Tenomodulin Inhibits Retinal Neovascularization in a Mouse Model of Oxygen-Induced Retinopathy
Abstract
:1. Introduction
2. Results
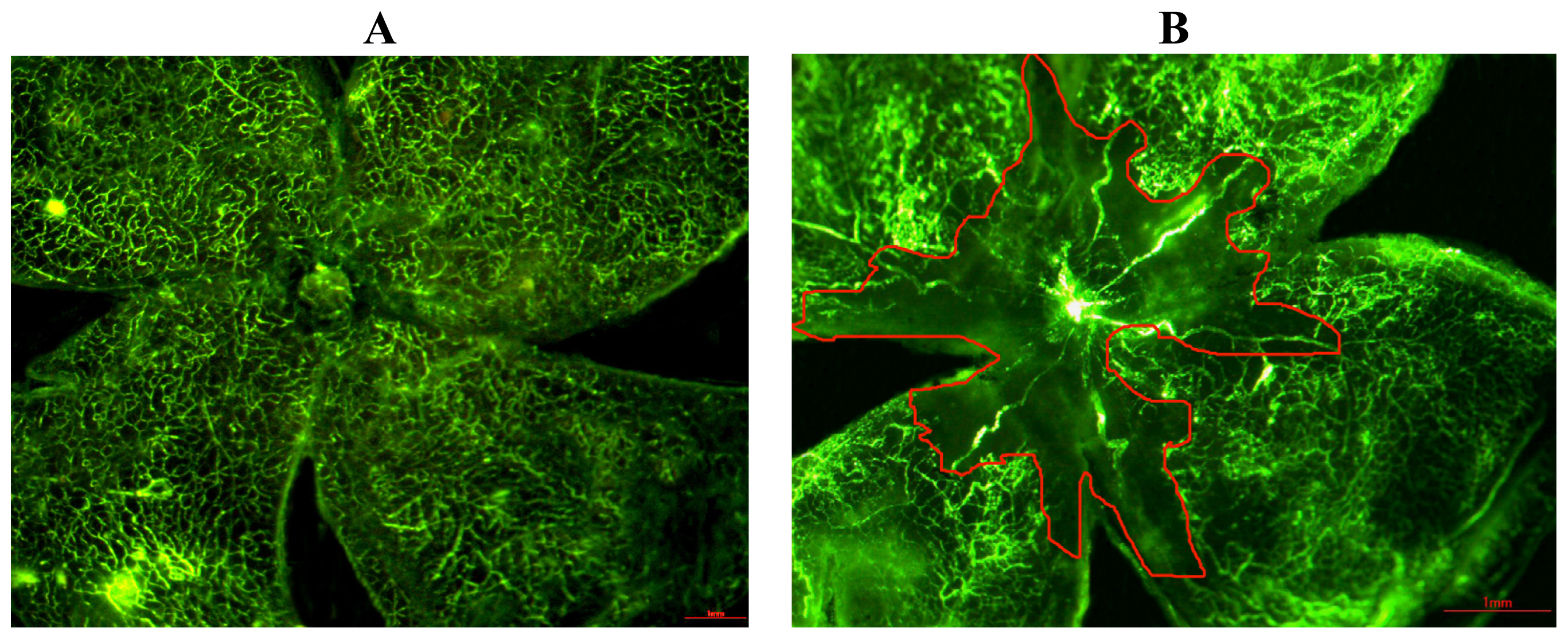
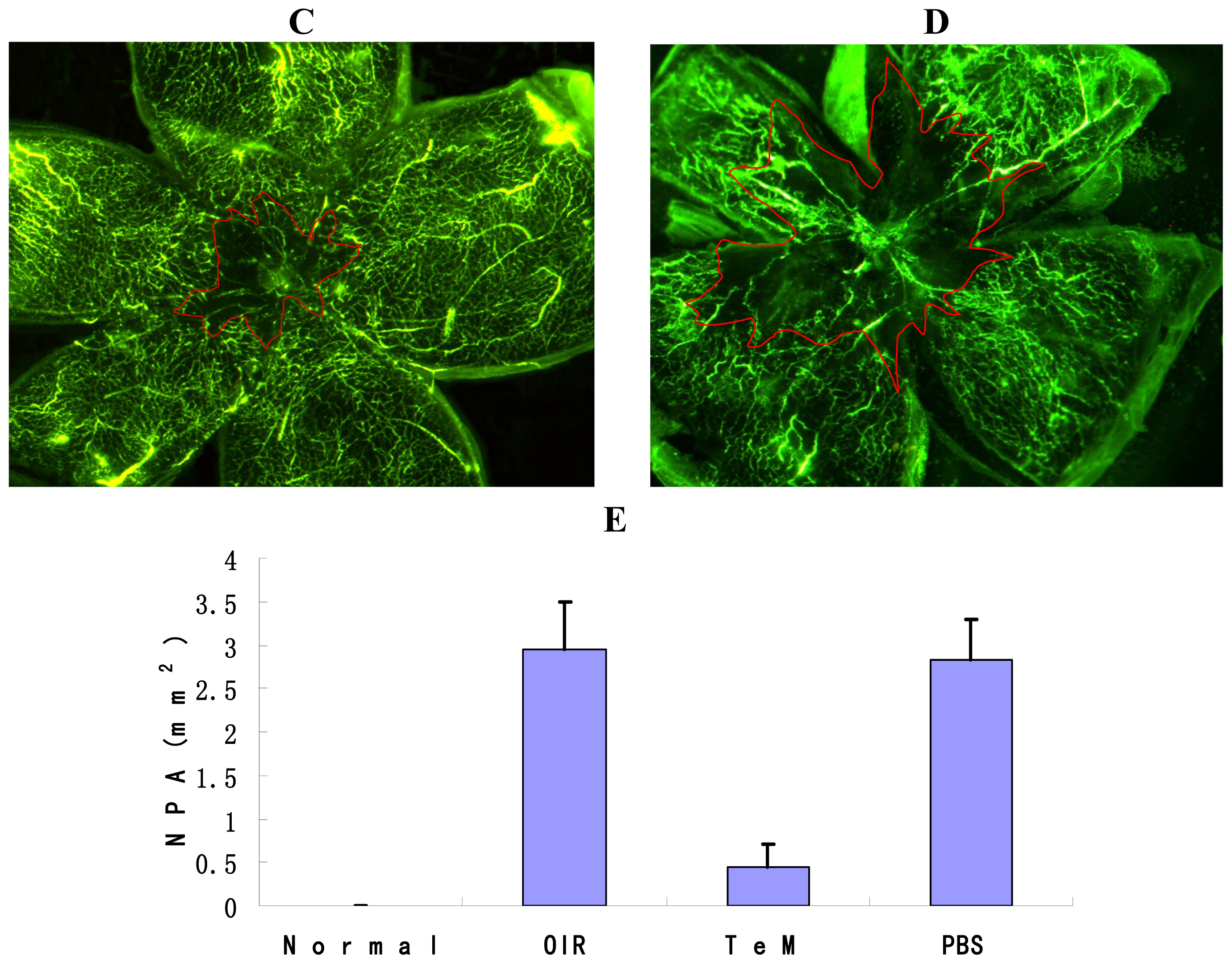
2.1. Quantification of the Central Nonperfusion Area of the Retina
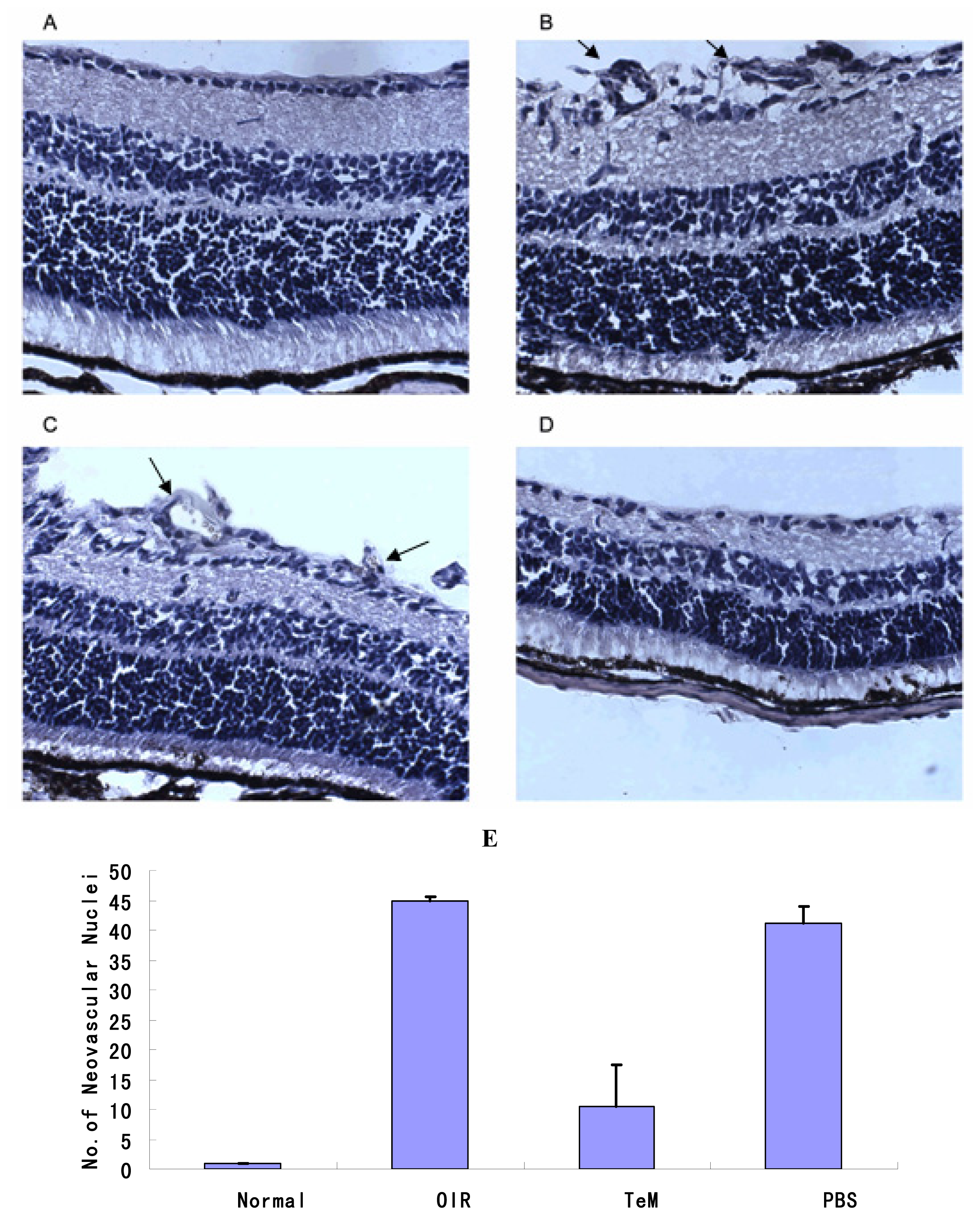
2.2. Histopathology
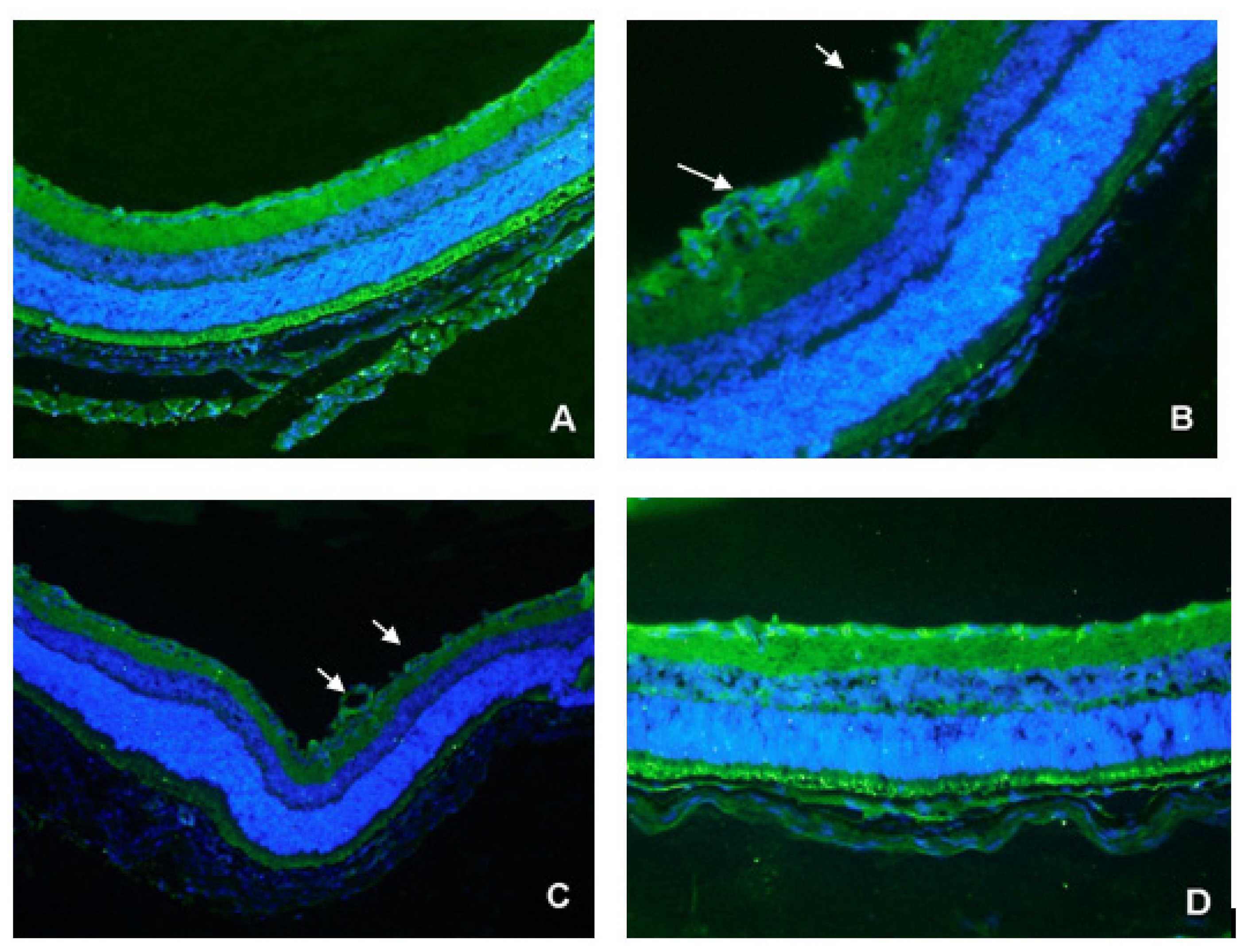
2.3. CD31 Staining of Vascular Cells
2.4. Detection of Exogenous TeM Protein in Retinas
3. Experimental Section
3.1. Preparation of Tenomodulin
3.2. Animal Model and Treatment
3.3. Angiography Using High Molecular Weight Fluorescein-Dextran for Retinal Flat-Mounting
3.4. HE Staining
3.5. Immunohistochemistry
3.6. Western Blot Analysis
3.7. Statistical Analysis
4. Discussion
5. Conclusions
Acknowledgements
References
- Kiyoshi, S.; Hitoshi, T.; Atsushi, O.; Hideyasu, O.; Yoshihito, H. Expression of Thrombospondin-1 in ischemia-induced retinal Neovascularization. Am. J. Pathol 1999, 154, 343–352. [Google Scholar]
- May, C.A.; Ohlmann, A.V.; Hammes, H.; Spandau, U.H.M. Proteins with an endostatin-like domain in a mouse model of oxygen-induced retinopathy. Exp. Eye Res 2006, 82, 341–348. [Google Scholar]
- Josef, N.; Aqeela, A.; Hao, P.; Esteban, G.; Nan, L.; Sergio, L.C.; Sergio, C.; Polyxenie, E.S.; Lynn, C.S.; Maria, B.G. The dipeptide Arg-Gln inhibits retinal neovascularization in the mouse model of Oxygen-induced retinopathy. Invest. Ophthalmol. Vis. Sci 2006, 47, 3151–3155. [Google Scholar]
