Abstract
Obesity and its related metabolic abnormalities, including insulin resistance, alterations in the insulin-like growth factor-1 (IGF-1)/IGF-1 receptor (IGF-1R) axis, and the state of chronic inflammation, increase the risk of colorectal cancer (CRC) and hepatocellular carcinoma (HCC). However, these findings also indicate that the metabolic disorders caused by obesity might be effective targets to prevent the development of CRC and HCC in obese individuals. Green tea catechins (GTCs) possess anticancer and chemopreventive properties against cancer in various organs, including the colorectum and liver. GTCs have also been known to exert anti-obesity, antidiabetic, and anti-inflammatory effects, indicating that GTCs might be useful for the prevention of obesity-associated colorectal and liver carcinogenesis. Further, branched-chain amino acids (BCAA), which improve protein malnutrition and prevent progressive hepatic failure in patients with chronic liver diseases, might be also effective for the suppression of obesity-related carcinogenesis because oral supplementation with BCAA reduces the risk of HCC in obese cirrhotic patients. BCAA shows these beneficial effects because they can improve insulin resistance. Here, we review the detailed relationship between metabolic abnormalities and the development of CRC and HCC. We also review evidence, especially that based on our basic and clinical research using GTCs and BCAA, which indicates that targeting metabolic abnormalities by either pharmaceutical or nutritional intervention may be an effective strategy to prevent the development of CRC and HCC in obese individuals.
1. Introduction
Obesity, which is the result of a positive energy balance, is a serious health problem throughout the world. The World Health Organization (WHO) estimates that currently, more than 1.5 billion adults worldwide are overweight, of which at least 500 million are obese [1]. Obesity is linked to several health disorders such as cardiovascular disease, hypertension, diabetes mellitus, and hyperlipidemia, which are collectively known as “metabolic syndrome”. In addition, mounting evidence indicates that obesity and its related metabolic abnormalities, especially diabetes mellitus, are associated with the development of certain types of human epithelial malignancies, including colorectal cancer (CRC) and hepatocellular carcinoma (HCC) [2–8]. On the basis of systematic reviews of epidemiological evidence as well as mechanistic interpretations and data from animal experimental models, the World Cancer Research Fund and American Institute for Cancer Research released a report in 2007 on the causal relationship between high body fatness and an increased risk of CRC [9]. A large-scale meta-analysis (221 datasets on 282,000 incidence cases) also revealed that the magnitude of risk for CRC was greater among obese men than non-obese men [10]. In a prospectively studied population of more than 900,000 American adults, the body mass index (BMI) was found to be significantly associated with higher rates of death from cancer, especially HCC, because the relative risk of death from HCC was significantly higher (4.52 times) among men with a BMI of at least 35.0 than those who had normal weight (95% confidence interval, 2.94–6.94) [11].
Several pathophysiological mechanisms that link obesity and colorectal and liver carcinogenesis have been shown, including the emergence of insulin resistance, alterations in the insulin-like growth factor-1 (IGF-1)/IGF-1 receptor (IGF-1R) axis, the state of chronic inflammation, induction of oxidative stress, and occurrence of adipocytokine imbalance [2–6]. On the other hand, these findings also suggest that targeting these pathophysiological disorders via nutritional or pharmaceutical intervention might be an effective and promising strategy to inhibit obesity-related carcinogenesis. For instance, a 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor pitavastatin, which is widely used to treat hyperlipidemia, prevents obesity-related colorectal and liver carcinogenesis by attenuating chronic inflammation [12,13]. Captopril and telmisartan, which are anti-hypertensive drugs, also suppress the development of colonic preneoplastic lesions in obese and diabetic mice, and this suppression is associated with the reduction of oxidative stress and chronic inflammation [14].
In recent years, green tea catechins (GTCs) have received considerable attention because of their beneficial effects: they improve metabolic abnormalities and prevent cancer development [15–19]. Dietary supplementation with branched-chain amino acids (BCAA; leucine, isoleucine, and valine), which can prevent progressive hepatic failure in patients with chronic liver disease by improving insulin resistance [20–22], also reduces the risk of HCC in such patients who are obese [8]. In this article, we review the many mechanisms by which obesity and the related metabolic abnormalities influence the development of CRC and HCC while especially focusing on the emergence of insulin resistance and the subsequent inflammatory cascade. We also prove that the nutraceutical approach using GTCs and BCAA might be effective in preventing obesity-related carcinogenesis in both the colorectum and liver.
