Circulating miR-208b and miR-34a Are Associated with Left Ventricular Remodeling after Acute Myocardial Infarction
Abstract
:1. Introduction
2. Results
2.1. Baseline Clinical Characteristics of the Study Population
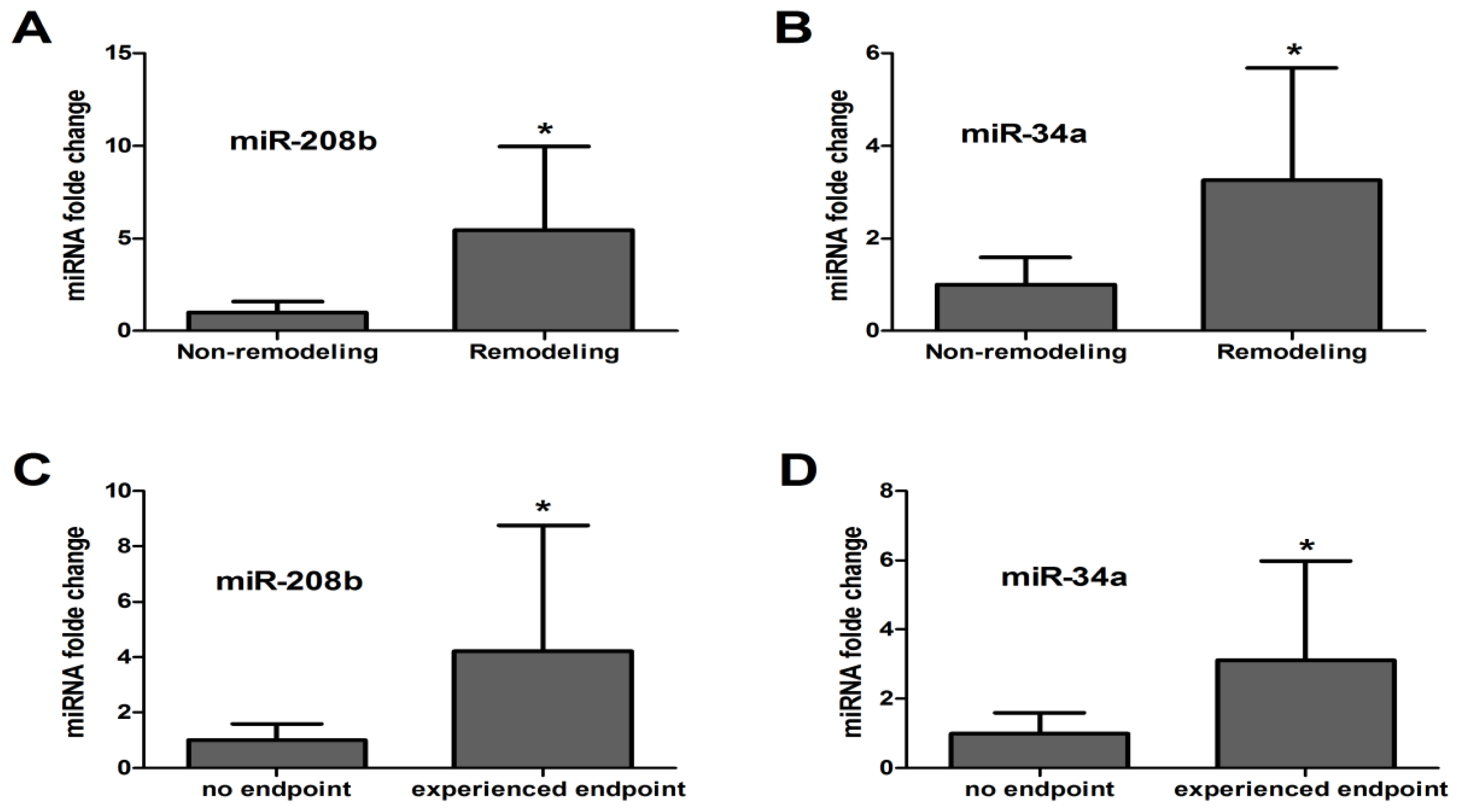
2.2. Circulating miRNA Levels Reflect LV Remodeling after AMI
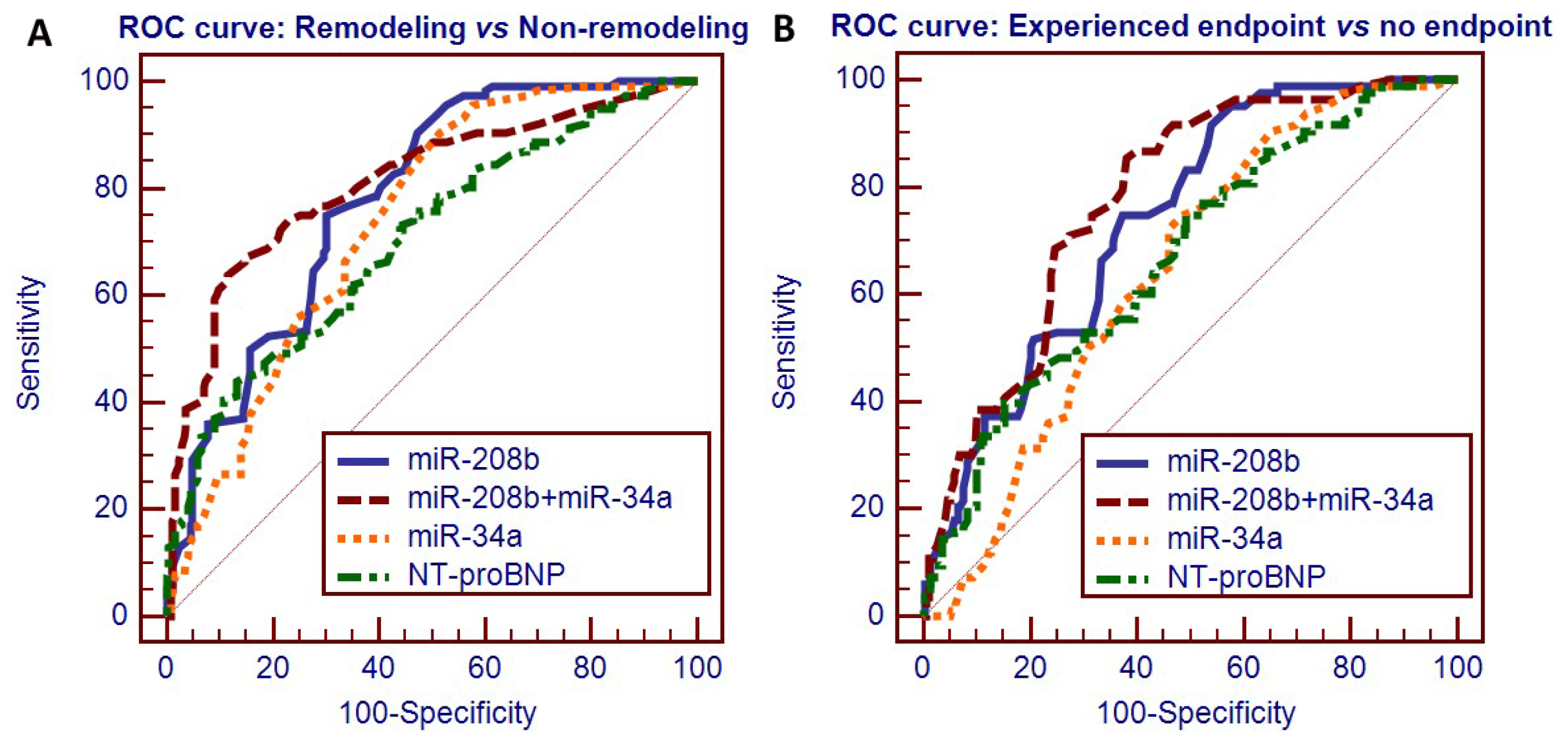
2.3. Circulating miRNAs as Potential Predictors of LV Remodeling after AMI
2.4. Prognostic Value of Circulating miRNAs after AMI
2.5. Reclassification Analyses for the Circulating miRNAs in Predicting LV Remodeling after AMI
3. Discussion
4. Experimental Section
4.1. Participants
4.2. Plasma Collection and Storage
4.3. RNA Preparation
4.4. MiRNA Determination
4.5. Statistical Analysis
4.6. Ethics Statement
5. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsConceived and designed the experiments: Fucheng He. Performed the experiments: Pin Lv, Mingxia Zhou, Jing He. Analyzed the data: Pin Lv, Weiwei Meng, Xuehan Ma. Contributed reagents/materials/analysis tools: Shuling Dong, Xianchun Meng, Xi Wang. Wrote the paper: Pin Lv, Xue Zhao.