- Anat, B.; Igor, M.; Millicent, S.; Helen, G.; Golda, D.; Sabrina, G.K.; Vicktoria, V.D.; Rami, S.; Paz, E.; Alexander, F.; et al. Inhibition of oxygen-induced retinopathy in RTP801-Deficient mice. Invest. Ophthalmol. Vis. Sci 2004, 45, 3796–3805. [Google Scholar]
- Frank, R.N. Diabetic retinopathy. N. Engl. J. Med 2004, 300, 48–58. [Google Scholar]
- Zhenhua, X.; Yilin, Y.; Elia, J.D. Vascular endothelial growth factor upregulates expression of ADAMTS1 in endothelial cells through protein Kinase C signaling. Invest. Ophthalmol. Vis. Sci 2006, 47, 4059–4066. [Google Scholar]
- Hanahan, D.; Folkman, J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 1996, 86, 353–364. [Google Scholar]
- Elia, J.D.; Hoseong, S.Y.; Izumi, S.; Masaru, M.; Elaine, Y.; Keisuke, M.; Miyuki, K.; Lin, Y.; Kiyoshi, S.; Karen, W.; et al. Pigment Epithelium-Derived Factor Suppresses Ischemia-induced retinal neovascularization and VEGF-induced migration and growth. Invest. Ophthalmol. Vis. Sci 2002, 43, 821–829. [Google Scholar]
- Smith, L.E.; Shen, W.; Perruzzi, C.; Soker, S.; Kinose, F.; Xu, X.; Robinson, G.; Driver, S.; Bischoff, J.; Zhang, B.; et al. Regulation of vascular endothelial growth factor-dependent retinal neovascularization by insulin-like growth factor-1 receptor. Nat. Med 1999, 5, 1390–1395. [Google Scholar]
- Smith, L.E.; Kopchick, J.J.; Chen, W.; Knapp, J.; Kinose, F.; Daley, D.; Foley, E.; Smith, R.G.; Schaeffer, J.M. Essential role of growth hormone in ischemia-induced retinal neovascularization. Science 1997, 276, 1706–1709. [Google Scholar]
