Lessons Learned about Human Stem Cell Responses to Ionizing Radiation Exposures: A Long Road Still Ahead of Us
Abstract
:1. Introduction
2. Human Stem Cell Radiosensitivity
2.1. Radiosensitivity of Human Embryonic Stem Cells
2.2. Radiosensitivity of Human Mesenchymal Stem Cells
2.3. Radiosensitivity of Human Hematopoietic Stem Cells
2.4. Radiosensitivity of Human Neural Stem Cells
3. Effects of Ionizing Radiation Exposures on the Cell Cycle of Human Stem Cells
3.1. Cell Cycle Alterations in Irradiated Human Embryonic Stem Cells
3.2. Changes in Cell Cycle in Human Mesenchymal Stem Cells Exposed to IR
3.3. Changes in Cell Cycle in Human Hematopoietic Stem Cells Exposed to IR
3.4. Changes in Cell Cycle in Human Neural Stem Cells Exposed to IR
4. Effects of Ionizing Radiation Exposures on DNA Damage Response of Human Stem Cells
4.1. DNA Damage Response in Irradiated Human Embryonic Stem Cells
4.2. DNA Damage response in Irradiated Human Mesenchymal Stem Cells
4.3. DNA Damage Response in Irradiated Human Hematopoietic Stem Cells
5. DNA Repair in Human Stem Cells after Exposures to Ionizing Radiation
5.1. DNA Repair in Irradiated Human Embryonic Stem Cells
5.2. DNA Repair in Irradiated Human Mesenchymal Stem Cells
5.3. DNA Repair in Irradiated Human Hematopoietic Stem Cells
5.4. DNA Repair in Irradiated Human Neural Stem/Progenitor Cells
6. Transcriptional Responses of Cultured Human Stem Cells to Ionizing Radiation
6.1. Changes in Gene Expression in Irradiated Human Embryonic Stem Cells
6.2. Gene Expression Alterations in Irradiated Human Mesenchymal Stem Cells
7. The Role of Epigenetics in Responses of Human Stem Cells to Ionizing Radiation Exposures
8. The Non-Targeted Effects of Ionizing Radiation on Human Stem Cells
9. The Effects of Ionizing Radiation on Human Colon/Intestinal Stem Cells
10. Conclusions
Acknowledgements
Conflict of Interest
References
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar]
- Reubinoff, B.E.; Pera, M.F.; Fong, C.Y.; Trounson, A.; Bongso, A. Embryonic stem cell lines from human blastocysts: Somatic differentiation in vitro. Nat. Biotechnol 2000, 18, 399–404. [Google Scholar]
- Pera, M.F.; Reubinoff, B.; Trounson, A. Human embryonic stem cells. J. Cell Sci 2000, 113, 5–10. [Google Scholar]
- Friedenstein, A.J.; Petrakova, K.V.; Kurolesova, A.I.; Frolova, G.P. Heterotopic of bone marrow. Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation 1968, 6, 230–247. [Google Scholar]
- Paul, G.; Ozen, I.; Christophersen, N.S.; Reinbothe, T.; Bengzon, J.; Visse, E.; Jansson, K.; Dannaeus, K.; Henriques-Oliveira, C.; Roybon, L.; et al. The adult human brain harbors multipotent perivascular mesenchymal stem cells. PLoS One 2012, 7, e35577. [Google Scholar]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar]
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 2008, 3, 301–313. [Google Scholar]
- Covas, D.T.; Panepucci, R.A.; Fontes, A.M.; Silva, W.A., Jr; Orellana, M.D.; Freitas, M.C.; Neder, L.; Santos, A.R.; Peres, L.C.; Jamur, M.C.; et al. Multipotent mesenchymal stromal cells obtained from diverse human tissues share functional properties and gene-expression profile with CD146+ perivascular cells and fibroblasts. Exp. Hematol. 2008, 36, 642–654. [Google Scholar]
- Blocki, A.; Wang, Y.; Koch, M.; Peh, P.; Beyer, S.; Law, P.; Hui, J.H.; Raghunath, M. Not all MSCs can act as pericytes: Functional in vitro assays to distinguish pericytes from other mesenchymal stem cells in angiogenesis. Stem Cells Dev. 2013, in press. [Google Scholar]
- Flores-Figueroa, E.; Varma, S.; Montgomery, K.; Greenberg, P.L.; Gratzinger, D. Distinctive contact between CD34+ hematopoietic progenitors and CXCL12+ CD271+ mesenchymal stromal cells in benign and myelodysplastic bone marrow. Lab. Invest 2012, 92, 1330–1341. [Google Scholar]
- Berardi, A.C.; Wang, A.; Levine, J.D.; Lopez, P.; Scadden, D.T. Functional isolation and characterization of human hematopoietic stem cells. Science 1995, 267, 104–108. [Google Scholar]
- Notta, F.; Doulatov, S.; Laurenti, E.; Poeppl, A.; Jurisica, I.; Dick, J.E. Isolation of single human hematopoietic stem cells capable of long-term multilineage engraftment. Science 2011, 333, 218–221. [Google Scholar]
- Haraguchi, N.; Ishii, H.; Mimori, K.; Ohta, K.; Uemura, M.; Nishimura, J.; Hata, T.; Takemasa, I.; Mizushima, T.; Yamamoto, H.; et al. CD49f-positive cell population efficiently enriches colon cancer-initiating cells. Int. J. Oncol 2013, 43, 425–430. [Google Scholar]
