A Co-Culture Model of Fibroblasts and Adipose Tissue-Derived Stem Cells Reveals New Insights into Impaired Wound Healing After Radiotherapy
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
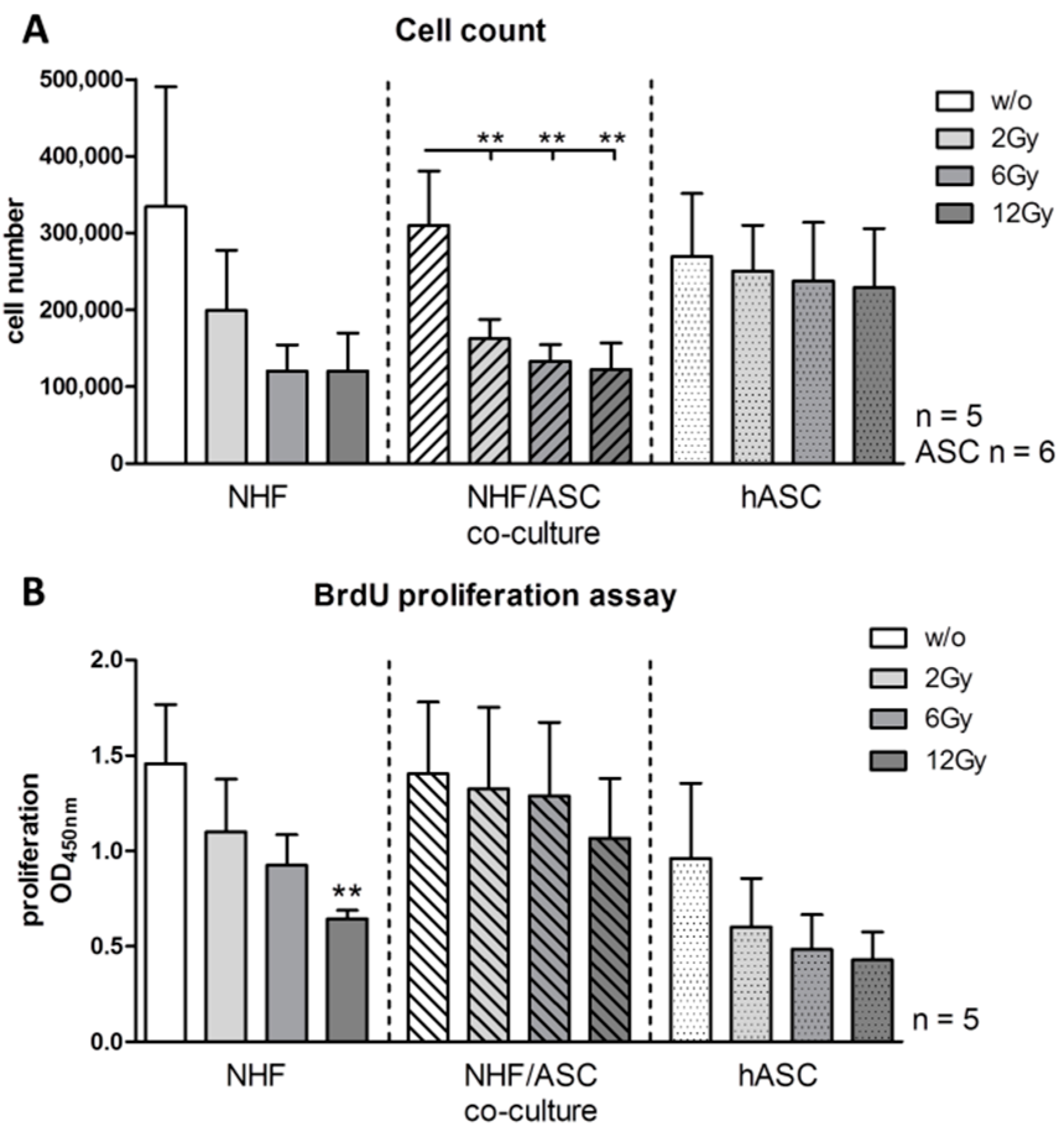
2.1.1. Effect of Irradiation on Cell Proliferation
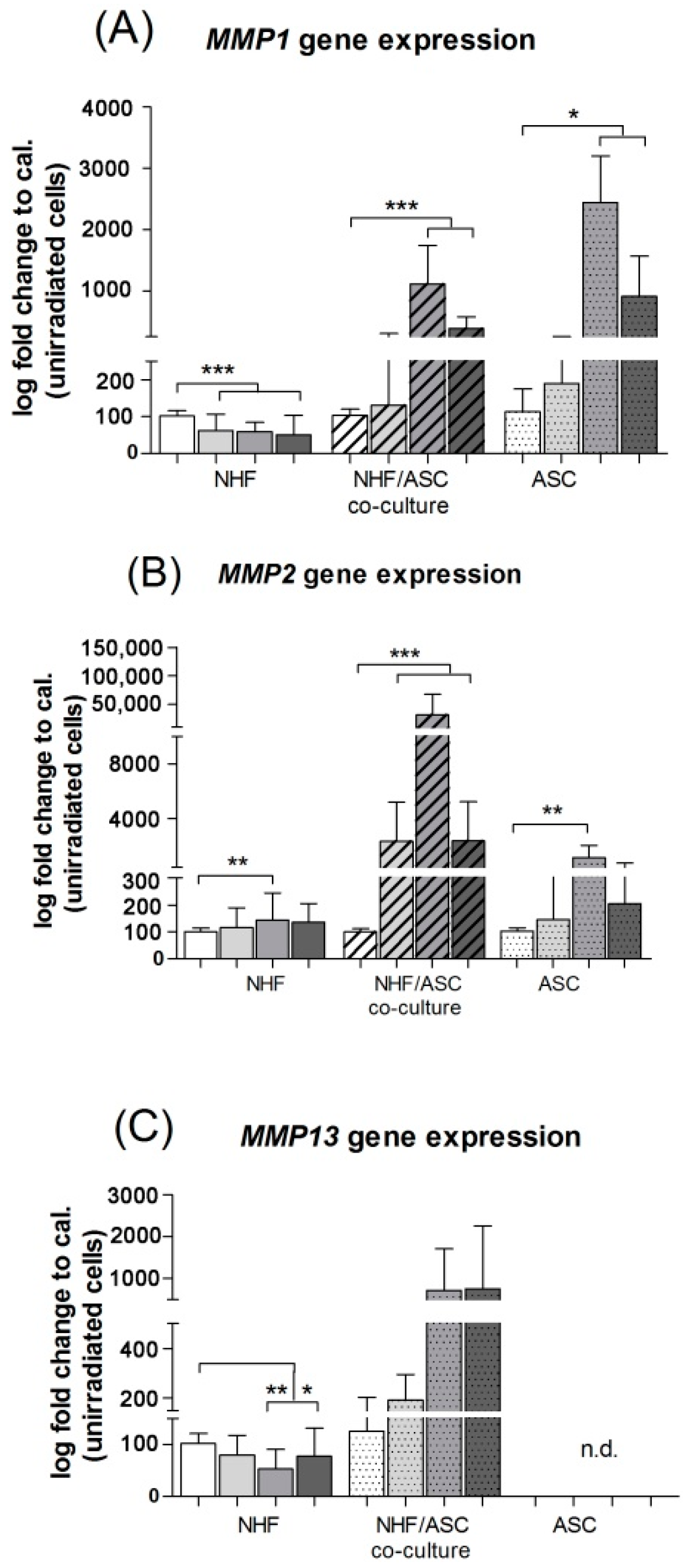
2.1.2. Gene Expression of MMP1 and MMP2 in Normal Human Fibroblasts (NHF), Adipose-Derived Stem Cells (ASC) and Co-Culture of NHF and ASC
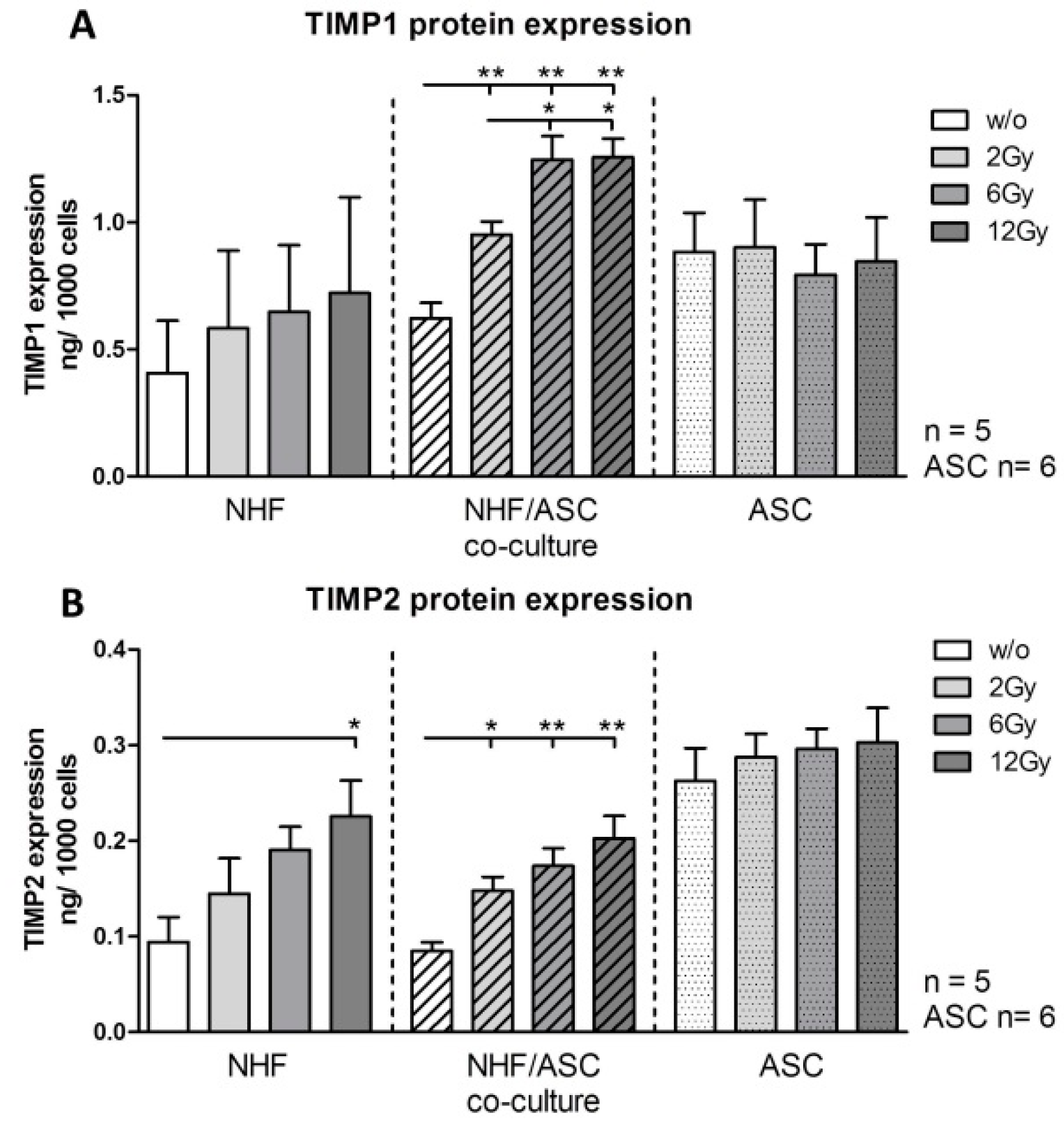
2.1.3. TIMP1 and TIMP2 Protein Expression in NHF, ASC and Co-Culture of NHF and ASC
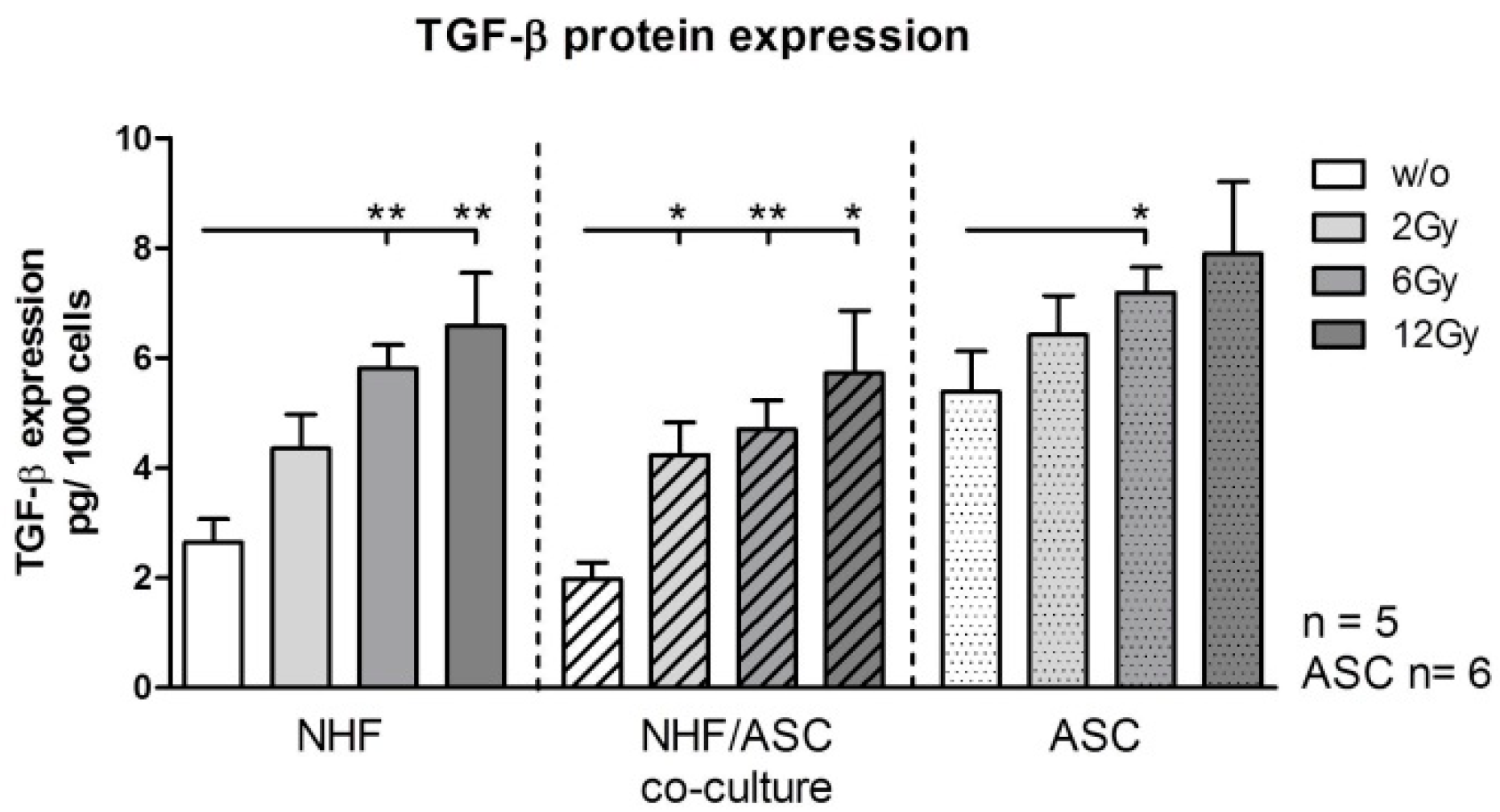
2.1.4. TGF-β Protein Expression in NHF, ASC and Co-Culture of NHF and ASC
2.2. Discussion
3. Experimental Section
3.1. Cell Culture
3.2. Co-Culture of NHFs and ASCs
3.3. Cell-Radiation
3.4. Cell-Harvesting
3.5. Cell Proliferation Assay
3.6. Enzyme-Linked Immunosorbent Assay (ELISA)
3.7. RNA Isolation and Reverse Transcriptase Quantitative Real-Time PCR
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Weber, R.S.; Berkey, B.A.; Forastiere, A.; Cooper, J.; Maor, M.; Goepfert, H.; Morrison, W.; Glisson, B.; Trotti, A.; Ridge, J.A.; et al. Outcome of salvage total laryngectomy following organ preservation therapy: The Radiation Therapy Oncology Group trial 91–11. Arch. Otolaryngol. Head Neck Surg. 2003, 129, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Marks, J.E.; Freeman, R.B.; Lee, F.; Ogura, J.H. Pharyngeal wall cancer: An analysis of treatment results complications and patterns of failure. Int. J. Radiat. Oncol. Biol. Phys. 1978, 4, 587–593. [Google Scholar] [CrossRef]
- Tang, Y.; Shen, Q.; Wang, Y.; Lu, K.; Peng, Y. A randomized prospective study of rehabilitation therapy in the treatment of radiation-induced dysphagia and trismus. Strahlenther. Onkol. 2011, 187, 39–44. [Google Scholar] [CrossRef] [PubMed]
