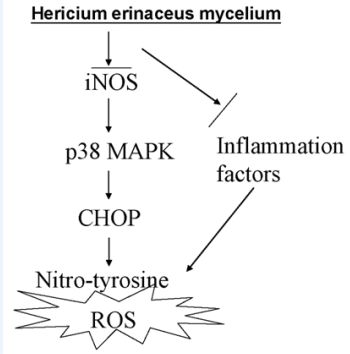
Protective Effects of Hericium erinaceus Mycelium and Its Isolated Erinacine A against Ischemia-Injury-Induced Neuronal Cell Death via the Inhibition of iNOS/p38 MAPK and Nitrotyrosine
Abstract
:1. Introduction
2. Results
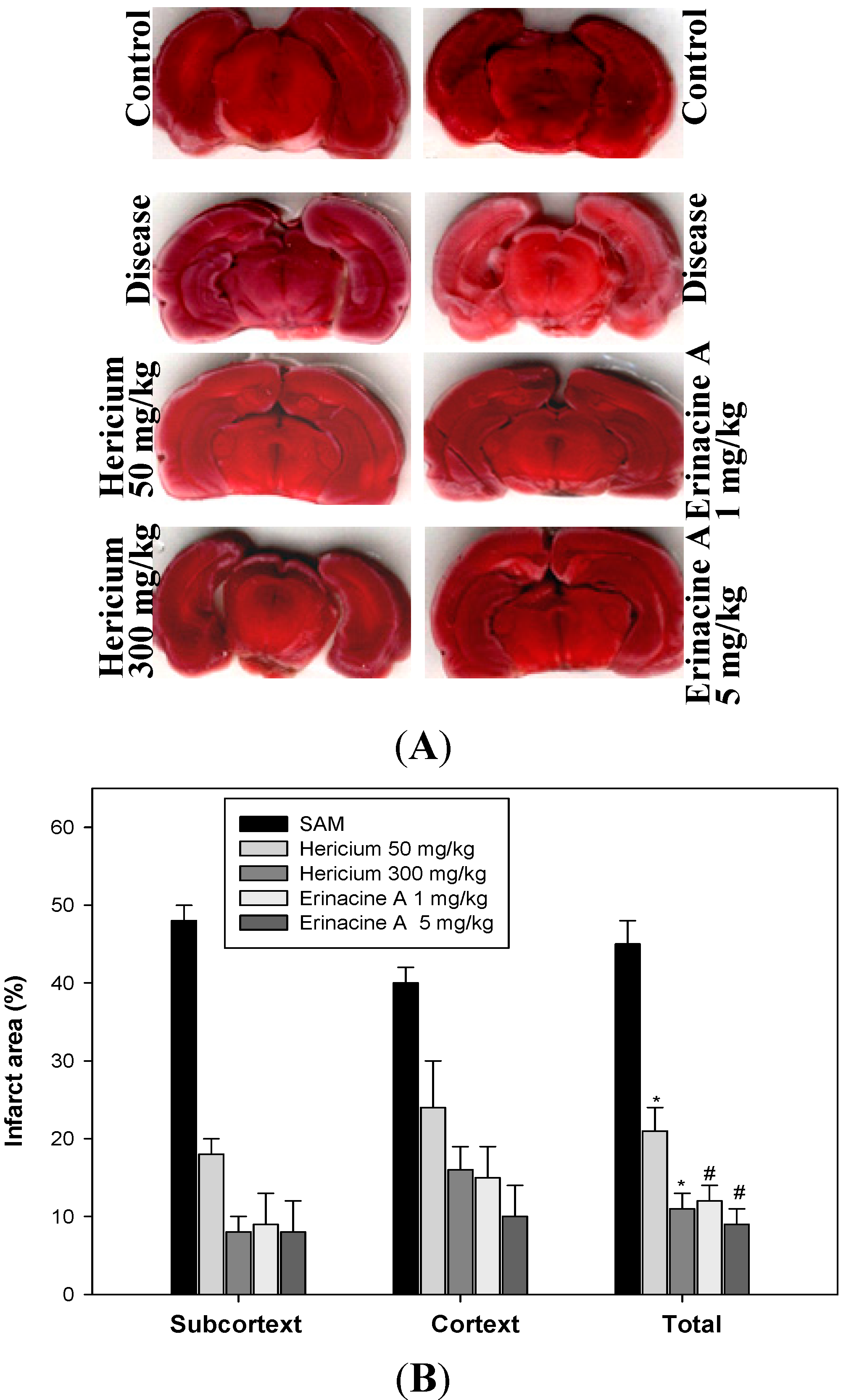
2.1. Effects of Hericium erinaceus Mycelium on the Suppression of Brain Damage in Ischemic Rats
2.2. Hericium erinaceus Mycelium Inhibits Neuronal Cell Death in a Transient Stroke Animal Model
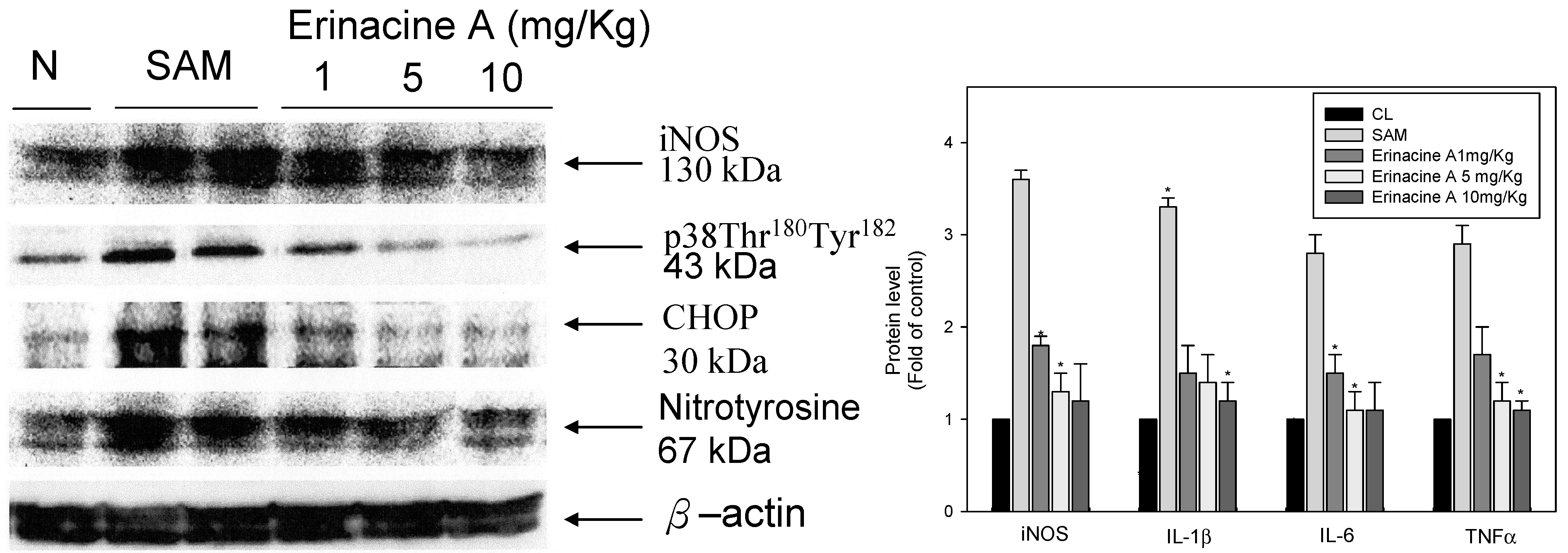
2.3. Effects of H. erinaceus Mycelium on Serum Protein ROS Oxidants and Acute Inflammation Cytokines
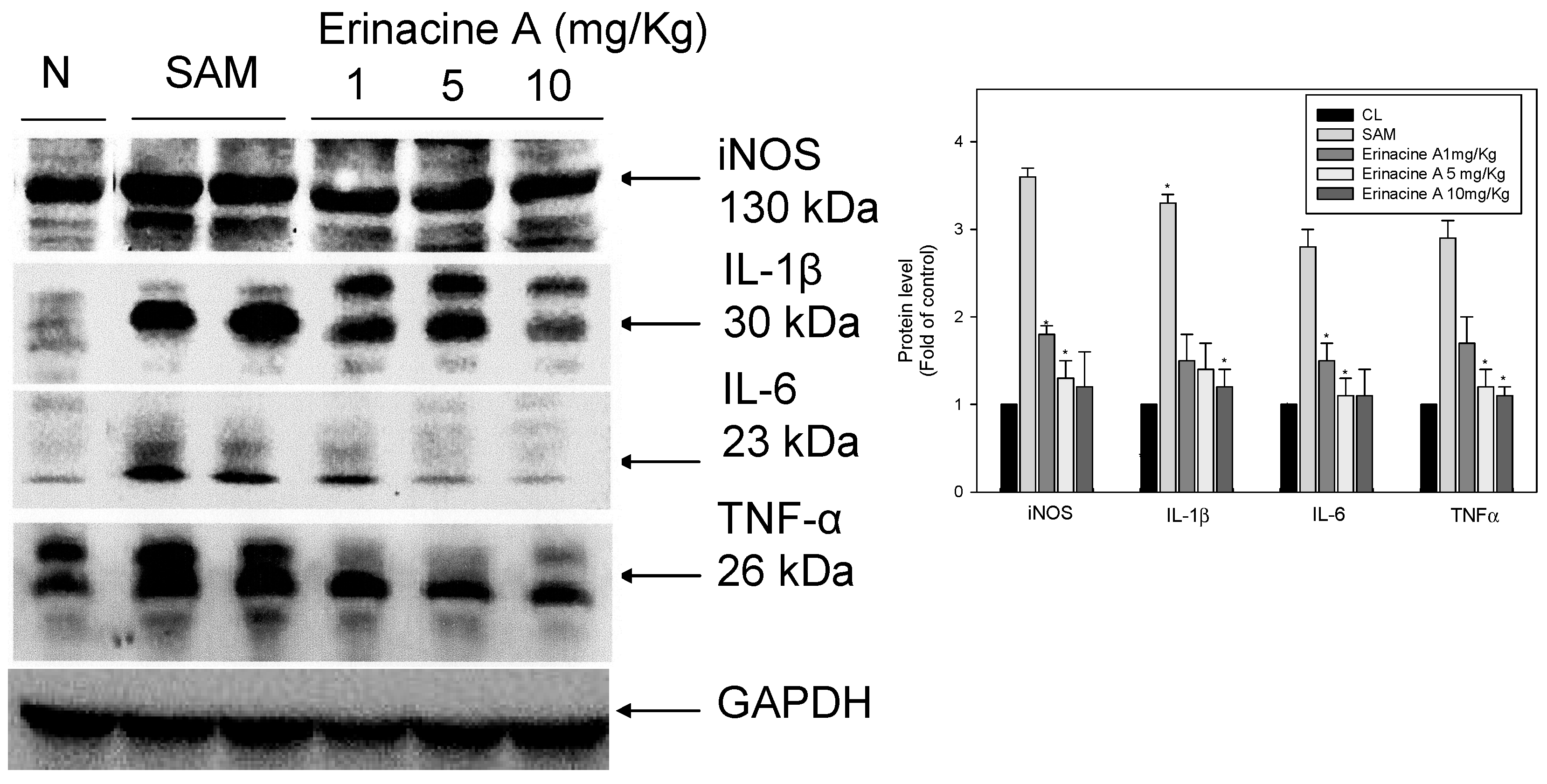
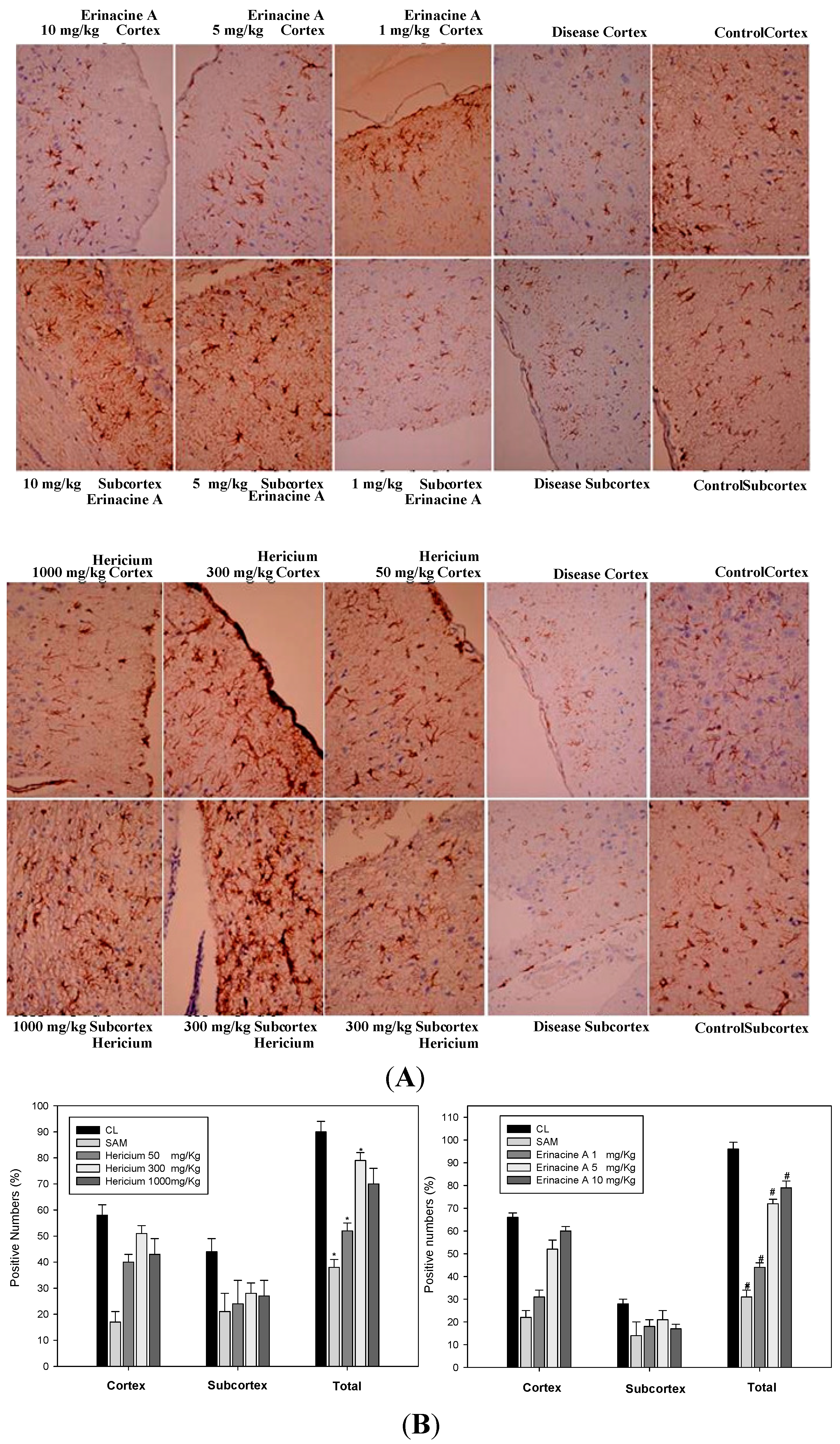
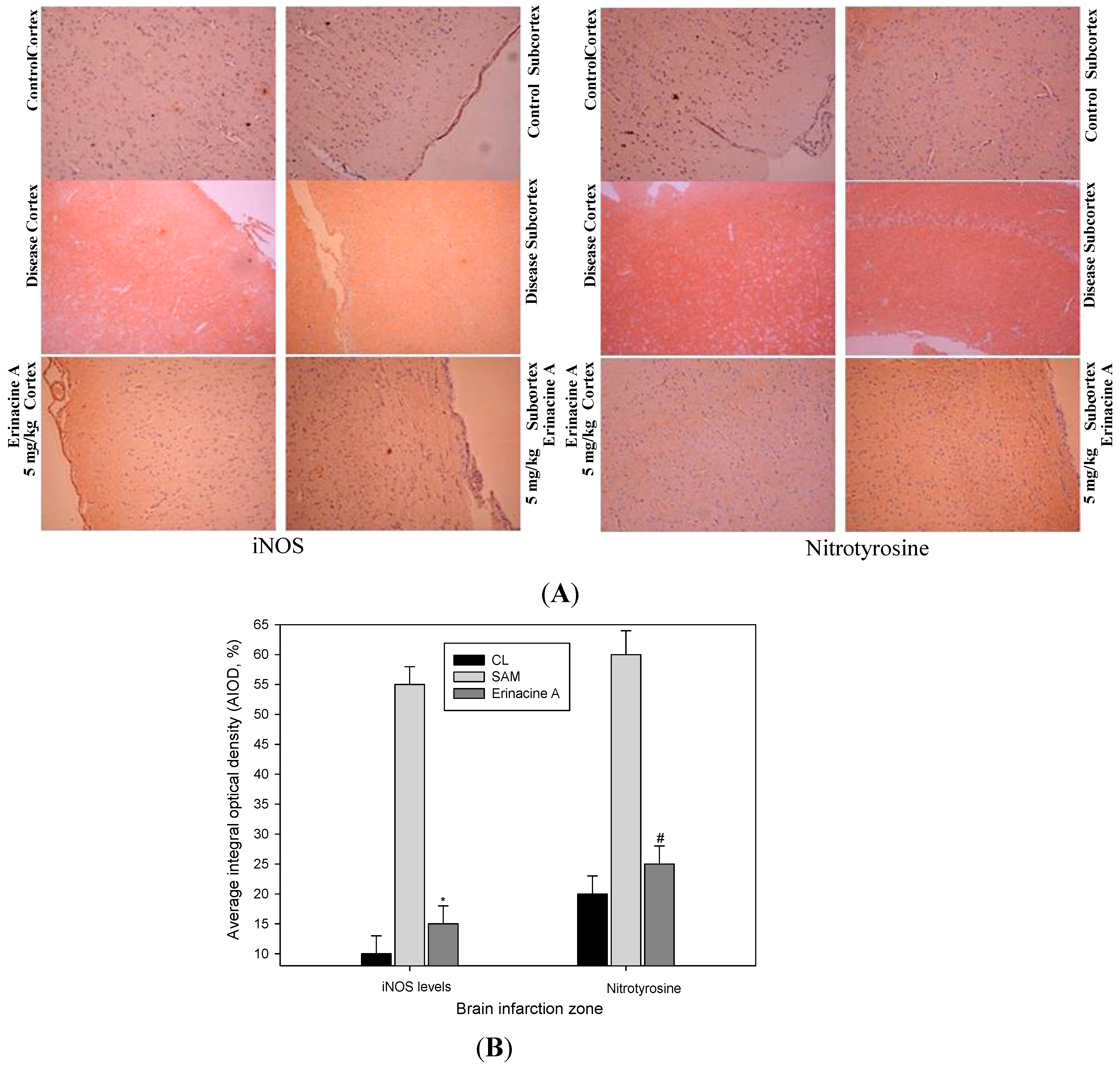
2.4. Immunohistochemistry Stain of iNOS and Nitrotyrosine Proteins on Brain Histopathology by Erinacine A in Transient Stroke as Seen in Animal Model Rats
2.5. Effects of Physiological Parameters Indicated by H. erinaceus Mycelium in a Transient Stroke Animal Model
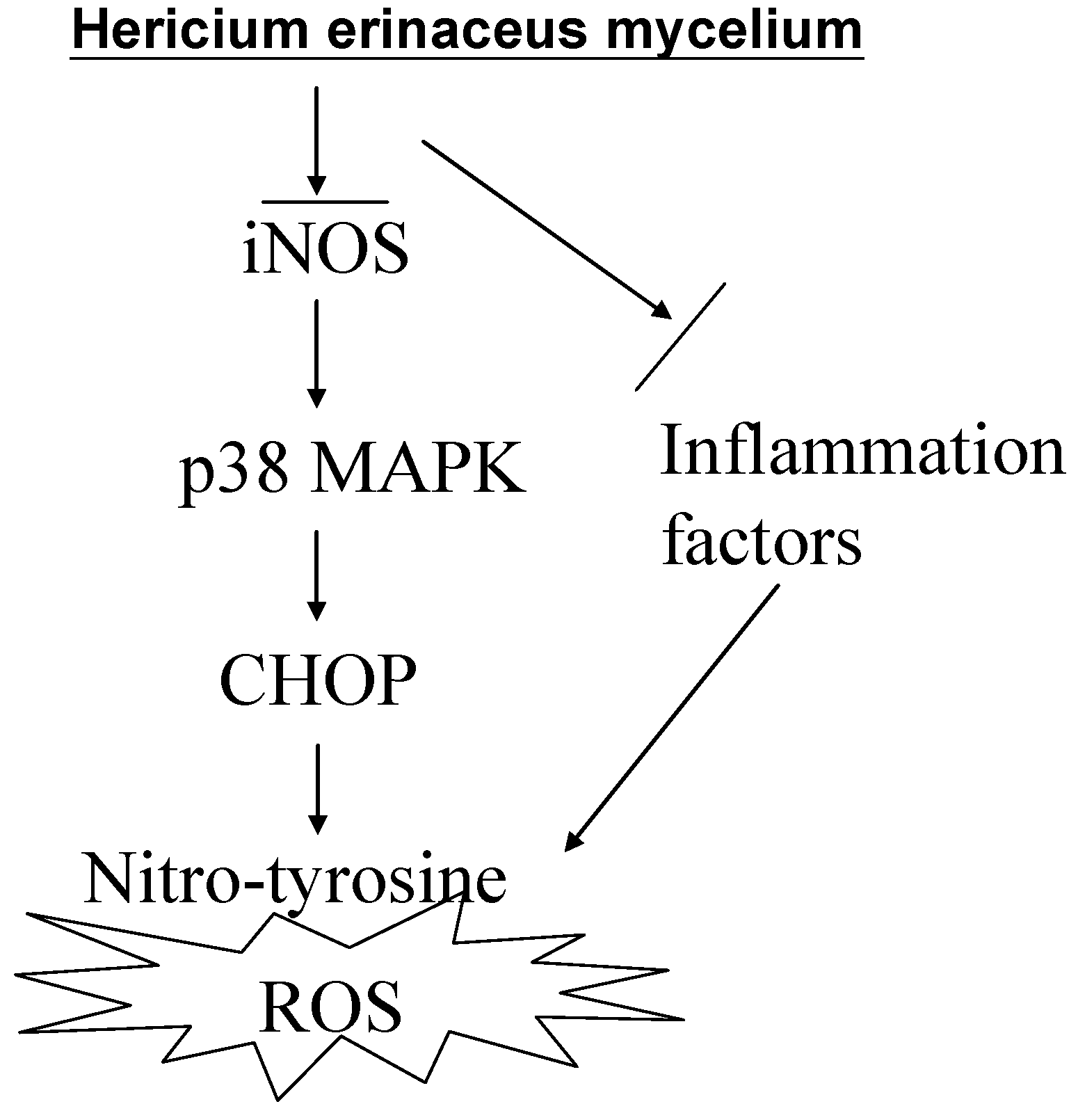
3. Discussion
4. Experimental Section
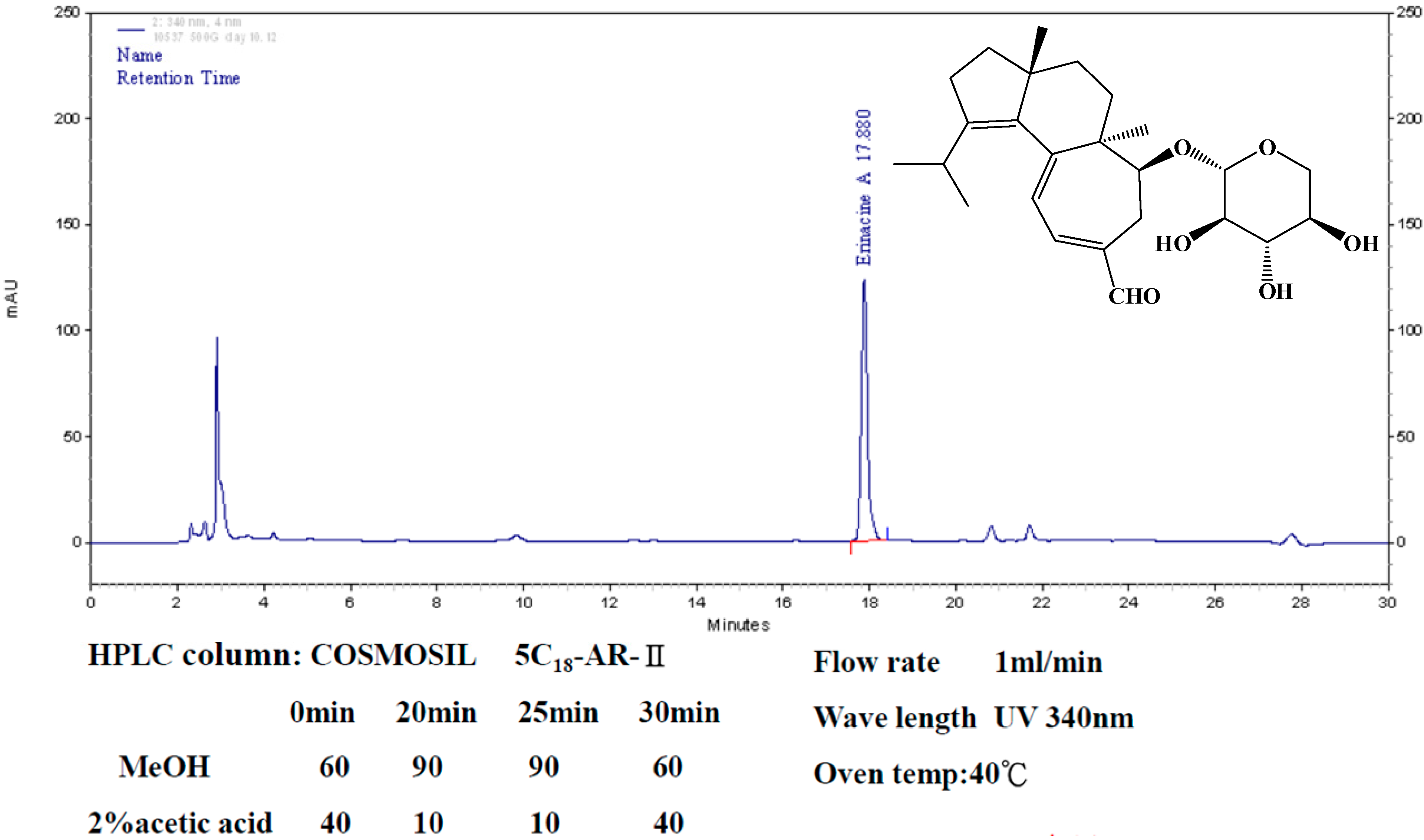
4.1. Hericium erinaceus Extracts and Analysis of Erinacine A
4.2. Induction Ischemia Reperfusion Brain Injury and Drug Administration
4.3. Chemical Reagents and Antibodies
4.4. Evaluation of the Size of the Ischemic Injury Using 2,3,5-Triphenyl tetrazolium chloride (TTC) Staining
4.5. Immunohistochemistry
4.6. Histopathological Evaluation
