The Kiss-and-Run Model of Intra-Golgi Transport
Abstract
:1. Introduction
- One of the problems of the CPM model is the mechanism of delivery of COPI vesicles filled with GRPs to the proximal compartment for the recycling of GRPs in yeast. In mammalian cells, COPI vesicles can be found as “strings” [21]. In contrast, in the yeast Saccharomyces cerevisiae, the different Golgi compartments are localized separately from each other, and hence they are divided by significant space. How COPI can be directed to the correct compartment in this case is not clear. Thus, it is necessary for a specific mechanism for the directed movement of COPI vesicles in yeast, because if their diffusion was not limited and targeting systems were lacking, COPI vesicles would fill the entire cytosol. However, this is not the case. Moreover, it has been shown that in mammalian cells, the ability of >50-nm particles (let us say, COPI vesicles) to diffuse throughout the cytosol is very limited [22].
- The movement of ManII during the polarization of Golgi stacks after washout of brefeldin A occurs when the Arf1/COPI machinery is blocked [23], which argues against the role of COPI vesicles for recycling of GRPs.
- In yeast containing the temperature-sensitive αCOP mutant ret1-1, which is known to be defective in COPI vesicle formation at the restrictive temperature, 37 °C [24], maturation of the Golgi compartments (as the replacement of the color particular to the proximal compartment by the color characteristic of the distal Golgi compartment) still occurs, even at the restrictive temperature [25].
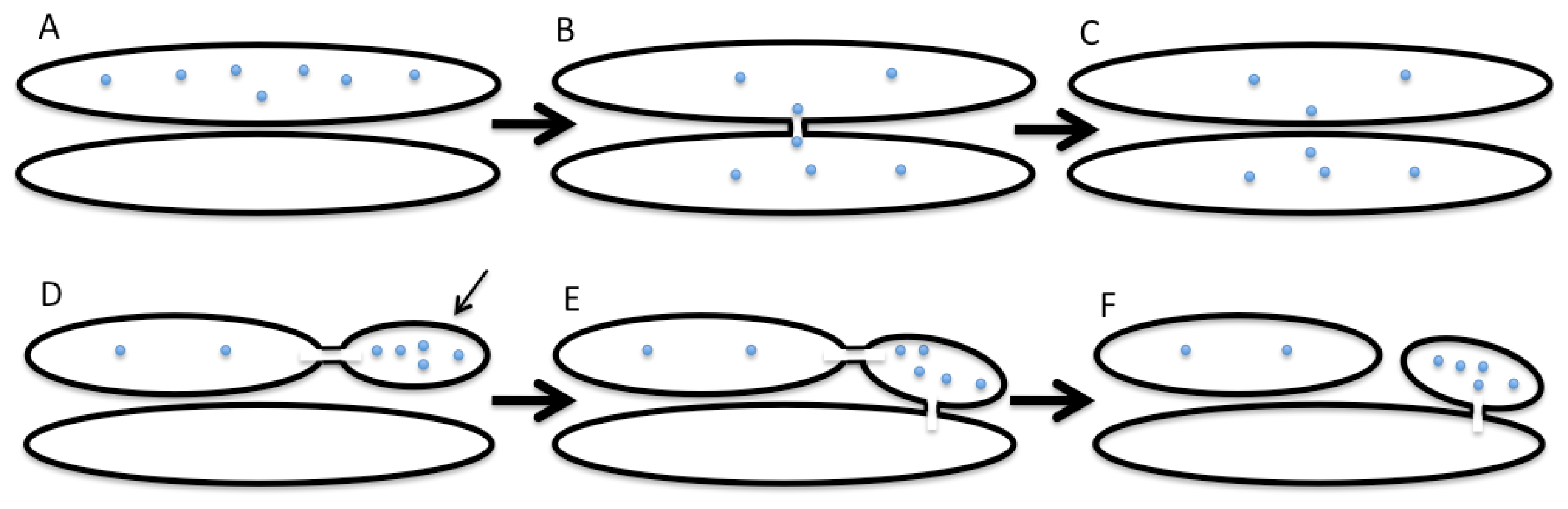
2. The Kiss-and-Run Model of Intra-Golgi Transport
- It requires a mechanism to ensure vectorial transport of the membrane within the framework of the asymmetric KAR model or the carrier-maturation model;
- There should be a mechanism for the concentration of the SNAREs in sites through which two compartments fuse with each other;
- The cells have to have a mechanism to break these membrane connections;
- To concentrate proteins in the distal Golgi compartments, there needs to be a mechanism that regulates the vectorial diffusion of proteins along intercisternal connections. For instance, this could be a gradient of ionic pumps or other protein machineries that can regulate the concentration of ions across the GA in order to create these gradients.