- White, H.N.; Golden, B.; Sweeny, L.; Carroll, W.R.; Magnuson, J.S.; Rosenthal, E.L. Assessment and incidence of salivary leak following laryngectomy. Laryngoscope 2012, 122, 1796–1799. [Google Scholar] [CrossRef] [PubMed]
- Salibian, A.A.; Widgerow, A.D.; Abrouk, M.; Evans, G.R. Stem cells in plastic surgery: A review of current clinical and translational applications. Arch. Plast. Surg. 2013, 40, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Behm, B.; Babilas, P.; Landthaler, M.; Schreml, S. Cytokines, chemokines and growth factors in wound healing. J. Eur. Acad. Dermatol. Venereol. 2012, 26, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Schreml, S.; Szeimies, R.M.; Prantl, L.; Landthaler, M.; Babilas, P. Wound healing in the 21st century. J. Am. Acad. Dermatol. 2010, 63, 866–881. [Google Scholar] [CrossRef] [PubMed]
- Singer, A.J.; Clark, R.A. Cutaneous wound healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [PubMed]
- Dormand, E.L.; Banwell, P.E.; Goodacre, T.E. Radiotherapy and wound healing. Int. Wound J. 2005, 2, 112–127. [Google Scholar] [CrossRef] [PubMed]
- Haubner, F.; Ohmann, E.; Pohl, F.; Prantl, L.; Strutz, J.; Gassner, H.G. Effects of radiation on the expression of adhesion molecules and cytokines in a static model of human dermal microvascular endothelial cells. Clin. Hemorheol. Microcirc. 2013, 54, 371–379. [Google Scholar] [PubMed]
- Rehman, J.; Traktuev, D.; Li, J.; Merfeld-Clauss, S.; Temm-Grove, C.J.; Bovenkerk, J.E.; Pell, C.L.; Johnstone, B.H.; Considine, R.V.; March, K.L. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 2004, 109, 1292–1298. [Google Scholar] [CrossRef] [PubMed]