- Jennifer, L.W.; Nicole, S.A.; Darren, J.K.; Richard, E. COX-2 inhibition and perinal angiogenesis in a mouse model of retinopathy of prematurity. Invest. Ophthalmol. Vis. Sci 2003, 44, 974–979. [Google Scholar]
- Moravski, C.J.; Kelly, D.J.; Cooper, M.E.; Gilbert, R.E.; Bertram, J.F.; Shahinfar, S.; Skinner, S.L.; Wilkinson-Berka, J.L. Retinal neovascularization is prevented by blockade of the renin-angiotensin system. Hypertension 2000, 36, 1099–1104. [Google Scholar]
- Gibert, R.E.; Vranes, D.; Berka, J.L.; Lelly, D.J.; Cox, A.; Wu, L.L.; Stacker, S.A.; Cooper, M.E. Vascular endothelial growth factor and its receptors in control and diabetic rat eyes. Lab. Invest 1998, 78, 1145–1155. [Google Scholar]
- Chabot, S.; Jabrane-Ferrat, N.; Bigot, K.; Tabiasco, J.; Provost, A.; Golzio, M.; Noman, M.Z.; Giustiniani, J.; Bellard, E.; Brayer, S.; et al. A novel antiangiogenic and vascular normalization therapy targeted against human CD160 receptor. J. Exp. Med 2011, 208, 973–986. [Google Scholar]
- Dawson, D.W.; Volpert, O.V.; Gillis, P.; Crawford, S.E.; Xu, H.; Benedict, W.; Bouck, N.P. Pigment epithelium-derived factor: a potent inhibitor of angiogenesis. Science 1999, 285, 245–248. [Google Scholar]
- Lutty, G.A.; Mello, R.J.; Chandler, C.; Fait, C.; Bennett, A.; Patz, A. Regulation of cell growth by vitreous humour. J. Cell. Sci 1985, 76, 53–65. [Google Scholar]
- Brem, S.; Preis, I.; Langer, R.; Brem, H.; Folkman, J. Inhibition of neo-vascularization by an extract derived from vitreous. Am. J. Ophthalmol 1997, 84, 323–328. [Google Scholar]
