,51,52,53,56] |
| Coniferaldehyde | A, D–F, I | 260 | 178 | 177 [M-H]−, 162, 159, 147 | [5,40,51,52,53,56] |
| Eugenol | D, E, G | 230 | 164 | 163 [M-H]−, 149, 137 | [51,52,54] |
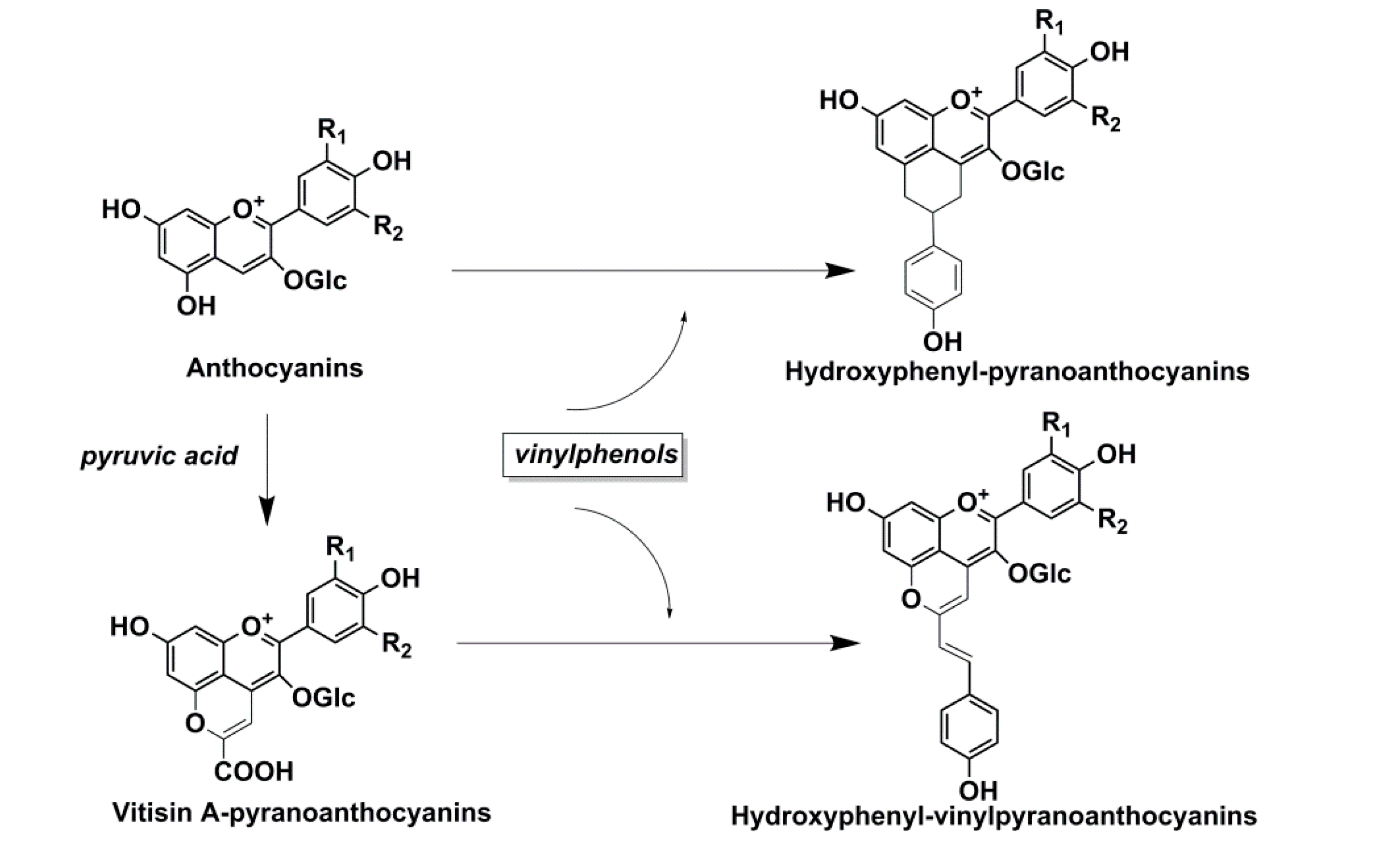
| Anthocyanins | pH | Hyperchromic Effect (%) | Bathochromic Shift (∆ λ max, nm) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Coumaric Acid | Caffeic Acid | Ferulic Acid | Sinapic Acid | Coumaric Acid | Caffeic Acid | Ferulic Acid | Sinapic Acid | ||
| Cy-3-glu | 3.2 | 11.6 | 18.8 | 25.3 | 29.7 | 2.5 | 4.5 | 6.6 | 8.7 |
| 4.0 | 27.7 | 41.0 | 63.9 | 110.5 | 2.2 | 4.0 | 6.8 | 10.2 | |
| Cy-3-soph | 3.2 | 9.9 | 11.2 | 16.5 | 23.9 | 2.5 | 3.8 | 5.2 | 5.5 |
| 4.0 | 16.3 | 22.7 | 40.7 | 60.9 | 2.3 | 2.6 | 6.5 | 7.7 | |
3.3. Ellagitannins
3.4. Polyphenolic Compounds in Non-Oak Woods
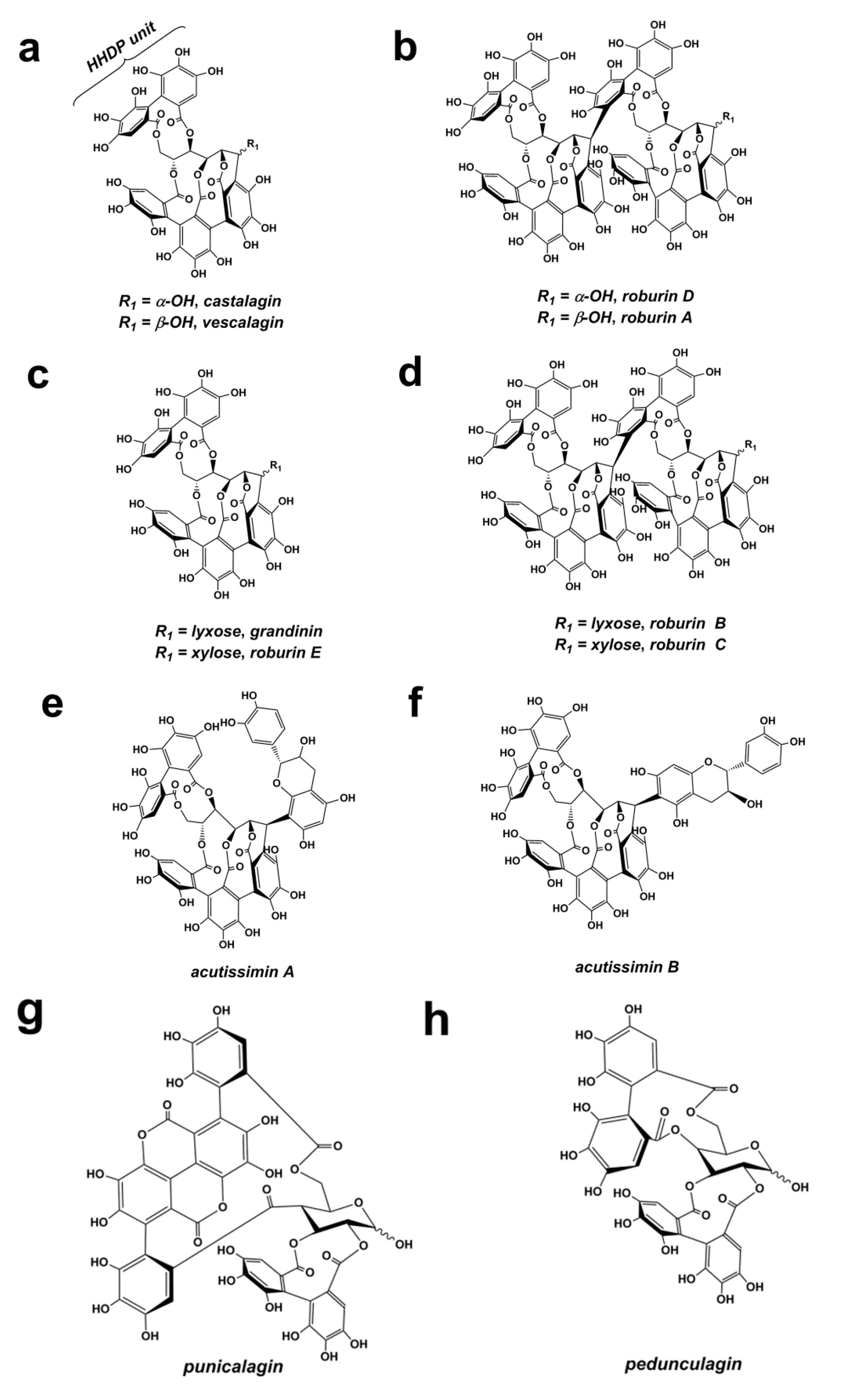
4. Some Factors Affecting the Polyphenolic Profile of Oak Woods
4.1. Oak Species and Geographical Origins
4.2. Cooperage Treatment
4.2.1. Seasoning
4.2.2. Toasting
4.2.3. Aging Process and Barrel Properties
4.2.4. Others
5. Analysis of Polyphenolics in Oak Woods
| Source | Sample Preparation | Analytical Techniques | Main Polyphenolic Compounds | References | |
|---|---|---|---|---|---|
| Oak heartwood (2006) | Extracted sawdust with methanol/water (1:1); Filtration and evaporated; Residue extracted in diethyl ether and ethyl acetate | HPLC-DAD | Gallic acid, vanillic acid, vanillin, syringic acid, syringaldehyde, ellagic acid, coniferylaldehyde, sinapic aldehyde, scopoletin, castalagin, vescalagin, grandinin, roburin A–E | [52] | |
| Oak and Brazilian woods (2009) | Extracted sawdust with sugarcane ethanol (47% v/v), followed by separation using C18 Sep-Pak | HPLC-DAD-fluorescence and HPLC-ESI-MS | (+)-Catechin, coniferaldehyde, coumarin, ellagic acid, (−)-epicatechin, eugenol, gallic acid, myricetin, quercetin, scopoletin, synapaldehyde, syringaldehyde, syringic acid, trans-resveratrol, vanillic acid, vanillin | [51] | |
| Oak heartwood (2011) | Extracted sawdust with methanol/water (1:1); Filtration and evaporated; Residue extracted in diethyl ether and ethyl acetate | HPLC-DAD | Gallic acids, ellagic acids, vanillic acids, syringic acids, ferulic acids, vanillin, syringaldehyde, coniferaldehyde, sinapaldehyde, aesculetin, scopoletin | [53] | |
| Oak heartwood (2011) | Extracted sawdust with methanol/water (1:1); Filtrated and evaporated; Residue extracted in diethyl ether and ethyl acetate | HPLC | Roburins A–E, grandinin, vescalagin, castalagin | [8] | |
| Cherry heartwood (2010) | Extracted sawdust with methanol/water (1:1); Filtrated and evaporated; Residue extracted in diethyl ether and ethyl acetate | HPLC-DAD/ESI-MS | Gallic acid, protocatechuic acid, vanillic acid, vanillin, syringic acid, syringaldehyde, 3,4,5-trimethoxyphenol, protocatechualdehyde, benzoic acid, p-hydroxybenzoic acid, p-coumaric acid, scopoletin, coniferaldehyde, coniferaldehyde, methyl vanillate, methyl syringate | [108] | |
| Black pine bark (2010) | Extracted with pressurized hot water; Filtrated; Washed with chloroform, ethyl ether and other solvents | HPLC, CC, TLC and FT-NMR | (+)-Catechin, (−)-epicatechin, quercetin, ferulic acid | [109] | |
| Acacia heartwood (2011) | Extracted sawdust with methanol/water (1:1); Followed by extraction with ethyl acetate | HPLC-DAD and LC-DAD/ESI-MS/MS | Gallic acid, gallic aldehyde, protocatechualdehyde, methyl gallate, β-resorcilyc acid, vanillic acid, β-resorcilyc aldehyde, caffeic acid, vanillin, syringic acid, syringaldehyde, coniferaldehyde, sinapaldehyde, ellagic acid, robtin etc. | [110] | |
| Red wine (2008) | Solid phase extraction | HPLC-UV | Monomeric, oligomeric and polymeric polyphenolic compounds | [111] | |
| Red wine vinegar produced in barrels made from different woods (2008) | Direct injected | HPLC-UV | Gallic acid, protocatechuic acid, tyrosol, caftaric acid, vanillic acid, (+)-catechin, caffeic acid, syringic acid, (−)-epicatechin, resveratrol glucoside, ellagic acid | [112] | |
| Vinegar (2012) | Extraction of vinegar with stir bar at 1250 rpm, 25 °C for 120 min | GC-MS | Benzaldehyde, benzaldehyde, phenol, 4-acetyl-2-methylphenol, 4-acetyl-2-methylphenol, p-ethylguaiacol, 4-ethylphenol, 2,4-ditertbutylphenol, benzoic acid | [113] | |
| Pomegranate beverage (2012) | Direct injection | HPLC-DAD-MS | Gallic acid, (+)-catechin, (−)-epicatechin, caftaric acid, ellagic acid, myricetin, quercetin, etc. | [114] | |
| Apple pomace (2009) | Extracted apple pomace powder with ethanol; Concentrated and vacuum dried; fractionated polyphenols using Sephadex LH-20; HSCCC separation | HPLC-MS | Chlorogenic acid, quercetin-3-glucoside, phloridzin, quercetin-3-glacaside, quercetin-3-xyloside, quercetin-3-arabinoside and quercetin-3-rhamnoside | [115] | |
| Apple pomace (2010) | Extracted with ethanol and assisted by microwave treatment | HPLC-UV | Chlorogenic acid, cafeic acid, syrigin, (−)-epicatechin, procyanidin B2, cinnamic acid, coumaric acid, phlorizin, quercetin | [116] | |
| Onion (2008) | Extract with 80% methanol; Filtrated | HPLC-UV | Kaempferol, quercetin, isoquercetin, quercetin monoglucoside, quercetin diglucoside | [117] | |
| Onion (2009) | Extracted with methanol:formic acid:water (50:5:45) | HPLC-DAD | Quercetin-3,4'-diglucoside, quercetin-4'-glucoside, cyanidin-3-glucoside, cyanidin-3-laminaribioside, cyanidin-3-(6''-malonyl-glucoside), cyanidin-3-(6''-malonyl-laminaribioside) | [118] | |
| Onion (2010) | Extracted with methanol:formic acid:water (50:5:45) | HPLC-DAD | Quercetin-3-glucoside, quercetin-3,4'-diglucoside, quercetin-4'-glucoside, quercetin-7,4-diglucoside, isorhamnetin-4-glucoside, isorhamnetin-3,4-diglucoside, cyanidin-3-glucoside, cyanidin-3-laminaribioside, cyanidin-3-(6''-malonyl-glucoside), cyanidin-3-(6''-malonyl-laminaribioside) | [119] | |
| Roasted wheat germ (2009) | Supercritical carbon dioxide extraction | HPLC-MS | Ferulic acid, vanillic acid | [120] | |
| Pistachio (2010) | Extracted crushed seeds and skins with methanol/water (2:1); ultrasonicated; Homogenate centrifuged and separated | HPLC-DAD | Gallic acid, eriodictyol-7-O-glucoside, catechin, naringenin-7-O-neohesperidoside, quercetin-3-O-rutinoside, eriodictyol | [121] | |
| Potato peels (2011) | Peels lyophilized and ground; Extracted with methanol assisted by microwave treatment | HPLC-UV | Chlorogenic acid, caffeic acid, ferulic acid, rutin | [122] | |
| Apple Seeds (2012) | Extraction of defatted apple seeds with aqueous acetone (30:70; v/v) | HPLC-DAD | Phenolic acids, chlorogenic acid, phloridzin, phloretin-2'-xyloglucoside, flavan-3-ols, quercetin-3-O-glucoside | [123] | |