- Zellner, J.; Mueller, M.; Berner, A.; Dienstknecht, T.; Kujat, R.; Nerlich, M.; Hennemann, B.; Koller, M.; Prantl, L.; Angele, M.; et al. Role of mesenchymal stem cells in tissue engineering of meniscus. J. Biomed. Mater. Res. A 2010, 94, 1150–1161. [Google Scholar] [CrossRef] [PubMed]
- Rigotti, G.; Marchi, A.; Galie, M.; Baroni, G.; Benati, D.; Krampera, M.; Pasini, A.; Sbarbati, A. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: A healing process mediated by adipose-derived adult stem cells. Plast. Reconstr. Surg. 2007, 119, 1409–1422. [Google Scholar] [CrossRef] [PubMed]
- Haubner, F.; Leyh, M.; Ohmann, E.; Pohl, F.; Prantl, L.; Gassner, H.G. Effects of external radiation in a co-culture model of endothelial cells and adipose-derived stem cells. Radiat. Oncol. 2013, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.B.; Jorgensen, L.N.; Adawi, D.; Blomqvist, P.; Asklof, G.B.; Gottrup, F.; Jeppsson, B. The effect of preoperative radiotherapy on systemic collagen deposition and postoperative infective complications in rectal cancer patients. Dis. Colon Rectum 2005, 48, 1573–1580. [Google Scholar] [CrossRef] [PubMed]
- Medrado, A.P.; Soares, A.P.; Santos, E.T.; Reis, S.R.; Andrade, Z.A. Influence of laser photobiomodulation upon connective tissue remodeling during wound healing. J. Photochem. Photobiol. B 2008, 92, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Riedel, F.; Philipp, K.; Sadick, H.; Goessler, U.; Hormann, K.; Verse, T. Immunohistochemical analysis of radiation-induced non-healing dermal wounds of the head and neck. In Vivo 2005, 19, 343–350. [Google Scholar] [PubMed]
- Haubner, F.; Ohmann, E.; Pohl, F.; Strutz, J.; Gassner, H.G. Wound healing after radiation therapy: Review of the literature. Radiat. Oncol. 2012, 7, 162. [Google Scholar] [CrossRef] [PubMed]