3. Concentration of Soluble Cargoes and Golgi Glycosylation Enzymes
4. The Kiss-and-Run Model and the Fusion and Fission Machineries
5. Potential Problems with the Kiss-and-Run Model
- Why does the exit of procollagen-I-GFP from the Golgi zone at steady-state occur according to an exponential decay, although the aggregates of procollagen-I-GFP cannot diffuse along the Golgi ribbon [35]? The reason for this might be the following. Most of the distensions containing procollagen-I are present within the TGN or the putative post-Golgi compartment and their emptying depends on the growth of microtubules. Indeed, as seen by figures 7, 8, 24 and 34 in Weinstock and Leblond [68], in situ at a steady-state, most of the procollagen-I distensions that are visible within the Golgi area are not integrated into the cisternae, but are localized out of the stacks, apparently within the putative post-Golgi compartment or the TGN.
- How can significant penetration of VSVG be explained when a large amount of cargo moves across the GA? Indeed, several studies [35,69,70] have found significant penetration of cargo into the GA (a cargo quickly reaches the trans-side of the GA) when large amounts of cargo are accumulated just before the GA. For instance, according to Bergman et al. [71], VSVG passes through the GA in 1–3 min. In only 10 min after release from a 40 °C temperature block, endo-H-sensitive VSVG (not processed by the medial Golgi enzyme ManII) appears in clathrin-coated vesicles [72]. If we consider that clathrin-coated buds are found exclusively on the trans cisternae [19] and that VSVG undergoes folding over 4–5 min [73], this suggests that VSVG reaches the trans-most cisterna 6 min after the moment when the first VSVG molecules reach the cis Golgi. This is faster than the well-established time necessary to cross the GA (10–20 min [7,53]). On the other hand, it is known that all of the medial cisternae have a more or less similar SNARE composition [74]. If the amounts of cargo traversing the GA simultaneously are high, the SNAREs within the first cisterna will be consumed quickly, and thus these cargoes will be forced to fuse with the subsequent cisterna. Even when the cargo domain arrives at the cis side of the Golgi stack, it might fuse not only with the first, but also with subsequent cisternae, by mistake. Thus, the deep penetration of VSVG and procollagen I through the GA can be explained without the assumption that they diffuse across the Golgi stack.
- Why does the diffusion of VSVG-GFP along the Golgi ribbon depend on the amount of VSVG-GFP passing through the GA [53]? Small domains of VSVG might not be able to diffuse through the Golgi enzymatic domains that are densely filled with the molecules of the GRPs [53]. However, when the amounts of membranes with VSVG-GFP are high, this might form some kind of a continuous parallel ribbon that is suitable for the VSVG-GFP diffusion.
- The KAR model assumes that the pH gradient across the compartments can contribute to some of the asymmetry in the intra-compartment transport that is needed for the KAR model to work. However, it is not clear if the gradient is actually sufficiently sharp to produce the observed behavior, or if the cargo in general would behave as needed for the KAR model to work. A similar argument can be made with respect to the concept of the Ca2+ gradient. The presence of a pH gradient along the GA is well known, starting from 6.9 at the cis side and reaching 4–5 at the trans side, and this gradient is important for the sorting of secretory proteins [75]. Similarly, lipid gradients across the GA are described [51].
6. Conclusions
Acknowledgments
Abbreviations
| CPM model | compartment progression/maturation model |