| Rooibos (2011) | Extraction with distilled water; Followed by filtration and re-extraction with ethanol (80%; v/v) | HPLC-ESI-MS | Esculin, rutin, quercetin, isoquercitrin, luteolin, nothofagin, secoisolariciresinol, etc. | [124] | |
| Rooibos (2012) | Extraction with boiled deionized water; Then filtrated | HPLC-DAD | Phenylpyruvic acid-2-O-glucoside, isoorientin, orientin, aspalathin, ferulic acid, quercetin-3-O-robinobioside, vitexin, hyperoside, rutin, isovitexin, isoquercitrin, nothofagin | [125] | |
| Mate tea (2012) | Extraction of leaves with ethanol/water; Followed by filtration | HPLC-DAD | Chlorogenic acid | [126] | |
| Grapes (V. vinifera L.) (2012) | SPME | GC-MS | Phenol, 2-methylphenol, eugenol, 2-methoxy-4-vinylphenol | [127] | |
| Mango (2012) | Extraction with 80% methanol and 2% formic acid; Followed by extraction with 80% methanol | HPLC-DAD-MS/MS-ESI | Gallic acid, protocatechuic acid, chlorogenic acid, vanillic acid | [128] | |
| Olive oil (2012) | Extraction with n-hexane and assisted with an ultrasonic probe; Centrifugation and separation; Followed by extraction with methanol | HPLC-DAD-FLD and LC-MS | Hydroxytyrosol, tyrosol, oleuropein aglycone derivative and ligstroside derivative | [129] | |
5.1. Extraction
5.2. Separation
5.3. Identification
6. Polyphenol Bioactive Ingredients in Other Plant Foods
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lnnhenheimer, F. Amphoras and Shipwrecks: Wine from the Tyrrhenian Coast at the End of the Republic and Its Distribution in Gaul; Wiley-Blackwell: Oxford, UK, 2013; p. 97. [Google Scholar]
- Sun, Z. Accelerated Seasoning of Manuka and Oak Wood Chips Destined for Wine and Spirit Flavour. Master’s Thesis, AUT University, Auckland, New Zealand, December 2013. [Google Scholar]
- Johnson, H.; Robinson, J. The World Atlas of Wine; Mitchell Beazley: London, UK, 2001; Volume 1. [Google Scholar]
- Charters, S. Wine and Society; Butterworth-Heinemann: Oxford, UK, 2006. [Google Scholar]
- Fernández de Simón, B.; Cadahía, E.; Conde, E.; García-Vallejo, M.C. Low molecular weight phenolic compounds in spanish oak woods. J. Agric. Food Chem. 1996, 44, 1507–1511. [Google Scholar] [CrossRef]
- Nonier, M.F.; Vivas, N.; Vivas de Gaulejac, N.; Absalon, C.; Soulié, P.; Fouquet, E. Pyrolysis–gas chromatography/mass spectrometry of Quercus sp. Wood: Application to structural elucidation of macromolecules and aromatic profiles of different species. J. Anal. Appl. Pyrolysis 2006, 75, 181–193. [Google Scholar]
- Glabasnia, A.; Hofmann, T. Sensory-directed identification of taste-active ellagitannins in American (Quercus alba L.) and European oak wood (Quercus robur L.) and quantitative analysis in bourbon whiskey and oak-matured red wines. J. Agric. Food Chem. 2006, 54, 3380–3390. [Google Scholar]
- Cadahía, E.; Varea, S.; Muñoz, L.; Fernández de Simón, B.; García-Vallejo, M.C. Evolution of ellagitannins in Spanish, French, and American oak woods during natural seasoning and toasting. J. Agric. Food Chem. 2001, 49, 3677–3684. [Google Scholar] [CrossRef] [PubMed]
- Hernández, T.; Estrella, I.; Dueñas, M.; de Simón, B.F.; Cadahía, E. Influence of wood origin in the polyphenolic composition of a Spanish red wine aging in bottle, after storage in barrels of Spanish, French and American oak wood. Eur. Food Res. Technol. 2007, 224, 695–705. [Google Scholar]
- De Simón, B.F.; Hernández, T.; Cadahía, E.; Dueñas, M.; Estrella, I. Phenolic compounds in a Spanish red wine aged in barrels made of Spanish, French and American oak wood. Eur. Food Res. Technol. 2003, 216, 150–156. [Google Scholar]
- Prida, A.; Puech, J.L. Influence of geographical origin and botanical species on the content of extractives in American, French, and East European oak woods. J. Agric. Food Chem. 2006, 54, 8115–8126. [Google Scholar] [CrossRef]
- Ibern-Gómez, M.; Andrés-Lacueva, C.; Lamuela-Raventós, R.M.; Lao-Luque, C.; Buxaderas, S.; de la Torre-Boronat, M.C. Differences in phenolic profile between oak wood and stainless steel fermentation in white wines. Am. J. Enol. Viticult. 2001, 52, 159–164. [Google Scholar]
- Canas, S.; Quaresma, H.; Belchior, A.P.; Spranger, M.I.; Bruno-de-Sousa, R. Evaluation of wine brandies authenticity by the relationships between benzoic and cinnamic aldehydes and between furanic aldehydes. Ciênc. Téc. Vitivinic. 2004, 19, 13–27. [Google Scholar]
- Fujieda, M.; Tanaka, T.; Suwa, Y.; Koshimizu, S.; Kouno, I. Isolation and structure of whiskey polyphenols produced by oxidation of oak wood ellagitannins. J. Agric. Food Chem. 2008, 56, 7305–7310. [Google Scholar] [CrossRef]
- Heras, M.O.; Rivero-Pérez, M.D.; Pérez-Magariño, S.; González-Huerta, C.; González-Sanjosé, M.L. Changes in the volatile composition of red wines during aging in oak barrels due to microoxygenation treatment applied before malolactic fermentation. Eur. Food Res. Technol. 2008, 226, 1485–1493. [Google Scholar] [CrossRef]
- Pérez-Magariño, S.; Sánchez-Iglesias, M.; Ortega-Heras, M.; González-Huerta, C.; González-Sanjosé, M.L. Colour stabilization of red wines by microoxygenation treatment before malolactic fermentation. Food Chem. 2007, 101, 881–893. [Google Scholar] [CrossRef]
- Sousa, A.; Mateus, N.; Silva, A.M.S.; Vivas, N.; Nonier, M.-F.; Pianet, I.; de Freitas, V. Isolation and structural characterization of anthocyanin-furfuryl pigments. J. Agric. Food Chem. 2010, 58, 5664–5669. [Google Scholar] [CrossRef]
- Saucier, C.; Jourdes, M.; Glories, Y.; Quideau, S. Extraction, detection, and quantification of flavano-ellagitannins and ethylvescalagin in a bordeaux red wine aged in oak barrels. J. Agric. Food Chem. 2006, 54, 7349–7354. [Google Scholar] [CrossRef]
- Pissarra, J.; Lourenco, S.; González-Paramás, A.M.; Mateus, N.; Buelga, C.S.; Silva, A.M.S.; de Freitas, V. Isolation and structural characterization of new anthocyanin-alkyl-catechin pigments. Food Chem. 2005, 90, 81–87. [Google Scholar] [CrossRef]
- Madrera, R.R.; Valles, B.S.; García, Y.D.; del Valle Argüelles, P.; Lobo, A.P. Alternative woods for aging distillates-an insight into their phenolic profiles and antioxidant activities. Food Sci. Biotechnol. 2010, 19, 1129–1134. [Google Scholar]
- Setzer, W.N. Lignin-derived oak phenolics: A theoretical examination of additional potential health benefits of red wine. J. Mol. Model. 2011, 17, 1841–1845. [Google Scholar] [CrossRef]
- Alañón, M.E.; Castro-Vázquez, L.; Díaz-Maroto, M.C.; Hermosín-Gutiérrez, I.; Gordon, M.H.; Pérez-Coello, M.S. Antioxidant capacity and phenolic composition of different woods used in cooperage. Food Chem. 2011, 129, 1584–1590. [Google Scholar] [CrossRef]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, S.S.; Sathe, S.K.; Salunkhe, D.K. Chemistry and safety of plant polyphenols. In Nutritional and Toxicological Aspects of Food Safety; Plenum Press: New York, NY, USA, 1984; pp. 457–495. [Google Scholar]