- Horton, J.A.; Hudak, K.E.; Chung, E.J.; White, A.O.; Scroggins, B.T.; Burkeen, J.F.; Citrin, D.E. Mesenchymal stem cells inhibit cutaneous radiation-induced fibrosis by suppressing chronic inflammation. Stem Cells 2013, 31, 2231–2241. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Jin, S.Y.; Song, J.S.; Seo, K.K.; Cho, K.H. Paracrine effects of adipose-derived stem cells on keratinocytes and dermal fibroblasts. Ann. Dermatol. 2012, 24, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Baum, C.L.; Arpey, C.J. Normal cutaneous wound healing: Clinical correlation with cellular and molecular events. Dermatol. Surg. 2005, 31, 674–686. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Cheng, T.; Shi, C.; Lin, Y.; Ran, X. A study on the activity of fibroblast cells in connection with tissue recovery in the wounds of skin injury after whole-body irradiation. J. Radiat. Res. 2004, 45, 341–344. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.H.; Rudolph, R. Healing in the irradiated wound. Clin. Plast. Surg. 1990, 17, 503–508. [Google Scholar] [PubMed]
- Lee, E.Y.; Xia, Y.; Kim, W.S.; Kim, M.H.; Kim, T.H.; Kim, K.J.; Park, B.S.; Sung, J.H. Hypoxia-enhanced wound-healing function of adipose-derived stem cells: Increase in stem cell proliferation and up-regulation of VEGF and bFGF. Wound Repair Regen. 2009, 17, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.S.; Park, B.S.; Sung, J.H.; Yang, J.M.; Park, S.B.; Kwak, S.J.; Park, J.S. Wound healing effect of adipose-derived stem cells: A critical role of secretory factors on human dermal fibroblasts. J. Dermatol. Sci. 2007, 48, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Ra, J.C.; Shin, I.S.; Jang, Y.H.; An, H.Y.; Choi, J.S.; Kim, W.C.; Kim, Y.M. Systemic transplantation of human adipose tissue-derived mesenchymal stem cells for the regeneration of irradiation-induced salivary gland damage. PLoS ONE 2013, 8, e71167. [Google Scholar] [CrossRef] [PubMed]
