Emerging Biomarkers in Heart Failure and Cardiac Cachexia
Abstract
:1. Introduction
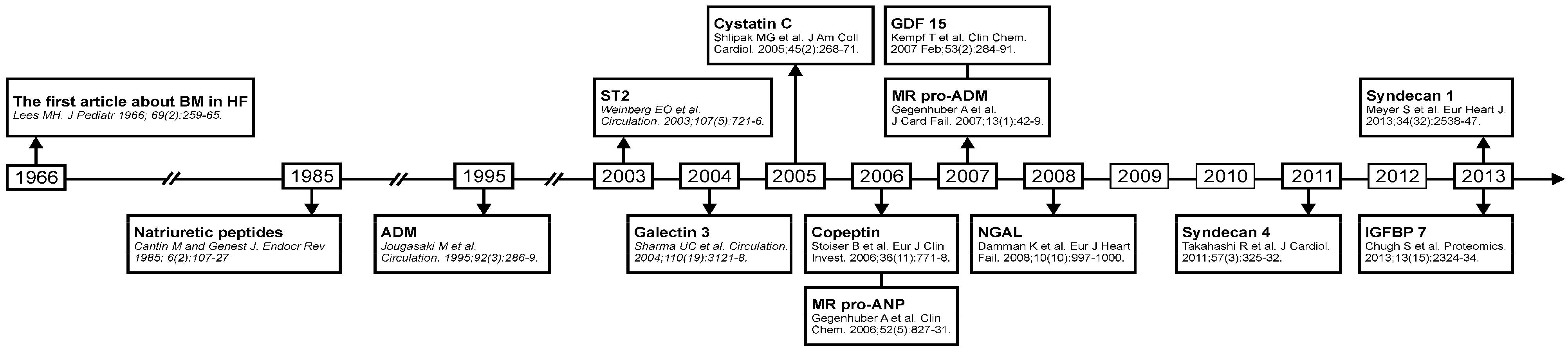
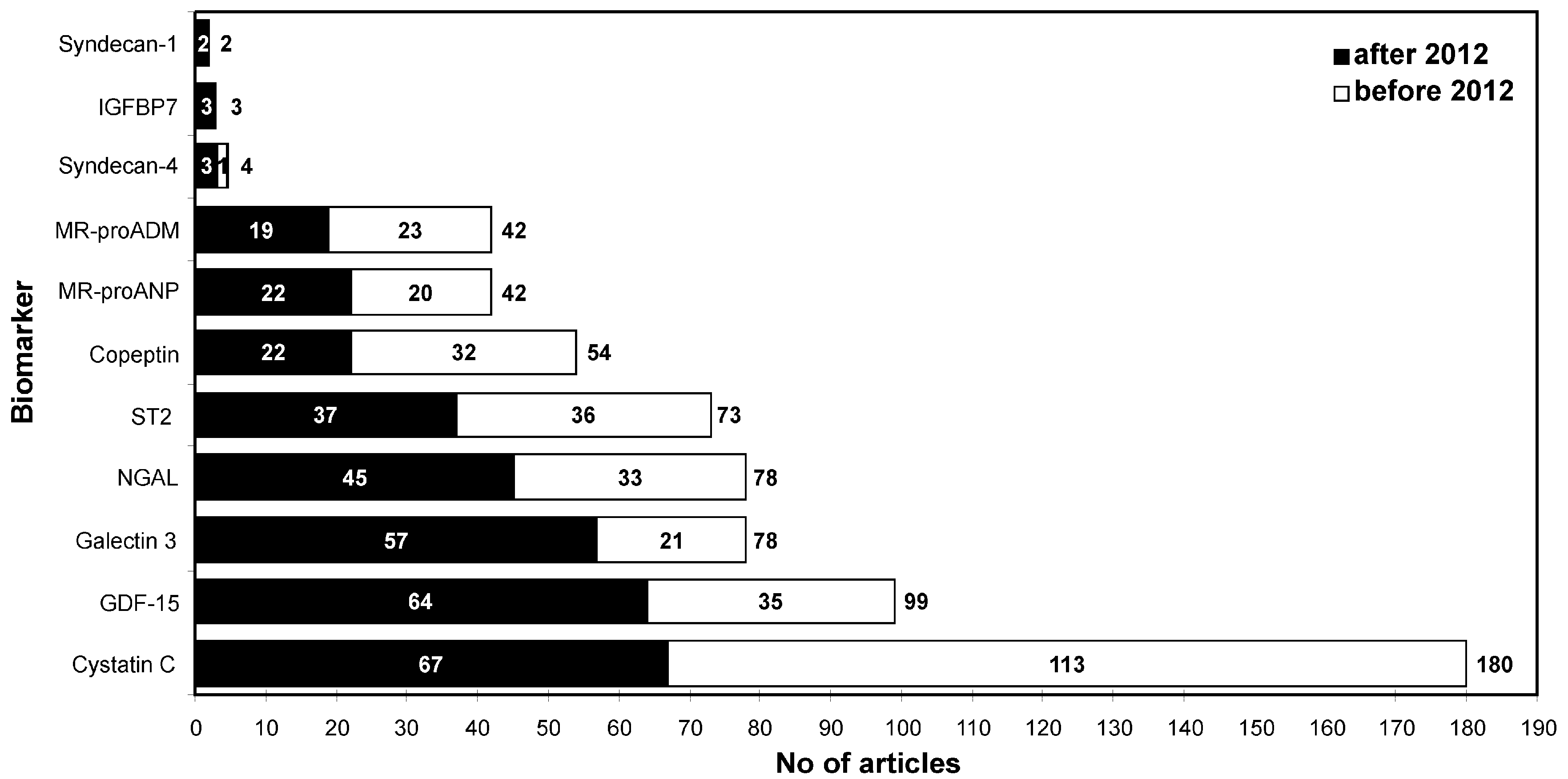
2. Biomarkers in Heart Failure
2.1. Mid-Regional Zone of proANP (MR-proANP)
| Biomarker | Diagnosis | Prognosis | Increased Risk of HF | Making Therapy Decision | |||
|---|---|---|---|---|---|---|---|
| AHF | HFPEF | AHF | CHF | HFPEF | |||
| MR-proANP | + | + | + | ||||
| ST2 | + | + | + | ||||
| Galectin 3 | + | + | + | + | + | ||
| MR-proADM | + | + | + | + | + | ||
| Copeptin | + | + | + | + | + | ||
| GDF15 | + | + | + | + | |||
| Cystatin C | + | + | + | + | |||
| NGAL | + | + | |||||
| Procalcitonin | + | ||||||
| Syndecan 1 | + | ||||||
| Syndecan 4 | + | ||||||
| IGFBP 7 | + | ||||||
2.2. ST2
| Biomarker | Cutoff Value | Sensitivity (%) | Specificity (%) | AUC |
|---|---|---|---|---|
| MR-proANP [7] | 120 pmol/L | 90–97 | 59.9–68 | 0.88 |
| ST2 [19] | 34.3 U/mL | 73.5 | 79.6 | 0.75 |
| Galectin 3 [21] | 17.8 ng/mL | 94.3 | 65.1 | 0.72 |
| GDF 15 [22] | 1306 ng/mL | 71.2 | 68.8 | 0.76 |
2.3. Galectin 3
2.4. Mid-Regional Pro-Hormone Fragment of Adrenomedullin (MR-proADM)
2.5. Copeptin
2.6. Growth-Differentiation Factor 15 (GDF 15)
2.7. Biomarkers of Extracardiac Involvement
2.7.1. Renal Impairment in HF
| Determining Therapy Approach | Monitoring Responses to Therapy | Therapy Guiding Potential |
|---|---|---|
| Galectin 3 | MR-proADM | MR-proANP |
| Cystatin-C | Copeptin | ST2 |
| NGAL |
2.7.2. Procalcitonin
2.8. The Newest Biomarkers of HF
3. Biomarkers of Cardiac Cachexia in Heart Failure
| Biomarkers of Cardiac Cachexia |
|---|
| Ghrelin |
| Adiponectin |
| C-terminal agrin fragment (CAF) |
| Growth differentiation factor 15 (GDF 15) |
| Atrial natriuretic peptide (ANP) |
| N-terminal propeptide of type III procollagen (P3NP) |
| Type VI collagen N-terminal globular domain epitope |
| Myostatin |
4. Conclusions and Future Research
Author Contributions
Conflicts of Interest
References
- McMurray, J.J.; Adamopoulos, S.; Anker, S.D.; Auricchio, A.; Bohm, M.; Dickstein, K.; Falk, V.; Filippatos, G.; Fonseca, C.; Gomez-Sanchez, M.A.; et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The task force for the diagnosis and treatment of acute and chronic heart failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 2012, 14, 803–869. [Google Scholar] [CrossRef] [PubMed]
- Cantin, M.; Genest, J. The heart and the atrial natriuretic factor. Endocr. Rev. 1985, 6, 107–127. [Google Scholar] [CrossRef] [PubMed]
- Lainscak, M.; Anker, M.S.; von Haehling, S.; Anker, S.D. Biomarkers for chronic heart failure: Diagnostic, prognostic, and therapeutic challenges. Herz 2009, 34, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Gaggin, H.K.; Januzzi, J.L., Jr. Biomarkers and diagnostics in heart failure. Biochim. Biophys. Acta 2013, 1832, 2442–2450. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Francis, G.S.; Morrow, D.A.; Newby, L.K.; Cannon, C.P.; Jesse, R.L.; Storrow, A.B.; Christenson, R.H.; Apple, F.S.; Ravkilde, J.; et al. National academy of clinical biochemistry laboratory medicine practice guidelines: Clinical utilization of cardiac biomarker testing in heart failure. Circulation 2007, 116, e99–e109. [Google Scholar] [CrossRef] [PubMed]
- Van Kimmenade, R.R.; Januzzi, J.L., Jr. Emerging biomarkers in heart failure. Clin. Chem. 2012, 58, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Maisel, A.; Mueller, C.; Nowak, R.; Peacock, W.F.; Landsberg, J.W.; Ponikowski, P.; Mockel, M.; Hogan, C.; Wu, A.H.; Richards, M.; et al. Mid-region pro-hormone markers for diagnosis and prognosis in acute dyspnea: Results from the BACH (Biomarkers in Acute Heart Failure) trial. J. Am. Coll. Cardiol. 2010, 55, 2062–2076. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.V.; Truong, Q.A.; Gaggin, H.K.; Pfannkuche, J.; Hartmann, O.; Januzzi, J.L., Jr. Mid-regional pro-atrial natriuretic peptide and pro-adrenomedullin testing for the diagnostic and prognostic evaluation of patients with acute dyspnoea. Eur. Heart J. 2012, 33, 2197–2205. