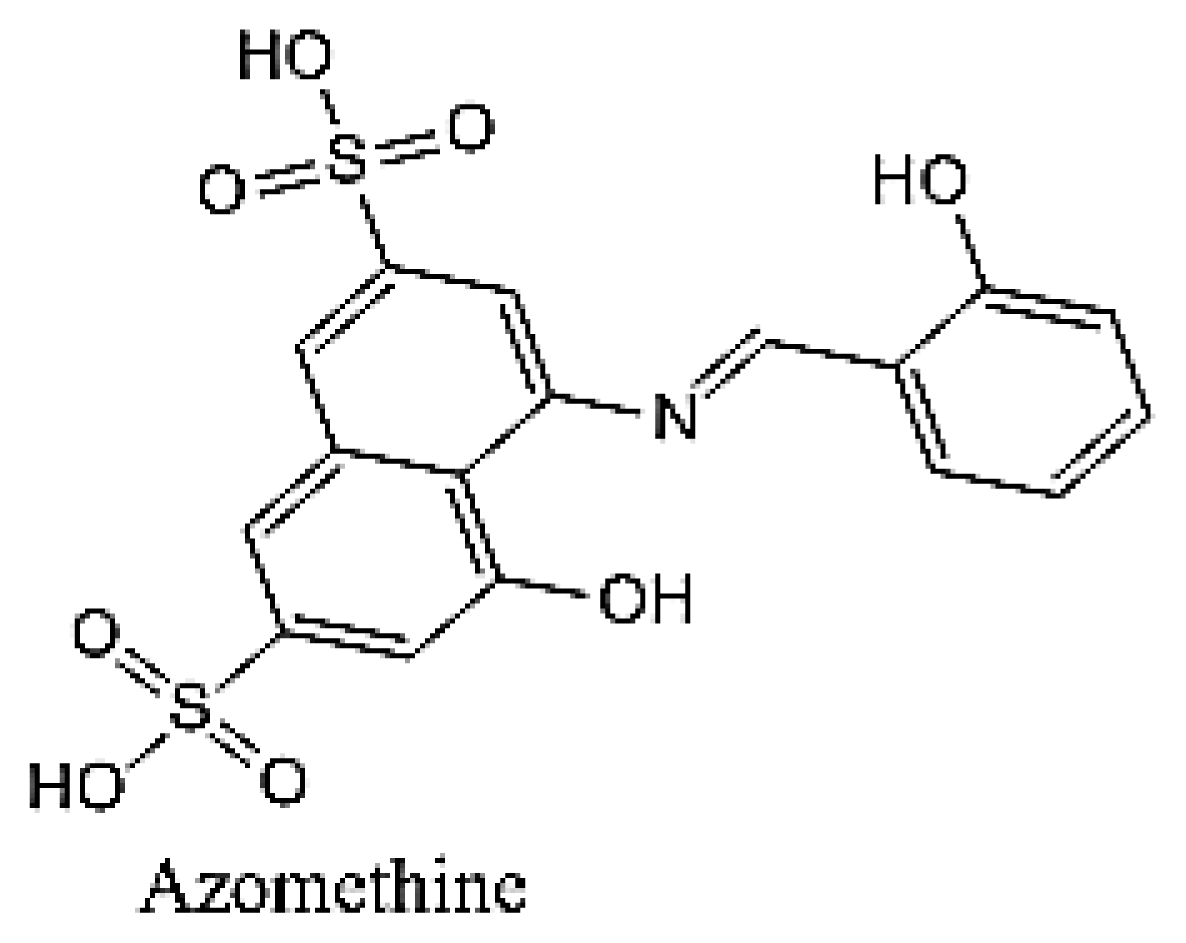
Synthesis and Characterization of the Inclusion Complex of β-cyclodextrin and Azomethine
Abstract
:1. Introduction
2. Results and Discussion
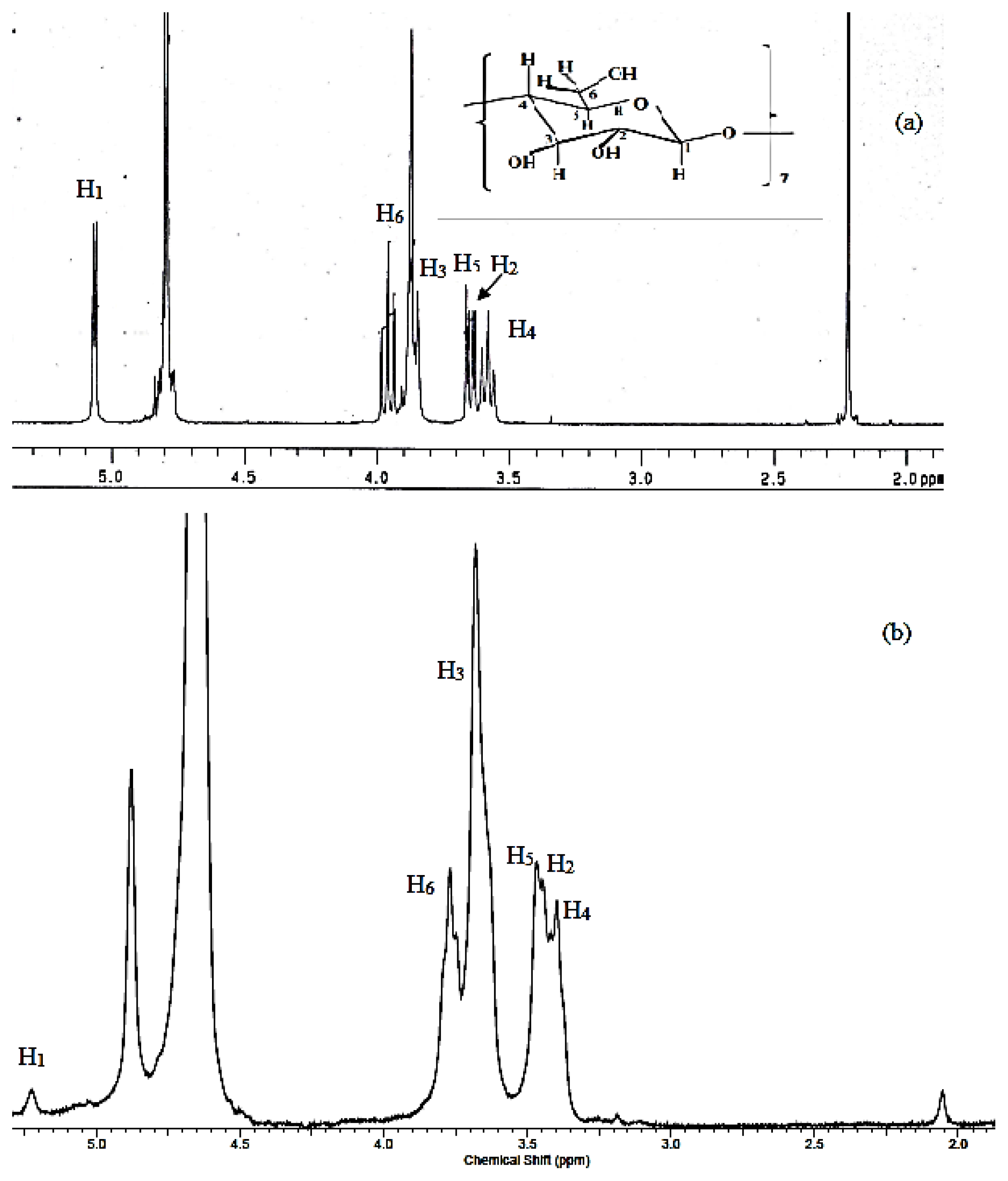
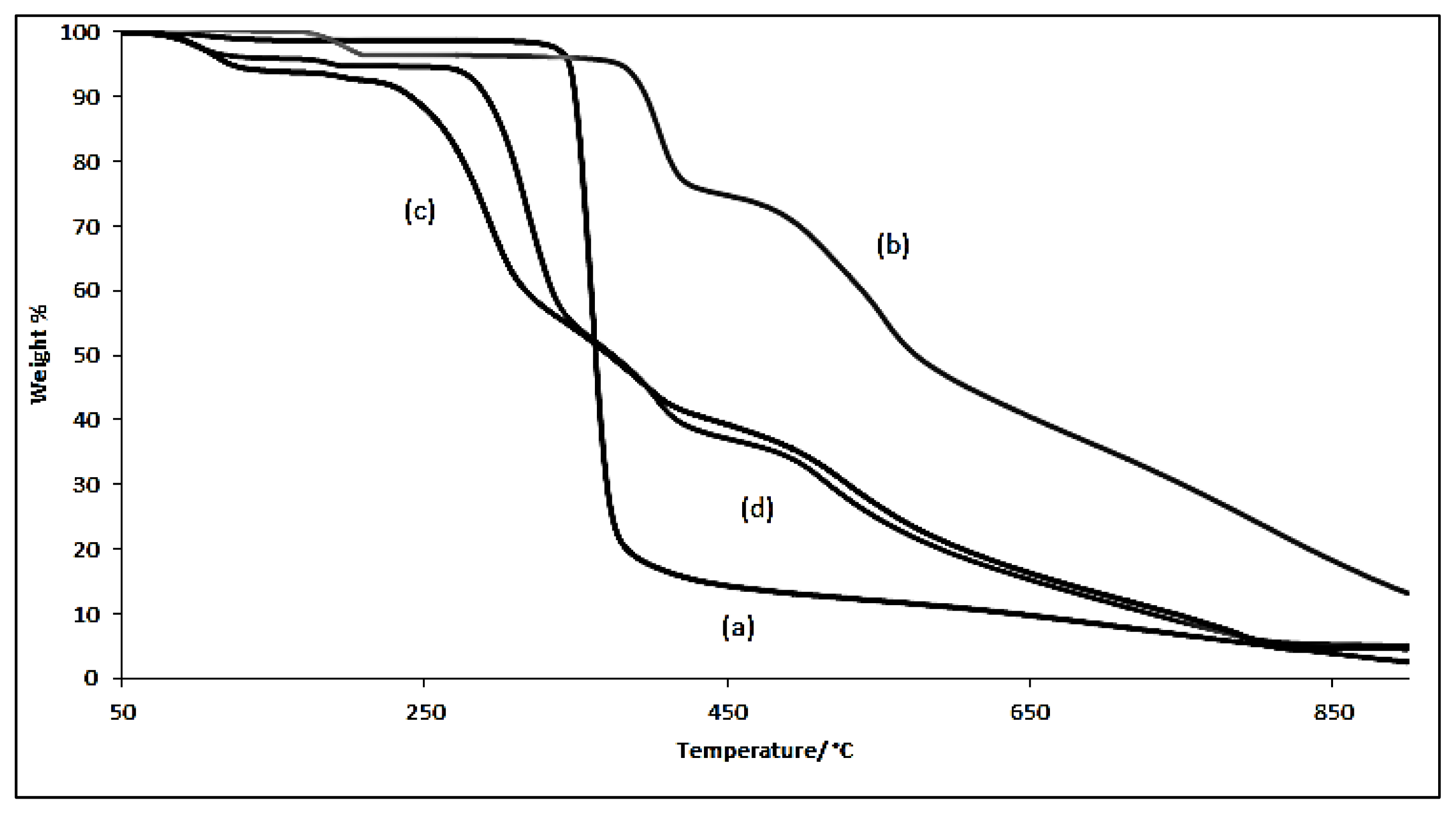
2.1. Characterizations
2.2. Spectroscopic Studies
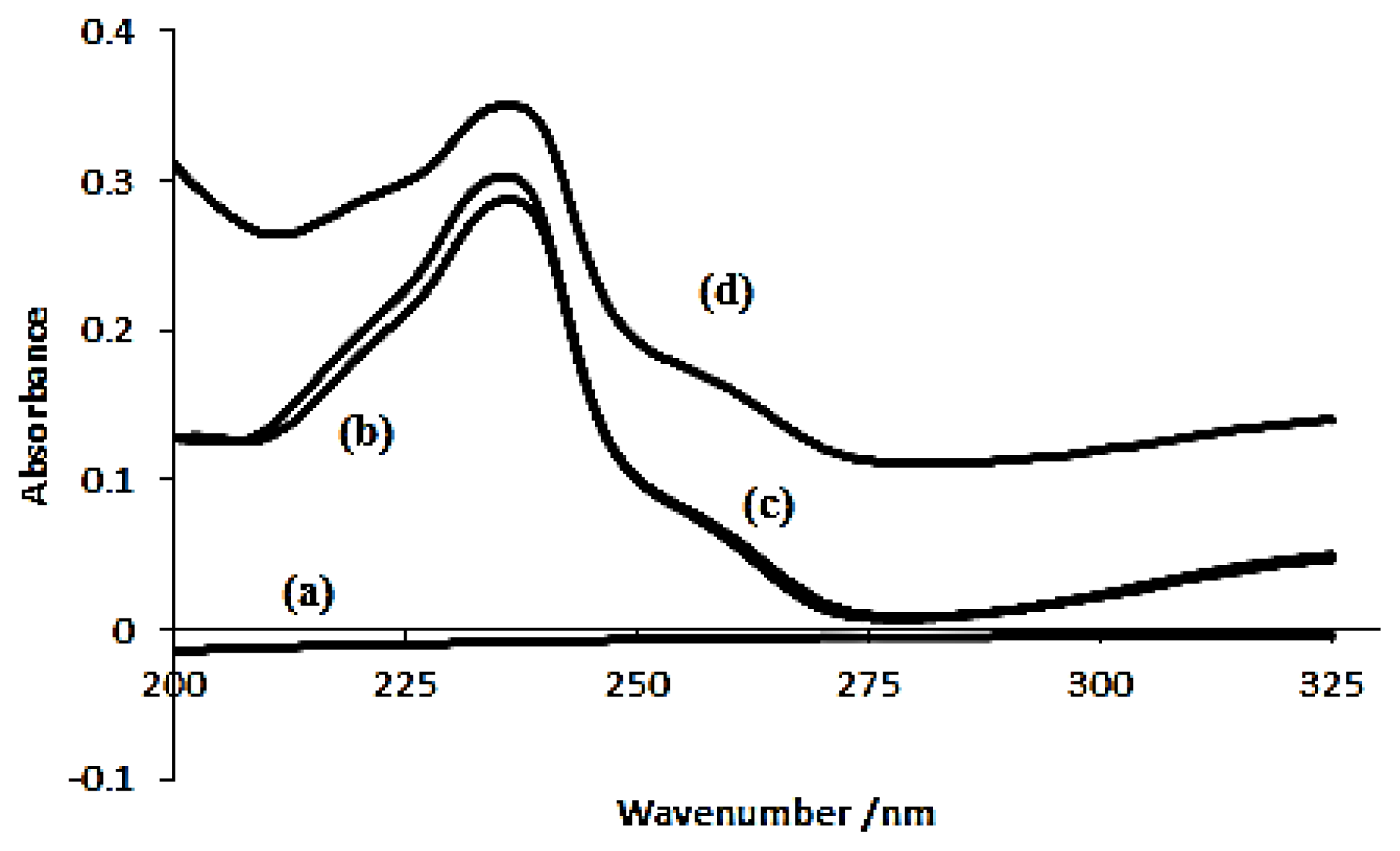
2.2.1. Effect of pH
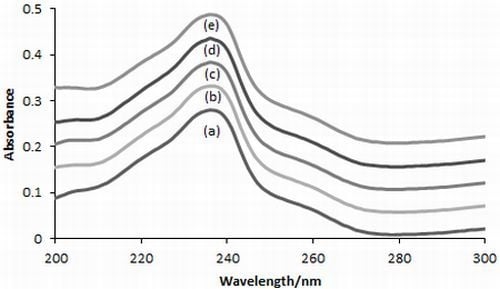
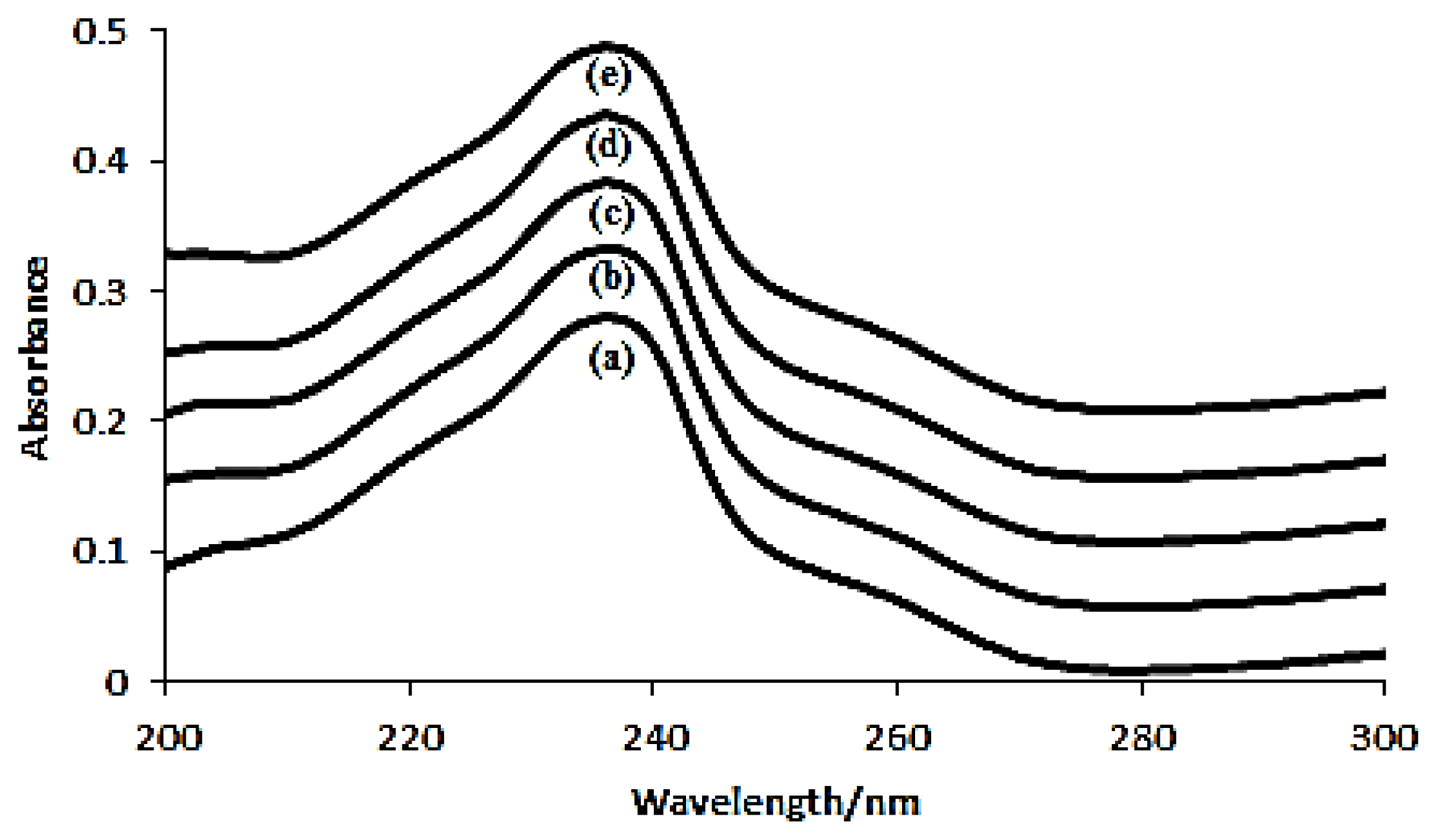
2.2.2. Absorption Spectra
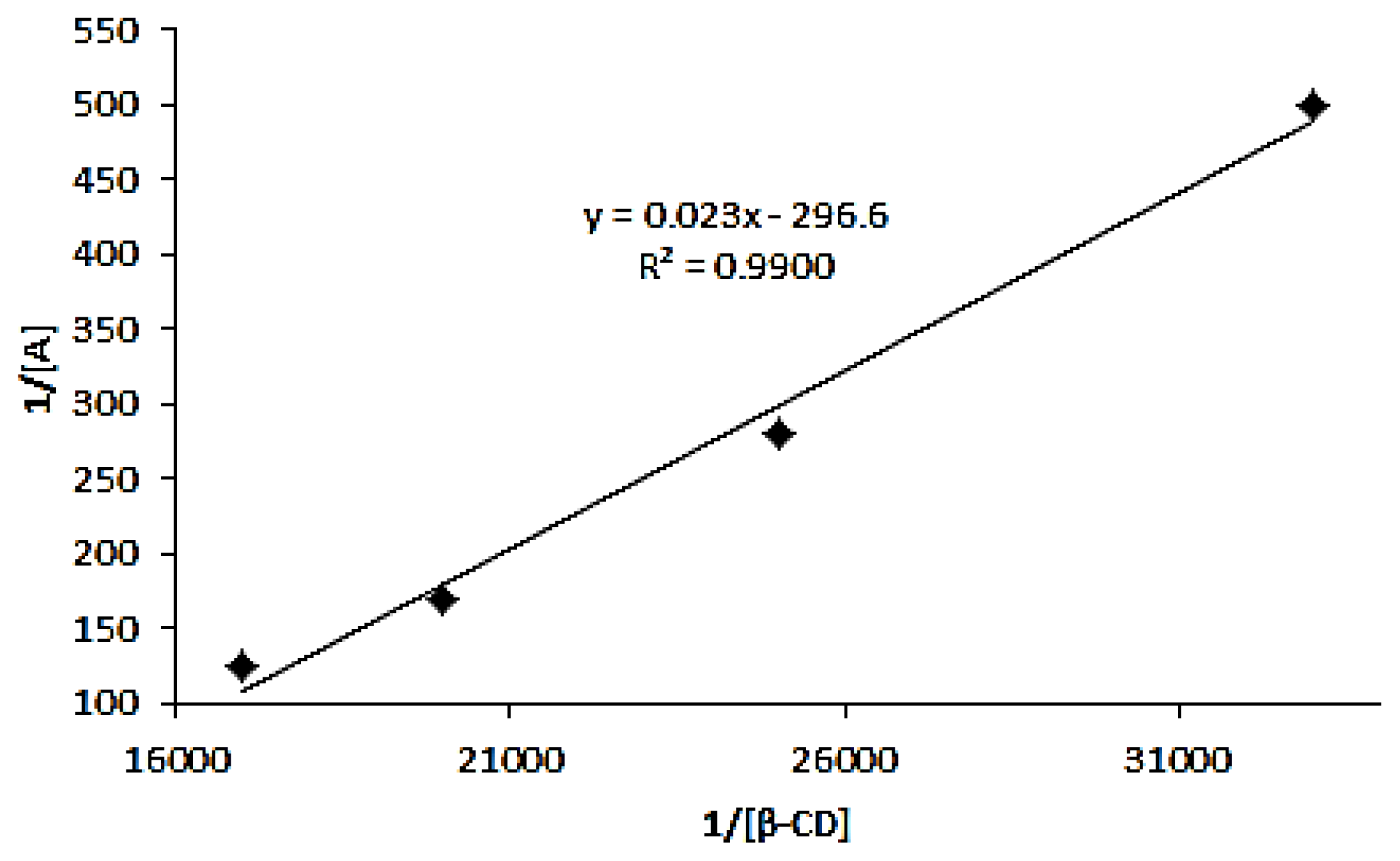
2.2.3. Stoichiometry and Apparent Formation Constant for Azomethine-β-Cyd Inclusion Complex
3. Experimental Section
3.1. Reagents and Solutions
3.2. Synthesis of the Inclusion Complex of β-Cyd and Azomethine
3.3. Characterizations
3.4. Spectroscopic Studies
3.4.1. Effect of pH
3.4.2. Absorption Spectra
3.4.3. Formation Constant
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Connors, K.A. The stability of cyclodextrin complexes in solution. Chem. Rev 1997, 97, 1325–1358. [Google Scholar]
- Szejtli, J. Introduction and general overview of cyclodextrin chemistry. Chem. Rev 1998, 98, 1743–1753. [Google Scholar]
