Methylphenidate Ameliorates Depressive Comorbidity in ADHD Children without any Modification on Differences in Serum Melatonin Concentration between ADHD Subtypes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Melatonin Serum Concentration by Attention-Deficit/Hyperactivity Disorder (ADHD) Subtypes and Subgroups
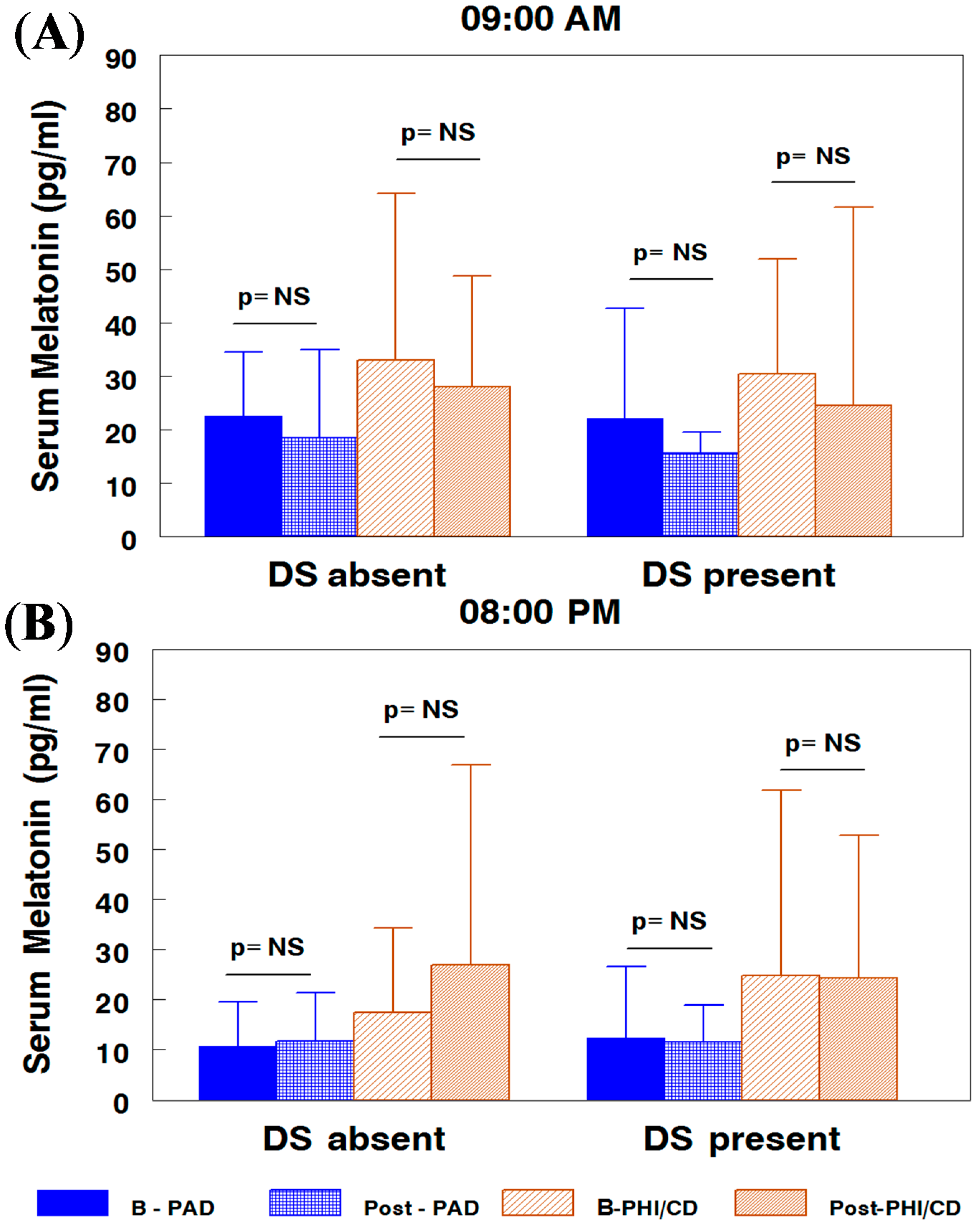
2.2. Nocturnal Excretion of 6-Sulphatoxy-melatonin by ADHD Subtypes

| Total CDI | Time | PAD (n = 37) | PHI/CD (n = 99) | Total (n = 136) | z | p | ||
|---|---|---|---|---|---|---|---|---|
| <18 (n = 104) | Baseline Post-PRMPH | 10.18 ± 4.74 | 10.48 ± 4.21 | 10.39 ± 4.34 | −1.743 | 0.081 | ||
| 9.47 ± 5.08 | 10.97 ± 6.34 | 10.45 ± 5.93 | ||||||
| 9.76 ± 4.49 | 10.65 ± 5.05 | |||||||
| z | p | z | p | |||||
| −2.09 | 0.037 | −0.84 | 0.40 | |||||
| >17 (n = 32) | Baseline Post-PRMPH | 22.40 ± 8.73 | 21.53 ± 2.59 | 21.71 ± 4.31 | −3.301 | 0.001 | ||
| 20.50 ± 8.96 | 14.86 ± 4.29 | 16.11 ± 5.84 | ||||||
| 22.00 ± 6.99 | 18.70 ± 4.74 | |||||||
| z | p | z | p | |||||
| −0.921 | 0.357 | −3.30 | 0.001 | |||||
| Total Simple (n = 136) | Baseline Post-PRMPH | 12.44 ± 7.30 | 13.44 ± 6.24 | 13.16 ± 6.52 | −3.596 | 0.0001 | ||
| 11.11 ± 6.12 | 12.33 ± 7.80 | 12.25 ± 6.19 | ||||||
| 11.89 ± 6.80 | 12.97 ± 6.06 | |||||||
| z | p | z | p | |||||
| −1.93 | 0.054 | −3.48 | 0.001 | |||||
3. Experimental Section
3.1. Sample
3.2. Clinical Method
| Sex | Depressive Symptoms | ADHD Subtype | Total (%) | |
|---|---|---|---|---|
| PAD | PHI/CD | |||
| Boys | No | 24 (77.42) | 61 (80.26) | 85 (79.43) |
| Yes | 7 (22.58) | 15 (19.74) | 22 (20.56) | |
| Total | 31 (28.97) | 76 (71.03) | 107 (78.67) | |
| Girls | No | 5 (83.33) | 14 (60.87) | 19 (65.52) |
| Yes | 1 (16.67) | 9 (39.13) | 10 (34.48) | |
| Total | 6 (20.69) | 23 (79.31) | 29 (21.33) | |
| Total | No | 29 (78.38) | 75 (75.75) | 104 (76.47) |
| Yes | 8 (21.62) | 24 (24.24) | 32 (23.53) | |
| Total | 37 (21.01) | 99 (72.99) | 136 (100) | |
3.2.1. Treatment
3.2.2. Measurements
3.3. Analytical Method
3.4. Statistical Method
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Caylak, E. Biochemical and genetic analyses of childhood attention deficit/hyperactivity disorder. Am. J. Med. Genet. 2012, 159, 613–627. [Google Scholar]
- Silva, D.; Colvin, L.; Hagemann, E.; Bower, C. Environmental risk factors by gender associated with attention-deficit/hyperactivity disorder. Pediatrics 2014, 133, e14–e22. [Google Scholar]
- Acosta, M.T.; Castellanos, F.X.; Bolton, K.L.; Balog, J.Z.; Eagen, P.; Nee, L.; Jones, J.; Palacio, L.; Sarampote, C.; Russell, H.F.; et al. Latent class subtyping of attention-deficit/hyperactivity disorder and comorbid conditions. J. Am. Acad. Child. Adolesc. Psychiatry 2008, 47, 797–807. [Google Scholar]