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, E.O.; Shimpo, M.; Hurwitz, S.; Tominaga, S.; Rouleau, J.L.; Lee, R.T. Identification of serum soluble ST2 receptor as a novel heart failure biomarker. Circulation 2003, 107, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Kakkar, R.; Lee, R.T. The IL-33/ST2 pathway: Therapeutic target and novel biomarker. Nat. Rev. Drug Discov. 2008, 7, 827–840. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Chan, W.L.; Leung, B.P.; Huang, F.; Wheeler, R.; Piedrafita, D.; Robinson, J.H.; Liew, F.Y. Selective expression of a stable cell surface molecule on type 2 but not type 1 helper T cells. J. Exp. Med. 1998, 187, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Seki, K.; Sanada, S.; Kudinova, A.Y.; Steinhauser, M.L.; Handa, V.; Gannon, J.; Lee, R.T. Interleukin-33 prevents apoptosis and improves survival after experimental myocardial infarction through ST2 signaling. Circ. Heart Fail. 2009, 2, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, E.O.; Shimpo, M.; de Keulenaer, G.W.; MacGillivray, C.; Tominaga, S.; Solomon, S.D.; Rouleau, J.L.; Lee, R.T. Expression and regulation of ST2, an interleukin-1 receptor family member, in cardiomyocytes and myocardial infarction. Circulation 2002, 106, 2961–2966. [Google Scholar] [CrossRef] [PubMed]
- Januzzi, J.L., Jr.; Peacock, W.F.; Maisel, A.S.; Chae, C.U.; Jesse, R.L.; Baggish, A.L.; O’Donoghue, M.; Sakhuja, R.; Chen, A.A.; van Kimmenade, R.R. Measurement of the interleukin family member ST2 in patients with acute dyspnea: Results from the PRIDE (Pro-Brain Natriuretic Peptide Investigation of Dyspnea in the Emergency Department) study. J. Am. Coll. Cardiol. 2007, 50, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.U.; Mueller, T.; Januzzi, J.L., Jr. Characteristics of the novel interleukin family biomarker ST2 in patients with acute heart failure. J. Am. Coll. Cardiol. 2008, 52, 1458–1465. [Google Scholar] [CrossRef] [PubMed]
- Boisot, S.; Beede, J.; Isakson, S.; Chiu, A.; Clopton, P.; Januzzi, J.; Maisel, A.S.; Fitzgerald, R.L. Serial sampling of ST2 predicts 90-day mortality following destabilized heart failure. J. Card. Fail. 2008, 14, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.D.; Su, G.H.; Lu, Y.X.; Shuai, X.X.; Tao, X.F.; Meng, Y.D.; Luo, P. Elevated soluble ST2 and depression increased the risk of all-cause mortality and hospitalization in patients with heart failure. Int. Heart J. 2014, 55, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Dieplinger, B.; Januzzi, J.L., Jr.; Steinmair, M.; Gabriel, C.; Poelz, W.; Haltmayer, M.; Mueller, T. Analytical and clinical evaluation of a novel high-sensitivity assay for measurement of soluble ST2 in human plasma—The Presage™ ST2 assay. Clin. Chim. Acta 2009, 409, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Aldous, S.J.; Richards, A.M.; Troughton, R.; Than, M. ST2 has diagnostic and prognostic utility for all-cause mortality and heart failure in patients presenting to the emergency department with chest pain. J. Card. Fail. 2012, 18, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Bayes-Genis, A.; Pascual-Figal, D.; Januzzi, J.L.; Maisel, A.; Casas, T.; Valdes, M.; Ordonez-Llanos, J. Soluble ST2 monitoring provides additional risk stratification for outpatients with decompensated heart failure. Rev. Esp. Cardiol. 2010, 63, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.S.; Shi, B.; Dong, L.; Bi, L. Comparative study of galectin-3 and B-type natriuretic peptide as biomarkers for the diagnosis of heart failure. J. Geriatr. Cardiol. 2014, 11, 79–82. [Google Scholar] [PubMed]
- Li, Y.; Wang, X.M.; Liu, Y.L.; Shi, K.; Yang, Y.F.; Guo, Y.H. Plasma concentration of growth-differentiation factor-15 in children with congenital heart disease: Relation ship to heart function and diagnostic value in heart failure. Zhongguo Dang Dai Er Ke Za Zhi 2013, 15, 95–98. [Google Scholar] [PubMed]