- Haubner, F.; Gassner, H.G. Potential of adipose-derived stem cells concerning the treatment of wound healing complications after radiotherapy. HNO 2015, 63, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Dasu, M.R.; Barrow, R.E.; Spies, M.; Herndon, D.N. Matrix metalloproteinase expression in cytokine stimulated human dermal fibroblasts. Burns 2003, 29, 527–531. [Google Scholar] [CrossRef]
- Tang, C.H.; Chen, C.F.; Chen, W.M.; Fong, Y.C. IL-6 increases MMP-13 expression and motility in human chondrosarcoma cells. J. Biol. Chem. 2011, 286, 11056–11066. [Google Scholar] [CrossRef] [PubMed]
- Boothe, D.L.; Coplowitz, S.; Greenwood, E.; Barney, C.L.; Christos, P.J.; Parashar, B.; Nori, D.; Chao, K.S.; Wernicke, A.G. Transforming growth factor β-1 (TGF-β1) is a serum biomarker of radiation induced fibrosis in patients treated with intracavitary accelerated partial breast irradiation: Preliminary results of a prospective study. Int. J. Radiat. Oncol. Biol. Phys. 2013, 87, 1030–1036. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vujaskovic, Z.; Anscher, M.S.; Feng, Q.F.; Rabbani, Z.N.; Amin, K.; Samulski, T.S.; Dewhirst, M.W.; Haroon, Z.A. Radiation-induced hypoxia may perpetuate late normal tissue injury. Int. J. Radiat. Oncol. Biol. Phys. 2001, 50, 851–855. [Google Scholar] [CrossRef]
- Iyer, N.V.; Kotch, L.E.; Agani, F.; Leung, S.W.; Laughner, E.; Wenger, R.H.; Gassmann, M.; Gearhart, J.D.; Lawler, A.M.; Yu, A.Y.; et al. Cellular and developmental control of O2 homeostasis by hypoxia-inducible factor 1α. Genes Dev. 1998, 12, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Schreml, S.; Szeimies, R.M.; Prantl, L.; Karrer, S.; Landthaler, M.; Babilas, P. Oxygen in acute and chronic wound healing. Br. J. Dermatol. 