- Williams, G.A.; Eisenstein, R.; Schumacher, B.; Hsiao, K.-C. Inhibitor of vascular endothelial cell growth in the lens. Am. J. Ophthalmol 1984, 97, 366–371. [Google Scholar]
- Zhai, Y.; Ni, J.; Jiang, G.W.; Lu, J.; Xing, L.; Lincoln, C.; Cart, K.C.; Janat, F.; Kozak, D.; Xu, S.; et al. VEGI, a novel cytokine of the tunor necrosis factor family, is an angiogenesis inhibitor that suppresses the growth of colon carcinomas in vivo. FASEB J 1999, 13, 181–189. [Google Scholar]
- Yusuke, O.; Chisa, S.; Junichi, H.; Koji, N.; Fumi, T.; Jun-ichi, M.; Yuji, H.; Yasuo, T. Expression and localization of Tenomodulin, a transmembrane type Chondromodulin-I-related angiogenesis inhibitor, in mouse eyes. Invest. Ophthalmol. Vis. Sci 2003, 44, 1814–1822. [Google Scholar]
- Brandau, O.; Meindl, A.; Fassler, R.; Aszodi, A. A novel gene, tendin, is strongly expressed in tendons and ligaments and shows high homology with chondromodulin-I. Dev. Dyn 2001, 221, 72–80. [Google Scholar]
- Pisani, D.F.; Pierson, P.M.; Massoudi, A.; Leclerc, L.; Chopard, A.; Marini, J.F.; Dechesne, C.A. Myodulin is a novel potential angiogenic factor in skeletal muscle. Exp. Cell. Res 2004, 292, 40–50. [Google Scholar]
- Shukunami, C.; Oshima, Y.; Hiraki, Y. Molecular cloning of tenomodulin, a novel chondromodulin-I related gene. Biochem. Biophys. Res. Commun 2001, 280, 1323–1327. [Google Scholar]