References
- Sutton, M.G.; Sharpe, N. Left ventricular remodeling after myocardial infarction: Pathophysiology and therapy. Circulation 2000, 101, 2981–2988. [Google Scholar]
- Parikh, N.I.; Gona, P.; Larson, M.G.; Fox, C.S.; Benjamin, E.J.; Murabito, J.M.; O’Donnell, C.J.; Vasan, R.S.; Levy, D. Long-term trends in myocardial infarction incidence and case fatality in the National Heart, Lung, and Blood Institute’s Framingham Heart study. Circulation 2009, 119, 1203–1210. [Google Scholar]
- Gajarsa, J.J.; Kloner, R.A. Left ventricular remodeling in the post-infarction heart: A review of cellular, molecular mechanisms, and therapeutic modalities. Heart Fail. Rev 2011, 16, 13–21. [Google Scholar]
- Gravning, J.; Smedsrud, M.K.; Omland, T. Sensitive troponin assays and N-terminal pro-B-type natriuretic peptide in acute coronary syndrome: Prediction of significant coronary lesions and long-term prognosis. Am. Heart J 2013, 165, 716–724. [Google Scholar]
- De Antonio, M.; Lupon, J.; Galan, A.; Vila, J.; Urrutia, A.; Bayes-Genis, A. Combined use of high-sensitivity cardiac troponin T and N-terminal pro-B type natriuretic peptide improves measurements of performance over established mortality risk factors in chronic heart failure. Am. Heart J 2012, 163, 821–828. [Google Scholar]
- Gianfranco, A.; Gianfranco, A.; Francesca, D.S. Cardiac biomarkers in acute coronary syndromes: A review. Curr. Vasc. Pharmacol 2010, 8, 388–393. [Google Scholar]
- Garg, R.; Singh, A.; Khaja, A.; Martin, A.; Aggarwal, K. How does volume status affect BNP and troponin levels as markers of cardiovascular status in peritoneal dialysis? Congest. Heart Fail 2009, 15, 240–244. [Google Scholar]
- Desai, A.S.; Toto, R.; Jarolim, P.; Uno, H.; Eckardt, K.-U.; Kewalramani, R.; Levey, A.S.; Lewis, E.F.; McMurray, J.J.V.; Parving, H.-H.; et al. Association between cardiac biomarkers and the development of ESRD in patients with type 2 diabetes mellitus, anemia, and CKD. Am. J. Kidney Dis 2011, 58, 717–728. [Google Scholar]
- Talwar, S.; Squire, I.B.; Downie, P.F.; Mccullough, A.M.; Campton, M.C.; Davies, J.E.; Barnett, D.B.; Ng, L.L. Profile of plasma N-terminal proBNP following acute myocardial infarction; correlation with left ventricular systolic dysfunction. Eur. Heart J 2000, 21, 1514–1521. [Google Scholar]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar]
- Small, E.M.; Olson, E.N. Pervasive roles of microRNAs in cardiovascular biology. Nature 2011, 469, 336–342. [Google Scholar]
- Meltzer, P.S. Cancer genomics: small RNAs with big impacts. Nature 2005, 435, 745–746. [Google Scholar]
- Sayed, D.; Abdellatif, M. MicroRNAs in development and disease. Physiol. Rev 2011, 91, 827–887. [Google Scholar]
- Gilad, S.; Meiri, E.; Yogev, Y.; Benjamin, S.; Lebanony, D.; Yerushalmi, N.; Benjamin, H.; Kushnir, M.; Cholakh, H.; Melamed, N.; et al. Serum microRNAs are promising novel biomarkers. PLoS One 2008, 3, e3148. [Google Scholar]