- Barkley, R.A. Behavioral inhibition, sustained attention, and executive functions: Constructing a unifying theory of adhd. Psychol. Bull. 1997, 121, 65–94. [Google Scholar]
- Nigg, J.T. Neuropsychologic theory and findings in attention-deficit/hyperactivity disorder: The state of the field and salient challenges for the coming decade. Biol. Psychiatry 2005, 57, 1424–1435. [Google Scholar]
- Wiltschko, A.B.; Pettibone, J.R.; Berke, J.D. Opposite effects of stimulant and antipsychotic drugs on striatal fast-spiking interneurons. Neuropsychopharmacology 2010, 35, 1261–1270. [Google Scholar]
- Nigg, J.T.; Casey, B.J. An integrative theory of attention-deficit/ hyperactivity disorder based on the cognitive and affective neurosciences. Dev. Psychopathol. 2005, 17, 785–806. [Google Scholar]
- Van, E.H.; Heslenfeld, D.J.; Zwiers, M.P.; Buitelaar, J.K.; Oosterlaan, J. Diffusion tensor imaging in attention deficit/hyperactivity disorder: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2012, 36, 1093–1106. [Google Scholar]
- Gupta, R.; Kar, B.R. Development of attentional processes in adhd and normal children. Prog. Brain Res. 2009, 176, 259–276. [Google Scholar]
- Matthews, M.; Nigg, J.T.; Fair, D.A. Attention deficit hyperactivity disorder. Curr. Top. Behav. Neurosci. 2014, 16, 235–266. [Google Scholar]
- Hardeland, R.; Madrid, J.A.; Tan, D.X.; Reiter, R.J. Melatonin, the circadian multioscillator system and health: The need for detailed analyses of peripheral melatonin signaling. J. Pineal Res. 2012, 52, 139–166. [Google Scholar]
- Alexiuk, N.A.; Vriend, J. Melatonin: Effects on dopaminergic and serotonergic neurons of the caudate nucleus of the striatum of male syrian hamsters. J. Neural Transm. 2007, 114, 549–554. [Google Scholar]
- Uberos, J.; Augustin-Morales, M.C.; Carballo, A.M.; Florido, J.; Narbona, E.; Munoz-Hoyos, A. Normalization of the sleep-wake pattern and melatonin and 6-sulphatoxy-melatonin levels after a therapeutic trial with melatonin in children with severe epilepsy. J. Pineal Res. 2011, 50, 192–196. [Google Scholar]
- Stehle, J.H.; Saade, A.; Rawashdeh, O.; Ackermann, K.; Jilg, A.; Sebestany, T.S.; Maronde, E. A survey of molecular details in the human pineal gland in the light of phylogeny, structure, function and chronobiological diseases. J. Pineal Res. 2011, 51, 17–43. [Google Scholar]
- Walters, A.S.; Silvestri, R.; Zucconi, M.; Chandrashekariah, R.; Konofal, E. Review of the possible relationship and hypothetical links between attention deficit hyperactivity disorder (ADHD) and the simple sleep related movement disorders, parasomnias, hypersomnias, and circadian rhythm disorders. J. Clin. Sleep Med. 2008, 4, 591–600. [Google Scholar]