- Gregorieff, A.; Clevers, H. Wnt signaling in the intestinal epithelium: From endoderm to cancer. Genes Dev 2005, 19, 877–890. [Google Scholar]
- Barker, N.; van Oudenaarden, A.; Clevers, H. Identifying the stem cell of the intestinal crypt: Strategies and pitfalls. Cell Stem Cell 2012, 11, 452–460. [Google Scholar]
- Labeed, F.H.; Lu, J.; Mulhall, H.J.; Marchenko, S.A.; Hoettges, K.F.; Estrada, L.C.; Lee, A.P.; Hughes, M.P.; Flanagan, L.A. Biophysical characteristics reveal neural stem cell differentiation potential. PLoS One 2011, 6, e25458. [Google Scholar]
- Sokolov, M.V.; Neumann, R.D. Human embryonic stem cell responses to ionizing radiation exposures: Current state of knowledge and future challenges. Stem Cells Int. 2012, 2012. [Google Scholar] [CrossRef]
- Wilson, K.D.; Sun, N.; Huang, M.; Zhang, W.Y.; Lee, A.S.; Li, Z.; Wang, S.X.; Wu, J.C. Effects of ionizing radiation on self-renewal and pluripotency of human embryonic stem cells. Cancer Res 2010, 70, 5539–5548. [Google Scholar]
- Momcilovic, O.; Choi, S.; Varum, S.; Bakkenist, C.; Schatten, G.; Navara, C. Ionizing radiation induces ataxia telangiectasia mutated-dependent checkpoint signaling and G(2) but not G(1) cell cycle arrest in pluripotent human embryonic stem cells. Stem Cells 2009, 27, 1822–1835. [Google Scholar]
- Filion, T.M.; Qiao, M.; Ghule, P.N.; Mandeville, M.; van Wijnen, A.J.; Stein, J.L.; Lian, J.B.; Altieri, D.C.; Stein, G.S. Survival responses of human embryonic stem cells to DNA damage. J. Cell. Physiol 2009, 220, 586–592. [Google Scholar]
- Sokolov, M.V.; Panyutin, I.V.; Onyshchenko, M.I.; Panyutin, I.G.; Neumann, R.D. Expression of pluripotency-associated genes in the surviving fraction of cultured human embryonic stem cells is not significantly affected by ionizing radiation. Gene 2010, 455, 8–15. [Google Scholar]
- Qin, H.; Yu, T.; Qing, T.; Liu, Y.; Zhao, Y.; Cai, J.; Li, J.; Song, Z.; Qu, X.; Zhou, P.; et al. Regulation of apoptosis and differentiation by p53 in human embryonic stem cells. J. Biol. Chem 2007, 282, 5842–5852. [Google Scholar]
- Xu, X.; Cowley, S.; Flaim, C.J.; James, W.; Seymour, L.; Cui, Z. The roles of apoptotic pathways in the low recovery rate after cryopreservation of dissociated human embryonic stem cells. Biotechnol. Progr 2010, 26, 827–837. [Google Scholar]
- Alekseenko, L.L.; Zemelko, V.I.; Zenin, V.V.; Pugovkina, N.A.; Kozhukharova, I.V.; Kovaleva, Z.V.; Grinchuk, T.M.; Fridlyanskaya, I.I.; Nikolsky, N.N. Heat shock induces apoptosis in human embryonic stem cells but a premature senescence phenotype in their differentiated progeny. Cell Cycle 2012, 11, 3260–3269. [Google Scholar]
- Grandela, C.; Pera, M.F.; Grimmond, S.M.; Kolle, G.; Wolvetang, E.J. p53 is required for etoposide-induced apoptosis of human embryonic stem cells. Stem Cell Res 2007, 1, 116–128. [Google Scholar]
- Madden, D.T.; Davila-Kruger, D.; Melov, S.; Bredesen, D.E. Human embryonic stem cells express elevated levels of multiple pro-apoptotic BCL-2 family members. PLoS One 2011, 6, e28530. [Google Scholar]
- Dumitru, R.; Gama, V.; Fagan, B.M.; Bower, J.J.; Swahari, V.; Pevny, L.H.; Deshmukh, M. Human Embryonic Stem Cells Have Constitutively Active Bax at the Golgi and Are Primed to Undergo Rapid Apoptosis. Mol. Cell 2012, 46, 573–583. [Google Scholar]
- Becker, K.A.; Ghule, P.N.; Therrien, J.A.; Lian, J.B.; Stein, J.L.; van Wijnen, A.J.; Stein, G.S. Self-renewal of human embryonic stem cells is supported by a shortened G1 cell cycle phase. J. Cell. Physiol 2006, 209, 883–893. [Google Scholar]
- Becker, K.A.; Stein, J.L.; Lian, J.B.; van Wijnen, A.J.; Stein, G.S. Human embryonic stem cells are pre-mitotically committed to self-renewal and acquire a lengthened G1 phase upon lineage programming. J. Cell. Physiol. 2010, 222, 103–110. [Google Scholar]
- Kapinas, K.; Grandy, R.; Ghule, P.; Medina, R.; Becker, K.; Pardee, A.; Zaidi, S.K.; Lian, J.; Stein, J.; van Wijnen, A.; et al. The Abbreviated Pluripotent Cell Cycle. J. Cell. Physiol 2013, 228, 9–20. [Google Scholar]
- Desmarais, J.A.; Hoffmann, M.J.; Bingham, G.; Gagou, M.E.; Meuth, M.; Andrews, P.W. Human embryonic stem cells fail to activate CHK1 and commit to apoptosis in response to DNA replication stress. Stem Cells 2012, 30, 1385–1393. [Google Scholar]
- Inniss, K.; Moore, H. Mediation of apoptosis and proliferation of human embryonic stem cells by sphingosine-1-phosphate. Stem Cells Dev 2006, 15, 789–796. [Google Scholar]