- Lee, C.-W.; Kim, S.-J.; Youn, Y-.S.; Widjojokusumo, E.; Lee, Y-H.; Kim, J.; Lee, Y-.W.; Tjandrawinata, R.R. Preparation of bitter taste masked cetirizine dihydrochloride/β-cyclodextrin inclusion complex by supercritical antisolvent (SAS) process. J. Supercrit. Fluids 2010, 55, 348–357. [Google Scholar]
- Zhu, X.; Sun, J.; Wu, J. Study on the inclusion interactions of β-cyclodextrin and its derivative with dyes by spectrofluorimetry and its analytical application. Talanta 2007, 72, 237–242. [Google Scholar]
- Nic, M.; Jirat, J.; Kosata, B. IUPAC, Compendium of Chemical Terminology; Blackwell Scientific Publications: Oxford, UK, 1997. [Google Scholar] [CrossRef]
- Namkung, M.J.; Fletcher, T.L. Derivatives of 2-hydroxy-p-phenetidine. I. Azomethines. J. Med. Chem 1970, 13, 329. [Google Scholar]
- Smith, H.E.; Chen, F.-M. Optically Active Amines 27. CNDO/S calculations on the azomethine and conjugated azomethine chromophores. J. Org. Chem. 1979, 44, 2775–2779. [Google Scholar]
- Morgan, P.W.; Kwolek, S.L.; Terry, C. Aromatic azomethine polymers and fibers. Macromolecules 1987, 20, 729–739. [Google Scholar]
- Catanescu, O.; Grigoras, M.; Colotin, G.; Dobreanu, A.; Hurduc, N.; Simionescu, C.I. Synthesis and characterization of some aliphatic-aromatic poly (Schiff base)s. Eur. Polym. J 2001, 37, 2213–2216. [Google Scholar]