| COP | coatomer protein |
| ER | endoplasmic reticulum |
| GA | Golgi apparatus |
| GRPs | Golgi resident proteins |
| Man | mannosidase |
| VSVG | G protein of the vesicular stomatitis virus |
References
- Glick, B.S.; Luini, A. Models for Golgi traffic: A critical assessment. Cold Spring Harb. Perspect. Biol 2011, 3. [Google Scholar] [CrossRef]
- Mironov, A.A.; Beznoussenko, G.V.; Polishchuk, R.S.; Trucco, A. Intra-Golgi transport. A way to a new paradigm? BBA Mol. Cell Res 2005, 1744, 340–350. [Google Scholar]
- Mironov, A.A.; Beznoussenko, G.V. Intra-Golgi Transport. In The Golgi Apparatus. State of the Art 110 Years after Camillo Golgi’s Discovery; Mironov, A.A., Pavelka, M., Eds.; Springer-Verlag: Berlin, Germany, 2008; pp. 342–357. [Google Scholar]
- Nakano, A.; Luini, A. Passage through the Golgi. Curr. Opin. Cell Biol 2010, 22, 471–478. [Google Scholar]
- Fierke, C.A.; Hammes, G.G. Transient kinetic approaches to enzyme mechanisms. Methods Enzymol 1995, 249, 3–37. [Google Scholar]
- Gilchrist, A.; Au, C.E.; Hiding, J.; Bell, A.W.; Fernandez-Rodriguez, J.; Lesimple, S.; Nagaya, H.; Roy, L.; Gosline, S.J.; Hallett, M.; et al. Quantitative proteomics analysis of the secretory pathway. Cell 2006, 127, 1265–1281. [Google Scholar]
- Bonfanti, L.; Mironov, A.A., Jr; Martínez-Menárguez, J.A.; Martella, O.; Fusella, A.; Baldassarre, M.; Buccione, R.; Geuze, H.J.; Mironov, A.A.; Luini, A. Procollagen traverses the Golgi stack without leaving the lumen of cisternae: Evidence for cisternal maturation. Cell 1998, 95, 993–1003. [Google Scholar]
- Orci, L.; Stamnes, M.; Ravazzola, M.; Amherdt, M.; Perrelet, A.; Sollner, T.H.; Rothman, J.E. Bidirectional transport by distinct populations of COPI-coated vesicles. Cell 1997, 90, 335–349. [Google Scholar]
- Glick, B.S.; Iston, E.T.; Oster, G. A cisternal maturation mechanism can explain the asymmetry of the Golgi stack. FEBS Lett 1997, 414, 177–181. [Google Scholar]
- Martinez-Menarguez, J.A.; Prekeris, R.; Oorschot, V.M.; Scheller, R.; Slot, J.W.; Geuze, H.J.; Klumperman, J. Peri-Golgi vesicles contain retrograde but not anterograde proteins consistent with the cisternal progression model of intra-Golgi transport. J. Cell Biol 2001, 155, 1213–1224. [Google Scholar]
- Cosson, P.; Amherdt, M.; Rothman, J.E.; Orci, L. A resident Golgi protein is excluded from peri-Golgi vesicles in NRK cells. Proc. Natl. Acad. Sci. USA 2002, 99, 12831–12834. [Google Scholar]
- Kweon, H.S.; Beznoussenko, G.V.; Micaroni, M.; Polishchuk, R.S.; Trucco, A.; Martella, O.; di Giandomenico, D.; Marra, P.; Fusella, A.; di Pentima, A.; et al. Golgi enzymes are enriched in perforated zones of Golgi cisternae but are depleted in COPI vesicles. Mol. Biol. Cell 2004, 15, 4710–4724. [Google Scholar]
- Lucocq, J.; Berger, E.; Hug, C. The pathway of Golgi cluster formation in okadaic acid-treated cells. J. Struct. Biol 1995, 115, 318–330. [Google Scholar]
- Orci, L.; Amherdt, M.; Ravazzola, M.; Perrelet, A.; Rothman, J.E. Exclusion of Golgi residents from transport vesicles budding from Golgi cisternae in intact cells. J. Cell Biol 2000, 150, 1263–1270. [Google Scholar]
- Fusella, A.; Micaroni, M.; Di Giandomenico, D.; Mironov, A.A.; Beznoussenko, G.V. COPI vesicles inhibit intra-Golgi transport by sequestration of Qb-SNAREs from Golgi cisternae. Traffic 2012. submitted for publication. [Google Scholar]
- Berger, E.G.; Hesford, F.J. Localization of galactosyl- and sialyltransferase by immunofluorescence: Evidence for different sites. Proc. Natl. Acad. Sci. USA 1985, 82, 4736–4739. [Google Scholar]
- Teasdale, R.D.; Matheson, F.; Gleeson, P.A. Post-translational modifications distinguish cell-surface from Golgi-retained beta-1,4 galactosyltransferase molecules. Golgi localization involves active retention. Glycobiology 1994, 4, 917–928. [Google Scholar]