4.7. Preparation of Total Cell Extracts and Immunoblotting Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Malinowska, E.; Krzyczkowski, W.; Łapienis, G.; Herold, F. Improved simultaneous production of mycelial biomass anpolysaccharides by submerged culture of Hericium erinaceum: Optimization using a central composite rotatable design (CCRD). J. Ind. Microbiol. Biotechnol. 2009, 36, 1513–1527. [Google Scholar] [CrossRef]
- Yang, B.K.; Park, J.B.; Song, C.H. Hypolipidemic effect of an exo-biopolymer produced from a submerged mycelial culture of Hericium erinaceus. Biosci. Biotechnol. Biochem. 2003, 67, 1292–1298. [Google Scholar] [CrossRef]
- Gong, M.; An, J.; Lü, H.Z.; Wu, C.F.; Li, Y.J.; Cheng, J.Q.; Bao, J.K. Effects of denaturation and amino acid modification on fluorescence spectrum and hemagglutinating activity of Hericium erinaceum Lectin. Acta Biochim. Biophys. Sin. 2004, 36, 343–350. [Google Scholar] [CrossRef]
- Yim, M.H.; Shin, J.W.; Son, J.Y.; Oh, S.M.; Han, S.H.; Cho, J.H.; Cho, C.K.; Yoo, H.S.; Lee, Y.W.; Son, C.G. Soluble components of Hericium erinaceum induce NK cell activation via production of interleukin-12 in mice splenocytes. Acta Pharmacol. Sin. 2004, 28, 901–907. [Google Scholar]
- Mizuno, T.; Wasa, T.; Ito, H.; Suzuki, C.; Ukai, N. Antitumor-active polysaccharides isolated from the fruiting body of Hericium erinaceum, an edible and medicinal mushroom called Yamabushitake or Houtou. Biosci. Biotechnol. Biochem. 1992, 56, 347–348. [Google Scholar] [CrossRef]