- Dutta, P.K.; Jain, P.; Sen, P.; Trivedi, R.; Sen, P.K.; Dutta, J. Synthesis and characterization of novel polyazomethine ether for NLO application. Eur. Polym. J 2003, 39, 1007–1011. [Google Scholar]
- Ribera, D.A.; Manteco’n, A.; Serra, A. Synthesis and crosslinking of a series of dimeric liquid-crystalline diglycidylester compounds containing imine groups. J. Polym. Sci. A 2002, 40, 4344–4356. [Google Scholar]
- Nepal, D.; Samal, S.; Geckeler, K.E. The first fullerene-terminated soluble poly(azomethine) rotaxene. Macromolecules 2003, 36, 3800–3802. [Google Scholar]
- Tang, B.; Liu, W.-L.; Wang, Y.; Chen, Z.-Z. Studies on the supramolecular interaction between napropamide and β-cyclodextrin by spectrofluorimetry and its analytical application. Anal. Chim. Acta 2004, 509, 145–150. [Google Scholar]
- Jiao, H.; Goh, S.H.; Valiyaveettil, S. Inclusion complexes of poly(neopentyl glycol sebacate) with cyclodextrins. Macromolecules 2011, 34, 8138–8142. [Google Scholar]
- Li, N.; Liu, J.; Zhao, X.; Gao, Y.; Zhang, L.; Zhang, J.; Yu, L. Complex formation of ionic liquid surfactant and β-cyclodextrin. Colloids Surf. A 2007, 292, 196–201. [Google Scholar]
- Li, W.; Lu, B.; Sheng, A.; Yang, F.; Wang, Z. Spectroscopic and theoretical study on inclusion complexation of beta-cyclodextrin with permethrin. J. Mol. Struct 2010, 981, 194–203. [Google Scholar]