- Yang, C.; Atkinson, S.P.; Vilella, F.; Lloret, M.; Armstrong, L.; Mann, D.A.; Lako, M. Opposing putative roles for canonical and noncanonical NFkappaB signaling on the survival, proliferation, and differentiation potential of human embryonic stem cells. Stem Cells 2010, 28, 1970–1980. [Google Scholar]
- Ardehali, R.; Inlay, M.A.; Ali, S.R.; Tang, C.; Drukker, M.; Weissman, I.L. Overexpression of BCL2 enhances survival of human embryonic stem cells during stress and obviates the requirement for serum factors. Proc. Natl. Acad. Sci. USA 2011, 108, 3282–3287. [Google Scholar]
- Bai, H.; Chen, K.; Gao, Y.X.; Arzigian, M.; Xie, Y.L.; Malcosky, C.; Yang, Y.G.; Wu, W.S.; Wang, Z.Z. Bcl-xL enhances single-cell survival and expansion of human embryonic stem cells without affecting self-renewal. Stem Cell Res 2012, 8, 26–37. [Google Scholar]
- Conklin, J.F.; Baker, J.; Sage, J. The RB family is required for the self-renewal and survival of human embryonic stem cells. Nat. Commun 2012, 3, 1244. [Google Scholar]
- Edel, M.J.; Menchon, C.; Menendez, S.; Consiglio, A.; Raya, A.; Izpisua Belmonte, J.C. Rem2 GTPase maintains survival of human embryonic stem cells as well as enhancing reprogramming by regulating p53 and cyclin D1. Genes Dev 2010, 24, 561–573. [Google Scholar] [Green Version]
- Eiselleova, L.; Matulka, K.; Kriz, V.; Kunova, M.; Schmidtova, Z.; Neradil, J.; Tichy, B.; Dvorakova, D.; Pospisilova, S.; Hampl, A.; et al. A complex role for FGF-2 in self-renewal, survival, and adhesion of human embryonic stem cells. Stem Cells 2009, 27, 1847–1857. [Google Scholar]
- Wong, R.C.; Dottori, M.; Koh, K.L.; Nguyen, L.T.; Pera, M.F.; Pebay, A. Gap junctions modulate apoptosis and colony growth of human embryonic stem cells maintained in a serum-free system. Biochem. Biophys. Res. Commun 2006, 344, 181–188. [Google Scholar]
- Wong, R.C.; Tellis, I.; Jamshidi, P.; Pera, M.; Pebay, A. Anti-apoptotic effect of sphingosine-1-phosphate and platelet-derived growth factor in human embryonic stem cells. Stem Cells Dev 2007, 16, 989–1001. [Google Scholar]
- Rieger, K.; Marinets, O.; Fietz, T.; Korper, S.; Sommer, D.; Mucke, C.; Reufi, B.; Blau, W.I.; Thiel, E.; Knauf, W.U. Mesenchymal stem cells remain of host origin even a long time after allogeneic peripheral blood stem cell or bone marrow transplantation. Exp. Hematol. 2005, 33, 605–611. [Google Scholar]
- Chen, M.F.; Lin, C.T.; Chen, W.C.; Yang, C.T.; Chen, C.C.; Liao, S.K.; Liu, J.M.; Lu, C.H.; Lee, K.D. The sensitivity of human mesenchymal stem cells to ionizing radiation. Int. J. Radiat. Oncol. Biol. Phys 2006, 66, 244–253. [Google Scholar]
- Li, J.; Kwong, D.L.; Chan, G.C. The effects of various irradiation doses on the growth and differentiation of marrow-derived human mesenchymal stromal cells. Pediatr. Transplant 2007, 11, 379–387. [Google Scholar]
- Kurpinski, K.; Jang, D.J.; Bhattacharya, S.; Rydberg, B.; Chu, J.; So, J.; Wyrobek, A.; Li, S.; Wang, D. Differential effects of x-rays and high-energy 56Fe ions on human mesenchymal stem cells. Int. J. Radiat. Oncol. Biol. Phys 2009, 73, 869–877. [Google Scholar]
- Wang, D.; Jang, D.J. Protein kinase CK2 regulates cytoskeletal reorganization during ionizing radiation-induced senescence of human mesenchymal stem cells. Cancer Res 2009, 69, 8200–8207. [Google Scholar]
- Cmielova, J.; Havelek, R.; Kohlerova, R.; Soukup, T.; Bruckova, L.; Suchanek, J.; Vavrova, J.; Mokry, J.; Rezacova, M. The effect of ATM kinase inhibition on the initial response of human dental pulp and periodontal ligament mesenchymal stem cells to ionizing radiation. Int. J. Radiat. Biol 2013, 89, 501–511. [Google Scholar]
- Prendergast, A.M.; Cruet-Hennequart, S.; Shaw, G.; Barry, F.P.; Carty, M.P. Activation of DNA damage response pathways in human mesenchymal stem cells exposed to cisplatin or gamma-irradiation. Cell Cycle 2011, 10, 3768–3777. [Google Scholar]
- Oliver, L.; Hue, E.; Sery, Q.; Lafargue, A.; Pecqueur, C.; Paris, F.; Vallette, F.M. Differentiation-related response to DNA breaks in human mesenchymal stem cells. Stem Cells 2012, 31, 800–807. [Google Scholar]
- Serakinci, N.; Christensen, R.; Graakjaer, J.; Cairney, C.J.; Keith, W.N.; Alsner, J.; Saretzki, G.; Kolvraa, S. Ectopically hTERT expressing adult human mesenchymal stem cells are less radiosensitive than their telomerase negative counterpart. Exp. Cell. Res 2007, 313, 1056–1067. [Google Scholar]
- Sokolov, M.V.; Neumann, R.D. Radiation-induced bystander effects in cultured human stem cells. PLoS One 2010, 5, e14195. [Google Scholar]