- Ananga, A.; Georgiev, V.; Tsolova, V. Manipulation and engineering of metabolic and biosynthetic pathway of plant polyphenols. Curr. Pharm. Des. 2013, 19, 6186–6206. [Google Scholar] [CrossRef] [PubMed]
- Hichri, I.; Barrieu, F.; Bogs, J.; Kappel, C.; Delrot, S.; Lauvergeat, V. Recent advances in the transcriptional regulation of the flavonoid biosynthetic pathway. J. Exp. Bot. 2011, 62, 2465–2483. [Google Scholar] [CrossRef] [PubMed]
- De Vries, G.E. Flavonoid biosynthetic pathway. Trends. Plant Sci. 2000, 5, 7. [Google Scholar]
- Wollgast, J.; Anklam, E. Review on polyphenols in Theobroma cacao changes in composition during the manufacture of chocolate and methodology for identification and quantification. Food Res. Int. 2000, 33, 423–447. [Google Scholar] [CrossRef]
- Ross, J.A.; Kasum, C.M. Dietary flavonoids: Bioavailability, metabolic effects, and safety. Annu. Rev. Nutr. 2002, 22, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Cuyckens, F.; Claeys, M. Mass spectrometry in the structural analysis of flavonoids. J. Mass Spectrom. 2004, 39, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, K. Flavonols and flavones in food plants: A review. Int. J. Food Sci. Technol. 1976, 11, 433–448. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Rentzsch, M.; Wilkens, A.; Winterhalter, P. Non-flavonoid phenolic compounds. In Wine Chemistry and Biochemistry; Springer: New York, NY, USA, 2009; pp. 509–527. [Google Scholar]
- Gonçalves, J.; Silva, C.L.; Castilho, P.C.; Câmara, J.S. An attractive, sensitive and high-throughput strategy based on microextraction by packed sorbent followed by UHPLC-PDA analysis for quantification of hydroxybenzoic and hydroxycinnamic acids in wines. Microchem. J. 2013, 106, 129–138. [Google Scholar] [CrossRef]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Phenolic compounds in brassica vegetables. Molecules 2010, 16, 251–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Phenols, polyphenols and tannins: An overview. In Plant Secondary Metabolites: Occurrence, Structure and Role in the Human Diet; Wiley-Blackwell: Oxford, UK, 2006; pp. 1–24. [Google Scholar]
- Kanerva, S. Plant Secondary Compounds and Soil Microbial Processes in Carbon and Nitrogen Cycling in Relation to Tree Species. Ph.D. Thesis, University of Helsinki, Helsinki, Finland, December 2007. [Google Scholar]
- Liu, P. Composition of Hawthorn (Crataegus spp.) Fruits and Leaves and Emblic Leafflower (Phyllanthus emblica) Fruits. Ph.D. Thesis, University of Turku, Turku, Finland, May 2012. [Google Scholar]
- Arapitsas, P. Hydrolyzable tannin analysis in food. Food Chem. 2012, 135, 1708–1717. [Google Scholar] [CrossRef] [PubMed]
- Alañón, M.E.; Castro-Vázquez, L.; Díaz-Maroto, M.C.; Gordon, M.H.; Pérez-Coello, M.S. A study of the antioxidant capacity of oak wood used in wine ageing and the correlation with polyphenol composition. Food Chem. 2011, 128, 997–1002. [Google Scholar] [CrossRef]
- Carrillo, J.D.; Garrido-López, Á.; Tena, M.T. Determination of volatile oak compounds in wine by headspace solid-phase microextraction and gas chromatography-mass spectrometry. J. Chromatogr. A 2006, 1102, 25–36. [Google Scholar]
- Pollnitz, A.P.; Pardon, K.H.; Sykes, M.; Sefton, M.A. The effects of sample preparation and gas chromatograph injection techniques on the accuracy of measuring guaiacol, 4-methylguaiacol and other volatile oak compounds in oak extracts by stable isotope dilution analyses. J. Agric. Food Chem. 2004, 52, 3244–3252. [Google Scholar] [CrossRef] [PubMed]
- Marín, J.; Zalacain, A.; de Miguel, C.; Alonso, G.L.; Salinas, M.R. Stir bar sorptive extraction for the determination of volatile compounds in oak-aged wines. J. Chromatogr. A 2005, 1098, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Guth, H. Quantitation and sensory studies of character impact odorants of different white wine varieties. J. Agric. Food Chem. 1997, 45, 3027–3032. [Google Scholar] [CrossRef]
- Jarauta, I.; Cacho, J.; Ferreira, V. Concurrent phenomena contributing to the formation of the aroma of wine during aging in oak wood: An analytical study. J. Agric. Food Chem. 2005, 53, 4166–4177. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, M.; Wabnitz, T.C.; Winterhalter, P. Pathway leading to the formation of anthocyanin-vinylphenol adducts and related pigments in red wines. J. Agric. Food Chem. 2003, 51, 3682–3687. [Google Scholar] [CrossRef] [PubMed]
- Natali, N.; Chinnici, F.; Riponi, C. Characterization of volatiles in extracts from oak chips obtained by accelerated solvent extraction (ASE). J. Agric. Food Chem. 2006, 54, 8190–8198. [Google Scholar] [CrossRef] [PubMed]
- Fernández de Simón, B.G.; Esteruelas, E.; Muñoz, A.N.M.; Cadahía, E.; Sanz, M. Volatile compounds in acacia, chestnut, cherry, ash, and oak woods, with a view to their use in cooperage. J. Agric. Food Chem. 2009, 57, 3217–3227. [Google Scholar]
- De Simón, B.F.; Muiño, I.; Cadahía, E. Characterization of volatile constituents in commercial oak wood chips. J. Agric. Food Chem. 2010, 58, 9587–9596. [Google Scholar]
- Del Alamo Sanza, M.; Nevares Domınguez, I.; Cárcel Cárcel, L.M.; Navas Gracia, L. Analysis for low molecular weight phenolic compounds in a red wine aged in oak chips. Anal. Chim. Acta 2004, 513, 229–237. [Google Scholar]
- Da Silva, A.A.; do Nascimento, E.S.P.; Cardoso, D.R.; Franco, D.W. Coumarins and phenolic fingerprints of oak and Brazilian woods extracted by sugarcane spirit. J. Sep. Sci. 2009, 32, 3681–3691. [Google Scholar]
- Fernández de Simón, B.; Sanz, M.; Cadahía, E.; Poveda, P.; Broto, M. Chemical characterization of oak heartwood from Spanish forests of Quercus pyrenaica (wild.). Ellagitannins, low molecular weight phenolic, and volatile compounds. J. Agric. Food Chem. 2006, 54, 8314–8321. [Google Scholar]
- Cadahía, E.; Muñoz, L.; Fernández de Simón, B.; García-Vallejo, M.C. Changes in low molecular weight phenolic compounds in Spanish, French, and American oak woods during natural seasoning and toasting. J. Agric. Food Chem. 2001, 49, 1790–1798. [Google Scholar] [CrossRef] [PubMed]
- Dos Anjos, J.P.; das Cardoso, M.G.; Saczk, A.A.; Dórea, H.S.; Santiago, W.D.; Machado, A.M.R.; Zacaroni, L.M.; Nelson, D.L. Evolution of the concentration of phenolic compounds in cachaça during aging in an oak (Quercus sp.) barrel. J. Braz. Chem. Soc. 2011, 22, 1307–1314. [Google Scholar]
- Regalado, E.L.; Tolle, S.; Pino, J.A.; Winterhalter, P.; Menendez, R.; Morales, A.R.; Rodríguez, J.L. Isolation and identification of phenolic compounds from rum aged in oak barrels by high-speed countercurrent chromatography/high-performance liquid chromatography-diode array detection-electrospray ionization mass spectrometry and screening for antioxidant activity. J. Chromatogr. A 2011, 1218, 7358–7364. [Google Scholar] [CrossRef] [PubMed]