- Yamana, K.; Wada, H.; Takahashi, Y.; Sato, H.; Kasahara, Y.; Kiyoki, M. Molecular cloning and characterization of CHM1L, a novel membrane molecule similar to chondromodulin-I. Biochem. Biophys. Res. Commun 2001, 280, 1101–1106. [Google Scholar]
- Docheva, D.; Hunziker, E.B.; Fassler, R.; Brandau, O. Tenomodulin is necessary for tenocyte proliferation and tendon maturation. Mol. Cell. Biol 2005, 25, 699–705. [Google Scholar]
- Oshima, Y.; Sato, K.; Tashiro, F.; Miyazaki, J.I.; Nishida, K.; Hiraki, Y.; Tano, Y.; Shukunami, C. Anti-angiogenic action of the C-terminal domain of tenomodulin that shares homology with chondromodulin-I. Cell. Sci. 2004. [Google Scholar] [CrossRef]
- Shukunami, C.; Oshima, Y.; Hiraki, Y. Chondromodulin-I and tenomodulin: A new class of tissue-specific angiogenesis inhibitors found in hypovascular connective tissues. Biochem. Biophys. Res. Commun 2005, 333, 299–307. [Google Scholar]
- Haruko, F.; Shoichi, S.; Kiyoshi, Y.; Koyama, Y.; Yaoita, E.; Funaki, S.; Shirakashi, M.; Oshima, Y.; Shukunami, C.; Hiraki, Y. Haruki Abe 2 and Tadashi Yamamoto Expression and localization of angiogenic inhibitory factor, chondromodulin-I, in adult rat eye. Invest. Ophthalmol. Vis. Sci 2001, 42, 1193–1200. [Google Scholar]
- Smith, L.E.; Wesolowski, E.; McLellan, A.; Kostyk, S.K.; D’Amato, R.; Sullivan, R.; D’Amore, P.A. Oxygen-induced retinopathy in the mouse. Invest. Ophthalmol. Vis. Sci 1994, 35, 101–111. [Google Scholar]
- Shibuya, M. Differential roles of vascular endothelial cell growth factor receptor-1 and Receptor-2 in Angiogenesis. Biochem. Mol. Biol 2006, 39, 469–478. [Google Scholar]