- Nagai, K.; Chiba, A.; Nishino, T.; Kubota, T.; Kawagishi, H. Dilinoleoyl-phosphatidylethanolamine from Hericium erinaceum protects against ER stress-dependent Neuro2a cell death via protein kinase C pathway. J. Nutr. Biochem. 2006, 17, 525–530. [Google Scholar] [CrossRef]
- Ueda, K.; Tsujimori, M.; Kodani, S.; Chiba, A.; Kubo, M.; Masuno, K.; Sekiya, A.; Nagai, K.; Kawagish, H. An endoplasmic reticulum (ER) stress-suppressive compound and its analogues from the mushroom Hericium erinaceum. Bioorg. Med. Chem. 2008, 16, 9467–9470. [Google Scholar] [CrossRef]
- Kawagishi, H.; Shimada, A.; Hosokawa, S.; Mori, H.; Sakamoto, H.; Ishiguro, Y.; Sakemi, S.; Bordner, J.; Kojima, N.; Furukawa, S. Erinacines E, F, and G, stimulators of nerve growth factor (NGF)-synthesis, from the mycelia of Hericium erinaceum. Tetrahedron Lett. 1996, 37, 7399–7402. [Google Scholar] [CrossRef]
- Kawagishia, H.; Shimadaa, A.; Shiraia, R.; Okamotob, K.; Ojimab, F.; Sakamotob, H.; Ishigurob, Y.; Furukawac, S. Erinacines A, B and C, strong stimulators of nerve growth factor (NGF)-synthesis, from the mycelia of Hericium erinaceum. Tetrahedron Lett. 1994, 35, 1569–1572. [Google Scholar] [CrossRef]
- Lee, E.W.; Shizuki, K.; Hosokawa, S.; Suzuki, M.; Suganuma, H.; Inakuma, T.; Li, J.; Ohnishi-Kameyama, M.; Nagata, T.; Furukawa, S.; et al. Two novel diterpenoids, erinacines H and I from the mycelia of Hericium erinaceum. Biosci. Biotechnol. Biochem. 2000, 64, 2402–2405. [Google Scholar] [CrossRef]
- Nagano, M.; Shimizu, K.; Kondo, R.; Hayashi, C.; Sato, D.; Kitagawa, K.; Ohnuki, K. Reduction of depression and anxiety by 4 weeks Hericium erinaceus intake. Biomed. Res. 2010, 31, 231–237. [Google Scholar] [CrossRef]