2010, 163, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Feghali, C.A.; Bost, K.L.; Boulware, D.W.; Levy, L.S. Control of IL-6 expression and response in fibroblasts from patients with systemic sclerosis. Autoimmunity 1994, 17, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Jansen, E.D.; Davidson, J.M. Comparison of mouse matrix metalloproteinase 13 expression in free-electron laser and scalpel incisions during wound healing. J. Investig. Dermatol. 2003, 121, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Davis, G.E.; Pintar Allen, K.A.; Salazar, R.; Maxwell, S.A. Matrix metalloproteinase-1 and -9 activation by plasmin regulates a novel endothelial cell-mediated mechanism of collagen gel contraction and capillary tube regression in three-dimensional collagen matrices. J. Cell Sci. 2001, 114, 917–930. [Google Scholar] [PubMed]
- Xue, M.; Le, N.T.; Jackson, C.J. Targeting matrix metalloproteases to improve cutaneous wound healing. Expert Opin. Ther. Targets 2006, 10, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Goessler, U.R.; Bugert, P.; Kassner, S.; Stern-Straeter, J.; Bran, G.; Sadick, H.; Hormann, K.; Riedel, F. In vitro analysis of radiation-induced dermal wounds. Otolaryngol. Head Neck Surg. 2010, 142, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Jourdan, M.M.; Lopez, A.; Olasz, E.B.; Duncan, N.E.; Demara, M.; Kittipongdaja, W.; Fish, B.L.; Mader, M.; Schock, A.; Morrow, N.V.; et al. Laminin 332 deposition is diminished in irradiated skin in an animal model of combined radiation and wound skin injury. Radiat. Res. 2011, 176, 636–648. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Thiele, C. Factors associated with free flap complications after head and neck reconstruction and the molecular basis of fibrotic tissue rearrangement in preirradiated soft tissue. J. Oral Maxillofac. Surg. 2010, 68, 2169–2178. [Google Scholar] [CrossRef] [PubMed]