- Henderson, N.C.; Mackinnon, A.C.; Farnworth, S.L.; Kipari, T.; Haslett, C.; Iredale, J.P.; Liu, F.T.; Hughes, J.; Sethi, T. Galectin-3 expression and secretion links macrophages to the promotion of renal fibrosis. Am. J. Pathol. 2008, 172, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Sharma, U.C.; Pokharel, S.; van Brakel, T.J.; van Berlo, J.H.; Cleutjens, J.P.; Schroen, B.; Andre, S.; Crijns, H.J.; Gabius, H.J.; Maessen, J.; et al. Galectin-3 marks activated macrophages in failure-prone hypertrophied hearts and contributes to cardiac dysfunction. Circulation 2004, 110, 3121–3128. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Ruifrok, W.P.; Meissner, M.; Bos, E.M.; van Goor, H.; Sanjabi, B.; van der Harst, P.; Pitt, B.; Goldstein, I.J.; Koerts, J.A.; et al. Genetic and pharmacological inhibition of galectin-3 prevents cardiac remodeling by interfering with myocardial fibrogenesis. Circ. Heart Fail. 2013, 6, 107–117. [Google Scholar] [CrossRef] [PubMed]
- McCullough, P.A. Practical experience using galectin-3 in heart failure. Clin. Chem. Lab. Med. 2014, 52, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- De Boer, R.A.; Lok, D.J.; Jaarsma, T.; van der Meer, P.; Voors, A.A.; Hillege, H.L.; van Veldhuisen, D.J. Predictive value of plasma galectin-3 levels in heart failure with reduced and preserved ejection fraction. Ann. Med. 2011, 43, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Van Kimmenade, R.R.; Januzzi, J.L., Jr.; Ellinor, P.T.; Sharma, U.C.; Bakker, J.A.; Low, A.F.; Martinez, A.; Crijns, H.J.; MacRae, C.A.; Menheere, P.P.; et al. Utility of amino-terminal pro-brain natriuretic peptide, galectin-3, and apelin for the evaluation of patients with acute heart failure. J. Am. Coll. Cardiol. 2006, 48, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Lok, D.J.; van der Meer, P.; de la Porte, P.W.; Lipsic, E.; van Wijngaarden, J.; Hillege, H.L.; van Veldhuisen, D.J. Prognostic value of galectin-3, a novel marker of fibrosis, in patients with chronic heart failure: Data from the DEAL-HF study. Clin. Res. Cardiol. 2010, 99, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Stolen, C.M.; Adourian, A.; Meyer, T.E.; Stein, K.M.; Solomon, S.D. Plasma galectin-3 and heart failure outcomes in MADIT-CRT (Multicenter Automatic Defibrillator Implantation Trial-Cardiac Resynchronization Therapy). J. Card. Fail. 2014, 20, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Gullestad, L.; Ueland, T.; Kjekshus, J.; Nymo, S.H.; Hulthe, J.; Muntendam, P.; Adourian, A.; Bohm, M.; van Veldhuisen, D.J.; Komajda, M.; et al. Galectin-3 predicts response to statin therapy in the controlled rosuvastatin multinational trial in heart failure (CORONA). Eur. Heart J. 2012, 33, 2290–2296. [Google Scholar] [CrossRef] [PubMed]
- Maisel, A.; Xue, Y.; van Veldhuisen, D.J.; Voors, A.A.; Jaarsma, T.; Pang, P.S.; Butler, J.; Pitt, B.; Clopton, P.; de Boer, R.A. Effect of spironolactone on 30-day death and heart failure rehospitalization (from the COACH Study). Am. J. Cardiol. 2014, 114, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Peacock, W.F. Novel biomarkers in acute heart failure: MR-pro-adrenomedullin. Clin. Chem. Lab. Med. 2014, 52, 1433–1435. [Google Scholar] [PubMed]
- Morgenthaler, N.G.; Struck, J.; Alonso, C.; Bergmann, A. Measurement of midregional proadrenomedullin in plasma with an immunoluminometric assay. Clin. Chem. 2005, 51, 1823–1829. [Google Scholar] [CrossRef] [PubMed]
- Bahrmann, P.; Bahrmann, A.; Hofner, B.; Christ, M.; Achenbach, S.; Sieber, C.C.; Bertsch, T. Multiple biomarker strategy for improved diagnosis of acute heart failure in older patients presenting to the emergency department. Acute Cardiovasc. Care 2014. [Google Scholar] [CrossRef]
- Funke-Kaiser, A.; Havulinna, A.S.; Zeller, T.; Appelbaum, S.; Jousilahti, P.; Vartiainen, E.; Blankenberg, S.; Sydow, K.; Salomaa, V. Predictive value of midregional pro-adrenomedullin compared to natriuretic peptides for incident cardiovascular disease and heart failure in the population-based FINRISK 1997 cohort. Ann. Med. 2014, 46, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.L.; Hartman, K.A.; Hodge, D.O.; Hartman, S.; Struck, J.; Morgenthaler, N.G.; Bergmann, A.; Jaffe, A.S. Response of novel biomarkers to BNP infusion in patients with decompensated heart failure: A multimarker paradigm. J. Cardiovasc. Transl. Res. 2009, 2, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, K. Neurohormonal activation in congestive heart failure and the role of vasopressin. Am. J. Cardiol. 2005, 95, 8b–13b. [Google Scholar] [CrossRef] [PubMed]