- Mori, K.; Inatomi, S.; Ouchi, K.; Azumi, Y.; Tuchida, T. Improving effects of the mushroom Yamabushitake (Hericium erinaceus) on mild cognitive impairment: A double-blind placebo-controlled clinical trial. Phytother. Res. 2009, 23, 367–372. [Google Scholar] [CrossRef]
- Iadecola, C.; Anrather, J. The immunology of stroke: From mechanisms to translation. Nat. Med. 2011, 17, 796–808. [Google Scholar] [CrossRef]
- Towfighi, A.; Saver, J.L. Stroke declines from third to fourth leading cause of death in the United States: Historical perspective and challenges ahead. Stroke 2011, 42, 2351–2235. [Google Scholar] [CrossRef]
- Gotoh, T.; Mori, M. Nitric oxide and endoplasmic reticulum stress. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 1439–1446. [Google Scholar] [CrossRef]
- Kohno, K.; Higuchi, T.; Ohta, S.; Kumon, Y.; Sakaki, S. Neuroprotective nitric oxide synthase inhibitor reduces intracellular calcium accumulation following transient global ischemia in the gerbil. Neurosci. Lett. 1997, 224, 17–20. [Google Scholar] [CrossRef]
- Zhu, C.; Wang, X.; Xu, F.; Bahr, B.A.; Shibata, M.; Uchiyama, Y.; Hagberg, H.; Blomgren, K. The influence of age on apoptotic and other mechanisms of cell death after cerebral hypoxia ischemia. Cell Death Differ. 2005, 12, 162–176. [Google Scholar] [CrossRef]
- Noshita, N.; Lewen, A.; Sugawara, T.; Chan, P.H. Evidence of phosphorylation of Akt and neuronal survival after transient focal cerebral ischemia in mice. J. Cereb. Blood Flow Metab. 2001, 21, 1442–1450. [Google Scholar]