- Li, J.; Ni, X.; Zhou, Z.; Leong, K.W. Preparation and characterization of polypseudorotaxanes based on block-selected inclusion complexation between poly(propylene oxide)-poly(ethylene oxide)-poly(propyleneoxide) triblock copolymers and α-cyclodextrin. J. Am. Chem. Soc 2003, 125, 1788–1795. [Google Scholar]
- Rusa, C.C.; Luca, C.; Tonelli, A.E. Polymer-cyclodextrin inclusion compounds: Toward new aspects of their inclusion mechanism. Macromolecules 2001, 34, 1318–1322. [Google Scholar]
- Tang, B.; Chen, Z.-Z.; Zhang, N.; Zhang, J.; Wang, Y. Synthesis and characterization of a novel cross-linking complex of β-cyclodextrin-o-vanilin furfaralhydrazone and highly selective spectrofluorimetric determination of trace gallium. Talanta 2006, 68, 575–580. [Google Scholar]
- Hamidi, H.; Abderrahim, R.; Meganem, F. Spectroscopic studies of inclusion complex of β-cyclodextrin and benzidine diammonium dipicrate. Spectrochim. Acta A 2010, 75, 32–36. [Google Scholar]
- Djedaini, F.; Lin, S.Z.; Perly, B.; Wouessidjewe, D. High-field nuclear magnetic resonance techniques for the investigation of a β-cyclodextrin:indomethacin inclusion complex. J. Pharm. Sci 1990, 79, 643–646. [Google Scholar]