- Chen, X.; Ba, Y.; Ma, L.; Cai, X.; Yin, Y.; Wang, K.; Guo, J.; Zhang, Y.; Chen, J.; Guo, X.; et al. Characterization of microRNAs in serum: A novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res 2008, 18, 997–1006. [Google Scholar]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar]
- Wang, K.; Zhang, S.; Weber, J.; Baxter, D.; Galas, D.J. Export of microRNAs and microRNA-protective protein by mammalian cells. Nucleic Acids. Res 2010, 38, 7248–7259. [Google Scholar]
- Long, G.; Wang, F.; Duan, Q.; Chen, F.; Yang, S.; Gong, W.; Wang, Y.; Chen, C.; Wang, D.W. Human circulating microRNA-1 and microRNA-126 as potential novel indicators for acute myocardial infarction. Int. J. Biol. Sci 2012, 8, 811–818. [Google Scholar]
- D’Alessandra, Y.; Devanna, P.; Limana, F.; Straino, S.; Carlo, A.D.; Brambilla, P.J.; Rubino, M.; Carena, M.C.; Spazzafumo, L.; de Simone, M. Circulating microRNAs are new and sensitive biomarkers of myocardial infarction. Eur. Heart J 2010, 31, 2765–2773. [Google Scholar]
- Devaux, Y.; Vausort, M.; Goretti, E.; Nazarov, P.V.; Azuaje, F.; Gilson, G.; Corsten, M.F.; Schroen, B.; Lair, M.-L.; Heymans, S.; et al. Use of circulating microRNAs to diagnose acute myocardial infarction. Clin. Chem 2012, 58, 559–567. [Google Scholar]
- Xiao, J.; Shen, B.; Li, J.; Lv, D.; Zhao, Y.; Wang, F.; Xu, J. Serum microRNA-499 and microRNA-208a as biomarkers of acute myocardial infarction. Int. J. Clin. Exp. Med 2014, 7, 136–141. [Google Scholar]
- Corsten, M.F.; Dennert, R.; Jochems, S.; Kuznetsova, T.; Devaux, Y.; Hofstra, L.; Wagner, D.R.; Staessen, J.A.; Heymans, S.; Schroen, B. Circulating MicroRNA-208b and MicroRNA-499 reflect myocardial damage in cardiovascular disease. Circulation 2010, 3, 499–506. [Google Scholar]
- Li, Y.-Q.; Zhang, M.-F.; Wen, H.-Y.; Hu, C.-L.; Liu, R.; Wei, H.-Y.; Ai, C.-M.; Wang, G.; Liao, X.-X.; Li, X.; et al. Comparing the diagnostic values of circulating microRNAs and cardiac troponin T in patients with acute myocardial infarction. Clinics 2013, 68, 75–80. [Google Scholar]
- Boon, R.A.; Iekushi, K.; Lechner, S.; Seeger, T.; Fischer, A.; Heydt, S.; Kaluza, D.; Tréguer, K.; Carmona, G.; Bonauer, A.; et al. MicroRNA-34a regulates cardiac ageing and function. Nature 2013, 495, 107–110. [Google Scholar]
- Fan, F.; Sun, A.; Zhao, H.; Liu, X.; Zhang, W.; Jin, X.; Wang, C.; Ma, X.; Shen, C.; Zou, Y.; et al. MicroRNA-34a promotes cardiomyocyte apoptosis post myocardial infarction through down-regulating aldehyde dehydrogenase 2. Curr. Pharm. Des 2013, 19, 4865–4873. [Google Scholar]
- Bernardo, B.C.; Gao, X.-M.; Winbanks, C.E.; Boeya, E.J.H.; Thama, Y.K.; Kiriazisa, H.; Gregorevica, P.; Obadb, S.; Kauppinenb, S.; Du, X.-J.; et al. Therapeutic inhibition of the miR-34 family attenuates pathological cardiac remodeling and improves heart function. Proc. Natl. Acad. Sci. USA 2012, 109, 17615–17620. [Google Scholar]
- Bernardo, B.C.; Gao, X.-M.; Tham, Y.K.; Kiriazis, H.; Winbanks, C.E.; Ooi, J.Y.Y.; Boey, E.J.H.; Obad, S.; Kauppinen, S.; Gregorevic, P. Silencing of miR-34a attenuates cardiac dysfunction in a setting of moderate, but not severe, hypertrophic cardiomyopathy. PLoS One 2014, 9, e90337. [Google Scholar]