- Camandola, S.; Mattson, M.P. NF-κB as a therapeutic target in neurodegenerative diseases. Expert Opin. Ther. Targets 2007, 11, 123–132. [Google Scholar] [CrossRef]
- Li, I.C.; Chen, Y.L.; Lee, L.Y.; Chen, W.P.; Tsai, Y.T.; Chen, C.C.; Chen, C.S. Evaluation of the toxicological safety of erinacine A-enriched Hericium erinaceus in a 28-day oral feeding study in Sprague–Dawley rats. Food Chem. Toxicol. 2014, 6, 61–67. [Google Scholar]
- Eltzschig, H.K.; Carmeliet, P. Hypoxia and inflammation. N. Engl. J. Med. 2011, 364, 656–665. [Google Scholar] [CrossRef]
- Kawahara, K.; Oyadomaria, S.; Gotoh, T.; Kohsaka, S.; Nakayama, H.; Mori, M. Induction of CHOP and apoptosis by nitric oxide in p53-deficient microglial cells. FEBS Lett. 2001, 506, 135–139. [Google Scholar]
- Shimbo, M.; Kawagishi, H.; Yokogoshi, H. Erinacine A increases catecholamine and nerve growth factor content in the central nervous system of rats. Nutr. Res. 2005, 25, 617–623. [Google Scholar] [CrossRef]
- Moskowitz, M.A.; Lo, E.H.; Iadecola, C. The science of stroke: Mechanisms in search of treatments. Neuron 2010, 67, 181–198. [Google Scholar] [CrossRef]
- Li, Y.; Gu, J.; Liu, Y.; Long, H.; Wang, G.; Yin, G.; Fan, J. iNOS participates in apoptosis of spinal cord neurons via p-BAD dephosphorylation following ischemia/reperfusion (I/R) injury in rat spinal cord. Neurosci. Lett. 2013, 545, 117–122. [Google Scholar] [CrossRef]
- McColl, B.W.; Allan, S.M.; Rothwell, N.J. Systemic infection, inflammation and acute ischemic stroke. Neuroscience 2009, 158, 1049–1061. [Google Scholar] [CrossRef]