- Fernandes, C.M.; Carvalho, R.A.; Pereira da Costa, S.; Veiga, F.J.B. Multimodal molecular encapsulation of nicardipine hydrochloride by β-cyclodextrin, hydroxypropyl-β-cyclodextrin and triacetyl-β-cyclodextrin in solution. Structural studies by 1H NMR and ROESY experiments. Eur. J. Pharm. Sci 2003, 18, 285–296. [Google Scholar]
- Youming, Z.; Xinrong, D.; Liangcheng, W.; Taibao, W. Synthesis and characterization of inclusion complexes of aliphatic-aromatic poly(Schiff base)s with β-cyclodextrin (highlight). J. Incl. Phenom. Macrocycl. Chem 2008, 60, 313–319. [Google Scholar]
- Chen, M.; Diao, G.; Zhang, E. Study of inclusion complex of β-cyclodextrin and nitrobenzene. Chemosphere 2006, 63, 522–529. [Google Scholar]
- Harata, K. The structure of the cyclodextrin complex. IV. The crystal structure of α-cyclodextrin-sodium 1-propanesulfonate nonahydrate. Bull. Chem. Soc. Jpn 1977, 50, 1259–1266. [Google Scholar]
- Saenger, W. Crystal packing patterns of cyclodextrin inclusion complexes. J. Incl. Phenom. Macrocycl. Chem 1984, 2, 445–454. [Google Scholar]
- Wang, J.; Cao, Y.; Sun, B.; Wang, C. Physicochemical and release characterization of garlic oil-β-cyclodextrin inclusion complexes. Food Chem 2011, 127, 1680–1685. [Google Scholar]