- Owens, J.A. The adhd and sleep conundrum: A review. J. Dev. Behav. Pediatr. 2005, 26, 312–322. [Google Scholar]
- De Berardis, D.; Marini, S.; Fornaro, M.; Srinivasan, V.; Iasevoli, F.; Tomasetti, C.; Valchera, A.; Perna, G.; Quera-Salva, M.A.; Martinotti, G.; et al. The melatonergic system in mood and anxiety disorders and the role of agomelatine: Implications for clinical practice. Int. J. Mol. Sci. 2013, 14, 12458–12483. [Google Scholar]
- Lanfumey, L.; Mongeau, R.; Hamon, M. Biological rhythms and melatonin in mood disorders and their treatments. Pharmacol. Ther. 2013, 138, 176–184. [Google Scholar]
- Connor, D.F.; Ford, J.D. Comorbid symptom severity in attention-deficit/hyperactivity disorder: A clinical study. J. Clin. Psychiatry. 2012, 73, 711–717. [Google Scholar]
- Molina-Carballo, A.; Justicia-Martinez, F.; Moreno-Madrid, F.; Cubero-Millan, I.; Machado-Casas, I.; Moreno-Garcia, L.; Leon, J.; Luna-Del-Castillo, J.-d.-D.; Uberos, J.; Munoz-Hoyos, A. Differential responses of two related neurosteroids to methylphenidate based on adhd subtype and the presence of depressive symptomatology. Psychopharmacology 2014, 231, 3635–3645. [Google Scholar]
- Molina-Carballo, A.; Naranjo-Gomez, A.; Uberos, J.; Justicia-Martinez, F.; Ruiz-Ramos, M.-J.; Cubero-Millan, I.; Contreras-Chova, F.; Augustin-Morales, M.-D.-C.; Khaldy-Belkadi, H.; Munoz-Hoyos, A. Methylphenidate effects on blood serotonin and melatonin levels may help to synchronise biological rhythms in children with adhd. J. Psychiatr. Res. 2013, 47, 377–383. [Google Scholar]
- Agil, A.; Rosado, I.; Ruiz, R.; Figueroa, A.; Zen, N.; Fern ndez-V zquez, G. Melatonin improves glucose homeostasis in young zucker diabetic fatty rats. J. Pineal Res. 2012, 52, 203–210. [Google Scholar]
- Carrillo-Vico, A.; Guerrero, J.M.; Lardone, P.J.; Reiter, R.J. A review of the multiple actions of melatonin on the immune system. Endocrine 2005, 27, 189–200. [Google Scholar]
- Molina-Carballo, A.; Munoz-Hoyos, A.; Sanchez-Forte, M.; Uberos-Fernandez, I.; Moreno-Madrid, F.; Acuna-Castroviejo, D. Melatonin increases following convulsive seizures may be related to its anticonvulsant properties at physiological concentrations. Neuropediatrics 2007, 38, 122–125. [Google Scholar]
- Muñoz-Hoyos, A.; Molina-Carballo, A.; Augustin-Morales, M.D.C.; Contreras-Chova, F.; Naranjo-Gómez, A.; Justicia-Martínez, F.; Uberos, J. Psychosocial dwarfism: Psychopathological aspects and putative neuroendocrine markers. Psychiatry Res. 2011, 188, 96–101. [Google Scholar]