- Cabrita, M.J.; Barrocas Dias, C.; Costa Freitas, A.M. Phenolic acids, phenolic aldehydes and furanic derivatives in oak chips: American vs. French oaks. S. Afr. J. Enol. Vitic. 2011, 32, 204–210. [Google Scholar]
- Nonier Bourden, M.F.; Vivas, N.; Absalon, C.; Vitry, C.; Fouquet, E.; Vivas de Gaulejac, N. Structural diversity of nucleophilic adducts from flavanols and oak wood aldehydes. Food Chem. 2008, 107, 1494–1505. [Google Scholar]
- Zhang, B.; Liu, R.; He, F.; Zhou, P.-P.; Duan, C.-Q. Copigmentation of malvidin-3-O-glucoside with five hydroxybenzoic acids in red wine model solutions: Experimental and theoretical investigations. Food Chem. 2015, 170, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Eiro, M.J.; Heinonen, M. Anthocyanin color behavior and stability during storage: Effect of intermolecular copigmentation. J. Agric. Food Chem. 2002, 50, 7461–7466. [Google Scholar] [CrossRef] [PubMed]
- Boulton, R. The copigmentation of anthocyanins and its role in the color of red wine: A critical review. Am. J. Enol. Vitic. 2001, 52, 67–87. [Google Scholar]
- Sun, J.; Cao, X.; Liao, X.; Hu, X. Comparative analyses of copigmentation of cyanidin 3-glucoside and cyanidin 3-sophoroside from red raspberry fruits. Food Chem. 2010, 120, 1131–1137. [Google Scholar] [CrossRef]
- Viriot, C.; Scalbert, A.; Lapierre, C.; Moutounet, M. Ellagitannins and lignins in aging of spirits in oak barrels. J. Agric. Food Chem. 1993, 41, 1872–1879. [Google Scholar] [CrossRef]
- Landete, J.M. Ellagitannins, ellagic acid and their derived metabolites: A review about source, metabolism, functions and health. Food Res. Int. 2011, 44, 1150–1160. [Google Scholar] [CrossRef]
- Ree, T. Tannins: Classification and definition. Nat. Prod. Rep. 2001, 18, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Niemetz, R.; Gross, G.G. Enzymology of gallotannin and ellagitannin biosynthesis. Phytochemistry 2005, 66, 2001–2011. [Google Scholar] [CrossRef] [PubMed]
- Michel, J.; Jourdes, M.; Silva, M.A.; Giordanengo, T.; Mourey, N.; Teissedre, P.-L. Impact of concentration of ellagitannins in oak wood on their levels and organoleptic influence in red wine. J. Agric. Food Chem. 2011, 59, 5677–5683. [Google Scholar] [CrossRef] [PubMed]
- Masson, G.; Moutounet, M.; Puech, J.L. Ellagitannin content of oak wood as a function of species and of sampling position in the tree. Am. J. Enol. Vitic. 1995, 46, 262–268. [Google Scholar]
- Doussot, F.; de Jéso, B.; Quideau, S.; Pardon, P. Extractives content in cooperage oak wood during natural seasoning and toasting; Influence of tree species, geographic location, and single-tree effects. J. Agric. Food Chem. 2002, 50, 5955–5961. [Google Scholar] [CrossRef] [PubMed]
- Mila, I.; Scalbert, A.; Expert, D. Iron withholding by plant polyphenols and resistance to pathogens and rots. Phytochemistry 1996, 42, 1551–1555. [Google Scholar] [CrossRef]
- Mosedale, J.R.; Puech, J.L.; Feuillat, F. The influence on wine flavor of the oak species and natural variation of heartwood components. Am. J. Enol. Vitic. 1999, 50, 503–512. [Google Scholar]
- Quideau, S.; Jourdes, M.; Lefeuvre, D.; Montaudon, D.; Saucier, C.; Glories, Y.; Pardon, P.; Pourquier, P. The chemistry of wine polyphenolic C-glycosidic ellagitannins targeting human topoisomerase II. Chem.-A Eur. J. 2005, 11, 6503–6513. [Google Scholar] [CrossRef]
- Chassaing, S.; Lefeuvre, D.; Jacquet, R.; Jourdes, M.; Ducasse, L.; Galland, S.; Grelard, A.; Saucier, C.; Teissedre, P.L.; Dangles, O. Physicochemical studies of new anthocyano-ellagitannin hybrid pigments: About the origin of the influence of oak C-glycosidic ellagitannins on wine color. Eur. J. Org. Chem. 2010, 2010, 55–63. [Google Scholar] [CrossRef]
- Alañón, M.E.; Schumacher, R.; Castro-Vázquez, L.; Díaz-Maroto, M.C.; Hermosín-Gutiérrez, I.; Pérez-Coello, M.S. Enological potential of chestnut wood for aging tempranillo wines part II: Phenolic compounds and chromatic characteristics. Food Res. Int. 2013, 51, 536–543. [Google Scholar] [CrossRef]
- Sanz, M.; Fernandez de Simon, B.; Esteruelas, E.; Munoz, A.M.; Cadahia, E.; Hernandez, M.T.; Estrella, I.; Martinez, J. Polyphenols in red wine aged in acacia (Robinia pseudoacacia) and oak (Quercus petraea) wood barrels. Anal. Chim. Acta 2012, 732, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Springmann, S.; Rogers, R.; Spiecker, H. Impact of artificial pruning on growth and secondary shoot development of wild cherry (Prunus avium L.). For. Ecol. Manag. 2011, 261, 764–769. [Google Scholar] [CrossRef]
- Fernandez de Simon, B.; Sanz, M.; Cadahia, E.; Martinez, J.; Esteruelas, E.; Munoz, A.M. Polyphenolic compounds as chemical markers of wine ageing in contact with cherry, chestnut, false acacia, ash and oak wood. Food Chem. 2014, 143, 66–76. [Google Scholar]
- Comandini, P.; Lerma-Garcia, M.J.; Simo-Alfonso, E.F.; Toschi, T.G. Tannin analysis of chestnut bark samples (Castanea sativa Mill.) by HPLC-DAD-MS. Food Chem. 2014, 157, 290–295. [Google Scholar] [CrossRef]
- Sanz, M.; Fernandez de Simon, B.; Cadahia, E.; Esteruelas, E.; Munoz, A.M.; Hernandez, M.T.; Estrella, I. Polyphenolic profile as a useful tool to identify the wood used in wine aging. Anal. Chim. Acta 2012, 732, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Fernandez de Simon, B.; Martinez, J.; Sanz, M.; Cadahia, E.; Esteruelas, E.; Munoz, A.M. Volatile compounds and sensorial characterisation of red wine aged in cherry, chestnut, false acacia, ash and oak wood barrels. Food Chem. 2014, 147, 346–356. [Google Scholar]
- Towey, J.P.; Waterhouse, A.L. Barrel-to-barrel variation of volatile oak extractives in barrel-fermented chardonnay. Am. J. Enol. Vitic. 1996, 47, 17–20. [Google Scholar]
- Puech, J.L.; Feuillat, F.; Mosedale, J.R. The tannins of oak heartwood: Structure, properties, and their influence on wine flavor. Am. J. Enol. Vitic. 1999, 50, 469–478. [Google Scholar]
- Fan, W.; Xu, Y.; Yu, A. Influence of oak chips geographical origin, toast level, dosage and aging time on volatile compounds of apple cider. J. Inst. Brew. 2006, 112, 255–263. [Google Scholar] [CrossRef]
- Guchu, E.; Díaz-Maroto, M.C.; Díaz-Maroto, I.J.; Vila-Lameiro, P.; Pérez-Coello, M.S. Influence of the species and geographical location on volatile composition of Spanish oak wood (Quercus petraea Liebl. and Quercus robur L.). J. Agric. Food Chem. 2006, 54, 3062–3066. [Google Scholar] [CrossRef] [PubMed]
- Fernández de Simón, B.; Cadahía, E.; Conde, E.; García-Vallejo, M.C. Evolution of phenolic compounds of Spanish oak wood during natural seasoning. First results. J. Agric. Food Chem. 1999, 47, 1687–1694. [Google Scholar]