- Rabouille, C.; Hui, N.; Hunte, F.; Kieckbusch, R.; Berger, E.G.; Warren, G.; Nilsson, T. Mapping the distribution of Golgi enzymes involved in the construction of complex oligosaccharides. J. Cell Sci 1995, 108, 1617–1627. [Google Scholar]
- Ladinsky, M.S.; Mastronarde, D.N.; McIntosh, J.R.; Howell, K.E.; Staehelin, L.A. Golgi structure in three dimensions: Functional insights from the normal rat kidney cell. J. Cell Biol 1999, 144, 1135–1149. [Google Scholar]
- Ladinsky, M.S.; Wu, C.C.; McIntosh, S.; McIntosh, J.R.; Howell, K. Structure of the Golgi and distribution of reporter molecules at 20 °C reveals the complexity of the exit compartments. Mol. Biol. Cell 2002, 13, 2810–2825. [Google Scholar]
- Orci, L.; Perrelet, A.; Rothman, J.E. Vesicles on strings: Morphological evidence for processive transport within the Golgi stack. Proc. Natl. Acad. Sci. USA 1998, 95, 2279–2283. [Google Scholar]
- Luby-Phelps, K. Physical properties of cytoplasm. Curr. Opin. Cell Biol 1994, 6, 3–9. [Google Scholar]
- Bannykh, S.I.; Plutner, H.; Matteson, J.; Balch, W.E. The role of ARF1 and rab GTPases in polarization of the Golgi stack. Traffic 2005, 6, 803–819. [Google Scholar]
- Letourneur, F.; Gaynor, E.C.; Hennecke, S.; Démollière, C.; Duden, R.; Emr, S.D.; Riezman, H.; Cosson, P. Coatomer is essential for retrieval of dilysine-tagged proteins to the endoplasmic reticulum. Cell 1994, 79, 1199–1207. [Google Scholar]
- Matsuura-Tokita, K.; Takeuchi, M.; Ichihara, A.; Mikuriya, K.; Nakano, A. Live imaging of yeast Golgi cisternal maturation. Nature 2006, 441, 1007–1010. [Google Scholar]
- Oprins, A.; Rabouille, C.; Posthuma, G.; Klumperman, J.; Geuze, H.J.; Slot, J.W. The ER to Golgi interface is the major concentration site of secretory proteins in the exocrine pancreatic cell. Traffic 2001, 2, 831–838. [Google Scholar]
- Mironov, A.A.; Arvan, P. Origin of the Regulated Secretory Pathway. In The Golgi Apparatus: State of the Art 110 Years after Camillo Golgi’s Discovery; Mironov, A.A., Pavelka, M., Eds.; Springer-Verlag: Berlin, Germany, 2008; pp. 482–515. [Google Scholar]
- Trucco, A.; Polishchuk, R.S.; Martella, O.; Di Pentima, A.; Fusella, A.; di Giandomenico, D.; San Pietro, E.; Beznoussenko, G.V.; Polishchuk, E.V.; Baldassarre, M.; et al. Secretory traffic triggers the formation of tubular continuities across Golgi sub-compartments. Nat. Cell Biol 2004, 6, 1071–1081. [Google Scholar]
- Beznoussenko, G.V.; Seetharaman Parashuraman, S.; Polishchuk, R.; Martella, O.; di Giandomenico, D.; Fusella, A.; Spaar, A.; Sallese, M.; Capestrano, M.G.; Pavelka, M.; et al. Secretion of soluble proteins by diffusion via inter-cisternal continuities: A novel intra-Golgi transport mechanism. Nat. Cell Biol 2012. submitted for publication. [Google Scholar]
- Marra, P.; Salvatore, L.; Mironov, A., Jr; di Campli, A.; di Tullio, G.; Trucco, A.; Beznoussenko, G.; Mironov, A.; de Matteis, M.A. The biogenesis of the Golgi ribbon: The roles of membrane input from the ER and of GM130. Mol. Biol. Cell 2007, 18, 1595–1608. [Google Scholar]
- Kondylis, V.; Spoorendonk, K.M.; Rabouille, C. dGRASP localization and function in the early exocytic pathway in Drosophila S2 cells. Mol. Biol. Cell 2005, 16, 4061–4072. [Google Scholar]
- Marsh, B.J.; Volkmann, N.; McIntosh, J.R.; Howell, K.E. Direct continuities between cisternae at different levels of the Golgi complex in glucose-stimulated mouse islet beta cells. Proc. Natl. Acad. Sci. USA 2004, 101, 5565–5570. [Google Scholar]