- Konopatskaya, O.; Churchill, A.J.; Harper, S.J.; Bates, D.O.; Gardiner, T.A. VEGF165b, an endogenous C-terminal splice variant of VEGF, inhibits retinal neovascularization in mice. Mol. Vis 2006, 12, 626–632. [Google Scholar]
- Lutty, G.A.; Mcleod, D.S. Retinal vascular development and oxygen-induced retinopathy: A role for adenosine. Prog. Ret. Eye Res 2003, 22, 95–111. [Google Scholar]
- Hidenori, I.; Masabaru, O.; Nobuyuki, K.; Takashi, K.; Onrai, M.; Yozo, M.; Takashi, M. Colocalization of neuropilin-1 and Flk-1 in retinal neovascularization in a mouse model of retinopathy. Invest. Ophthalmol. Vis. Sci 2001, 42, 1172–1178. [Google Scholar]
- Irene, N.; Rosemary, D.; Lucia, Z.; Peter, S.; Stephen, P.G. C-Ab1 is required for the development of hyperoxia-induced retinopathy. J. Exp. Med 2001, 193, 1383–1391. [Google Scholar]
- Aiello, L.P.; Avery, R.L.; Arrigg, P.G.; Keyt, B.A.; Jampel, H.D.; Shah, S.T.; Pasquale, L.R.; Thieme, H.; Iwamoto, M.A.; Park, J.E.; et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. King N. Engl. Med. 1994, 331, 1480–1487. [Google Scholar]
- Aiello, L.P.; Pierce, E.A.; Foley, E.D.; Takaqi, H.; Chen, H.; Riddle, L.; Ferrara, N.; King, G.L.; Smith, L.E. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc. Natl. Acad. Sci. USA 1995, 92, 10457–10461. [Google Scholar]
- Adamis, A.P.; Shima, D.T.; Tolentino, M.J.; Gragoudas, E.S.; Ferrara, N.; Folkmam, J.; D’Amore, P.A.; Miller, J.W. Inhibition of vascular endothelial growth factor prevents retinal ischemia-associated iris neovascularization in a nonhuman primate. Arch. Ophthamol 1996, 114, 66–71. [Google Scholar]