- Chern, C.M.; Wang, Y.H.; Liou, K.T.; Hou, Y.C.; Chen, C.C.; Shen, Y.C. 2-Methoxystypandrone ameliorates brain function through preserving BBB integrity and promoting neurogenesis in mice with acute ischemic stroke. Biochem. Pharmacol. 2014, 87, 502–514. [Google Scholar] [CrossRef]
- Kim, Y.S.; Lee, J.Y.; Park, J.; Hwang, W.; Lee, J.; Park, D. Synthesis and microbiological evaluation of honokiol derivatives as new antimicrobial agents. Arch. Pharm. Res. 2010, 33, 61–65. [Google Scholar] [CrossRef]
- Engin, F.; Hotamisligil, G.S. Restoring endoplasmic reticulum function by chemical chaperones: An emerging therapeutic approach for metabolic diseases. Diabetes Obes. Metab. 2010, 2, 108–115. [Google Scholar] [CrossRef]
- Thompson, J.W.; Narayanan, S.V.; Perez-Pinzon, M.A. Redox signaling pathways involved in neuronal ischemic preconditioning. Curr. Neuropharmacol. 2012, 10, 354–369. [Google Scholar] [CrossRef]
- Zhang, S.; Qi, Y.; Xu, Y.; Han, X.; Peng, J.; Liu, K.; Sun, C.K. Protective effect of flavonoid-rich extract from Rosa laevigata Michx on cerebral ischemia-reperfusion injury through suppression of apoptosis and inflammation. Neurochem. Int. 2013, 63, 522–532. [Google Scholar] [CrossRef]
- Yilmaz, G.; Granger, D.N. Leukocyte recruitment and ischemic brain injury. Neuromol. Med. 2010, 12, 193–204. [Google Scholar] [CrossRef]
- McCormick, M.T.; Muir, K.W.; Gray, C.S.; Walters, M.R. Management of hyperglycemia in acute stroke: How, when, and for whom? Stroke 2008, 39, 2177–2185. [Google Scholar] [CrossRef]