- Cmielova, J.; Havelek, R.; Soukup, T.; Jiroutova, A.; Visek, B.; Suchanek, J.; Vavrova, J.; Mokry, J.; Muthna, D.; Bruckova, L.; et al. Gamma radiation induces senescence in human adult mesenchymal stem cells from bone marrow and periodontal ligaments. Int. J. Radiat. Biol 2012, 88, 393–404. [Google Scholar]
- Ko, E.; Lee, K.Y.; Hwang, D.S. Human umbilical cord blood-derived mesenchymal stem cells undergo cellular senescence in response to oxidative stress. Stem Cells Dev 2012, 21, 1877–1886. [Google Scholar]
- Bruder, S.P.; Jaiswal, N.; Haynesworth, S.E. Growth kinetics, self-renewal, and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryopreservation. J. Cell. Biochem 1997, 64, 278–294. [Google Scholar]
- Shibata, K.R.; Aoyama, T.; Shima, Y.; Fukiage, K.; Otsuka, S.; Furu, M.; Kohno, Y.; Ito, K.; Fujibayashi, S.; Neo, M.; Nakayama, T.; Nakamura, T.; Toguchida, J. Expression of the p16INK4A gene is associated closely with senescence of human mesenchymal stem cells and is potentially silenced by DNA methylation during in vitro expansion. Stem Cells 2007, 25, 2371–2382. [Google Scholar]
- Scaffidi, P.; Misteli, T. Lamin A-dependent misregulation of adult stem cells associated with accelerated ageing. Nat. Cell Biol 2008, 10, 452–459. [Google Scholar]
- Wagner, W.; Horn, P.; Castoldi, M.; Diehlmann, A.; Bork, S.; Saffrich, R.; Benes, V.; Blake, J.; Pfister, S.; Eckstein, V.; et al. Replicative senescence of mesenchymal stem cells: A continuous and organized process. PLoS One 2008, 3, e2213. [Google Scholar]
- Kim, J.S.; Kim, E.J.; Kim, H.J.; Yang, J.Y.; Hwang, G.S.; Kim, C.W. Proteomic and metabolomic analysis of H2O2-induced premature senescent human mesenchymal stem cells. Exp. Gerontol 2011, 46, 500–510. [Google Scholar]
- Akino, K.; Mineta, T.; Fukui, M.; Fujii, T.; Akita, S. Bone morphogenetic protein-2 regulates proliferation of human mesenchymal stem cells. Wound Repair Regener 2003, 11, 354–360. [Google Scholar]
- Yuan, H.F.; Zhai, C.; Yan, X.L.; Zhao, D.D.; Wang, J.X.; Zeng, Q.; Chen, L.; Nan, X.; He, L.J.; Li, S.T.; et al. SIRT1 is required for long-term growth of human mesenchymal stem cells. J. Mol. Med 2012, 90, 389–400. [Google Scholar]
- Tsai, C.C.; Su, P.F.; Huang, Y.F.; Yew, T.L.; Hung, S.C. Oct4 and Nanog directly regulate Dnmt1 to maintain self-renewal and undifferentiated state in mesenchymal stem cells. Mol. cell 2012, 47, 169–182. [Google Scholar]
- Alves, H.; Munoz-Najar, U.; De Wit, J.; Renard, A.J.; Hoeijmakers, J.H.; Sedivy, J.M.; van Blitterswijk, C.; De Boer, J. A link between the accumulation of DNA damage and loss of multi-potency of human mesenchymal stromal cells. J. Cell. Mol. Med 2010, 14, 2729–2738. [Google Scholar]
- Katoh, O.; Tauchi, H.; Kawaishi, K.; Kimura, A.; Satow, Y. Expression of the vascular endothelial growth factor (VEGF) receptor gene, KDR, in hematopoietic cells and inhibitory effect of VEGF on apoptotic cell death caused by ionizing radiation. Cancer Res 1995, 55, 5687–5692. [Google Scholar]
- Becker, D.; Elsasser, T.; Tonn, T.; Seifried, E.; Durante, M.; Ritter, S.; Fournier, C. Response of human hematopoietic stem and progenitor cells to energetic carbon ions. Int. J. Radiat. Biol 2009, 85, 1051–1059. [Google Scholar]
- Marone, M.; Bonanno, G.; Rutella, S.; Leone, G.; Scambia, G.; Pierelli, L. Survival and cell cycle control in early hematopoiesis: Role of bcl-2, and the cyclin dependent kinase inhibitors P27 and P21. Leuk. Lymphoma 2002, 43, 51–57. [Google Scholar]
- Milyavsky, M.; Gan, O.I.; Trottier, M.; Komosa, M.; Tabach, O.; Notta, F.; Lechman, E.; Hermans, K.G.; Eppert, K.; Konovalova, Z.; et al. A distinctive DNA damage response in human hematopoietic stem cells reveals an apoptosis-independent role for p53 in self-renewal. Cell Stem Cell 2010, 7, 186–197. [Google Scholar]
- Batard, P.; Monier, M.N.; Fortunel, N.; Ducos, K.; Sansilvestri-Morel, P.; Phan, T.; Hatzfeld, A.; Hatzfeld, J.A. TGF-(beta)1 maintains hematopoietic immaturity by a reversible negative control of cell cycle and induces CD34 antigen up-modulation. J. Cell Sci 2000, 113, 383–390. [Google Scholar]
- Yahata, T.; Muguruma, Y.; Yumino, S.; Sheng, Y.; Uno, T.; Matsuzawa, H.; Ito, M.; Kato, S.; Hotta, T.; Ando, K. Quiescent human hematopoietic stem cells in the bone marrow niches organize the hierarchical structure of hematopoiesis. Stem Cells 2008, 26, 3228–3236. [Google Scholar]