- Bolignano, D.; Cabassi, A.; Fiaccadori, E.; Ghigo, E.; Pasquali, R.; Peracino, A.; Peri, A.; Plebani, M.; Santoro, A.; Settanni, F.; et al. Copeptin (CTproAVP), a new tool for understanding the role of vasopressin in pathophysiology. Clin. Chem. Lab. Med. 2014, 52, 1447–1456. [Google Scholar] [CrossRef] [PubMed]
- Vasile, V.C.; Jaffe, A.S. Emerging biomarkers for acute heart conditions. Curr. Opin. Cardiol. 2014, 29, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Vetrone, F.; Santarelli, S.; Russo, V.; Lalle, I.; de Berardinis, B.; Magrini, L.; di Stasio, E.; Salerno, G.; Cardelli, P.; Piccoli, A.; et al. Copeptin decrease from admission to discharge has favorable prognostic value for 90-day events in patients admitted with dyspnea. Clin. Chem. Lab. Med. 2014, 52, 1457–1464. [Google Scholar] [CrossRef] [PubMed]
- Loncar, G.; von Haehling, S.; Tahirovic, E.; Inkrot, S.; Mende, M.; Sekularac, N.; Lainscak, M.; Apostolovic, S.; Putnikovic, B.; Edelmann, F.; et al. Effect of beta blockade on natriuretic peptides and copeptin in elderly patients with heart failure and preserved or reduced ejection fraction: Results from the CIBIS-ELD trial. Clin. Biochem. 2012, 45, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Kempf, T.; Horn-Wichmann, R.; Brabant, G.; Peter, T.; Allhoff, T.; Klein, G.; Drexler, H.; Johnston, N.; Wallentin, L.; Wollert, K.C. Circulating concentrations of growth-differentiation factor 15 in apparently healthy elderly individuals and patients with chronic heart failure as assessed by a new immunoradiometric sandwich assay. Clin. Chem. 2007, 53, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Lind, L.; Wallentin, L.; Kempf, T.; Tapken, H.; Quint, A.; Lindahl, B.; Olofsson, S.; Venge, P.; Larsson, A.; Hulthe, J.; et al. Growth-differentiation factor-15 is an independent marker of cardiovascular dysfunction and disease in the elderly: Results from the Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) Study. Eur. Heart J. 2009, 30, 2346–2353. [Google Scholar] [CrossRef] [PubMed]
- Lok, S.I.; Winkens, B.; Goldschmeding, R.; van Geffen, A.J.; Nous, F.M.; van Kuik, J.; van der Weide, P.; Klöpping, C.; Kirkels, J.H.; Lahpor, J.R.; et al. Circulating growth differentiation factor-15 correlates with myocardial fibrosis in patients with non-ischaemic dilated cardiomyopathy and decreases rapidly after left ventricular assist device support. Eur. J. Heart Fail. 2012, 14, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Guo, Y.; Yu, H.; Zheng, L.; Mi, L.; Gao, W. Growth differentiation factor 15 in different stages of heart failure: Potential screening implications. Biomarkers 2010, 15, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Baessler, A.; Strack, C.; Rousseva, E.; Wagner, F.; Bruxmeier, J.; Schmiedel, M.; Riegger, G.; Lahmann, C.; Loew, T.; Schmitz, G.; et al. Growth-differentiation factor-15 improves reclassification for the diagnosis of heart failure with normal ejection fraction in morbid obesity. Eur. J. Heart Fail. 2012, 14, 1240–1248. [Google Scholar] [CrossRef] [PubMed]
- Lok, D.J.; Klip, I.T.; Lok, S.I.; Bruggink-Andre de la Porte, P.W.; Badings, E.; van Wijngaarden, J.; Voors, A.A.; de Boer, R.A.; van Veldhuisen, D.J.; van der Meer, P. Incremental prognostic power of novel biomarkers (growth-differentiation factor-15, high-sensitivity C-reactive protein, galectin-3, and high-sensitivity troponin-T) in patients with advanced chronic heart failure. Am. J. Cardiol. 2013, 112, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Izumiya, Y.; Hanatani, S.; Kimura, Y.; Takashio, S.; Yamamoto, E.; Kusaka, H.; Tokitsu, T.; Rokutanda, T.; Araki, S.; Tsujita, K.; et al. Growth differentiation factor-15 is a useful prognostic marker in patients with heart failure with preserved ejection fraction. Can. J. Cardiol. 