- Ito, T.; Yagi, S.; Yamakuchi, M. MicroRNA-34a regulation of endothelial senescence. Biochem. Biophys. Res. Commun 2010, 398, 735–740. [Google Scholar]
- Pfeffer, M.A.; Braunwald, E. Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation 1990, 81, 1161–1172. [Google Scholar]
- Tijsen, A.J.; Pinto, Y.M.; Creemers, E.E. Circulating microRNAs as diagnostic biomarkers for cardiovascular diseases. Am. J. Physiol. Heart Circ. Physiol 2012, 303, H1085–H1095. [Google Scholar]
- Gao, Y.; Schug, J.; McKenna, L.B.; Lay, J.L.; Kaestner, K.H.; Greenbaum, L.E. Tissue-specific regulation of mouse microRNA genes in endoderm-derived tissues. Nucleic Acids. Res 2011, 39, 454–463. [Google Scholar]
- Mendell, J.T.; Olson, E.N. MicroRNAs in stress signaling and human disease. Cell 2012, 148, 1172–1187. [Google Scholar]
- Matsumoto, S.; Sakata, Y.; Suna, S.; Nakatani, D.; Usami, M.; Hara, M.; Kitamura, Y.; Hamasaki, Y.; Nanto, S.; Kawahara, Y. Circulating p53-responsive microRNAs are predictive indicators of heart failure after acute myocardial infarction. Circ. Res 2013, 113, 322–326. [Google Scholar]
- Zile, M.R.; Mehurg, S.M.; Arroyo, J.E.; Stroud, R.E.; DeSantis, S.M.; Spinale, F.G. Relationship between the temporal profile of plasma microRNA and left ventricular remodeling in patients after myocardial infarction. Circ. Cardiovasc. Genet 2011, 4, 614–619. [Google Scholar]
- Widera, C.; Gupta, S.K.; Lorenzen, J.M.; Bang, C.; Bauersachs, J.; Bethmann, K.; Kempf, T.; Woller, K.C.; Thum, Y. Diagnostic and prognostic impact of six circulating microRNAs in acute coronary syndrome. J. Mol. Cell. Cardiol 2011, 51, 872–875. [Google Scholar]
- Matsumoto, S.; Sakata, Y.; Nakatani, D.; Sunaa, S.; Mizunoa, H.; Shimizua, M.; Usamia, M.; Sasakid, T.; Satoe, H.; Kawaharaf, Y. A subset of circulating microRNAs are predictive for cardiac death after discharge for acute myocardial infarction. Biochem. Biophys. Res. Commun 2012, 427, 280–284. [Google Scholar]
- Devaux, Y.; Vausort, M.; McCann, G.P.; Zangrando, J.; Kelly, D.; Razvi, N.; Zhang, L.; Ng, L.L.; Wagner, D.R.; Squire, L.B. MicroRNA-150: A novel marker of left ventricular remodeling after acute myocardial infarction. Circ. Cardiovasc. Genet 2013, 6, 290–298. [Google Scholar]
- Devaux, Y.; Vausort, M.; McCann, G.P.; Kelly, D.; Collignon, O.; Ng, L.L.; Wagner, D.R.; Squire, L.B. A panel of 4 microRNAs facilitates the prediction of left ventricular contractility after acute myocardial infarction. PLoS One 2013, 8, e70644. [Google Scholar]
- Morrow, D.A.; Cannon, C.P.; Jesse, R.L.; Newby, L.K.; Ravkilde, J.; Storrow, A.B.; Wu, A.H.B.; Christenson, R.H. National Academy of Clinical Biochemistry Laboratory Medicine Practice Guidelines: Clinical characteristics and utilization of biochemical markers in acute coronary syndromes. Circulation 2007, 115, e356–e375. [Google Scholar]