- Ducos, K.; Panterne, B.; Fortunel, N.; Hatzfeld, A.; Monier, M.N.; Hatzfeld, J. p21(cip1) mRNA is controlled by endogenous transforming growth factor-beta1 in quiescent human hematopoietic stem/progenitor cells. J. Cell. Physiol 2000, 184, 80–85. [Google Scholar]
- Miyata, Y.; Liu, Y.; Jankovic, V.; Sashida, G.; Lee, J.M.; Shieh, J.H.; Naoe, T.; Moore, M.; Nimer, S.D. Cyclin C regulates human hematopoietic stem/progenitor cell quiescence. Stem Cells 2010, 28, 308–317. [Google Scholar]
- Gibbs, K.D., Jr; Gilbert, P.M.; Sachs, K.; Zhao, F.; Blau, H.M.; Weissman, I.L.; Nolan, G.P.; Majeti, R. Single-cell phospho-specific flow cytometric analysis demonstrates biochemical and functional heterogeneity in human hematopoietic stem and progenitor compartments. Blood 2011, 117, 4226–4233. [Google Scholar]
- Acharya, M.M.; Lan, M.L.; Kan, V.H.; Patel, N.H.; Giedzinski, E.; Tseng, B.P.; Limoli, C.L. Consequences of ionizing radiation-induced damage in human neural stem cells. Free Radical Biol. Med 2010, 49, 1846–1855. [Google Scholar]
- Lan, M.L.; Acharya, M.M.; Tran, K.K.; Bahari-Kashani, J.; Patel, N.H.; Strnadel, J.; Giedzinski, E.; Limoli, C.L. Characterizing the radioresponse of pluripotent and multipotent human stem cells. PLoS One 2012, 7, e50048. [Google Scholar]
- Acharya, M.M.; Christie, L.A.; Lan, M.L.; Giedzinski, E.; Fike, J.R.; Rosi, S.; Limoli, C.L. Human neural stem cell transplantation ameliorates radiation-induced cognitive dysfunction. Cancer Res 2011, 71, 4834–4845. [Google Scholar]
- Acharya, M.M.; Christie, L.A.; Lan, M.L.; Limoli, C.L. Comparing the functional consequences of human stem cell transplantation in the irradiated rat brain. Cell Transplant 2013, 22, 55–64. [Google Scholar]
- Zou, Y.; Zhang, N.; Ellerby, L.M.; Davalos, A.R.; Zeng, X.; Campisi, J.; Desprez, P.Y. Responses of human embryonic stem cells and their differentiated progeny to ionizing radiation. Biochem. Biophys. Res. Commun 2012, 426, 100–105. [Google Scholar]
- Schneider, L.; Fumagalli, M.; d’Adda di Fagagna, F. Terminally differentiated astrocytes lack DNA damage response signaling and are radioresistant but retain DNA repair proficiency. Cell Death Differ 2012, 19, 582–591. [Google Scholar]
- Becker, K.A.; Stein, J.L.; Lian, J.B.; van Wijnen, A.J.; Stein, G.S. Establishment of histone gene regulation and cell cycle checkpoint control in human embryonic stem cells. J. Cell. Physiol 2007, 210, 517–526. [Google Scholar]
- Choi, H.S.; Lee, H.M.; Jang, Y.J.; Kim, C.H.; Ryu, C.J. Heterogeneous Nuclear Ribonucleoprotein A2/B1 Regulates the Selfrenewal and Pluripotency of Human Embryonic Stem Cells via the Control of the G1/S Transition. Stem Cells 2013. [Google Scholar] [CrossRef]
- Qi, J.; Yu, J.Y.; Shcherbata, H.R.; Mathieu, J.; Wang, A.J.; Seal, S.; Zhou, W.; Stadler, B.M.; Bourgin, D.; Wang, L.; et al. microRNAs regulate human embryonic stem cell division. Cell Cycle 2009, 8, 3729–3741. [Google Scholar]
- Ghule, P.N.; Becker, K.A.; Harper, J.W.; Lian, J.B.; Stein, J.L.; van Wijnen, A.J.; Stein, G.S. Cell cycle dependent phosphorylation and subnuclear organization of the histone gene regulator p220(NPAT) in human embryonic stem cells. J. Cell. Physiol 2007, 213, 9–17. [Google Scholar]
- Ghule, P.N.; Dominski, Z.; Yang, X.C.; Marzluff, W.F.; Becker, K.A.; Harper, J.W.; Lian, J.B.; Stein, J.L.; van Wijnen, A.J.; Stein, G.S. Staged assembly of histone gene expression machinery at subnuclear foci in the abbreviated cell cycle of human embryonic stem cells. Proc. Natl. Acad. Sci. USA 2008, 105, 16964–16969. [Google Scholar]
- Medina, R.; Ghule, P.N.; Cruzat, F.; Barutcu, A.R.; Montecino, M.; Stein, J.L.; van Wijnen, A.J.; Stein, G.S. Epigenetic control of cell cycle-dependent histone gene expression is a principal component of the abbreviated pluripotent cell cycle. Mol. Cell. Biol 2012, 32, 3860–3871. [Google Scholar]
- Stein, G.S.; Stein, J.L.; van Wijnen, A.; Lian, J.B.; Montecino, M.; Medina, R.; Kapinas, K.; Ghule, P.; Grandy, R.; Zaidi, S.K.; et al. The architectural organization of human stem cell cycle regulatory machinery. Curr. Pharm. Des 2012, 18, 1679–1685. [Google Scholar]
- Momcilovic, O.; Navara, C.; Schatten, G. Cell cycle adaptations and maintenance of genomic integrity in embryonic stem cells and induced pluripotent stem cells. Results Probl. Cell. Differ 2011, 53, 415–458. [Google Scholar]