- Beznoussenko, G.V.; Scanu, T.; Parashuraman, S.; di Giandomenico, D.; Deerink, T.J.; Martone, M.E.; Vos, M.R.; Rikers, Y.G.M.; Mironov, A.A. In situ, COPII and COPI vesicles are depleted of anterograde and retrograde cargo correspondingly. Mol. Cell Biol 2012. to be submitted for publication. [Google Scholar]
- Bouchet-Marquis, C.; Starkuviene, V.; Grabenbauer, M. Golgi apparatus studied in vitreous sections. J. Microsc 2008, 230, 308–316. [Google Scholar]
- Patterson, G.H.; Hirschberg, K.; Polishchuk, R.S.; Gerlich, D.; Phair, R.D.; Lippincott-Schwartz, J. Transport through the Golgi apparatus by rapid partitioning within a two-phase membrane system. Cell 2008, 133, 1055–1067. [Google Scholar]
- Pagano, R.E.; Sepanski, M.A.; Martin, O.C. Molecular trapping of a fluorescent ceramide analogue at the Golgi apparatus of fixed cells: Interaction with endogenous lipids provides a trans-Golgi marker for both light and electron microscopy. J. Cell Biol 1989, 109, 2067–2079. [Google Scholar]
- Mironov, A.A.; Colanzi, A.; Polishchuk, R.S.; Beznoussenko, G.V.; Mironov, A.A., Jr; Fusella, A.; di Tullio, G.; Silletta, M.G.; Corda, D.; de Matteis, M.A.; Luini, A. Dicumarol, an inhibitor of ADP-ribosylation of CtBP3/BARS, fragments Golgi non-compact tubular zones and inhibits intra-Golgi transport. Eur. J. Cell Biol. 2004, 83, 263–279. [Google Scholar]
- Mavillard, F.; Hidalgo, J.; Megias, D.; Levitsky, K.L.; Velasco, A. PKA-mediated Golgi remodeling during cAMP signal transmission. Traffic 2010, 11, 90–109. [Google Scholar]
- Pagano, R.E.; Longmuir, K.J. Phosphorylation, transbilayer movement, and facilitated intracellular transport of diacylglycerol are involved in the uptake of a fluorescent analog of phosphatidic acid by cultured fibroblasts. J. Biol. Chem 1985, 260, 1909–1916. [Google Scholar]
- Sleight, R.G.; Pagano, R.E. Rapid appearance of newly synthesized phosphatidylethanolamine at the plasma membrane. J. Biol. Chem 1983, 258, 9050–9058. [Google Scholar]
- Stinchcombe, J.C.; Nomoto, H.; Cutler, D.F.; Hopkins, C.R. Anterograde and retrograde traffic between the rough endoplasmic reticulum and the Golgi complex. J. Cell Biol 1995, 131, 1387–1401. [Google Scholar]
- Hermo, L.; Smith, C.E. The structure of the Golgi apparatus: A sperm’s eye view in principal epithelial cells of the rat epididymis. Histochem. Cell Biol 1998, 109, 431–447. [Google Scholar]
- Rambourg, A.; Clermont, Y. Three-Dimensional Structure of the Golgi Apparatus in Mammalian Cells. In The Golgi Apparatus; Roth, J., Berger, E.G., Eds.; Birkhauser: Basel, Switzerland, 1997; pp. 37–61. [Google Scholar]
- Krijnse-Locker, J.; Ericsson, M.; Rottier, P.J.; Griffiths, G. Characterization of the budding compartment of mouse hepatitis virus: Evidence that transport from the RER to the Golgi complex requires only one vesicular transport step. J. Cell Biol 1994, 124, 55–70. [Google Scholar]
- Ladinsky, M.S.; Kremer, J.R.; Furcinitti, P.S.; McIntosh, J.R.; Howell, K.E. HVEM tomography of the trans-Golgi network: Structural insights and identification of a lace-like vesicle coat. J. Cell Biol 1994, 127, 29–38. [Google Scholar]
- Clermont, Y.; Rambourg, A.; Hermo, L. Connections between the various elements of the cis- and mid-compartments of the Golgi apparatus of early rat spermatids. Anat. Rec 1994, 240, 469–480. [Google Scholar]
- Griffiths, G.; Pepperkok, R.; Locker, J.K.; Kreis, T.E. Immunocytochemical localization of beta-COP to the ER-Golgi boundary and the TGN. J. Cell Sci 1995, 108, 2839–2856. [Google Scholar]
- Griffiths, G.; Ericsson, M.; Krijnse-Locker, J.; Nilsson, T.; Goud, B.; Söling, H.D.; Tang, B.L.; Wong, S.H.; Hong, W. Localization of the Lys, Asp, Glu, Leu tetrapeptide receptor to the Golgi complex and the intermediate compartment in mammalian cells. J. Cell Biol 1994, 127, 1557–1574. [Google Scholar]