- Paclt, I.; Ptacek, R.; Kuzelova, H.; Cermakova, N.; Trefilova, A.; Kollarova, P.; Calkova, T.; Csemy, L.; Cihal, L. Circadian rhythms of saliva melatonin in ADHD, anxious and normal children. Neuro Endocrinol. Lett. 2011, 32, 790–798. [Google Scholar]
- Sánchez-Forte, M.; MorenoMadrid, F.; Munoz-Hoyos, A.; Molina-Carballo, A.; Acuña-Castroviejo, D.; Molina-Font, J.A. The effect of melatonin as an anti-convulsant and neurone protector. Rev. Neurol. 1997, 25, 1229–1234. [Google Scholar]
- Muñoz-Hoyos, A.; Molina-Carballo, A.; Uberos, J.; Contreras-Chova, F.; Augustin-Morales, M.C.; Ruiz-Alba, M.; Galdó-Muñoz, G. Serum melatonin concentration in the child with non-organic failure to thrive: Comparison with other types of stress. J. Biol. Regul. Homeost. Agents 2009, 23, 15–22. [Google Scholar]
- Charalampopoulos, I.; Remboutsika, E.; Margioris, A.N.; Gravanis, A. Neurosteroids as modulators of neurogenesis and neuronal survival. Trends Endocrinol. Metab. 2008, 19, 300–307. [Google Scholar]
- Presman, D.M.; Levi, V.; Pignataro, O.P.; Pecci, A. Melatonin inhibits glucocorticoid-dependent GR-TIF2 interaction in newborn hamster kidney (BHK) cells. Mol. Cell. Endocrinol. 2012, 349, 214–221. [Google Scholar]
- McNelis, J.C.; Manolopoulos, K.N.; Gathercole, L.L.; Bujalska, I.J.; Stewart, P.M.; Tomlinson, J.W.; Arlt, W. Dehydroepiandrosterone exerts antiglucocorticoid action on human preadipocyte proliferation, differentiation, and glucose uptake. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E1134–E1144. [Google Scholar]
- Gallagher, P.; Watson, S.; Smith, M.S.; Young, A.H.; Ferrier, I.N. Plasma cortisol-dehydroepiandrosterone (DHEA) ratios in schizophrenia and bipolar disorder. Schizophr. Res. 2007, 90, 258–265. [Google Scholar]
- Sung, V.; Hiscock, H.; Sciberras, E.; Efron, D. Sleep problems in children with attention-deficit/hyperactivity disorder: Prevalence and the effect on the child and family. Arch. Pediatr. Adolesc. Med. 2008, 162, 336–342. [Google Scholar]
- Mayes, S.D.; Calhoun, S.L.; Bixler, E.O.; Vgontzas, A.N.; Mahr, F.; Hillwig-Garcia, J.; Elamir, B.; Edhere-Ekezie, L.; Parvin, M. Adhd subtypes and comorbid anxiety, depression, and oppositional-defiant disorder: Differences in sleep problems. J. Pediatr. Psychol. 2009, 34, 328–337. [Google Scholar]
- Efron, D.; Lycett, K.; Sciberras, E. Use of sleep medication in children with ADHD. Sleep Med. 2014, 15, 472–475. [Google Scholar]
- Cortese, S.; Brown, T.E.; Corkum, P.; Gruber, R.; OʼBrien, L.M.; Stein, M.; Weiss, M.; Owens, J. Assessment and management of sleep problems in youths with attention-deficit/hyperactivity disorder. J. Am. Acad. Child Adolesc. Psychiatry 2013, 52, 784–796. [Google Scholar]
- Hardeland, R.; Tan, D.X.; Reiter, R.J. Kynuramines, metabolites of melatonin and other indoles: The resurrection of an almost forgotten class of biogenic amines. J. Pineal Res. 2009, 47, 109–126. [Google Scholar]