- Alessio, N.; Bohn, W.; Rauchberger, V.; Rizzolio, F.; Cipollaro, M.; Rosemann, M.; Irmler, M.; Beckers, J.; Giordano, A.; Galderisi, U. Silencing of RB1 but not of RB2/P130 induces cellular senescence and impairs the differentiation potential of human mesenchymal stem cells. Cell. Mol. Life Sci 2013, 70, 1637–1651. [Google Scholar]
- Adams, B.R.; Golding, S.E.; Rao, R.R.; Valerie, K. Dynamic dependence on ATR and ATM for double-strand break repair in human embryonic stem cells and neural descendants. PLoS One 2010, 5, e10001. [Google Scholar]
- Sokolov, M.V.; Panyutin, I.V.; Panyutin, I.G.; Neumann, R.D. Dynamics of the transcriptome response of cultured human embryonic stem cells to ionizing radiation exposure. Mutat. Res. 2011, 709–710, 40–48. [Google Scholar]
- Bieging, K.T.; Attardi, L.D. Deconstructing p53 transcriptional networks in tumor suppression. Trends Cell Biol 2012, 22, 97–106. [Google Scholar]
- Kuerbitz, S.J.; Plunkett, B.S.; Walsh, W.V.; Kastan, M.B. Wild-type p53 is a cell cycle checkpoint determinant following irradiation. Proc. Natl. Acad. Sci. USA 1992, 89, 7491–7495. [Google Scholar]
- Mantel, C.; Guo, Y.; Lee, M.R.; Kim, M.K.; Han, M.K.; Shibayama, H.; Fukuda, S.; Yoder, M.C.; Pelus, L.M.; Kim, K.S.; et al. Checkpoint-apoptosis uncoupling in human and mouse embryonic stem cells: A source of karyotpic instability. Blood 2007, 109, 4518–4527. [Google Scholar]
- Barta, T.; Vinarsky, V.; Holubcova, Z.; Dolezalova, D.; Verner, J.; Pospisilova, S.; Dvorak, P.; Hampl, A. Human embryonic stem cells are capable of executing G1/S checkpoint activation. Stem Cells 2010, 28, 1143–1152. [Google Scholar]
- Dolezalova, D.; Mraz, M.; Barta, T.; Plevova, K.; Vinarsky, V.; Holubcova, Z.; Jaros, J.; Dvorak, P.; Pospisilova, S.; Hampl, A. MicroRNAs regulate p21(Waf1/Cip1) protein expression and the DNA damage response in human embryonic stem cells. Stem Cells 2012, 30, 1362–1372. [Google Scholar]
- Yahata, T.; Takanashi, T.; Muguruma, Y.; Ibrahim, A.A.; Matsuzawa, H.; Uno, T.; Sheng, Y.; Onizuka, M.; Ito, M.; Kato, S.; et al. Accumulation of oxidative DNA damage restricts the selfrenewal capacity of human hematopoietic stem cells. Blood 2011, 118, 2941–2950. [Google Scholar]
- Svilar, D.; Goellner, E.M.; Almeida, K.H.; Sobol, R.W. Base excision repair and lesion-dependent subpathways for repair of oxidative DNA damage. Antioxid Redox Signal 2011, 14, 2491–2507. [Google Scholar]
- Wilson, D.M., III; Kim, D.; Berquist, B.R.; Sigurdson, A.J. Variation in base excision repair capacity. Mutat. Res. 2011, 711, 100–112. [Google Scholar]
- Vermeulen, W. Dynamics of mammalian NER proteins. DNA Repair 2011, 10, 760–771. [Google Scholar]
- Liu, L.; Lee, J.; Zhou, P. Navigating the nucleotide excision repair threshold. J. Cell. Physiol 2010, 224, 585–589. [Google Scholar]
- Jiricny, J. The multifaceted mismatch-repair system. Nat. Rev. Mol. Cell. Biol 2006, 7, 335–346. [Google Scholar]
- Mladenov, E.; Iliakis, G. Induction and repair of DNA double strand breaks: The increasing spectrum of non-homologous end joining pathways. Mutat. Res 2011, 711, 61–72. [Google Scholar]
- Lieber, M.R. The mechanism of double-strand DNA break repair by the nonhomologous DNA end-joining pathway. Annu. Rev. Biochem 2010, 79, 181–211. [Google Scholar]
- Kass, E.M.; Jasin, M. Collaboration and competition between DNA double-strand break repair pathways. FEBS Lett 2010, 584, 3703–3708. [Google Scholar]
- Holthausen, J.T.; Wyman, C.; Kanaar, R. Regulation of DNA strand exchange in homologous recombination. DNA Repair 2010, 9, 1264–1272. [Google Scholar]
- Kruta, M.; Balek, L.; Hejnova, R.; Dobsakova, Z.; Eiselleova, L.; Matulka, K.; Barta, T.; Fojtik, P.; Fajkus, J.; Hampl, A.; et al. Decrease in abundance of apurinic/apyrimidinic endonuclease causes failure of base excision repair in culture-adapted human embryonic stem cells. Stem Cells 2012, 31, 693–702. [Google Scholar]
- Hyka-Nouspikel, N.; Desmarais, J.; Gokhale, P.J.; Jones, M.; Meuth, M.; Andrews, P.W.; Nouspikel, T. Deficient DNA damage response and cell cycle checkpoints lead to accumulation of point mutations in human embryonic stem cells. Stem Cells 2012, 30, 1901–1910. [Google Scholar]
- Luo, L.Z.; Gopalakrishna-Pillai, S.; Nay, S.L.; Park, S.W.; Bates, S.E.; Zeng, X.; Iverson, L.E.; O’Connor, T.R. DNA repair in human pluripotent stem cells is distinct from that in non-pluripotent human cells. PLoS One 2012, 7, e30541. [Google Scholar]
- Moynahan, M.E.; Jasin, M. Mitotic homologous recombination maintains genomic stability and suppresses tumorigenesis. Nat. Rev. Mol. Cell. Biol 2010, 11, 196–207. [Google Scholar]