- Fernandes, J.A.; Lima, S.; Braga, S.S.; Claro, P.R.; Borges, J.E.R.; Teixeira, C.; Pilinger, M.; Dias, J.C.C.T.; Goncalves, I.S. Inclusion Complex formation of diferrocenyldimethylsilane with β-cyclodextrin. J. Organomet Chem 2005, 690, 4801–4808. [Google Scholar]
- Salustio, P.J.; Feio, G.; Figueirinhas, J.L.; Pinto, J.F.; Marques, H.M.C. The influence of the preparation methods on the inclusion of model drugs in a β-cyclodextrin cavity. Eur. J. Pharm. Biopharm 2009, 71, 377–386. [Google Scholar] [Green Version]
- Cesla, P.; Fischer, J.; Tesarova, E.; Jandera, P.; Stanek, V. Effects of capillary coating and β-cyclodextrin additive to the background electrolyte on separation of sulphonated azodyes by capillary zone electrophoresis. J. Chromatogr. A 2007, 1149, 358–367. [Google Scholar]
- Li, N.; Zang, Y.-H.; Wu, Y.-N.; Xiong, X.-L.; Zhang, Y.-H. Inclusion complex of trimethoprim with β-cyclodextrin. J. Pharmaceut. Biomed 2005, 39, 824–829. [Google Scholar]
- Wang, H.Y.; Han, J.; Feng, X.G. Spectroscopic study of orange G-β-cyclodextrin complex and its analytical application. Spectrochim. Acta A 2007, 66, 578–585. [Google Scholar]