2. Potential Pathophysiological Mechanisms Linking Obesity and the Development of CRC
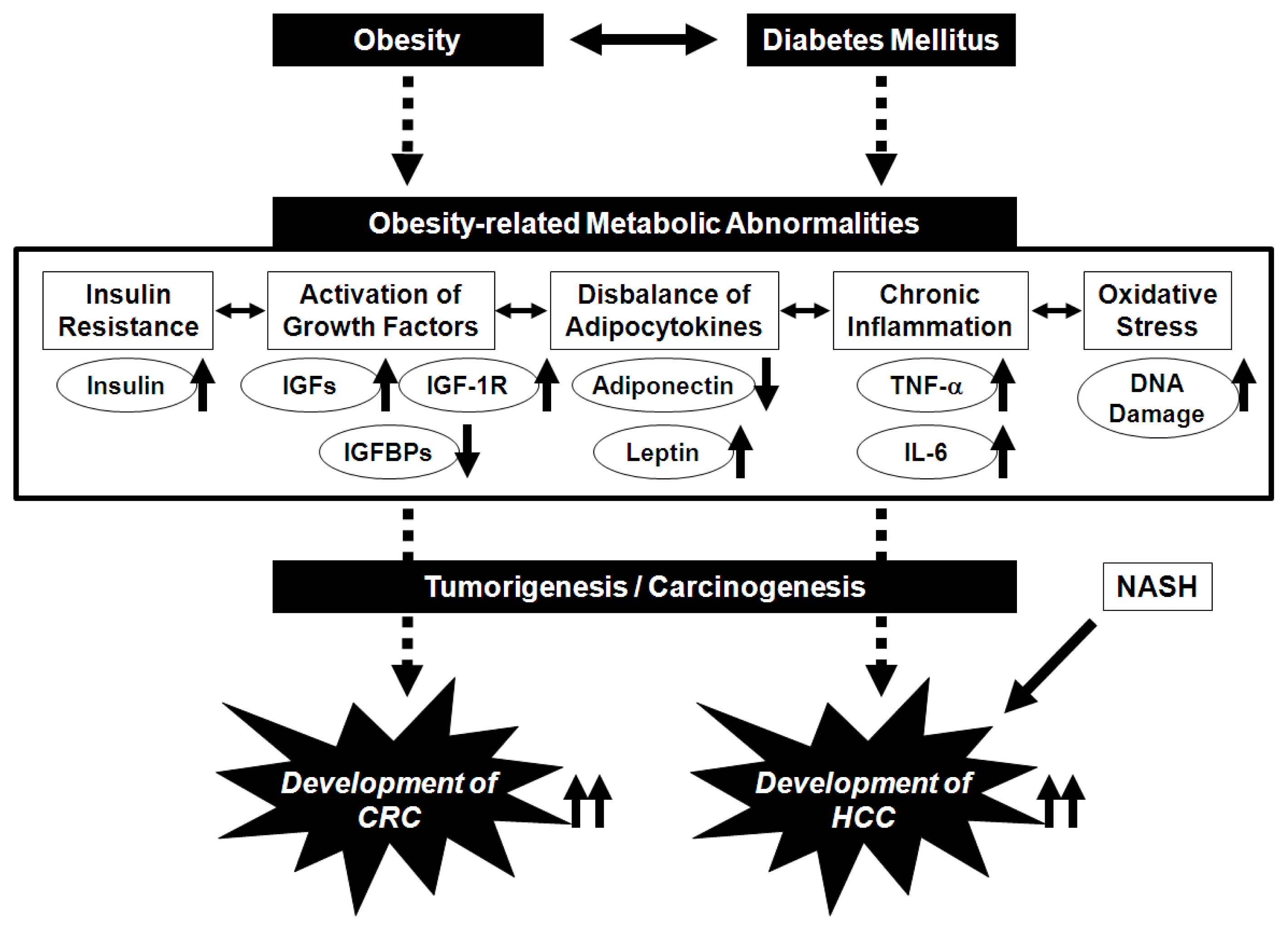
Obesity is the main determinant of insulin resistance and hyperinsulinemia, which is a risk factor for CRC [23]. Insulin itself and the signal transduction network it regulates have important roles in oncogenesis [24,25]. In animal models, insulin stimulates the growth of CRC cells while also promoting CRC tumor growth [26,27]. In addition, insulin resistance increases the biological activity of IGF-1, an important endocrine and paracrine regulator of tissue growth and metabolism. The binding of insulin and IGF-1 to the cell-surface receptors, insulin receptor and IGF-1R, respectively, on tumors and precancerous cells activates the phosphatidylinositol 3-kinase (PI3K)/Akt pathway, which is responsible for cellular processes like growth, proliferation, and survival [24,25]. Alterations in the IGF/IGF-1R axis caused by insulin resistance contribute to the development of CRC [28]. IGF-1 is positively correlated with body fat and waist circumference [29]. Moreover, insulin resistance and increased adipose mass create an oxidative environment in the tissues that upregulates the expression of various pro-inflammatory cytokines, including tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6), which stimulate tumor growth and progression [30–34]. Increased oxidative stress promotes damage to cell structures, including DNA, and activates the PI3K/Akt pathway, and both these processes play a key role in cancer development [35,36]. Therefore, insulin resistance and the subsequent inflammatory cascade involving increased oxidative stress are regarded as important factors in the development of obesity-associated CRC.