- Diderich, K.; Alanazi, M.; Hoeijmakers, J.H. Premature aging and cancer in nucleotide excision repair-disorders. DNA Repair 2011, 10, 772–780. [Google Scholar]
- Maynard, S.; Swistowska, A.M.; Lee, J.W.; Liu, Y.; Liu, S.T.; da Cruz, A.B.; Rao, M.; de Souza-Pinto, N.C.; Zeng, X.; Bohr, V.A. Human embryonic stem cells have enhanced repair of multiple forms of DNA damage. Stem Cells 2008, 26, 2266–2274. [Google Scholar]
- Adams, B.R.; Hawkins, A.J.; Povirk, L.F.; Valerie, K. ATM-independent, high-fidelity nonhomologous end joining predominates in human embryonic stem cells. Aging (Albany NY) 2010, 2, 582–596. [Google Scholar]
- Fung, H.; Weinstock, D.M. Repair at single targeted DNA double-strand breaks in pluripotent and differentiated human cells. PLoS One 2011, 6, e20514. [Google Scholar]
- Bogomazova, A.N.; Lagarkova, M.A.; Tskhovrebova, L.V.; Shutova, M.V.; Kiselev, S.L. Error-prone nonhomologous end joining repair operates in human pluripotent stem cells during late G2. Aging (Albany NY) 2011, 3, 584–596. [Google Scholar]
- Worku, M.; Fersht, N.; Martindale, C.; Funes, J.M.; Short, S.C. Sequential Transformation of Mesenchymal Stem Cells is Associated with Increased Radiosensitivity and Reduced DNA Repair Capacity. Radiat. Res 2013, 179, 698–706. [Google Scholar]
- Mohrin, M.; Bourke, E.; Alexander, D.; Warr, M.R.; Barry-Holson, K.; Le Beau, M.M.; Morrison, C.G.; Passegue, E. Hematopoietic stem cell quiescence promotes error-prone DNA repair and mutagenesis. Cell Stem Cell 2010, 7, 174–185. [Google Scholar]
- de Laval, B.; Pawlikowska, P.; Petit-Cocault, L.; Bilhou-Nabera, C.; Aubin-Houzelstein, G.; Souyri, M.; Pouzoulet, F.; Gaudry, M.; Porteu, F. Thrombopoietin-increased DNA-PK-dependent DNA repair limits hematopoietic stem and progenitor cell mutagenesis in response to DNA damage. Cell Stem Cell 2013, 12, 37–48. [Google Scholar]
- Rieger, K.E.; Chu, G. Portrait of transcriptional responses to ultraviolet and ionizing radiation in human cells. Nucleic Acids Res 2004, 32, 4786–4803. [Google Scholar]
- Sokolov, M.; Panyutin, I.G.; Neumann, R. Genome-wide gene expression changes in normal human fibroblasts in response to low-LET gamma-radiation and high-LET-like 125IUdR exposures. Radiat. Prot. Dosim 2006, 122, 195–201. [Google Scholar]
- Sokolov, M.V.; Smirnova, N.A.; Camerini-Otero, R.D.; Neumann, R.D.; Panyutin, I.G. Microarray analysis of differentially expressed genes after exposure of normal human fibroblasts to ionizing radiation from an external source and from DNA-incorporated iodine-125 radionuclide. Gene 2006, 382, 47–56. [Google Scholar]
- Amundson, S.A.; Do, K.T.; Fornace, A.J., Jr. Induction of stress genes by low doses of gamma rays. Radiat. Res. 1999, 152, 225–231. [Google Scholar]
- Rodningen, O.K.; Overgaard, J.; Alsner, J.; Hastie, T.; Borresen-Dale, A.L. Microarray analysis of the transcriptional response to single or multiple doses of ionizing radiation in human subcutaneous fibroblasts. Radiother. Oncol 2005, 77, 231–240. [Google Scholar]
- Jin, Y.W.; Na, Y.J.; Lee, Y.J.; An, S.; Lee, J.E.; Jung, M.; Kim, H.; Nam, S.Y.; Kim, C.S.; Yang, K.H.; et al. Comprehensive analysis of time- and dose-dependent patterns of gene expression in a human mesenchymal stem cell line exposed to low-dose ionizing radiation. Oncol. Rep 2008, 19, 135–144. [Google Scholar]
- Calvanese, V.; Fraga, M.F. Epigenetics of embryonic stem cells. Adv. Exp. Med. Biol 2012, 741, 231–253. [Google Scholar]
- Mattout, A.; Meshorer, E. Chromatin plasticity and genome organization in pluripotent embryonic stem cells. Curr. Opin. Cell Biol 2010, 22, 334–341. [Google Scholar]
- Meissner, A. Epigenetic modifications in pluripotent and differentiated cells. Nat. Biotechnol 2010, 28, 1079–1088. [Google Scholar]
- Bibikova, M.; Chudin, E.; Wu, B.; Zhou, L.; Garcia, E.W.; Liu, Y.; Shin, S.; Plaia, T.W.; Auerbach, J.M.; Arking, D.E.; et al. Human embryonic stem cells have a unique epigenetic signature. Genome Res 2006, 16, 1075–1083. [Google Scholar]
- Altun, G.; Loring, J.F.; Laurent, L.C. DNA methylation in embryonic stem cells. J. Cell. Biochem 2010, 109, 1–6. [Google Scholar]
- Bock, C.; Kiskinis, E.; Verstappen, G.; Gu, H.; Boulting, G.; Smith, Z.D.; Ziller, M.; Croft, G.F.; Amoroso, M.W.; Oakley, D.H.; et al. Reference Maps of human ES and iPS cell variation enable high-throughput characterization of pluripotent cell lines. Cell 2011, 144, 439–452. [Google Scholar]