- Grundy, S.M.; Cleeman, J.I.; Bairey, C.N.; Brewer, H.B., Jr.; Clark, L.T.; Hunninghake, D.B.; Pasternak, R.C.; Smith, S.C., Jr.; Stone, N.J. National Heart, Lung, and Blood Institute. Implications of recent clinical trials for the national cholesterol education pro-gram adult treatment panel III guidelines. Circulation 2004, 110, 227–239. [Google Scholar]
- Aoki, S.; Nakagomi, A.; Asai, K.; Takano, H.; Yasutake, M.; Seino, Y.; Mizuno, K. Elevated peripheral blood mononuclear cell count is an independent predictor of left ventricular remodeling in patients with acute myocardial infarction. J. Cardiol 2011, 57, 202–207. [Google Scholar]
- Januzzi, J.L.; van Kimmenade, R.; Lainchbury, J.; Bayes-Genis, A.; Ordonez-Llanos, J.; Santalo-Bel, M.; Pinto, Y.M.; Richards, M. NT-proBNP testing for diagnosis and short-term prognosis in acute destabilized heart failure: An international pooled analysis of 1256 patients. Eur. Heart J 2006, 27, 330–337. [Google Scholar]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc 2008, 3, 1101–1108. [Google Scholar]
- Pencina, M.J.; D’Agostino, R.B., Sr.; D’Agostino, R.B., Jr.; Vasan, R.S. Evaluating the added predictive ability of a new marker: From area under the ROC curve to reclassification and beyond. Stat. Med 2008, 27, 157–172. [Google Scholar]
| Characteristics | Total patients (n = 359) | Remodeling (n = 116) | Non-remodeling (n = 243) | p1 | Experienced endpoint (n = 83) | No endpoint (n = 276) | p2 |
|---|---|---|---|---|---|---|---|
| Age (years) | 58 ± 14 | 59 ± 12 | 57 ± 15 | 0.587 | 57 ± 11 | 58 ± 14 | 0.805 |
| Male/female (n/n) | 301/58 | 97/19 | 204/39 | 0.937 | 75/8 | 226/50 | 0.066 |
| Current smoking, n (%) | 190 (53%) | 64 (55%) | 124 (51%) | 0.462 | 44 (53%) | 146 (53%) | 0.986 |
| Diabetes mellitus, n (%) | 57 (16%) | 22 (19%) | 35 (14%) | 0.269 | 15 (18%) | 42 (15%) | 0.533 |
| Hypertension, n (%) | 172 (48%) | 60 (52%) | 109 (45%) | 0.223 | 33 (40%) | 139 (50%) | 0.090 |
| Hyperlipidaemia, n (%) | 126 (35%) | 44 (38%) | 78 (32%) | 0.275 | 32 (38%) | 94 (34%) | 0.452 |
| SBP (mmHg) | 123 ± 21 | 127 ± 25 | 120 ± 16 | 0.155 | 122 ± 28 | 124 ± 18 | 0.824 |
| DBP (mmHg) | 76 ± 12 | 77 ± 9 | 74 ± 12 | 0.949 | 74 ± 15 | 77 ± 12 | 0.360 |
| TC (mmol/L) | 3.99 ± 1.07 | 3.80 ± 0.94 | 4.16 ± 1.15 | 0.112 | 3.73 ± 1.15 | 4.07 ± 1.04 | 0.199 |
| TG (mmol/L) | 1.55 ± 0.91 | 1.61 ± 0.98 | 1.50 ± 0.85 | 0.564 | 1.58 ± 1.01 | 1.54 ± 0.89 | 0.859 |
| HDL (mmol/L) | 1.03 ± 0.30 | 1.00 ± 0.26 | 1.07 ± 0.32 | 0.289 | 0.95 ± 0.24 | 1.07 ± 0.31 | 0.125 |
| LDL (mmol/L) | 2.41 ± 0.82 | 2.23 ± 0.57 | 2.43 ± 0.97 | 0.093 | 2.15 ± 0.70 | 2.49 ± 0.84 | 0.089 |
| AMI onset to sample (h; median(range)) | 6 (2–10) | 6 (2–10) | 6 (2–10) | 0.473 | 6 (3–10) | 6 (2–10) | 0.293 |
| discharge to follow up (days; median(range)) | 176 (121–226) | 170 (121–214) | 182 (133–226) | 0.248 | 179 (134–214) | 172 (121–226) | 0.322 |
| Serum biomarkers during admission (median(IQR)) | |||||||
| Peak CK (U/L) | 1536 (239,6839) | 1474 (191,5805) | 1609 (286,7017) | 0.119 | 1616 (253,6378) | 1390 (193,7082) | 0.094 |