Excess production of storage lipids causes an adipocytokine imbalance, which entails increased levels of leptin and decreased levels of adiponectin in the serum, for example. This imbalance may also be related to obesity-associated carcinogenesis [37,38]. Leptin stimulates cell growth in CRC [39]. An epidemiologic study by Stattin et al. [40] suggested an association between circulating leptin levels and the development of CRC. TNF-α and IL-6 increase the levels of leptin, while leptin influences inflammatory responses, possibly by triggering the release of TNF-α and IL-6 [41–43]. These findings suggest that the pathophysiological abnormalities caused by obesity cooperatively aggravate the risk of cancers, including CRC, in obese individuals (Figure 1).
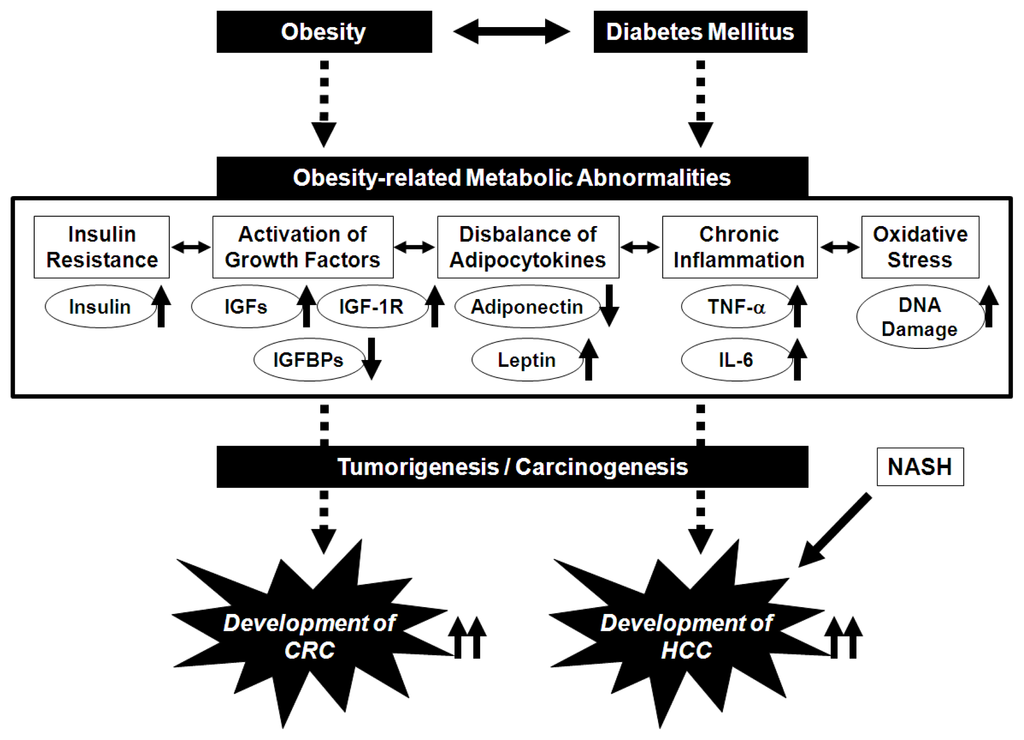
Figure 1.
Proposed mechanisms linking obesity and its related metabolic abnormalities to the development of colorectal cancer (CRC) and hepatocellular carcinoma (HCC).