2014, 30, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Wollert, K.C.; Kempf, T. Growth differentiation factor 15 in heart failure: An update. Curr. Heart Fail. Rep. 2012, 9, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Stahrenberg, R.; Edelmann, F.; Mende, M.; Kockskamper, A.; Dungen, H.D.; Luers, C.; Binder, L.; Herrmann-Lingen, C.; Gelbrich, G.; Hasenfuss, G.; et al. The novel biomarker growth differentiation factor 15 in heart failure with normal ejection fraction. Eur. J. Heart Fail. 2010, 12, 1309–1316. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.; Sethi, S.K. Biomarkers in cardiorenal syndromes. Transl. Res. 2014, 164, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Shlipak, M.G.; Katz, R.; Fried, L.F.; Jenny, N.S.; Stehman-Breen, C.; Newman, A.B.; Siscovick, D.; Psaty, B.M.; Sarnak, M.J. Cystatin-C and mortality in elderly persons with heart failure. J. Am. Coll. Cardiol. 2005, 45, 268–271. [Google Scholar] [CrossRef] [PubMed]
- Sarnak, M.J.; Katz, R.; Stehman-Breen, C.O.; Fried, L.F.; Jenny, N.S.; Psaty, B.M.; Siscovick, D.; Shlipak, M.G. Cystatin C concentration as a risk factor for heart failure in older adults. Ann. Intern. Med. 2005, 142, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Z.B.; Zhu, L.; Yin, Y.G.; Chen, G.C. Cystatin C, N-terminal probrain natriuretic peptides and outcomes in acute heart failure with acute kidney injury in a 12-month follow-up: Insights into the cardiorenal syndrome. J. Res. Med. Sci. 2014, 19, 404–409. [Google Scholar] [PubMed]
- Gao, C.; Zhong, L.; Gao, Y.; Li, X.; Zhang, M.; Wei, S. Cystatin C levels are associated with the prognosis of systolic heart failure patients. Arch. Cardiovasc. Dis. 2011, 104, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Shlipak, M.G.; Katz, R.; Sarnak, M.J.; Seliger, S.L.; Kestenbaum, B.; Psaty, B.; Tracy, R.P.; Siscovick, D.S. Cystatin C and prognosis for cardiovascular and kidney outcomes in elderly persons without chronic kidney disease. Ann. Intern. Med. 2006, 145, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Stevens, P.E.; Levin, A. Evaluation and management of chronic kidney disease: Synopsis of the kidney disease: Improving global outcomes 2012 clinical practice guideline. Ann. Intern. Med. 2013, 158, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Matana, A.; Zaninović Jurjević, T.; Matana Kaštelan, Z. Can the difference in serum concentration of urea and cystatin C be used in diagnosis and prognosis of heart failure? Med. Hypotheses 2014, 83, 401–403. [Google Scholar] [CrossRef] [PubMed]
- Mishra, J.; Dent, C.; Tarabishi, R.; Mitsnefes, M.M.; Ma, Q.; Kelly, C.; Ruff, S.M.; Zahedi, K.; Shao, M.; Bean, J.; et al. Neutrophil gelatinase associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 2005, 365, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Yndestad, A.; Landro, L.; Ueland, T.; Dahl, C.P.; Flo, T.H.; Vinge, L.E.; Espevik, T.; Frøland, S.S.; Husberg, C.; Christensen, G.; et al. Increased systemic and myocardial expression of neutrophil gelatinaseassociated lipocalin in clinical and experimental heart failure. Eur. Heart J. 2009, 30, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- Devarajan, P. Review: Neutrophil gelatinase-associated lipocalin: A troponin-like biomarker for human acute kidney injury. Nephrology 2010, 15, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Kuster, N.; Moréna, M.; Bargnoux, A.S.; Leray, H.; Chenine, L.; Dupuy, A.M.; Canaud, B.; Cristol, J.P. Biomarkers of cardiorenal syndrome. Ann. Biol. Clin. 2013, 71, 409–418. [Google Scholar]
- Damman, K.; van Veldhuisen, D.J.; Navis, G.; Voors, A.A.; Hillege, H.L. Urinary neutrophil gelatinase associated lipocalin (NGAL), a marker of tubular damage, is increased in patients with chronic heart failure. Eur. J. Heart Fail. 2008, 10, 997–1000. [Google Scholar] [CrossRef] [PubMed]