- Acuña-Castroviejo, D.; López, L.C.; Escames, G.; López, A.; García, J.A.; Reiter, R.J. Melatonin-mitochondria interplay in health and disease. Curr. Top. Med. Chem. 2011, 11, 221–240. [Google Scholar]
- Manev, H.; Uz, T. Dosing time-dependent actions of psychostimulants. Int. Rev. Neurobiol. 2009, 88, 25–41. [Google Scholar]
- Kita, T.; Miyazaki, I.; Asanuma, M.; Takeshima, M.; Wagner, G.C. Dopamine-induced behavioral changes and oxidative stress in methamphetamine-induced neurotoxicity. Int. Rev. Neurobiol. 2009, 88, 43–64. [Google Scholar]
- Ares-Santos, S.; Granado, N.; Moratalla, R. The role of dopamine receptors in the neurotoxicity of methamphetamine. J. Int. Med. 2013, 273, 437–453. [Google Scholar]
- Imam, S.Z.; El-Yazal, J.; Newport, G.D.; Itzhak, Y.; Cadet, J.L.; Slikker, W., Jr.; Ali, S.F. Methamphetamine-induced dopaminergic neurotoxicity: Role of peroxynitrite and neuroprotective role of antioxidants and peroxynitrite decomposition catalysts. Ann. N. Y. Acad. Sci. 2001, 939, 366–380. [Google Scholar]
- Jumnongprakhon, P.; Govitrapong, P.; Tocharus, C.; Tungkum, W.; Tocharus, J. Protective effect of melatonin on methamphetamine-induced apoptosis in glioma cell line. Neurotox. Res. 2014, 25, 286–294. [Google Scholar]
- Brookes, K.J.; Hawi, Z.; Kirley, A.; Barry, E.; Gill, M.; Kent, L. Association of the steroid sulfatase (STS) gene with attention deficit hyperactivity disorder. Am. J. Med. Genet. 2008, 147, 1531–1535. [Google Scholar]
- Trent, S.; Dean, R.; Veit, B.; Cassano, T.; Bedse, G.; Ojarikre, O.A.; Humby, T.; Davies, W. Biological mechanisms associated with increased perseveration and hyperactivity in a genetic mouse model of neurodevelopmental disorder. Psychoneuroendocrinology 2013, 38, 1370–1380. [Google Scholar]
- Strous, R.D.; Spivak, B.; Yoran-Hegesh, R.; Maayan, R.; Averbuch, E.; Kotler, M.; Mester, R.; Weizman, A. Analysis of neurosteroid levels in attention deficit hyperactivity disorder. Int. J. Neuropsychopharmacol. 2001, 4, 259–264. [Google Scholar]
- Wang, L.J.; Hsiao, C.C.; Huang, Y.S.; Chiang, Y.L.; Ree, S.C.; Chen, Y.C.; Wu, Y.W.; Wu, C.C.; Shang, Z.Y.; Chen, C.K. Association of salivary dehydroepiandrosterone levels and symptoms in patients with attention deficit hyperactivity disorder during six months of treatment with methylphenidate. Psychoneuroendocrinology 2011, 36, 1209–1216. [Google Scholar]
- Wang, L.J.; Huang, Y.S.; Hsiao, C.C.; Chiang, Y.L.; Wu, C.C.; Shang, Z.Y.; Chen, C.K. Salivary dehydroepiandrosterone, but not cortisol, is associated with attention deficit hyperactivity disorder. World J. Biol. Psychiatry 2011, 12, 99–109. [Google Scholar]