3. Potential Pathophysiological Mechanisms Linking Obesity, Non-Alcoholic Fatty Liver Disease/Non-Alcoholic Steatohepatitis, and the Development of HCC
Several pathophysiological mechanisms linking obesity, steatosis, and liver carcinogenesis have been shown, including insulin resistance and the subsequent inflammatory cascade. Insulin induces HCC cells to proliferate and resist apoptosis [44,45]. Insulin resistance raises the risk for recurrence of HCC after curative radiofrequency ablation in hepatitis C virus-positive patients [46]. Insulin resistance also leads to an increased expression of TNF-α and its dysregulation is associated with the development of steatosis and inflammation within the liver [47]. Activation of the IGF/IGF-1R axis is involved with liver carcinogenesis [48,49]. High levels of serum leptin, which stimulates the growth of HCC cells [50], increase the risk of HCC recurrence after curative treatment [51]. These findings suggest that in addition to colorectal carcinogenesis, obesity and its related metabolic abnormalities also play an important role in the development of HCC (Figure 1).
Non-alcoholic fatty liver disease (NAFLD), which is known to be a hepatic manifestation of metabolic syndrome, is the most common form of chronic liver disease in developed countries [52,53]. It covers a spectrum of disorders ranging from simple steatosis to non-alcoholic steatohepatitis (NASH), which can progress to cirrhosis and thus HCC (Figure 1) [52,53]. Retrospective data suggest that in as many as 4–27% of cases, NASH progresses to HCC after cirrhosis develops [53,54]. Insulin resistance is considered a critical factor in the etiology of NASH [55]. The flux of free fatty acids to the liver and insulin resistance lead to hepatic fat accumulation, which causes inflammatory changes in the liver [56,57]. Enhanced TNF-α expression and increased leptin levels are also found in patients with NASH [58,59]. In addition, Wong et al. [60] recently reported interesting results from a cross-sectional study, indicating that NASH is associated with a high prevalence of colorectal adenomas and advanced neoplasms. This finding may suggest that in addition to HCC, NASH may be associated with an increased risk of CRC.
4. Preventive Effects of GTCs on the Metabolic Abnormalities and Cancer Development
Numerous studies have indicated that tea catechins, especially GTCs, are beneficial for various reasons, such as their anti-obesity effects [15]. A recent meta-analysis of clinical trials reported that GTCs help reduce body weight [61]. The underlying mechanisms include an increase in energy expenditure, stimulation of fatty acid oxidation, and reduction of nutrient absorption [62]. The effects of GTCs whereby they suppress metabolic syndrome have also been investigated in laboratory, epidemiological, and intervention studies [63,64]. In a rodent model of obesity and diabetes, treatment with green tea or its constituents was found to result in significantly reduced body weight and, therefore, improved hyperglycemia, hyperinsulinemia, hyperleptinemia, hepatic steatosis, and liver dysfunction [65–67]. GTCs supplementation was also found to decrease plasma levels of insulin, TNF-α, and IL-6 in a rat insulin resistance model [68]. These reports suggest that long-term treatment with GTCs may be effective for preventing the progression of obesity-related metabolic disorders.
In addition to the anti-obesity effects, GTCs possess anti-cancer and cancer-preventive properties [16–19]. Intervention studies provide clear evidence of the chemopreventive effects of tea preparations [69,70]. A pilot study also showed that GTCs successfully prevent colorectal adenomas, the precancerous lesions of CRC, after polypectomy [71]. Several properties of GTCs are responsible for their anti-cancer and cancer-preventive effects, including their antioxidant and anti-inflammatory properties [16,72]. An increasing number of studies have reported that GTCs, especially the major biologically active component in green tea (−)-epigallocatechin gallate (EGCG), inhibit proliferation of and induce apoptosis among cancer cells by modulating the activities of different receptor tyrosine kinases (RTKs) and their downstream signaling pathways, including the Ras/extracellular signal-regulated kinase (ERK) and PI3K/Akt signaling pathways [17–19,73,74]. EGCG suppresses cell growth by inhibiting the activation of IGF-1R, a member of the RTK family, in human CRC and HCC cells, and this inhibition is associated with a decrease in the expression of IGF-1/2, but an increase in the expression of IGF-binding protein-3 (IGFBP-3), which negatively controls the function of the IGF/IGF-1R axis [49,75]. EGCG also prevents carbon tetrachloride-induced hepatic fibrosis in rats by inhibiting IGF-1R expression [76]. These reports indicate that the IGF/IGF-1R axis, which plays a critical role in both cancer development and obesity-induced pathological events [24,25], might be a critical target of GTCs.