- Mueller, C.K.; Thorwarth, M.; Schultze-Mosgau, S. Late changes in cutaneous gene expression patterns after adjuvant treatment of oral squamous cell carcinoma (OSCC) by radiation therapy. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 109, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Lozito, T.P.; Jackson, W.M.; Nesti, L.J.; Tuan, R.S. Human mesenchymal stem cells generate a distinct pericellular zone of MMP activities via binding of MMPs and secretion of high levels of TIMPs. Matrix Biol. 2014, 34, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Meier, R.J.; Schreml, S.; Wang, X.D.; Landthaler, M.; Babilas, P.; Wolfbeis, O.S. Simultaneous photographing of oxygen and pH in vivo using sensor films. Angew. Chem. 2011, 50, 10893–10896. [Google Scholar] [CrossRef] [PubMed]
- Schreml, S.; Meier, R.J.; Wolfbeis, O.S.; Landthaler, M.; Szeimies, R.M.; Babilas, P. 2D luminescence imaging of pH in vivo. Proc. Natl. Acad. Sci. USA 2011, 108, 2432–2437. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liu, T.; Song, K.; Fan, X.; Ma, X.; Cui, Z. Ex vivo expansion of adipose tissue-derived stem cells in spinner flasks. Biotechnol. J. 2009, 4, 1198–1209. [Google Scholar] [CrossRef] [PubMed]
- Gehmert, S.; Hidayat, M.; Sultan, M.; Berner, A.; Klein, S.; Zellner, J.; Muller, M.; Prantl, L. Angiogenesis: The role of PDGF-BB on adipose-tissue derived stem cells (ASCs). Clin. Hemorheol. Microcirc. 2011, 48, 5–13. [Google Scholar] [PubMed]