- Escalona, H.; Birkmyre, L.; Piggott, J.R.; Paterson, A. Effect of maturation in small oak casks on the volatility of red wine aroma compounds. Anal. Chim. Acta 2002, 458, 45–54. [Google Scholar] [CrossRef]
- Miller, D.P.; Howell, G.S.; Michaelis, C.S.; Dickmann, D.I. The content of phenolic acid and aldehyde flavor components of white oak as affected by site and species. Am. J. Enol. Vitic. 1992, 43, 333–338. [Google Scholar]
- Glabasnia, A.; Hofmann, T. Identification and sensory evaluation of dehydro-and deoxy-ellagitannins formed upon toasting of oak wood (Quercus alba L.). J. Agric. Food Chem. 2007, 55, 4109–4118. [Google Scholar] [CrossRef] [PubMed]
- Sefton, M.A.; Francis, I.L.; Pocock, K.F.; Williams, P.J. The influence of natural seasoning on the concentrations of eugenol, vanillin, and cis-and trans-β-methyl-γ-octalactone extracted from French and American oakwood. Sci. Des. Aliment. 1993, 13, 629–643. [Google Scholar]
- Pérez-Coello, M.S.; Díaz-Maroto, M.C. Volatile compounds and wine aging. In Wine Chemistry and Biochemistry; Springer: New York, NY, USA, 2009; pp. 295–311. [Google Scholar]
- Pérez-Coello, M.S.; Sanz, J.; Cabezudo, M.D. Determination of volatile compounds in hydroalcoholic extracts of french and american oak wood. Am. J. Enol. Vitic. 1999, 50, 162–165. [Google Scholar]
- Prida, A.; Boulet, J.-C.; Ducousso, A.; Nepveu, G.; Puech, J.-L. Effect of species and ecological conditions on ellagitannin content in oak wood from an even-aged and mixed stand of Quercus robur L. And Quercus petraea Liebl. Ann. For. Sci. 2006, 63, 415–424. [Google Scholar]
- Gallego, L.; del Alamo, M.; Nevares, I.; Fernández, J.A.; de Simón, B.F.; Cadahía, E. Phenolic compounds and sensorial characterization of wines aged with alternative to barrel products made of Spanish oak wood (Quercus pyrenaica Willd.). Food Sci. Technol. Int. 2012, 18, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Chatonnet, P.; Boidron, J.N.; Dubourdieu, D.; Pons, M. Evolution of oakwood polyphenolic compounds during seasoning. First results. J. Int. Sci. Vigne Vin 1994, 28, 337–357. [Google Scholar]
- Bate-Smith, E.C. The commoner phenolic constituents of plants and their systematic distribution. Proc. R. Dublin Sci. Soc. 1956, 27, 165–176. [Google Scholar]
- Matricardi, L.; Waterhouse, A.L. Influence of toasting technique on color and ellagitannins of oak wood in barrel making. Am. J. Enol. Vitic. 1999, 50, 519–526. [Google Scholar]
- Frangipane, M.T.; Santis, D.D.; Ceccarelli, A. Influence of oak woods of different geographical origins on quality of wines aged in barriques and using oak chips. Food Chem. 2007, 103, 46–54. [Google Scholar] [CrossRef]
- Chatonnet, P.; Cutzach, I.; Pons, M.; Dubourdieu, D. Monitoring toasting intensity of barrels by chromatographic analysis of volatile compounds from toasted oak wood. J. Agric. Food Chem. 1999, 47, 4310–4318. [Google Scholar] [CrossRef] [PubMed]
- Sarni, F.; Moutounet, M.; Puech, J.-L.; Rabier, P. Effect of heat treatment of oak wood extractable compounds. Holzforschung 1990, 44, 461–466. [Google Scholar] [CrossRef]
- Jordão, A.M.; Correia, A.C.; DelCampo, R.; SanJosé, M.L.G. Antioxidant capacity, scavenger activity, and ellagitannins content from commercial oak pieces used in winemaking. Eur. Food Res. Technol. 2012, 235, 817–825. [Google Scholar] [CrossRef]
- Tao, Y.; García, J.F.; Sun, D.-W. Advances in wine ageing technologies for enhancing wine quality and accelerating wine ageing process. Crit. Rev. Food Sci. Nutr. 2014, 54, 817–835. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Prieto, L.J.; López-Roca, J.M.; Martínez-Cutillas, A.; Pardo-Mínguez, F.; Gómez-Plaza, E. Extraction and formation dynamic of oak-related volatile compounds from different volume barrels to wine and their behavior during bottle storage. J. Agric. Food Chem. 2003, 51, 5444–5449. [Google Scholar] [CrossRef] [PubMed]
- Spillman, P.J.; Iland, P.G.; Sefton, M.A. Accumulation of volatile oak compounds in a model wine stored in American and Limousin oak barrels. Aust. J. Grape Wine Res. 1998, 4, 67–73. [Google Scholar] [CrossRef]
- Rodríguez-Rodríguez, P.; Gómez-Plaza, E. Effect of volume and toast level of french oak barrels (Quercus petraea L.) on cabernet sauvignon wine characteristics. Am. J. Enol. Vitic. 2011, 62, 359–365. [Google Scholar] [CrossRef]
- Charrier, B.; Janin, G.; Haluk, J.P.; Mosedale, J.R. Colour and chemical characteristics of moon rings in oakwood. Holzforschung 1995, 49, 287–292. [Google Scholar] [CrossRef]
- Viriot, C.; Scalbert, A.; Hervé du Penhoat, C.L.M.; Moutounet, M. Ellagitannins in woods of sessile oak and sweet chestnut dimerization and hydrolysis during wood ageing. Phytochemistry 1994, 36, 1253–1260. [Google Scholar] [CrossRef]
- Mosedale, J.R.; Savill, P.S. Variation of heartwood phenolics and oak lactones between the species and phenological types of Quercus petraea and Q. robur. Forestry 1996, 69, 47–55. [Google Scholar] [CrossRef]
- Mosedale, J.R.; Charrier, B.; Janin, G. Genetic control of wood colour, density and heartwood ellagitannin concentration in european oak (Quercus petraea and Q. Robur). Forestry 1996, 69, 111–124. [Google Scholar] [CrossRef]
- Sanz, M.; Cadahía, E.; Esteruelas, E.; Muñoz, A.N.M.; Fernández De Simón, B.G.; Hernández, T.; Estrella, I. Phenolic compounds in cherry (Prunus avium) heartwood with a view to their use in cooperage. J. Agric. Food Chem. 2010, 58, 4907–4914. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Jun, H.; Choi, S.; Kim, Y.; Jung, E.; Oh, G.; Joo, S.; Kim, S.; Kim, I. Evaluation of antioxidant activities and active compounds separated from water soluble extracts of Korean black pine barks. Bull. Korean Chem. Soc. 2010, 31, 3567–3572. [Google Scholar] [CrossRef]
- Sanz, M.; Fernández de Simón, B.G.; Esteruelas, E.; Muñoz, A.N.M.; Cadahía, E.; Hernández, T.; Estrella, I.; Pinto, E. Effect of toasting intensity at cooperage on phenolic compounds in acacia (Robinia pseudoacacia) heartwood. J. Agric. Food Chem. 2011, 59, 3135–3145. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, D.W.; Mercurio, M.D.; Herderich, M.J.; Hayasaka, Y.; Smith, P.A. Rapid isolation of red wine polymeric polyphenols by solid-phase extraction. J. Agric. Food Chem. 2008, 56, 2571–2580. [Google Scholar] [CrossRef] [PubMed]
- Cerezo, A.B.; Tesfaye, W.; Torija, M.J.; Mateo, E.; García-Parrilla, M.C.; Troncoso, A.M. The phenolic composition of red wine vinegar produced in barrels made from different woods. Food Chem. 2008, 109, 606–615. [Google Scholar] [CrossRef]
- Marrufo-Curtido, A.; Cejudo-Bastante, M.J.; Durán-Guerrero, E.; Castro-Mejías, R.; Natera-Marín, R.; Chinnici, F.; García-Barroso, C. Characterization and differentiation of high quality vinegars by stir bar sorptive extraction coupled to gas chromatography-mass spectrometry (SBSE-GC-MS). LWT-Food Sci. Technol. 2012, 47, 332–341. [Google Scholar] [CrossRef]