| Cardiac troponin T (ng/mL) | 12.33 (0.088,53.32) | 15.65 (0.35,58.44) | 10.65 (0.004,47.21) | 0.015 | 13.13 (0.54,63.46) | 12.07 (0.005,49.57) | 0.075 |
| Nt-pro-BNP (pg/mL) | 350 (145,807) | 507 (212,1057) | 279 (81,733) | 0.003 | 567 (253,1189) | 233 (116,773) | 0.001 |
| Medications, n (%) | |||||||
| Beta-blockers | 305 (85%) | 100 (86%) | 205 (84%) | 0.674 | 67 (81%) | 238 (86%) | 0.218 |
| Calcium antagonists | 118 (33%) | 44 (38%) | 74 (30%) | 0.158 | 33 (40%) | 85 (31%) | 0.128 |
| ACEI/ARB | 219 (61%) | 66 (57%) | 153 (63%) | 0.270 | 44 (53%) | 175 (63%) | 0.089 |
| Statins | 352 (98%) | 114 (98%) | 238 (98%) | 0.831 a | 83 (100%) | 269 (97%) | 0.143 a |
| Anti-platelet therapy | 359 (100%) | 116 (100%) | 243 (100%) | 1.000 | 83 (100%) | 276 (100%) | 1.000 |
| Diuretic | 126 (35%) | 47 (41%) | 79 (33%) | 0.137 | 35 (42%) | 91 (33%) | 0.124 |
| Treatment, n (%) | |||||||
| CAG | 291 (81%) | 93 (80%) | 198 (81%) | 0.767 | 68 (82%) | 223 (81%) | 0.818 |
| Thrombolysis | 183 (51%) | 60 (52%) | 123 (51%) | 0.844 | 41 (49%) | 142 (51%) | 0.743 |
| PCI | 244 (68%) | 74 (64%) | 170 (70%) | 0.242 | 55 (66%) | 189 (68%) | 0.705 |
| Pre-discharge echo (median(IQR)) | |||||||
| LVEDV (mL) | 108 (97,119) | 108 (89,119) | 108 (103,119) | 0.595 | 106 (89,121) | 109 (98,119) | 0.334 |
| LVESV (mL) | 47 (37,54) | 48 (37,56) | 47 (38,53) | 0.489 | 45 (37,54) | 47 (38,54) | 0.772 |
| LVEF (%) | 60 (56,64) | 60 (56,64) | 60 (56,63) | 0.412 | 60 (56,66) | 60 (56,63) | 0.394 |
| Follow-up echo (median(IQR)) | |||||||
| LVEDV (mL) | 120 (102,131) | 124 (110,132) | 112 (100,122) | 0.034 | 121 (104,132) | 120 (102,130) | 0.850 |
| LVESV (mL) | 49 (41,55) | 48 (38,55) | 49 (45,55) | 0.427 | 51 (44,57) | 48 (39,55) | 0.366 |
| LVEF (%) | 55 (49,61) | 51 (47,58) | 59 (55,63) | 0.027 | 54 (48,62) | 56 (52,60) | 0.575 |
| Change between discharge and follow-up (median(IQR)) | |||||||
| ΔLVEDV (mL) | 10 (2,18) | 18 (14,24) | 3 (−4,5) | 0.000 | 17 (13,26) | 5 (−3,15) | 0.000 |
| ΔLVESV (mL) | 2 (−1,4) | 4 (1,6) | 1 (−2,4) | 0.005 | 4 (2,7) | 2 (−1,4) | 0.022 |
| ΔLVEF (%) | −6 (−8,2) | −9 (−13,2) | 1 (−4,4) | 0.000 | −6 (−11,2) | −1 (−3,3) | 0.003 |
| MiRNAs | ΔCt/ΔΔCt | Remodeling (n = 116) | Non-remodeling (n = 243) | p1 | Experienced endpoint (n = 83) | No endpoint (n = 276) | p2 |
|---|---|---|---|---|---|---|---|
| miR-208b | ΔCt | 2.86 ± 1.30 | 4.04 ± 1.61 | 0.000 | 2.47 ± 1.48 | 3.50 ± 1.39 | 0.004 |
| ΔΔCt | −1.94 ± 1.27 | 0 | −1.35 ± 1.48 | 0 | |||
| miR-34a | ΔCt | 3.06 ± 1.12 | 4.06 ± 1.59 | 0.001 | 2.93 ± 1.63 | 3.77 ± 1.54 | 0.035 |
| ΔΔCt | −1.32 ± 1.12 | 0 | −0.94 ± 1.63 | 0 |
| Model a without miR-208b | Model a with miR-208b | Reclassification | ||||||
|---|---|---|---|---|---|---|---|---|
| Predicted risk | <10% | 10%–30% | >30% | Total | Increased risk, n (%) | Decreased risk, n (%) | NRI b | p |
| Patients with remodeling (n = 116) | ||||||||
| <10% | 27 | 9 | 5 | 41 | ||||
| 10%–30% | 4 | 35 | 4 | 43 | ||||