- Moreno, M.; Economidou, D.; Mar, A.C.; Lopez-Granero, C.; Caprioli, D.; Theobald, D.E.; Fernando, A.; Newman, A.H.; Robbins, T.W.; Dalley, J.W. Divergent effects of D2/3 receptor activation in the nucleus accumbens core and shell on impulsivity and locomotor activity in high and low impulsive rats. Psychopharmacology 2013, 228, 19–30. [Google Scholar]
- Muñoz-Hoyos, A.; Bonillo-Perales, A.; Avila-Villegas, R.; Gonzalez-Ripoll, M.; Uberos, J.; Florido-Navío, J.; Molina-Carballo, A. Melatonin levels during the first week of life and their relation with the antioxidant response in the perinatal period. Neonatology 2007, 92, 209–216. [Google Scholar]
- Esposito, E.; Cuzzocrea, S. Antiinflammatory activity of melatonin in central nervous system. Curr. Neuropharmacol. 2010, 8, 228–242. [Google Scholar]
- Wang, X. The antiapoptotic activity of melatonin in neurodegenerative diseases. CNS Neurosci. Ther. 2009, 15, 345–357. [Google Scholar]
- Cardinali, D.P.; Srinivasan, V.; Brzezinski, A.; Brown, G.M. Melatonin and its analogs in insomnia and depression. J. Pineal Res. 2012, 52, 365–375. [Google Scholar]
- Maldonado, M.D.; Reiter, R.J.; Perez-San-Gregorio, M.A. Melatonin as a potential therapeutic agent in psychiatric illness. Hum. Psychopharmacol. 2009, 24, 391–400. [Google Scholar]
- Sarlak, G.; Jenwitheesuk, A.; Chetsawang, B.; Govitrapong, P. Effects of melatonin on nervous system aging: Neurogenesis and neurodegeneration. J. Pharmacol. Sci. 2013, 123, 9–24. [Google Scholar]
- Strickland, A.D. Prevention of cerebral palsy, autism spectrum disorder, and attention deficit-hyperactivity disorder. Med. Hypotheses 2014, 82, 522–528. [Google Scholar]
- Reiter, R.J.; Manchester, L.C.; Tan, D.X. Neurotoxins: Free radical mechanisms and melatonin protection. Curr. Neuropharmacol. 2010, 8, 194–210. [Google Scholar]
- McLoughlin, G.; Palmer, J.A.; Rijsdijk, F.; Makeig, S. Genetic overlap between evoked frontocentral theta-band phase variability, reaction time variability, and attention-deficit/hyperactivity disorder symptoms in a twin study. Biol. Psychiatry 2014, 75, 238–247. [Google Scholar]
- López-Ibor Aliño, J. J.; Miyar, M.V. DSM-IV-TR. Manual diagnóstico y estadístico de los trastornos mentales; American Psychiatric Association: Barcelona, Spain, 2002; Volume 1, pp. 1–1049. [Google Scholar]
- Sánchez, C.R.; Díaz, F.; Ramos, C. Trastorno por dé‚ficit de atención/hiperactividad en la adolescencia: Baremación de la escala EDAH (a). Rev. Neurol. 2010, 51, 337–346. [Google Scholar]