- Miller, J.W.; Adamis, A.P.; Aiello, L.P. Vascular endothelial growth factor in ocular neovascularization and proliferative diabetic retinopathy. Diabetes Metab. Rev 1997, 13, 37–50. [Google Scholar]
- Kim, I.; Ryan, A.M.; Rohan, R.; Amano, S.; Aqular, S.; Miller, J.W.; Adamis, A.P. Constitutive expression of VEGF, VEGFR-1, and VEGFR-2 in normal eyes. Invest. Ophthalmol. Vis. Sci 1999, 40, 2115–2121. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, W.; Li, Z.; Sato, T.; Oshima, Y. Tenomodulin Inhibits Retinal Neovascularization in a Mouse Model of Oxygen-Induced Retinopathy. Int. J. Mol. Sci. 2012, 13, 15373-15386. https://doi.org/10.3390/ijms131115373
Wang W, Li Z, Sato T, Oshima Y. Tenomodulin Inhibits Retinal Neovascularization in a Mouse Model of Oxygen-Induced Retinopathy. International Journal of Molecular Sciences. 2012; 13(11):15373-15386. https://doi.org/10.3390/ijms131115373
Chicago/Turabian StyleWang, Wei, Zhongqiu Li, Tatsuhiko Sato, and Yusuke Oshima. 2012. "Tenomodulin Inhibits Retinal Neovascularization in a Mouse Model of Oxygen-Induced Retinopathy" International Journal of Molecular Sciences 13, no. 11: 15373-15386. https://doi.org/10.3390/ijms131115373
APA StyleWang, W., Li, Z., Sato, T., & Oshima, Y. (2012). Tenomodulin Inhibits Retinal Neovascularization in a Mouse Model of Oxygen-Induced Retinopathy. International Journal of Molecular Sciences, 13(11), 15373-15386. https://doi.org/10.3390/ijms131115373