- Mironov, A.A.; Weidman, P.; Luini, A. Variations on the intracellular transport theme: Maturing cisternae and trafficking tubules. J. Cell Biol 1997, 138, 481–484. [Google Scholar]
- Tanaka, K.; Mitsushima, A.; Fukudome, H.; Kashima, Y. Three-dimensional architecture of the Golgi complex observed by high resolution scanning electron microscopy. J. Submicrosc. Cytol 1986, 18, 1–9. [Google Scholar]
- Cluett, E.B.; Kuismanen, E.; Machamer, C.E. Heterogeneous distribution of the unusual phospholipid semilysobisphosphatidic acid through the Golgi complex. Mol. Biol. Cell 1997, 8, 2233–2240. [Google Scholar]
- Orci, L.; Montesano, R.; Meda, P.; Malaisse-Lagae, F.; Brown, D.; Perrelet, A.; Vassalli, P. Heterogeneous distribution of filipin-cholesterol complexes across the cisternae of the Golgi apparatus. Proc. Natl. Acad. Sci. USA 1981, 78, 293–297. [Google Scholar]
- Mironov, A.A.; Beznoussenko, G.V.; Nicoziani, P.; Martella, O.; Trucco, A.; Kweon, H.S.; di Giandomenico, D.; Polishchuk, R.S.; Fusella, A.; Lupetti, P.; et al. Small cargo proteins and large aggregates can traverse the Golgi by a common mechanism without leaving the lumen of cisternae. J. Cell Biol 2001, 155, 1225–1238. [Google Scholar]
- Volchuk, A.; Amherdt, M.; Ravazzola, M.; Brügger, B.; Rivera, V.M.; Clackson, T.; Perrelet, A.; Söllner, T.H.; Rothman, J.E.; Orci, L. Megavesicles implicated in the rapid transport of intracisternal aggregates across the Golgi stack. Cell 2000, 102, 335–348. [Google Scholar]
- Perinetti, G.; Müller, T.; Spaar, A.; Polishchuk, R.; Luini, A.; Egner, A. Correlation of 4Pi- and electron microscopy to study transport through single Golgi stacks in living cells with super resolution. Traffic 2009, 10, 379–391. [Google Scholar]
- Ceccarelli, B.; Hurlbut, W.P.; Mauro, A. Depletion of vesicles from frog neuromuscular junctions by prolonged tetanic stimulation. J. Cell Biol 1972, 54, 30–38. [Google Scholar]
- Roberts, R.L.; Barbieri, M.A.; Pryse, K.M.; Chua, M.; Morisaki, J.H.; Stahl, P.D. Endosome fusion in living cells overexpressing GFP-rab5. J. Cell Sci. 1999, 112, 3667–3675. [Google Scholar]
- Van Kempen, G.T.; van der Leest, H.T.; van den Berg, R.J.; Eilers, P.; Westerink, R.H. Three distinct modes of exocytosis revealed by amperometry in neuroendocrine cells. Biophys. J 2011, 100, 968–977. [Google Scholar]
- Jaiswal, J.K.; Rivera, V.M.; Simon, S.M. Exocytosis of post-Golgi vesicles is regulated by components of the endocytic machinery. Cell 2009, 137, 1308–1319. [Google Scholar]
- Losev, E.; Reinke, C.A.; Jellen, J.; Strongin, D.E.; Bevis, B.J.; Glick, B.S. Golgi maturation visualized in living yeast. Nature 2006, 441, 1002–1006. [Google Scholar]
- Quinn, P.; Griffiths, G.; Warren, G. Density of newly synthesized plasma membrane proteins in intracellular membranes II. Biochemical studies. J. Cell Biol 1984, 98, 2142–2147. [Google Scholar]
- Beznoussenko, G.V.; Fusella, A.; Ragnini, A.; Ellinger, A.; Wilson, C.; Mironov, A.A. Three-dimensional structure of the Golgi in yeast S. cerevisiae and mechanisms of intracellular transport. Traffic 2012. to be submitted for publication. [Google Scholar]
- Micaroni, M.; Perinetti, G.; Di Giandomenico, D.; Bianchi, K.; Spaar, A.; Mironov, A.A. Synchronous intra-Golgi transport induces the release of Ca2+ from the Golgi apparatus. Exp. Cell Res 2010, 316, 2071–2086. [Google Scholar]
- Beznoussenko, G.V.; Vetterlein, M.; Neumuller, J.; Giandomenico, D.; Bao Cutrona, M.; Ellinger, A.; Pavelka, M.; Mironov, A.A. In the transporting Golgi stacks, Golgin-97 facilitates the attachment of the trans-most cisternae to the last medial cisterna. Nat. Cell Biol 2012. to be submitted for publication. [Google Scholar]