- Zambrano-Sánchez, E.; Martínez-Cortés, J.A.; del Río-Carlos, Y.; Martínez-Wbaldo, M.D.; Poblano, A. Identification of attention-deficit-hyperactivity disorder and conduct disorder in mexican children by the scale of evaluation of deficit of attention and hyperactivity scale. Psychiatry Res. 2011, 187, 437–440. [Google Scholar]
- Farré-Riba, A.; Narbona, J. Escalas de conners en la evaluación del trastorno por d‚éficit de atención con hiperactividad: Nuevo estudio factorial en niños españoles. Rev. Neurol. 1997, 25, 200–204. [Google Scholar]
- Brickenkamp, R. D2, Test de Atención. Adaptación Española; TEA ediciones: Madrid, Spain, 1997. [Google Scholar]
- Kovacs, M. Manual for the Children's Depression Inventory; Multi-Health Systems.: North Tonawanda, NJ, USA, 1992. [Google Scholar]
- Kaufman, A.S.; Kaufman, N.D. Kbit. In Test Breve de Inteligencia de Kaufmann; TEA Ediciones: Madrid, Spain, 1996. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cubero-Millán, I.; Molina-Carballo, A.; Machado-Casas, I.; Fernández-López, L.; Martínez-Serrano, S.; Tortosa-Pinto, P.; Ruiz-López, A.; Luna-del-Castillo, J.-d.-D.; Uberos, J.; Muñoz-Hoyos, A. Methylphenidate Ameliorates Depressive Comorbidity in ADHD Children without any Modification on Differences in Serum Melatonin Concentration between ADHD Subtypes. Int. J. Mol. Sci. 2014, 15, 17115-17129. https://doi.org/10.3390/ijms150917115
Cubero-Millán I, Molina-Carballo A, Machado-Casas I, Fernández-López L, Martínez-Serrano S, Tortosa-Pinto P, Ruiz-López A, Luna-del-Castillo J-d-D, Uberos J, Muñoz-Hoyos A. Methylphenidate Ameliorates Depressive Comorbidity in ADHD Children without any Modification on Differences in Serum Melatonin Concentration between ADHD Subtypes. International Journal of Molecular Sciences. 2014; 15(9):17115-17129. https://doi.org/10.3390/ijms150917115
Chicago/Turabian StyleCubero-Millán, Isabel, Antonio Molina-Carballo, Irene Machado-Casas, Luisa Fernández-López, Sylvia Martínez-Serrano, Pilar Tortosa-Pinto, Aida Ruiz-López, Juan-de-Dios Luna-del-Castillo, José Uberos, and Antonio Muñoz-Hoyos. 2014. "Methylphenidate Ameliorates Depressive Comorbidity in ADHD Children without any Modification on Differences in Serum Melatonin Concentration between ADHD Subtypes" International Journal of Molecular Sciences 15, no. 9: 17115-17129. https://doi.org/10.3390/ijms150917115
APA StyleCubero-Millán, I., Molina-Carballo, A., Machado-Casas, I., Fernández-López, L., Martínez-Serrano, S., Tortosa-Pinto, P., Ruiz-López, A., Luna-del-Castillo, J.-d.-D., Uberos, J., & Muñoz-Hoyos, A. (2014). Methylphenidate Ameliorates Depressive Comorbidity in ADHD Children without any Modification on Differences in Serum Melatonin Concentration between ADHD Subtypes. International Journal of Molecular Sciences, 15(9), 17115-17129. https://doi.org/10.3390/ijms150917115