- Jiang, H.; Li, D.; Shanshan, Y. Synthesis and characterization of β-cyclodextrin inclusion complex containing di(8-hydroxyquinoline)magnesium. Spectrochim. Acta A 2008, 70, 878–883. [Google Scholar]
- Davies, J.E.D.; Ripmeester, J.A. Physical Methods in Supramolecular Chemistry; Atwood, J.L., Lehn, J.-M., Eds.; Pergamon Press: Oxford, UK, 1996; p. 426. [Google Scholar]
- Wang, J.; Cao, Y.; Sun, B.; Wang, C. Characterisation of inclusion complex of trans-ferulic acid and hydroxypropyl-β-cyclodextrin. Food Chem 2011, 124, 1069–1075. [Google Scholar]
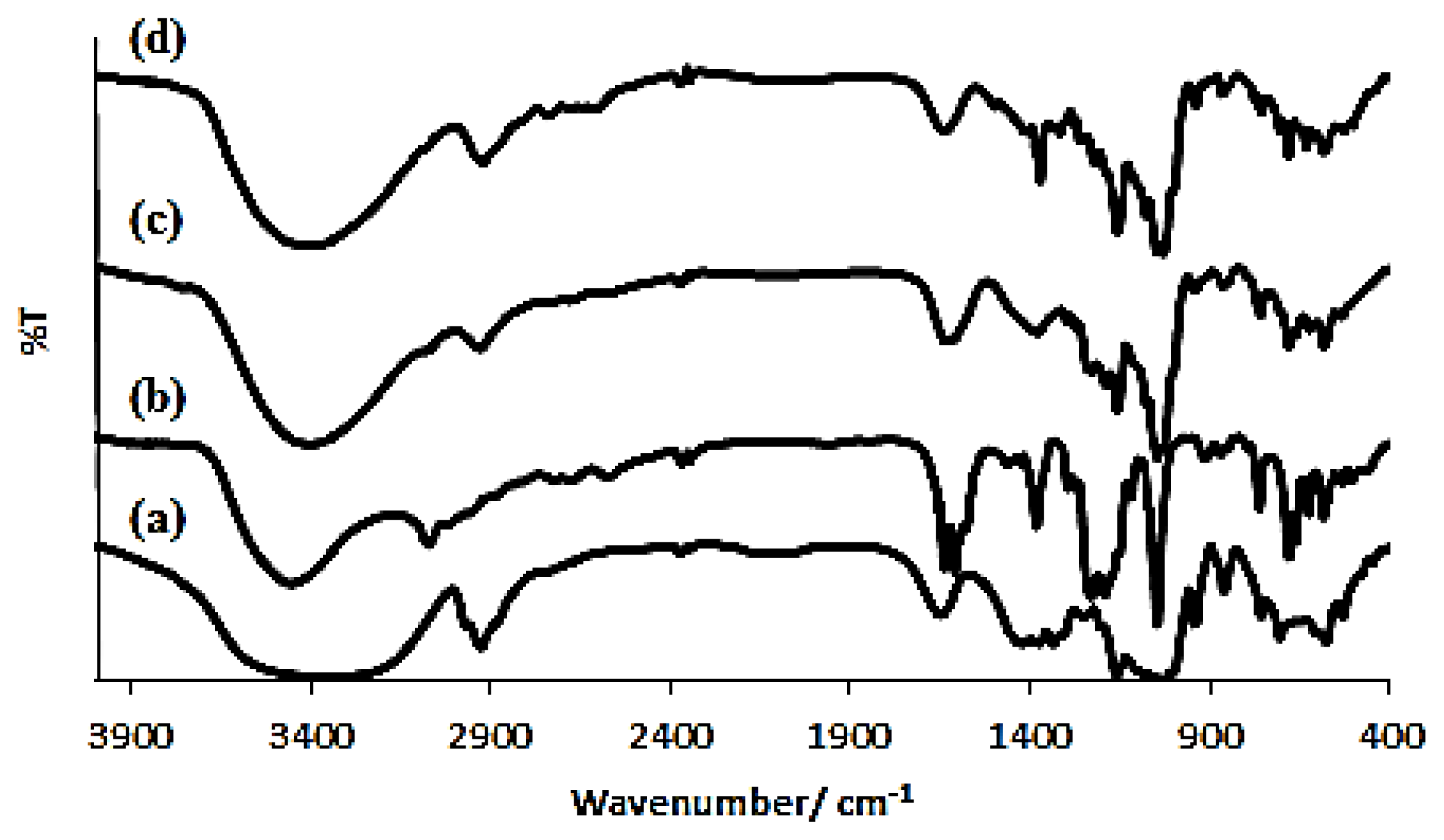
| Functional Group | Wavenumber (cm−1) | Changes Δδ | |
|---|---|---|---|
| β-Cyd | Inclusion complex | ||
| ν[OH] symmetric and antisymmetric | 3370.72 | 3379.88 | +9.16 |
| ν[CH2] | 2928.53 | 2923.12 | −5.53 |
| ν[C–C] | 1157.84 | 1157.66 | −0.18 |
| ν[O–H] bending vibration | 1029.24 | 1079.36 | +50.12 |
| Functional Group | Wavenumber (cm−1) | Changes Δδ | |
|---|---|---|---|
| Azomethine | Inclusion complex | ||
| ν[OH] | 3449.28 | 3379.88 | −69.4 |
| ν[C=N] stretching vibration | 1608.52 | 1639.04 | +30.52 |
| ν[=C–H] | 681.39 | 760.28 | +78.89 |
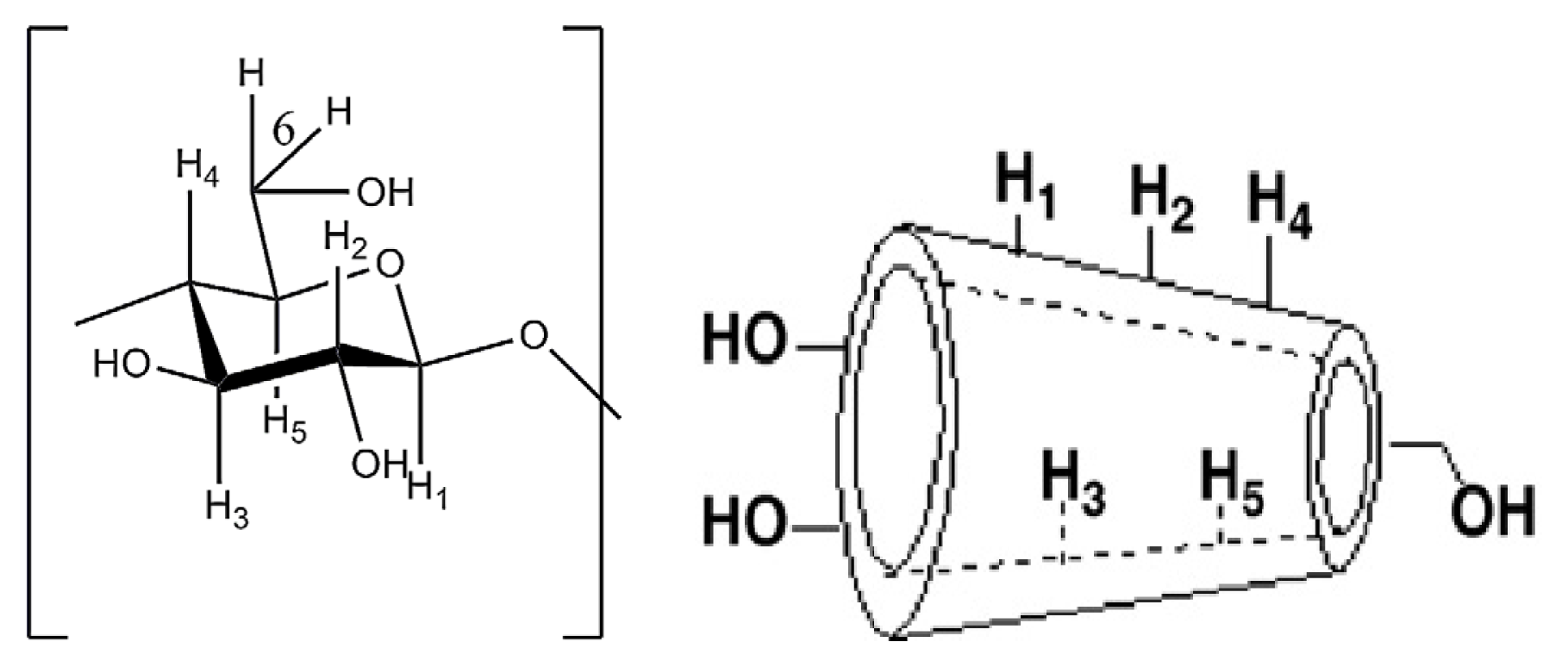 | ||||||
|---|---|---|---|---|---|---|
| H1 | H2 | H3 | H4 | H5 | H6 | |
| β-Cyd 5.0642 | 3.6309 | 3.9616 | 3.5833 | 3.6650 | 3.9616 | |