- Borges, G.; Crozier, A. HPLC-PDA-MS fingerprinting to assess the authenticity of pomegranate beverages. Food Chem. 2012, 135, 1863–1867. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Wang, C.; Pei, H.; Sun, B. Separation and identification of polyphenols in apple pomace by high-speed counter-current chromatography and high-performance liquid chromatography coupled with mass spectrometry. J. Chromatogr. A 2009, 1216, 4268–4274. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.L.; Yue, T.L.; Yuan, Y.H.; Zhang, H.W. Optimization of microwave-passisted extraction of polyphenols from apple pomace using response surface methodology and HPLC analysis. J. Sep. Sci. 2010, 33, 3751–3758. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez Galdón, B.; Rodríguez Rodríguez, E.M.; Díaz Romero, C. Flavonoids in onion cultivars (Allium cepa L.). J. Food Sci. 2008, 73, C599–C605. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.S.; Pérez-Gregorio, M.R.; García-Falcón, M.S.; Simal-Gándara, J. Effect of curing and cooking on flavonols and anthocyanins in traditional varieties of onion bulbs. Food Res. Int. 2009, 42, 1331–1336. [Google Scholar] [CrossRef]
- Rodrigues, A.S.; Pérez-Gregorio, M.R.; García-Falcón, M.S.; Simal-Gándara, J.; Almeida, D.P.F. Effect of post-harvest practices on flavonoid content of red and white onion cultivars. Food Control 2010, 21, 878–884. [Google Scholar] [CrossRef]
- Gelmez, N.; Kıncal, N.S.; Yener, M.E. Optimization of supercritical carbon dioxide extraction of antioxidants from roasted wheat germ based on yield, total phenolic and tocopherol contents, and antioxidant activities of the extracts. J. Supercrit. Fluids 2009, 48, 217–224. [Google Scholar] [CrossRef]
- Tomaino, A.; Martorana, M.; Arcoraci, T.; Monteleone, D.; Giovinazzo, C.; Saija, A. Antioxidant activity and phenolic profile of pistachio (Pistacia vera L., variety Bronte) seeds and skins. Biochimie 2010, 92, 1115–1122. [Google Scholar]
- Singh, A.; Sabally, K.; Kubow, S.; Donnelly, D.J.; Gariepy, Y.; Orsat, V.; Raghavan, G.S.V. Microwave-assisted extraction of phenolic antioxidants from potato peels. Molecules 2011, 16, 2218–2232. [Google Scholar] [CrossRef] [PubMed]
- Fromm, M.; Bayha, S.; Carle, R.; Kammerer, D.R. Characterization and quantitation of low and high molecular weight phenolic compounds in apple seeds. J. Agric. Food Chem. 2012, 60, 1232–1242. [Google Scholar] [CrossRef] [PubMed]
- Iswaldi, I.; Arráez-Román, D.; Rodríguez-Medina, I.; Beltrán-Debón, R.; Joven, J.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Identification of phenolic compounds in aqueous and ethanolic rooibos extracts (Aspalathus linearis) by HPLC-ESI-MS (TOF/IT). Anal. Bioanal. Chem. 2011, 400, 3643–3654. [Google Scholar] [CrossRef] [PubMed]
- Beelders, T.; Sigge, G.O.; Joubert, E.; de Beer, D.; de Villiers, A. Kinetic optimisation of the reversed phase liquid chromatographic separation of rooibos tea (Aspalathus linearis) phenolics on conventional high performance liquid chromatographic instrumentation. J. Chromatogr. A 2012, 1219, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Grujic, N.; Lepojevic, Z.; Srdjenovic, B.; Vladic, J.; Sudji, J. Effects of different extraction methods and conditions on the phenolic composition of mate tea extracts. Molecules 2012, 17, 2518–2528. [Google Scholar] [CrossRef] [PubMed]
- Perestrelo, R.; Caldeira, M.; Barros, A.S.; Rocha, S.M.; Câmara, J.S. Solid phase microextraction as a reliable alternative to conventional extraction techniques to evaluate the pattern of hydrolytically released components in of Vitis vinifera L. grapes. Talanta 2012, 95, 1–11. [Google Scholar] [CrossRef]
- Palafox-Carlos, H.; Yahia, E.M.; González-Aguilar, G.A. Identification and quantification of major phenolic compounds from mango (Mangifera indica, cv. Ataulfo) fruit by HPLC-DAD-MS/MS-ESI and their individual contribution to the antioxidant activity during ripening. Food Chem. 2012, 135, 105–111. [Google Scholar]
- Jerman Klen, T.; Mozetič Vodopivec, B. Optimisation of olive oil phenol extraction conditions using a high-power probe ultrasonication. Food Chem. 2012, 134, 2481–2488. [Google Scholar] [CrossRef] [PubMed]
- Naczk, M.; Shahidi, F. Extraction and analysis of phenolics in food. J. Chromatogr. A 2004, 1054, 95–111. [Google Scholar] [CrossRef] [PubMed]
- Naczk, M.; Shahidi, F. Phenolics in cereals, fruits and vegetables: Occurrence, extraction and analysis. J. Pharm. Biomed. Anal. 2006, 41, 1523–1542. [Google Scholar] [CrossRef] [PubMed]
- Antolovich, M.; Prenzler, P.; Robards, K.; Ryan, D. Sample preparation in the determination of phenolic compounds in fruits. Analyst 2000, 125, 989–1009. [Google Scholar] [CrossRef]
- Pinelo, M.; Fabbro, P.D.; Manzocco, L.; Nuñez, M.J.; Nicoli, M.C. Optimization of continuous phenol extraction from Vitis vinifera byproducts. Food Chem. 2005, 92, 109–117. [Google Scholar] [CrossRef]
- Nardini, M.; Cirillo, E.; Natella, F.; Mencarelli, D.; Comisso, A.; Scaccini, C. Detection of bound phenolic acids: Prevention by ascorbic acid and ethylenediaminetetraacetic acid of degradation of phenolic acids during alkaline hydrolysis. Food Chem. 2002, 79, 119–124. [Google Scholar] [CrossRef]
- Aaby, K.; Ekeberg, D.; Skrede, G. Characterization of phenolic compounds in strawberry (Fragaria× ananassa) fruits by different HPLC detectors and contribution of individual compounds to total antioxidant capacity. J. Agric. Food. Chem. 2007, 55, 4395–4406. [Google Scholar] [CrossRef] [PubMed]
- Hayouni, E.A.; Abedrabba, M.; Bouix, M.; Hamdi, M. The effects of solvents and extraction method on the phenolic contents and biological activities in vitro of Tunisian Quercus coccifera L. and Juniperus phoenicea L. fruit extracts. Food Chem. 2007, 105, 1126–1134. [Google Scholar]
- Paniwnyk, L.; Beaufoy, E.; Lorimer, J.P.; Mason, T.J. The extraction of rutin from flower buds of Sophora japonica. Ultrason. Sonochem. 2001, 8, 299–301. [Google Scholar] [CrossRef] [PubMed]
- Paniwnyk, L.; Cai, H.; Albu, S.; Mason, T.J.; Cole, R. The enhancement and scale up of the extraction of anti-oxidants from Rosmarinus officinalis using ultrasound. Ultrason. Sonochem. 2009, 16, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, G.; Pawliszyn, J. SPME in environmental analysis. Anal. Bioanal. Chem. 2006, 386, 1059–1073. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Maroto, M.C.; Sánchez-Palomo, E.; Pérez-Coello, M.S. Fast screening method for volatile compounds of oak wood used for aging wines by headspace SPME-GC-MS (SIM). J. Agric. Food Chem. 2004, 52, 6857–6861. [Google Scholar] [CrossRef] [PubMed]