- Palazzuoli, A.; Ruocco, G.; Beltrami, M.; Franci, B.; Pellegrini, M.; Lucani, B.; Nuti, R.; Ronco, C. Admission plasma neutrophil gelatinase associated lipocalin (NGAL) predicts worsening renal function during hospitalization and post discharge outcome in patients with acute heart failure. Acute Card. Care 2014, 16, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Van Deursen, V.M.; Damman, K.; Voors, A.A.; van der Wal, M.H.; Jaarsma, T.; van Veldhuisen, D.J.; Hillege, H.L. Prognostic value of plasma neutrophil gelatinase-associated lipocalin for mortality in patients with heart failure. Circ. Heart Fail. 2014, 7, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Maisel, A.S.; Mueller, C.; Fitzgerald, R.; Brikhan, R.; Hiestand, B.C.; Iqbal, N.; Clopton, P.; van Veldhuisen, D.J. Prognostic utility of plasma neutrophil gelatinase-associated lipocalin in patients with acute heart failure: The NGAL EvaLuation Along with B-type NaTriuretic Peptide in acutely decompensated heart failure (GALLANT) trial. Eur. J. Heart Fail. 2011, 13, 846–851. [Google Scholar] [CrossRef] [PubMed]
- Aghel, A.; Shrestha, K.; Mullens, W.; Borowski, A.; Tang, W.H. Serum neutrophil gelatinase-associated lipocalin (NGAL) in predicting worsening renal function in acute decompensated heart failure. J. Card. Fail. 2010, 16, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Niebauer, J.; Volk, H.D.; Kemp, M.; Dominguez, M.; Schumann, R.R.; Rauchhaus, M.; Poole-Wilson, P.A.; Coats, A.J.; Anker, S.D. Endotoxin and immune activation in chronic heart failure: A prospective cohort study. Lancet 1999, 353, 1838–1842. [Google Scholar] [CrossRef] [PubMed]
- Sandek, A.; Springer, J.; Habedank, D.; Brunkhorst, F.; Anker, S.D. Procalcitonin-guided antibiotic treatment in heart failure. Lancet 2004, 363, 1555–1556. [Google Scholar] [CrossRef] [PubMed]
- Cinar, O.; Cevik, E.; Acar, A.; Kaya, C.; Ardic, S.; Comert, B.; Yokusoglu, M.; Bilgi, C.; Meisner, M.; Madsen, T. Evaluation of mid-regional pro-atrial natriuretic peptide, procalcitonin, and mid-regional pro-adrenomedullin for the diagnosis and risk stratification of dyspneic ED patients. Am. J. Emerg. Med. 2012, 30, 1915–1920. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, X.; Ge, N.; Liu, J.; Yuan, H.; Zhang, P.; Liu, W.; Wen, D. Procalcitonin testing for diagnosis and short-term prognosis in bacterial infection complicated by congestive heart failure: A multicenter analysis of 4,698 cases. Crit. Care 2014, 18, R4. [Google Scholar] [CrossRef] [PubMed]
- Tromp, J.; van der Pol, A.; Klip, I.J.T.; de Boer, R.A.; Jaarsma, T.; van Gilst, W.H.; Voors, A.A.; van Veldhuisen, D.J.; van der Meer, P. Fibrosis marker syndecan-1 and outcome in patients with heart failure with reduced and preserved ejection fraction. Circ. Heart Fail. 2014, 7, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.; van der Meer, P.; van Deursen, V.M.; Jaarsma, T.; van Veldhuisen, D.J.; van der Wal, M.H.; Hillege, H.L.; Voors, A.A. Neurohormonal and clinical sex differences in heart failure. Eur. Heart J. 2013, 34, 2538–2547. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Ouyang, P.; Zhang, Z.; Lai, W.; Xu, D. Changes and clinical significance of serum level of syndecan-4 protein in patients with chronic congestive heart failure. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2013, 29, 866–869. [Google Scholar] [PubMed]
- Hoste, E.A.; McCullough, P.A.; Kashani, K.; Chawla, L.S.; Joannidis, M.; Shaw, A.D.; Feldkamp, T.; Uettwiller-Geiger, D.L.; McCarthy, P.; Shi, J.; et al. Derivation and validation of cutoffs for clinical use of cell cycle arrest biomarkers. Nephrol. Dial. Transplant. 2014, 29, 2054–2061. [Google Scholar]
- Chugh, S.; Ouzounian, M.; Lu, Z.; Mohamed, S.; Li, W.; Bousette, N.; Liu, P.P.; Gramolini, A.O. Pilot study identifying myosin heavy chain 7, desmin, insulin-like growth factor 7, and annexin A2 as circulating biomarkers of human heart failure. Proteomics 2013, 13, 2324–2334. [Google Scholar] [CrossRef] [PubMed]
- Motiwala, S.R.; Szymonifka, J.; Belcher, A.; Weiner, R.B.; Baggish, A.L.; Gaggin, H.K.; Bhardwaj, A.; Januzzi, J.L., Jr. Measurement of novel biomarkers to predict chronic heart failure outcomes and left ventricular remodeling. J. Cardiovasc. Transl. Res. 2014, 7, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Kudumula, C.R. Regulatory noncoding RNAs in cardiovascular disease: Shedding light on “Dark Matter”. J. Cardiovasc. Dis. 2014, in press. [Google Scholar]