- Weidman, P.; Roth, R.; Heuser, J. Golgi membrane dynamics imaged by freeze-etch electron microscopy: Views of different membrane coatings involved in tubulation versus vesiculation. Cell 1993, 75, 123–133. [Google Scholar]
- Yang, J.S.; Lee, S.Y.; Spanò, S.; Gad, H.; Zhang, L.; Nie, Z.; Bonazzi, M.; Corda, D.; Luini, A.; Hsu, V.W. A role for BARS at the fission step of COPI vesicle formation from Golgi membrane. EMBO J 2005, 24, 4133–4143. [Google Scholar]
- Yang, J.S.; Zhang, L.; Lee, S.Y.; Luini, A.; Hsu, V.W. Key components of the fission machinery are interchangeable. Nat. Cell Biol 2006, 8, 1376–1382. [Google Scholar]
- Weinstock, M.; Leblond, C.P. Synthesis, migration, and release of precursor collagen by odontoblasts as visualized by radioautography after (3H)proline administration. J. Cell Biol 1974, 60, 92–127. [Google Scholar]
- Bergmann, J.E. Using temperature-sensitive mutants of VSV to study membrane protein biogenesis. Methods Cell Biol 1989, 32, 85–110. [Google Scholar]
- Griffiths, G. Gut thoughts on the Golgi complex. Traffic 2000, 1, 738–745. [Google Scholar]
- Bergmann, J.E.; Tokuyasu, K.T.; Singer, S.J. Passage of an integral membrane protein, the vesicular stomatitis virus glycoprotein, through the Golgi apparatus en route to the plasma membrane. Proc. Natl. Acad. Sci. USA 1981, 78, 1746–1750. [Google Scholar]
- Rothman, J.E.; Bursztyn-Pettegrew, H.; Fine, R.E. Transport of the membrane glycoprotein of vesicular stomatitis virus to the cell surface in two stages by clathrin-coated vesicles. J. Cell Biol 1980, 86, 162–171. [Google Scholar]
- Bannykh, S.; Aridor, M.; Plutner, H.; Rowe, T.; Balch, W.E. Regulated export of cargo from the endoplasmic reticulum of mammalian cells. Cold Spring Harb. Symp. Quant. Biol 1995, 60, 127–137. [Google Scholar]
- Orci, L.; Ravazzola, M.; Volchuk, A.; Engel, T.; Gmachl, M.; Amherdt, M.; Perrelet, A.; Sollner, T.H.; Rothman, J.E. Anterograde flow of cargo across the golgi stack potentially mediated via bidirectional “percolating” COPI vesicles. Proc. Natl. Acad. Sci. USA 2000, 97, 10400–10405. [Google Scholar]
- Huang, C.; Chang, A. pH-dependent cargo sorting from the Golgi. J. Biol. Chem 2011, 286, 10058–10065. [Google Scholar]
- Beznoussenko, G.V.; Mironov, A.A. Models of intracellular transport and evolution of the Golgi complex. Anat. Rec 2002, 268, 226–238. [Google Scholar]
- Mironov, A.A.; Luini, A.; Buccione, R. Constitutive transport between the trans-Golgi network and the plasma membrane according to the maturation model. A hypothesis. FEBS Lett 1998, 440, 99–102. [Google Scholar]
- Mironov, A., Jr; Luini, A.; Mironov, A. A synthetic model of intra-Golgi traffic. FASEB J. 1998, 12, 249–252. [Google Scholar]
| Number | ER | IC | cis | med1 | med2 | med3 | med4 | TGN | PGC |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 10 | 20 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2 | 10 | 7 | 13 | 0 | 0 | 0 | 0 | 0 | 0 |
| 3 | 10 | 20 | 4 | 9 | 0 | 0 | 0 | 0 | 0 |
| 4 | 10 | 8 | 16 | 3 | 6 | 0 | 0 | 0 | 0 |
| 5 | 10 | 20 | 6 | 13 | 2 | 4 | 0 | 0 | 0 |
| 6 | 10 | 9 | 17 | 5 | 10 | 1 | 3 | 0 | 0 |
| 7 | 10 | 20 | 7 | 15 | 4 | 7 | 1 | 2 | 0 |
| 8 | 10 | 9 | 18 | 6 | 13 | 3 | 5 | 1 | 1 |
| 9 | 10 | 20 | 8 | 16 | 5 | 11 | 2 | 4 | 1 |
| 10 | 10 | 9 | 19 | 7 | 14 | 4 | 9 | 2 | 3 |
| 11 | 10 | 20 | 9 | 17 | 6 | 12 | 4 | 7 | 3 |
| 12 | 10 | 10 | 19 | 8 | 15 | 5 | 11 | 3 | 7 |
| 13 | 10 | 20 | 9 | 18 | 7 | 13 | 5 | 9 | 7 |
| 14 | 10 | 10 | 19 | 8 | 17 | 6 | 12 | 5 | 11 |
| 15 | 10 | 20 | 9 | 18 | 8 | 15 | 6 | 11 | 11 |
| 16 | 10 | 10 | 19 | 9 | 17 | 7 | 14 | 7 | 15 |
| 17 | 10 | 20 | 9 | 19 | 8 | 16 | 7 | 14 | 15 |
| 18 | 10 | 10 | 19 | 9 | 18 | 8 | 15 | 10 | 19 |
| 19 | 10 | 20 | 10 | 19 | 9 | 17 | 8 | 17 | 19 |