- Kadim, D.; Mannheim, C.H. Kinetics of phenolic extraction during aging of model wine solution and white wine in oak barrels. Am. J. Enol. Vitic. 1999, 50, 33–39. [Google Scholar]
- Robbins, R.J. Phenolic acids in foods: An overview of analytical methodology. J. Agric. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef] [PubMed]
- Barton, G.M.; Evans, R.S.; Gardner, J.A.F. Paper chromatography of phenolic substances. Nature 1952, 170, 249–250. [Google Scholar] [CrossRef]
- Quinn, M.K.; Singleton, V.L. Isolation and identification of ellagitannins from white oak wood and an estimation of their roles in wine. Am. J. Enol. Vitic. 1985, 36, 148–155. [Google Scholar]
- Bele, A.A.; Khale, A. An overview on thin layer chromatography. Int. J. Pharm. Pharm. Sci. 2011, 2, 256–267. [Google Scholar]
- Lepri, L.; Desideri, P.G.; Heimler, D. Reversed-phase and soap thin-layer chromatography of phenols. J. Chromatogr. A 1980, 195, 339–348. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef] [PubMed]
- De Beer, D.; Harbertson, J.F.; Kilmartin, P.A.; Roginsky, V.; Barsukova, T.; Adams, D.O.; Waterhouse, A.L. Phenolics: A comparison of diverse analytical methods. Am. J. Enol. Vitic. 2004, 55, 389–400. [Google Scholar]
- Hellström, J.K.; Mattila, P.H. Hplc determination of extractable and unextractable proanthocyanidins in plant materials. J. Agric. Food Chem. 2008, 56, 7617–7624. [Google Scholar] [CrossRef] [PubMed]
- Herderich, M.J.; Smith, P.A. Analysis of grape and wine tannins: Methods, applications and challenges. Aust. J. Grape Wine Res. 2005, 11, 205–214. [Google Scholar] [CrossRef]
- Escarpa, A.; Gonzalez, M.C. Evaluation of high-performance liquid chromatography for determination of phenolic compounds in pear horticultural cultivars. Chromatographia 2000, 51, 37–43. [Google Scholar] [CrossRef]
- Mattila, P.; Kumpulainen, J. Determination of free and total phenolic acids in plant-derived foods by HPLC with diode-array detection. J. Agric. Food Chem. 2002, 50, 3660–3667. [Google Scholar] [CrossRef] [PubMed]
- Cui, T.; Li, J.-Z.; Kayahara, H.; Ma, L.; Wu, L.-X.; Nakamura, K. Quantification of the polyphenols and triterpene acids in Chinese hawthorn fruit by high-performance liquid chromatography. J. Agric. Food Chem. 2006, 54, 4574–4581. [Google Scholar] [CrossRef] [PubMed]
- Hathway, D.E. Oak-bark tannins. Biochem. J. 1958, 70, 34. [Google Scholar] [PubMed]
- He, Z.; Xia, W.; Chen, J. Isolation and structure elucidation of phenolic compounds in Chinese olive (Canarium album L.) fruit. Eur. Food Res. Technol. 2008, 226, 1191–1196. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef] [PubMed]
- Du, F.; Zhang, B.; Zhou, H.; Yan, B.; Chen, L. Structure elucidation of nanoparticle-bound organic molecules by 1 H NMR. TrAC Trends Anal. Chem. 2009, 28, 88–95. [Google Scholar] [CrossRef]
- Sudjaroen, Y.; Hull, W.E.; Erben, G.; Würtele, G.; Changbumrung, S.; Ulrich, C.M.; Owen, R.W. Isolation and characterization of ellagitannins as the major polyphenolic components of Longan (Dimocarpus longan Lour) seeds. Phytochemistry 2012, 77, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Vivas, N.; Laguerre, M.; Glories, Y.; Bourgeois, G.; Vitry, C. Structure simulation of two ellagitannins from Quercus robur l. Phytochemistry 1995, 39, 1193–1199. [Google Scholar] [CrossRef]
- Mabry, T.J.; Markham, K.R.; Thomas, M.B. The NMR spectra of flavonoids. In The Systematic Identification of Flavonoids; Springer: New York, NY, USA, 1970; pp. 274–343. [Google Scholar]
- Slimestad, R. Flavonoids in buds and young needles of Picea, Pinus and Abies. Biochem. Syst. Ecol. 2003, 31, 1247–1255. [Google Scholar] [CrossRef]
- Perez-Gregorio, M.R.; Regueiro, J.; Simal-Gandara, J.; Rodrigues, A.S.; Almeida, D.P. Increasing the added-value of onions as a source of antioxidant flavonoids: A critical review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1050–1062. [Google Scholar] [CrossRef] [PubMed]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep. 2009, 26, 1001–1043. [Google Scholar] [CrossRef] [PubMed]
- Buendía, B.a.; Gil, M.a.I.; Tudela, J.A.; Gady, A.L.; Medina, J.J.; Soria, C.; López, J.M.; Tomás-Barberán, F.A. HPLC-MS analysis of proanthocyanidin oligomers and other phenolics in 15 strawberry cultivars. J. Agric. Food Chem. 2009, 58, 3916–3926. [Google Scholar] [CrossRef]
- Basu, A.; Nguyen, A.; Betts, N.M.; Lyons, T.J. Strawberry as a functional food: An evidence-based review. Crit. Rev. Food Sci. Nutr. 2014, 54, 790–806. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Guo, Y.; Fu, C.; Li, J.; Li, Z. Simultaneous separation and purification of total polyphenols, chlorogenic acid and phlorizin from thinned young apples. Food Chem. 2013, 136, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- Kammerer, D.R.; Kammerer, J.; Valet, R.; Carle, R. Recovery of polyphenols from the by-products of plant food processing and application as valuable food ingredients. Food Res. Int. 2014, 65, 2–12. [Google Scholar] [CrossRef]
- Gı̂rzu, M.; Fraisse, D.; Carnat, A.-P.; Carnat, A.; Lamaison, J.-L. High-performance liquid chromatographic method for the determination of juglone in fresh walnut leaves. J. Chromatogr. A 1998, 805, 315–318. [Google Scholar] [CrossRef]
- Marceneiro, S.; Coimbra, P.; Braga, M.E.M.; Dias, A.M.A.; de Sousa, H.C. Measurement and correlation of the solubility of juglone in supercritical carbon dioxide. Fluid Phase Equilibria 2011, 311, 1–8. [Google Scholar] [CrossRef]
- Ruiz-Montañez, G.; Ragazzo-Sánchez, J.A.; Calderón-Santoyo, M.; Velázquez-de la Cruz, G.; de León, J.A.R.; Navarro-Ocaña, A. Evaluation of extraction methods for preparative scale obtention of mangiferin and lupeol from mango peels (Mangifera indica L.). Food Chem. 2014, 159, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Jyotshna; Srivastava, P.; Killadi, B.; Shanker, K. Uni-dimensional double development HPTLC-densitometry method for simultaneous analysis of mangiferin and lupeol content in mango (Mangifera indica) pulp and peel during storage. Food Chem. 2015, 176, 91–98. [Google Scholar] [CrossRef]
- Liu, C.; Wang, L.; Wang, J.; Wu, B.; Liu, W.; Fan, P.; Liang, Z.; Li, S. Resveratrols in Vitis berry skins and leaves: Their extraction and analysis by HPLC. Food Chem. 2013, 136, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Alfaro, I.; Gonzalez-Arenzana, L.; Lopez, N.; Santamaria, P.; Lopez, R.; Garde-Cerdan, T. Pulsed electric field treatment enhanced stilbene content in Graciano, Tempranillo and Grenache grape varieties. Food Chem. 2013, 141, 3759–3765. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.-F.; Li, C.-M.; Xu, Y.-J.; Hu, W.-F.; Chen, M.-H.; Wan, Q.-H. Structural features and antioxidant activity of tannin from persimmon pulp. Food Res. Int. 2008, 41, 208–217. [Google Scholar] [CrossRef]