| >30% | 0 | 3 | 29 | 32 | ||||
| Total | 31 | 47 | 38 | 116 | 18 (15.5) | 7 (6.0) | ||
| Patients without remodeling (n = 243) | ||||||||
| <10% | 98 | 3 | 2 | 103 | ||||
| 10%–30% | 5 | 99 | 3 | 107 | ||||
| >30% | 0 | 3 | 30 | 33 | ||||
| Total | 103 | 105 | 35 | 243 | 8 (3.3) | 8 (3.3) | ||
| NRI b | 0.095 | 0.039 | ||||||
| Model a without miR-34a | Model a with miR-34a | Reclassification | ||||||
|---|---|---|---|---|---|---|---|---|
| Predicted risk | <10% | 10%–30% | >30% | Total | Increased risk, n (%) | Decreased risk, n (%) | NRI b | p |
| Patients with remodeling (n = 116) | ||||||||
| <10% | 26 | 7 | 3 | 36 | ||||
| 10%–30% | 5 | 35 | 7 | 47 | ||||
| >30% | 0 | 4 | 29 | 33 | ||||
| Total | 31 | 46 | 39 | 116 | 17 (14.7) | 9 (7.8) | ||
| Patients without remodeling (n = 243) | ||||||||
| <10% | 93 | 4 | 3 | 100 | ||||
| 10%–30% | 4 | 97 | 5 | 106 | ||||
| >30% | 0 | 7 | 30 | 37 | ||||
| Total | 97 | 108 | 38 | 243 | 12 (4.9) | 11 (4.5) | ||
| NRI b | 0.065 | 0.177 | ||||||
| Model a without miR-208b and miR-34a | Model a with miR-208b and miR-34a | Reclassification | ||||||
|---|---|---|---|---|---|---|---|---|
| Predicted risk | <10% | 10%–30% | >30% | Total | Increased risk, n (%) | Decreased risk, n (%) | NRI b | p |
| Patients with remodeling (n = 116) | ||||||||
| <10% | 25 | 11 | 3 | 39 | ||||
| 10%–30% | 4 | 35 | 8 | 47 | ||||
| >30% | 0 | 4 | 26 | 30 | ||||
| Total | 29 | 50 | 37 | 116 | 22 (19.0) | 8 (6.9) | ||
| Patients without remodeling (n = 243) | ||||||||
| <10% | 89 | 6 | 3 | 98 | ||||
| 10%–30% | 8 | 95 | 6 | 109 | ||||
| >30% | 0 | 6 | 30 | 36 | ||||
| Total | 97 | 107 | 39 | 243 | 15 (6.2) | 14 (5.8) | ||
| NRI b | 0.117 | 0.025 | ||||||
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lv, P.; Zhou, M.; He, J.; Meng, W.; Ma, X.; Dong, S.; Meng, X.; Zhao, X.; Wang, X.; He, F. Circulating miR-208b and miR-34a Are Associated with Left Ventricular Remodeling after Acute Myocardial Infarction. Int. J. Mol. Sci. 2014, 15, 5774-5788. https://doi.org/10.3390/ijms15045774
Lv P, Zhou M, He J, Meng W, Ma X, Dong S, Meng X, Zhao X, Wang X, He F. Circulating miR-208b and miR-34a Are Associated with Left Ventricular Remodeling after Acute Myocardial Infarction. International Journal of Molecular Sciences. 2014; 15(4):5774-5788. https://doi.org/10.3390/ijms15045774
Chicago/Turabian StyleLv, Pin, Mingxia Zhou, Jing He, Weiwei Meng, Xuehan Ma, Shuling Dong, Xianchun Meng, Xue Zhao, Xi Wang, and Fucheng He. 2014. "Circulating miR-208b and miR-34a Are Associated with Left Ventricular Remodeling after Acute Myocardial Infarction" International Journal of Molecular Sciences 15, no. 4: 5774-5788. https://doi.org/10.3390/ijms15045774
APA StyleLv, P., Zhou, M., He, J., Meng, W., Ma, X., Dong, S., Meng, X., Zhao, X., Wang, X., & He, F. (2014). Circulating miR-208b and miR-34a Are Associated with Left Ventricular Remodeling after Acute Myocardial Infarction. International Journal of Molecular Sciences, 15(4), 5774-5788. https://doi.org/10.3390/ijms15045774