| 20 | 10 | 10 | 20 | 9 | 19 | 8 | 17 | 12 | 24 |
| 21 | 10 | 20 | 10 | 19 | 9 | 18 | 10 | 19 | 24 |
| 22 | 10 | 10 | 20 | 9 | 19 | 9 | 19 | 14 | 29 |
| 23 | 10 | 20 | 10 | 19 | 9 | 19 | 11 | 22 | 29 |
| 24 | 10 | 10 | 20 | 9 | 19 | 10 | 20 | 17 | 34 |
| 25 | 10 | 20 | 10 | 19 | 10 | 19 | 12 | 25 | 34 |
| 26 | 10 | 10 | 20 | 10 | 19 | 11 | 20 | 20 | 39 |
| 27 | 10 | 20 | 10 | 20 | 10 | 20 | 13 | 37 | 39 |
| 28 | 10 | 10 | 20 | 10 | 20 | 11 | 22 | 25 | 51 |
| Departure | |||||||||
| 29 | 10 | 20 | 10 | 20 | 10 | 21 | 16 | 31 | 25 |
| 30 | 10 | 10 | 20 | 10 | 20 | 12 | 35 | 19 | 37 |
| 31 | 10 | 20 | 10 | 20 | 11 | 21 | 15 | 29 | 37 |
| 32 | 10 | 10 | 20 | 10 | 21 | 12 | 24 | 22 | 44 |
| 33 | 10 | 20 | 10 | 20 | 11 | 22 | 15 | 31 | 44 |
| 34 | 10 | 10 | 20 | 10 | 21 | 12 | 35 | 25 | 49 |
| 35 | 10 | 20 | 10 | 20 | 11 | 22 | 20 | 40 | 49 |
| 36 | 10 | 10 | 20 | 10 | 21 | 14 | 28 | 29 | 60 |
| Departure | |||||||||
| 37 | 10 | 20 | 10 | 20 | 12 | 23 | 16 | 31 | 29 |
| 38 | 10 | 10 | 20 | 11 | 21 | 13 | 26 | 20 | 40 |
| 39 | 10 | 20 | 10 | 31 | 11 | 23 | 15 | 31 | 40 |
| 40 | 10 | 10 | 20 | 14 | 28 | 16 | 32 | 24 | 47 |
| 41 | 10 | 20 | 11 | 23 | 15 | 29 | 25 | 51 | 47 |
| 42 | 10 | 10 | 21 | 13 | 25 | 18 | 36 | 33 | 65 |
| Departure | |||||||||
| 43 | 10 | 20 | 11 | 23 | 14 | 29 | 23 | 46 | 33 |
| 44 | 10 | 10 | 21 | 12 | 25 | 17 | 35 | 26 | 53 |
| Departure | |||||||||
| 45 | 10 | 20 | 11 | 22 | 14 | 28 | 20 | 41 | 26 |
| 46 | 10 | 10 | 21 | 12 | 24 | 16 | 32 | 22 | 45 |
| 47 | 10 | 20 | 11 | 22 | 13 | 27 | 18 | 36 | 45 |
| 48 | 10 | 10 | 21 | 12 | 23 | 15 | 30 | 27 | 54 |
| Departure | |||||||||
| ER | cis | med1 | med2 | med3 | med4 | med5 | TGN | PGC |
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 1 | 2 | 1 | 2 | 2 | 1 | 1 |
| 1 | 2 | 1 | 2 | 2 | 1 | 2 | 1 | |
| 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| 10 | 20 | 7 | 13 | 7 | 13 | 13 | 7 | 10 |
| 10 | 7 | 18 | 7 | 13 | 17 | 9 | 11 | 6 |
| 10 | 20 | 8 | 17 | 10 | 20 | 13 | 7 | 6 |
| 10 | 9 | 19 | 9 | 18 | 22 | 11 | 9 | 4 |
| 10 | 20 | 9 | 19 | 13 | 27 | 13 | 7 | 4 |
| 10 | 10 | 19 | 11 | 21 | 27 | 13 | 7 | 4 |
| 10 | 20 | 10 | 20 | 16 | 32 | 13 | 7 | 4 |
| 10 | 10 | 20 | 12 | 24 | 30 | 15 | 7 | 4 |
| 10 | 20 | 11 | 21 | 18 | 36 | 15 | 7 | 4 |
| 10 | 10 | 21 | 13 | 26 | 34 | 17 | 7 | 4 |
| 10 | 20 | 11 | 23 | 20 | 40 | 16 | 8 | 4 |
| 10 | 10 | 21 | 14 | 29 | 38 | 18 | 8 | 4 |
| 9 | 18 | 12 | 23 | 22 | 45 | 17 | 9 | 4 |
| 9 | 10 | 20 | 15 | 30 | 41 | 21 | 9 | 4 |
| 8 | 16 | 12 | 23 | 24 | 49 | 20 | 10 | 4 |
| 8 | 9 | 19 | 16 | 31 | 46 | 23 | 9 | 5 |
| 7 | 14 | 12 | 23 | 26 | 51 | 21 | 11 | 5 |
| 7 | 8 | 16 | 16 | 33 | 48 | 24 | 11 | 5 |
| 6 | 12 | 14 | 28 | 27 | 54 | 23 | 12 | 5 |
| 6 | 9 | 17 | 15 | 30 | 52 | 26 | 11 | 6 |
| 5 | 10 | 11 | 21 | 27 | 55 | 25 | 12 | 6 |
| 5 | 7 | 14 | 16 | 32 | 53 | 26 | 12 | 6 |
| 4 | 8 | 10 | 20 | 28 | 57 | 25 | 13 | 6 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mironov, A.A.; Beznoussenko, G.V. The Kiss-and-Run Model of Intra-Golgi Transport. Int. J. Mol. Sci. 2012, 13, 6800-6819. https://doi.org/10.3390/ijms13066800
Mironov AA, Beznoussenko GV. The Kiss-and-Run Model of Intra-Golgi Transport. International Journal of Molecular Sciences. 2012; 13(6):6800-6819. https://doi.org/10.3390/ijms13066800
Chicago/Turabian StyleMironov, Alexander A., and Galina V. Beznoussenko. 2012. "The Kiss-and-Run Model of Intra-Golgi Transport" International Journal of Molecular Sciences 13, no. 6: 6800-6819. https://doi.org/10.3390/ijms13066800
APA StyleMironov, A. A., & Beznoussenko, G. V. (2012). The Kiss-and-Run Model of Intra-Golgi Transport. International Journal of Molecular Sciences, 13(6), 6800-6819. https://doi.org/10.3390/ijms13066800