| Inclusion complex | 5.0300 | 3.4400 | 3.6800 | 3.4000 | 3.3900 | 3.7700 |
| Δδ | 0.0342 | 0.1909 | 0.2816 | 0.1833 | 0.2750 | 0.1916 |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sambasevam, K.P.; Mohamad, S.; Sarih, N.M.; Ismail, N.A. Synthesis and Characterization of the Inclusion Complex of β-cyclodextrin and Azomethine. Int. J. Mol. Sci. 2013, 14, 3671-3682. https://doi.org/10.3390/ijms14023671
Sambasevam KP, Mohamad S, Sarih NM, Ismail NA. Synthesis and Characterization of the Inclusion Complex of β-cyclodextrin and Azomethine. International Journal of Molecular Sciences. 2013; 14(2):3671-3682. https://doi.org/10.3390/ijms14023671
Chicago/Turabian StyleSambasevam, Kavirajaa Pandian, Sharifah Mohamad, Norazilawati Muhamad Sarih, and Nor Atiqah Ismail. 2013. "Synthesis and Characterization of the Inclusion Complex of β-cyclodextrin and Azomethine" International Journal of Molecular Sciences 14, no. 2: 3671-3682. https://doi.org/10.3390/ijms14023671
APA StyleSambasevam, K. P., Mohamad, S., Sarih, N. M., & Ismail, N. A. (2013). Synthesis and Characterization of the Inclusion Complex of β-cyclodextrin and Azomethine. International Journal of Molecular Sciences, 14(2), 3671-3682. https://doi.org/10.3390/ijms14023671