- Santulli, G.; Iaccarino, G.; de Luca, N.; Trimarco, B.; Condorelli, G. Atrial fibrillation and microRNAs. Front. Physiol. 2014, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Kalozoumi, G.; Yacoub, M.; Sanoudou, D. MicroRNAs in heart failure: Small molecules with major impact. Glob. Cardiol. Sci. Pract. 2014, 2014, 79–102. [Google Scholar] [PubMed]
- Braunwald, E. The war against heart failure: The Lancet lecture. Lancet 2014. [Google Scholar] [CrossRef]
- Evans, W.J.; Morley, J.E.; Argiles, J.; Bales, C.; Baracos, V.; Guttridge, D.; Jatoi, A.; Kalantar-Zadeh, K.; Lochs, H.; Mantovani, G.; et al. Cachexia: A new definition. Clin. Nutr. 2008, 7, 793–799. [Google Scholar] [CrossRef]
- Farkas, J.; von Haehling, S.; Kalantar-Zadeh, K.; Morley, J.E.; Anker, S.D.; Lainscak, M. Cachexia as a major public health problem: Frequent, costly, and deadly. J. Cachexia Sarcopenia Muscle 2013, 4, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Okoshi, M.P.; Romeiro, F.G.; Paiva, S.A.; Okoshi, K. Heart failure-induced cachexia. Arq. Bras. Cardiol. 2013, 100, 476–482. [Google Scholar] [PubMed]
- Anker, S.D.; Sharma, R. The syndrome of cardiac cachexia. Int. J. Cardiol. 2002, 85, 51–66. [Google Scholar] [CrossRef] [PubMed]
- Martins, T.; Vitorino, R.; Moreira-Gonçalves, D.; Amado, F.; Duarte, J.A.; Ferreira, R. Recent insights on the molecular mechanisms and therapeutic approaches for cardiac cachexia. Clin. Biochem. 2014, 47, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Sandek, A.; Swidsinski, A.; Schroedl, W.; Watson, A.; Valentova, M.; Herrmann, R.; Scherbakov, N.; Cramer, L.; Rauchhaus, M.; Grosse-Herrenthey, A.; et al. Intestinal blood flow in patients with chronic heart failure: A link with bacterial growth, gastrointestinal symptoms, and cachexia. J. Am. Coll. Cardiol. 2014, 64, 1092–1102. [Google Scholar] [CrossRef] [PubMed]
- Palus, S.; von Haehling, S.; Springer, J. Muscle wasting: An overview of recent developments in basic research. J. Cachexia Sarcopenia Muscle 2014, 5, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Heymsfield, S.B.; Adamek, M.; Gonzalez, M.C.; Jia, G.; Thomas, D.M. Assessing skeletal muscle mass: Historical overview and state of the art. J. Cachexia Sarcopenia Muscle 2014, 5, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Attanasio, P.; Anker, S.D.; Doehner, W.; von Haehling, S. Hormonal consequences and prognosis of chronic heart failure. Curr. Opin. Endocrinol. Diabetes Obes. 2011, 18, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.T.; Kral, J.G. Ghrelin: Integrative neuroendocrine peptide in health and disease. Ann. Surg. 2004, 239, 464–474. [Google Scholar] [CrossRef] [PubMed]
- Strasser, F. Clinical application of ghrelin. Curr. Pharm. Des. 2012, 18, 4800–4812. [Google Scholar] [CrossRef] [PubMed]
- Szabó, T.; Scherbakov, N.; Sandek, A.; Kung, T.; von Haehling, S.; Lainscak, M.; Jankowska, E.A.; Rudovich, N.; Anker, S.D.; Frystyk, J.; et al. Plasma adiponectin in heart failure with and without cachexia: Catabolic signal linking catabolism, symptomatic status, and prognosis. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 50–56. [Google Scholar] [CrossRef] [PubMed]
- McEntegart, M.B.; Awede, B.; Petrie, M.C.; Sattar, N.; Dunn, F.G.; MacFarlane, N.G.; McMurray, J.J. Increase in serum adiponectin concentration in patients with heart failure and cachexia: Relationship with leptin, other cytokines, and B-type natriuretic peptide. Eur. Heart J. 2007, 28, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Loncar, G.; Bozic, B.; von Haehling, S.; Dungen, H.D.; Prodanovic, N.; Lainscak, M.; Arandjelovic, A.; Dimkovic, S.; Radojicic, Z.; Popovic, V. Association of adiponectin with peripheral muscle status in elderly patients with heart failure. Eur. J. Intern. Med. 2013, 24, 818–823. [Google Scholar] [CrossRef] [PubMed]