- Dicker, A.; Le Blanc, K.; Astrom, G.; van Harmelen, V.; Gotherstrom, C.; Blomqvist, L.; Arner, P.; Ryden, M. Functional studies of mesenchymal stem cells derived from adult human adipose tissue. Exp. Cell Res. 2005, 308, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Pohl, F.; Hassel, S.; Nohe, A.; Flentje, M.; Knaus, P.; Sebald, W.; Koelbl, O. Radiation-induced suppression of the Bmp2 signal transduction pathway in the pluripotent mesenchymal cell line C2C12: An in vitro model for prevention of heterotopic ossification by radiotherapy. Radiat. Res. 2003, 159, 345–350. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haubner, F.; Muschter, D.; Pohl, F.; Schreml, S.; Prantl, L.; Gassner, H.G. A Co-Culture Model of Fibroblasts and Adipose Tissue-Derived Stem Cells Reveals New Insights into Impaired Wound Healing After Radiotherapy. Int. J. Mol. Sci. 2015, 16, 25947-25958. https://doi.org/10.3390/ijms161125935
Haubner F, Muschter D, Pohl F, Schreml S, Prantl L, Gassner HG. A Co-Culture Model of Fibroblasts and Adipose Tissue-Derived Stem Cells Reveals New Insights into Impaired Wound Healing After Radiotherapy. International Journal of Molecular Sciences. 2015; 16(11):25947-25958. https://doi.org/10.3390/ijms161125935
Chicago/Turabian StyleHaubner, Frank, Dominique Muschter, Fabian Pohl, Stephan Schreml, Lukas Prantl, and Holger G. Gassner. 2015. "A Co-Culture Model of Fibroblasts and Adipose Tissue-Derived Stem Cells Reveals New Insights into Impaired Wound Healing After Radiotherapy" International Journal of Molecular Sciences 16, no. 11: 25947-25958. https://doi.org/10.3390/ijms161125935
APA StyleHaubner, F., Muschter, D., Pohl, F., Schreml, S., Prantl, L., & Gassner, H. G. (2015). A Co-Culture Model of Fibroblasts and Adipose Tissue-Derived Stem Cells Reveals New Insights into Impaired Wound Healing After Radiotherapy. International Journal of Molecular Sciences, 16(11), 25947-25958. https://doi.org/10.3390/ijms161125935