- Gardiner, M.; Smith, M.L; Kagstrom, E.; Shohami, E.; Siesjo, B.K. Influence of blood glucose concentration on brain lactate accumulation during severe hypoxia and subsequent recovery of brain energy metabolism. J. Cereb. Blood Flow Metab. 1982, 2, 429–438. [Google Scholar] [CrossRef]
- Krzyczkowski, W.; Malinowska, E.; Herold, F. Erinacine A biosynthesis in submerged cultivation of Hericium erinaceum: Quantification and improved cultivation. Eng. Life Sci. 2010, 10, 446–457. [Google Scholar] [CrossRef]
- Smith, M.L.; Bendek, G.; Dahlgren, N.; Rosén, I.; Wieloch, T.; Siesjö, B.K. Models for studying long-term recovery following forebrain ischemia in the rat. 2. A 2-vessel occlusion model. Acta Neurol. Scand. 2007, 69, 385–401. [Google Scholar]
- Swanson, R.A.; Sharp, F.R. Infarct measurement methodology. J. Cereb. Blood Flow Metab. 1994, 14, 697–698. [Google Scholar] [CrossRef]
- Chiu, Y.W.; Lin, T.H.; Huang, W.S.; Teng, C.Y.; Liou, Y.S.; Kuo, W.H.; Lin, W.L.; Huang, H.I.; Tung, J.N.; Huang, C.Y.; et al. Baicalein inhibits the migration and invasive properties of human hepatoma cells. Toxicol. Appl. Pharmacol. 2011, 255, 316–326. [Google Scholar] [CrossRef]
- Tsai, J.J.; Kuo, H.C.; Lee, K.F.; Tsai, T.H. Glycyrrhizin represses total parenteral nutrition-associated acute liver injury in rats by suppressing endoplasmic reticulum stress. Int. J. Mol. Sci. 2013, 14, 12563–12580. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, K.-F.; Chen, J.-H.; Teng, C.-C.; Shen, C.-H.; Hsieh, M.-C.; Lu, C.-C.; Lee, K.-C.; Lee, L.-Y.; Chen, W.-P.; Chen, C.-C.; et al. Protective Effects of Hericium erinaceus Mycelium and Its Isolated Erinacine A against Ischemia-Injury-Induced Neuronal Cell Death via the Inhibition of iNOS/p38 MAPK and Nitrotyrosine. Int. J. Mol. Sci. 2014, 15, 15073-15089. https://doi.org/10.3390/ijms150915073