- Ebner, N.; Steinbeck, L.; Doehner, W.; Anker, S.D.; von Haehling, S. Highlights from the 7th cachexia conference: Muscle wasting pathophysiological detection and novel treatment strategies. J. Cachexia Sarcopenia Muscle 2014, 5, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Steinbeck, L.; Ebner, N.; Valentova, M.; Sandek, A.; Bekfani, T.; Doehner, W.; Anker, S.D.; von Haehling, S. C-terminal agrin fragment as a novel diagnostic marker for muscle wasting in patients with chronic heart failure: Results from the studies investigating co-morbidities aggravating heart failure. J. Cachexia Sarcopenia Muscle 2014, 5, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Lerner, L.; Guillory, B.; Chen, J.; Winston, W.; Weiler, S.; Gyuris, J.; Garcia, J. Growth differentiating factor-15 (GDF-15) induces anorexia and cachexia in mice: A novel pathway for cachexia. J. Cachexia Sarcopenia Muscle 2013, 4, 295–343. [Google Scholar] [CrossRef] [PubMed]
- Martins, T.; Vitorino, R.; Amado, F.; Duarte, J.A.; Ferreira, R. Biomarkers for cardiac cachexia: Reality or utopia. Clin. Chim. Acta 2014, 436, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Fragala, M.S.; Jajtner, A.R.; Beyer, K.S.; Townsend, J.R.; Emerson, N.S.; Scanlon, T.C.; Oliveira, L.P.; Hoffman, J.R.; Stout, J.R. Biomarkers of muscle quality: N-terminal propeptide of type III procollagen and C-terminal agrin fragment responses to resistance exercise training in older adults. J. Cachexia Sarcopenia Muscle 2014, 5, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Bhasin, S.; He, E.J.; Kawakubo, M.; Schroeder, E.T.; Yarasheski, K.; Opiteck, G.J.; Reicin, A.; Chen, F.; Lam, R.; Tsou, J.A.; et al. N-terminal propeptide of type III procollagen as a biomarker of anabolic response to recombinant human GH and testosterone. J. Clin. Endocrinol. Metab. 2009, 94, 4224–4233. [Google Scholar] [CrossRef] [PubMed]
- Nedergaard, A.; Sun, S.; Karsdal, M.A.; Henriksen, K.; Kjaer, M.; Lou, Y.; He, Y.; Zheng, Q.; Suetta, C. Type VI collagen turnover-related peptides-novel serological biomarkers of muscle mass and anabolic response to loading in young men. J. Cachexia Sarcopenia Muscle 2013, 4, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Nedergaard, A.; Karsdal, M.A.; Sun, S.; Henriksen, K. Serological muscle loss biomarkers: An overview of current concepts and future possibilities. J. Cachexia Sarcopenia Muscle 2013, 4, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Scott, I.C.; Tomlinson, W.; Walding, A.; Isherwood, B.; Dougall, I.G. Large-scale isolation of human skeletal muscle satellite cells from post-mortem tissue and development of quantitative assays to evaluate modulators of myogenesis. J. Cachexia Sarcopenia Muscle 2013, 4, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Loncar, G.; Fulster, S.; von Haehling, S.; Popovic, V. Metabolism and the heart: An overview of muscle, fat, and bone metabolism in heart failure. Int. J. Cardiol. 2013, 162, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Stimpson, S.A.; Leonard, M.S.; Clifton, L.G.; Poole, J.C.; Turner, S.M.; Shearer, T.W.; Remlinger, K.S.; Clark, R.V.; Hellerstein, M.K.; Evans, W.J. Longitudinal changes in total body creatine pool size and skeletal muscle mass using the d-creatine dilution method. J. Cachexia Sarcopenia Muscle 2013, 4, 217–223. [Google Scholar] [CrossRef]
- Giannessi, D. Multimarker approach for heart failure management: Perspectives and limitations. Pharmacol. Res. 2011, 64, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Takeishi, Y. Biomarkers in Heart Failure. Int. Heart J. 2014. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loncar, G.; Omersa, D.; Cvetinovic, N.; Arandjelovic, A.; Lainscak, M. Emerging Biomarkers in Heart Failure and Cardiac Cachexia. Int. J. Mol. Sci. 2014, 15, 23878-23896. https://doi.org/10.3390/ijms151223878
Loncar G, Omersa D, Cvetinovic N, Arandjelovic A, Lainscak M. Emerging Biomarkers in Heart Failure and Cardiac Cachexia. International Journal of Molecular Sciences. 2014; 15(12):23878-23896. https://doi.org/10.3390/ijms151223878
Chicago/Turabian StyleLoncar, Goran, Daniel Omersa, Natasa Cvetinovic, Aleksandra Arandjelovic, and Mitja Lainscak. 2014. "Emerging Biomarkers in Heart Failure and Cardiac Cachexia" International Journal of Molecular Sciences 15, no. 12: 23878-23896. https://doi.org/10.3390/ijms151223878
APA StyleLoncar, G., Omersa, D., Cvetinovic, N., Arandjelovic, A., & Lainscak, M. (2014). Emerging Biomarkers in Heart Failure and Cardiac Cachexia. International Journal of Molecular Sciences, 15(12), 23878-23896. https://doi.org/10.3390/ijms151223878