- Wu, P.-W.; Hwang, L.S. Determination of soluble persimmon tannin by high performance gel permeation chromatography. Food Res. Int. 2002, 35, 793–800. [Google Scholar] [CrossRef]
- Hajiaghaalipour, F.; Kanthimathi, M.S.; Sanusi, J.; Rajarajeswaran, J. White tea (Camellia sinensis) inhibits proliferation of the colon cancer cell line, HT-29, activates caspases and protects DNA of normal cells against oxidative damage. Food Chem. 2015, 169, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Sahpazidou, D.; Geromichalos, G.D.; Stagos, D.; Apostolou, A.; Haroutounian, S.A.; Tsatsakis, A.M.; Tzanakakis, G.N.; Hayes, A.W.; Kouretas, D. Anticarcinogenic activity of polyphenolic extracts from grape stems against breast, colon, renal and thyroid cancer cells. Toxicol. Lett. 2014, 230, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.S.; Pérez-Gregorio, M.R.; García-Falcón, M.S.; Simal-Gándara, J.; Almeida, D.P.F. Effect of meteorological conditions on antioxidant flavonoids in Portuguese cultivars of white and red onions. Food Chem. 2011, 124, 303–308. [Google Scholar] [CrossRef]
- Ruiz-Canela, M.; Martínez-González, M.A. Olive oil in the primary prevention of cardiovascular disease. Maturitas 2011, 68, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J.; Kaur, G. An insight into the role of citrus bioactives in modulation of colon cancer. J. Funct. Foods 2015, 13, 239–261. [Google Scholar] [CrossRef]
- Yamanaka, F.; Hatano, T.; Ito, H.; Taniguchi, S.; Takahashi, E.; Okamoto, K. Antibacterial effects of guava tannins and related polyphenols on Vibrio and Aeromonas species. Nat. Prod. Commun. 2008, 3, 711–720. [Google Scholar]
- Tripoli, E.; Guardia, M.L.; Giammanco, S.; Majo, D.D.; Giammanco, M. Citrus flavonoids: Molecular structure, biological activity and nutritional properties: A review. Food Chem. 2007, 104, 466–479. [Google Scholar] [CrossRef]
- Wen, X.-B.; Miao, F.; Zhou, L.; Zhang, M.; He, Q.-L. In vitro antioxidant activity of Parnassia wightiana W. extracts. Chin. J. Nat. Med. 2012, 10, 190–195. [Google Scholar] [CrossRef]
- Pérez-Gregorio, M.R.; González-Barreiro, C.; Rial-Otero, R.; Simal-Gándara, J. Comparison of sanitizing technologies on the quality appearance and antioxidant levels in onion slices. Food Control 2011, 22, 2052–2058. [Google Scholar] [CrossRef]
- Pérez-Gregorio, M.R.; Regueiro, J.; González-Barreiro, C.; Rial-Otero, R.; Simal-Gándara, J. Changes in antioxidant flavonoids during freeze-drying of red onions and subsequent storage. Food Control 2011, 22, 1108–1113. [Google Scholar] [CrossRef]
- Figueiredo-González, M.; Cancho-Grande, B.; Simal-Gándara, J. Effects on colour and phenolic composition of sugar concentration processes in dried-on- or dried-off-vine grapes and their aged or not natural sweet wines. Trends Food Sci. Technol. 2013, 31, 36–54. [Google Scholar] [CrossRef]
- Quijada-Morin, N.; Regueiro, J.; Simal-Gandara, J.; Tomas, E.; Rivas-Gonzalo, J.C.; Escribano-Bailon, M.T. Relationship between the sensory-determined astringency and the flavanolic composition of red wines. J. Agric. Food Chem. 2012, 60, 12355–12361. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Hydamaka, A.; Lowry, L. Comparison of antioxidant capacity and phenolic compounds of berries, chokecherry and seabuckthorn. Open Life Sci. 2009, 4, 499–506. [Google Scholar]
- Pieszka, M.; Tombarkiewicz, B.; Roman, A.; Migdal, W.; Niedziolka, J. Effect of bioactive substances found in rapeseed, raspberry and strawberry seed oils on blood lipid profile and selected parameters of oxidative status in rats. Environ. Toxicol. Pharmacol. 2013, 36, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, S.; Cimmino, A.; Raimo, M.; Salvatore, A.; Zappia, V.; Galletti, P. Effect of reddening-ripening on the antioxidant activity of polyphenol extracts from cv. “Annurca” apple fruits. J. Agric. Food Chem. 2007, 55, 9977–9985. [Google Scholar] [CrossRef]
- Stracke, B.A.; Rüfer, C.E.; Weibel, F.P.; Bub, A.; Watzl, B. Three-year comparison of the polyphenol contents and antioxidant capacities in organically and conventionally produced apples (Malus domestica bork. Cultivar “golden delicious”). J. Agric. Food Chem. 2009, 57, 4598–4605. [Google Scholar] [CrossRef]
- Reque, P.M.; Steffens, R.S.; Jablonski, A.; Flôres, S.H.; de O. Rios, A.; de Jong, E.V. Cold storage of blueberry (Vaccinium spp.) fruits and juice: Anthocyanin stability and antioxidant activity. J. Food Compos. Anal. 2014, 33, 111–116. [Google Scholar]
- Wang, S.Y.; Chen, H.; Camp, M.J.; Ehlenfeldt, M.K. Flavonoid constituents and their contribution to antioxidant activity in cultivars and hybrids of rabbiteye blueberry (Vaccinium ashei reade). Food Chem. 2012, 132, 855–864. [Google Scholar] [CrossRef]
- Kosinska-Cagnazzo, A.; Diering, S.; Prim, D.; Andlauer, W. Identification of bioaccessible and uptaken phenolic compounds from strawberry fruits in in vitro digestion/Caco-2 absorption model. Food Chem. 2015, 170, 288–294. [Google Scholar] [CrossRef] [PubMed]
- McKay, D.L.; Chen, C.Y.; Zampariello, C.A.; Blumberg, J.B. Flavonoids and phenolic acids from cranberry juice are bioavailable and bioactive in healthy older adults. Food Chem. 2015, 168, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Hyson, D.A. A comprehensive review of apples and apple components and their relationship to human health. Adv. Nutr. 2011, 2, 408–420. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.; Ismail, A.; Esa, N.M.; Pei, C.P. Transcriptomics expression analysis to unveil the molecular mechanisms underlying the cocoa polyphenol treatment in diet-induced obesity rats. Genomics 2015, 105, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Sanbongi, C.; Suzuki, N.; Sakane, T. Polyphenols in chocolate, which have antioxidant activity, modulate immune functions in humans in vitro. Cell. Immunol. 1997, 177, 129–136. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, B.; Cai, J.; Duan, C.-Q.; Reeves, M.J.; He, F. A Review of Polyphenolics in Oak Woods. Int. J. Mol. Sci. 2015, 16, 6978-7014. https://doi.org/10.3390/ijms16046978
Zhang B, Cai J, Duan C-Q, Reeves MJ, He F. A Review of Polyphenolics in Oak Woods. International Journal of Molecular Sciences. 2015; 16(4):6978-7014. https://doi.org/10.3390/ijms16046978
Chicago/Turabian StyleZhang, Bo, Jian Cai, Chang-Qing Duan, Malcolm J. Reeves, and Fei He. 2015. "A Review of Polyphenolics in Oak Woods" International Journal of Molecular Sciences 16, no. 4: 6978-7014. https://doi.org/10.3390/ijms16046978
APA StyleZhang, B., Cai, J., Duan, C.-Q., Reeves, M. J., & He, F. (2015). A Review of Polyphenolics in Oak Woods. International Journal of Molecular Sciences, 16(4), 6978-7014. https://doi.org/10.3390/ijms16046978