- Chen, P.Y.; Feng, S.; Joo, J.W.; Jacobsen, S.E.; Pellegrini, M. A comparative analysis of DNA methylation across human embryonic stem cell lines. Genome Biol 2011, 12, R62. [Google Scholar]
- Laurent, L.C.; Chen, J.; Ulitsky, I.; Mueller, F.J.; Lu, C.; Shamir, R.; Fan, J.B.; Loring, J.F. Comprehensive microRNA profiling reveals a unique human embryonic stem cell signature dominated by a single seed sequence. Stem Cells 2008, 26, 1506–1516. [Google Scholar]
- Aranda, P.; Agirre, X.; Ballestar, E.; Andreu, E.J.; Roman-Gomez, J.; Prieto, I.; Martin-Subero, J.I.; Cigudosa, J.C.; Siebert, R.; Esteller, M.; et al. Epigenetic signatures associated with different levels of differentiation potential in human stem cells. PLoS One 2009, 4, e7809. [Google Scholar]
- Dasa, D.; Marek, M.; Tomas, B.; Karla, P.; Vladimir, V.; Zuzana, H.; Josef, J.; Petr, D.; Sarka, P.; Ales, H. MicroRNAs Regulate p21(Waf1/cip1) Protein Expression and the DNA Damage Response in Human Embryonic Stem Cells. Stem Cells 2012, 30, 1362–1372. [Google Scholar]
- Sokolov, M.V.; Panyutin, I.V.; Neumann, R.D. Unraveling the global microRNAome responses to ionizing radiation in human embryonic stem cells. PLoS One 2012, 7, e31028. [Google Scholar]
- Cheasley, D.; Pereira, L.; Lightowler, S.; Vincan, E.; Malaterre, J.; Ramsay, R.G. Myb controls intestinal stem cell genes and self-renewal. Stem Cells 2011, 29, 2042–2050. [Google Scholar]
- Yan, K.S.; Chia, L.A.; Li, X.; Ootani, A.; Su, J.; Lee, J.Y.; Su, N.; Luo, Y.; Heilshorn, S.C.; Amieva, M.R.; et al. The intestinal stem cell markers Bmi1 and Lgr5 identify two functionally distinct populations. Proc. Natl. Acad. Sci. USA 2012, 109, 466–471. [Google Scholar]
- Otsuka, K.; Hamada, N.; Magae, J.; Matsumoto, H.; Hoshi, Y.; Iwasaki, T. Ionizing Radiation Leads to the Replacement and de novo Production of Colonic Lgr5 Stem Cells. Radiat. Res 2013, 179, 637–646. [Google Scholar]
- Walker, F.; Zhang, H.H.; Odorizzi, A.; Burgess, A.W. LGR5 is a negative regulator of tumourigenicity, antagonizes Wnt signalling and regulates cell adhesion in colorectal cancer cell lines. PLoS One 2011, 6, e22733. [Google Scholar]
- Plaks, V.; Brenot, A.; Lawson, D.A.; Linnemann, J.R.; Van Kappel, E.C.; Wong, K.C.; de Sauvage, F.; Klein, O.D.; Werb, Z. Lgr5-expressing cells are sufficient and necessary for postnatal mammary gland organogenesis. Cell Rep 2013, 3, 70–78. [Google Scholar]
- Van der Flier, L.G.; van Gijn, M.E.; Hatzis, P.; Kujala, P.; Haegebarth, A.; Stange, D.E.; Begthel, H.; van den Born, M.; Guryev, V.; Oving, I.; et al. Transcription factor achaete scute-like 2 controls intestinal stem cell fate. Cell 2009, 136, 903–912. [Google Scholar]
- Zhu, R.; Yang, Y.; Tian, Y.; Bai, J.; Zhang, X.; Li, X.; Peng, Z.; He, Y.; Chen, L.; Pan, Q.; et al. Ascl2 knockdown results in tumor growth arrest by miRNA-302b-related inhibition of colon cancer progenitor cells. PLoS One 2012, 7, e32170. [Google Scholar]
- Hua, G.; Thin, T.H.; Feldman, R.; Haimovitz-Friedman, A.; Clevers, H.; Fuks, Z.; Kolesnick, R. Crypt base columnar stem cells in small intestines of mice are radioresistant. Gastroenterology 2012, 143, 1266–1276. [Google Scholar]
- Qiu, W.; Leibowitz, B.; Zhang, L.; Yu, J. Growth factors protect intestinal stem cells from radiation-induced apoptosis by suppressing PUMA through the PI3K/AKT/p53 axis. Oncogene 2010, 29, 1622–1632. [Google Scholar]
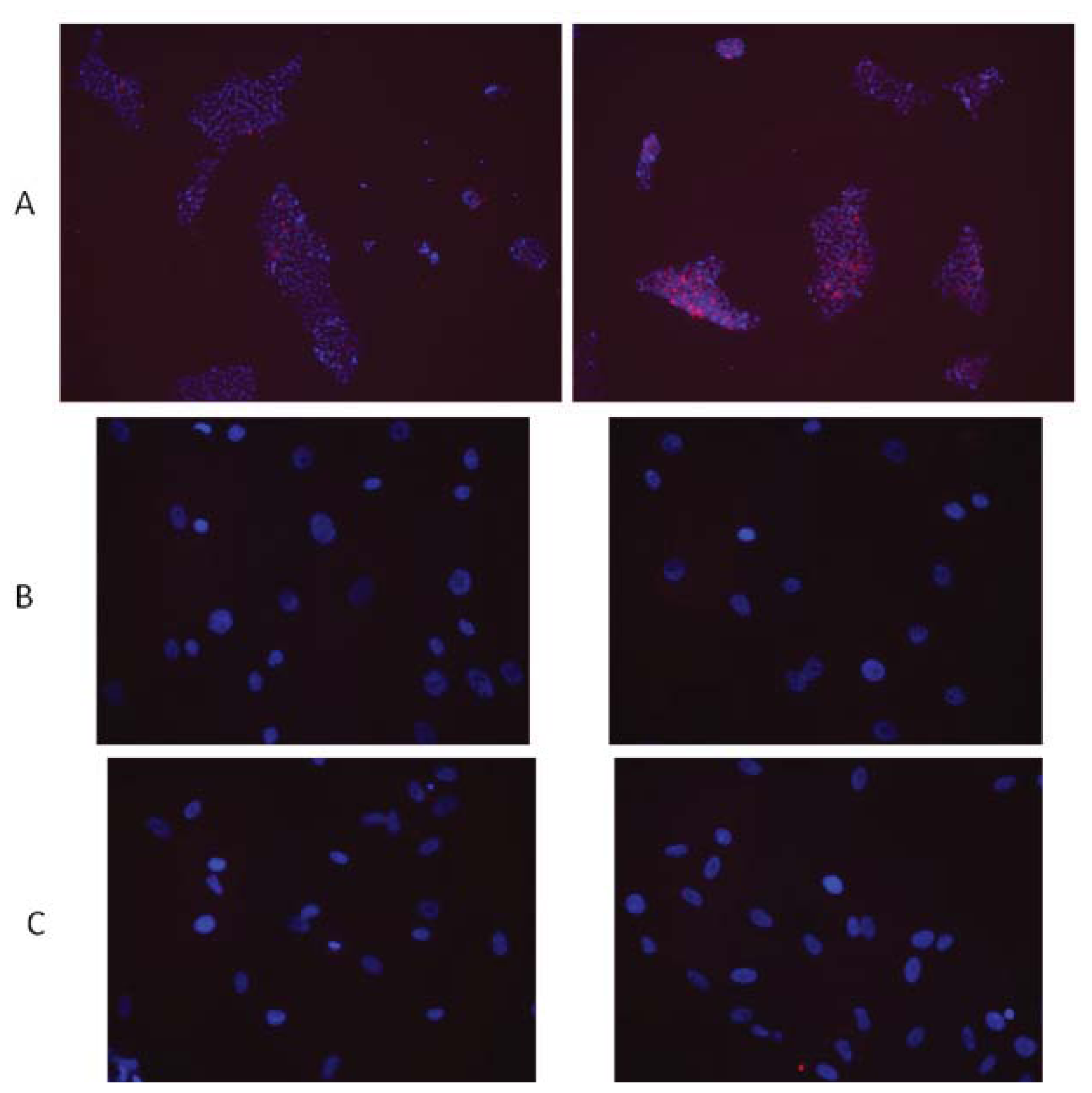
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sokolov, M.; Neumann, R. Lessons Learned about Human Stem Cell Responses to Ionizing Radiation Exposures: A Long Road Still Ahead of Us. Int. J. Mol. Sci. 2013, 14, 15695-15723. https://doi.org/10.3390/ijms140815695
Sokolov M, Neumann R. Lessons Learned about Human Stem Cell Responses to Ionizing Radiation Exposures: A Long Road Still Ahead of Us. International Journal of Molecular Sciences. 2013; 14(8):15695-15723. https://doi.org/10.3390/ijms140815695
Chicago/Turabian StyleSokolov, Mykyta, and Ronald Neumann. 2013. "Lessons Learned about Human Stem Cell Responses to Ionizing Radiation Exposures: A Long Road Still Ahead of Us" International Journal of Molecular Sciences 14, no. 8: 15695-15723. https://doi.org/10.3390/ijms140815695
APA StyleSokolov, M., & Neumann, R. (2013). Lessons Learned about Human Stem Cell Responses to Ionizing Radiation Exposures: A Long Road Still Ahead of Us. International Journal of Molecular Sciences, 14(8), 15695-15723. https://doi.org/10.3390/ijms140815695