Lee K-F, Chen J-H, Teng C-C, Shen C-H, Hsieh M-C, Lu C-C, Lee K-C, Lee L-Y, Chen W-P, Chen C-C, et al. Protective Effects of Hericium erinaceus Mycelium and Its Isolated Erinacine A against Ischemia-Injury-Induced Neuronal Cell Death via the Inhibition of iNOS/p38 MAPK and Nitrotyrosine. International Journal of Molecular Sciences. 2014; 15(9):15073-15089. https://doi.org/10.3390/ijms150915073
Chicago/Turabian StyleLee, Kam-Fai, Jiann-Hwa Chen, Chih-Chuan Teng, Chien-Heng Shen, Meng-Chiao Hsieh, Chien-Chang Lu, Ko-Chao Lee, Li-Ya Lee, Wan-Ping Chen, Chin-Chu Chen, and et al. 2014. "Protective Effects of Hericium erinaceus Mycelium and Its Isolated Erinacine A against Ischemia-Injury-Induced Neuronal Cell Death via the Inhibition of iNOS/p38 MAPK and Nitrotyrosine" International Journal of Molecular Sciences 15, no. 9: 15073-15089. https://doi.org/10.3390/ijms150915073
APA StyleLee, K.-F., Chen, J.-H., Teng, C.-C., Shen, C.-H., Hsieh, M.-C., Lu, C.-C., Lee, K.-C., Lee, L.-Y., Chen, W.-P., Chen, C.-C., Huang, W.-S., & Kuo, H.-C. (2014). Protective Effects of Hericium erinaceus Mycelium and Its Isolated Erinacine A against Ischemia-Injury-Induced Neuronal Cell Death via the Inhibition of iNOS/p38 MAPK and Nitrotyrosine. International Journal of Molecular Sciences, 15(9), 15073-15089. https://doi.org/10.3390/ijms150915073