5. Preventive Effects of BCAA on Metabolic Abnormalities and HCC in Obese, Cirrhotic Patients: Results Form the LOTUS Study
Because the liver, an important target organ of insulin, plays a critical role in regulating metabolism, patients with chronic liver diseases often suffer from several nutritional and metabolic disorders, such as protein-energy malnutrition and insulin resistance [77–80]. Decreased serum levels of BCAA and albumin are associated with a high incidence of liver cirrhosis, while supplementation with BCAA has been shown to improve protein malnutrition and increase the serum albumin concentration in cirrhotic patients [20,77,78]. In addition, recent experimental studies have revealed that BCAA improves insulin resistance and glucose tolerance [81–83]. She et al. [81] reported that mitochondrial branched-chain aminotransferase knock out mice, which show a significant elevation in the serum BCAA level, exhibit decreased adiposity and remarkable improvements in glucose and insulin tolerance. BCAA has favorable effects on glucose metabolism not just in the liver but also in skeletal muscle and adipose tissue [84–86]. In the liver, BCAA activates liver-type glucokinase and glucose transporter (GLUT)-2, while suppressing the expression of glucose-6-phosphatase, which catalyzes the final steps of gluconeogenesis [84]. On the other hand, BCAA promotes glucose uptake through activation of PI3K and subsequent translocation of GLUT1 and GLUT4 to the plasma membrane in the skeletal muscle [86]. Moreover, in mice fed a high-fat diet, BCAA supplementation ameliorated insulin resistance by improving adipocytokine imbalance, inhibiting lipid accumulation in the liver, and increasing the hepatic levels of peroxisome proliferator-activated receptor-α [87,88]. Several clinical trials have also reported that oral BCAA supplementation improves glucose tolerance and insulin resistance in patients with chronic liver disease [22,89,90].
The Long-Term Survival Study (LOTUS) was a large-scale (n = 622) multicenter randomized controlled trial conducted from 1997 to 2003 in Japan to investigate the effects of supplemental BCAA therapy on event-free survival in patients with decompensated cirrhosis. In this trial, oral supplementation with a BCAA preparation was found to significantly prevent progressive hepatic failure and improve event-free survival [20]. Moreover, subset analysis from this trial demonstrated that long-term oral supplementation with BCAA is associated with a reduced frequency of HCC in obese patients (BMI score ≥ 25, P = 0.008) with decompensated cirrhosis [8]. What could the mechanisms of action of BCAA in the prevention of HCC have been? It seems reasonable to consider that the improvement of glucose metabolism by BCAA contributes to a decrease in the HCC incidence in obese cirrhotic patients because these patients generally have a particularly high incidence of hyperinsulinemia and insulin resistance [79,80]. In addition, Hagiwara et al. [91] recently reported significant findings that BCAA suppresses insulin-induced proliferation of HCC cells by inhibiting the insulin-induced activation of the PI3K/Akt pathway and the subsequent anti-apoptotic pathway. The precise mechanisms of action of BCAA in relation to carcinogenesis are explained in detail in the following sections.
6. Prevention of Obesity-Related CRC via the Nutraceutical Approach—GTCs and BCAA Effectively Prevent Obesity-Related Colorectal Carcinogenesis
Recent evidence indicates that increased body fatness and BMI are associated with an increased risk of CRC [4,5,9–11]. In contrast, studies have provided convincing evidence that dietary habits, especially high fruit and vegetable consumption, may reduce the risk of this malignancy [92]. Hirose et al. [93] established a useful preclinical model to determine the underlying mechanisms of how specific agents prevent the development of obesity-related CRC. The model used was C57BL/KsJ-db/db (db/db) mice, which are a genetically altered animal model with phenotypes of obesity and diabetes mellitus [94]. These mice have hyperlipidemia, hyperinsulinemia, and hyperleptinemia and are susceptible to the colonic carcinogen azoxymethane (AOM) because AOM-induced colonic precancerous lesions, aberrant crypt foci (ACF) and β-catenin accumulated crypts (BCAC), develop to a significantly greater extent in these mice than in the genetic control mice [93]. The colonic mucosa of db/db mice expresses high levels of IGF-1R, the phosphorylated (activated) form of IGF-1R (p-IGF-1R), β-catenin, and cyclooxygenase-2 (COX-2) [95]. Dietary supplementation with certain types of flavonoids, such as citrus compounds, suppresses the development of these putative lesions for CRC in the db/db mice [96–98].
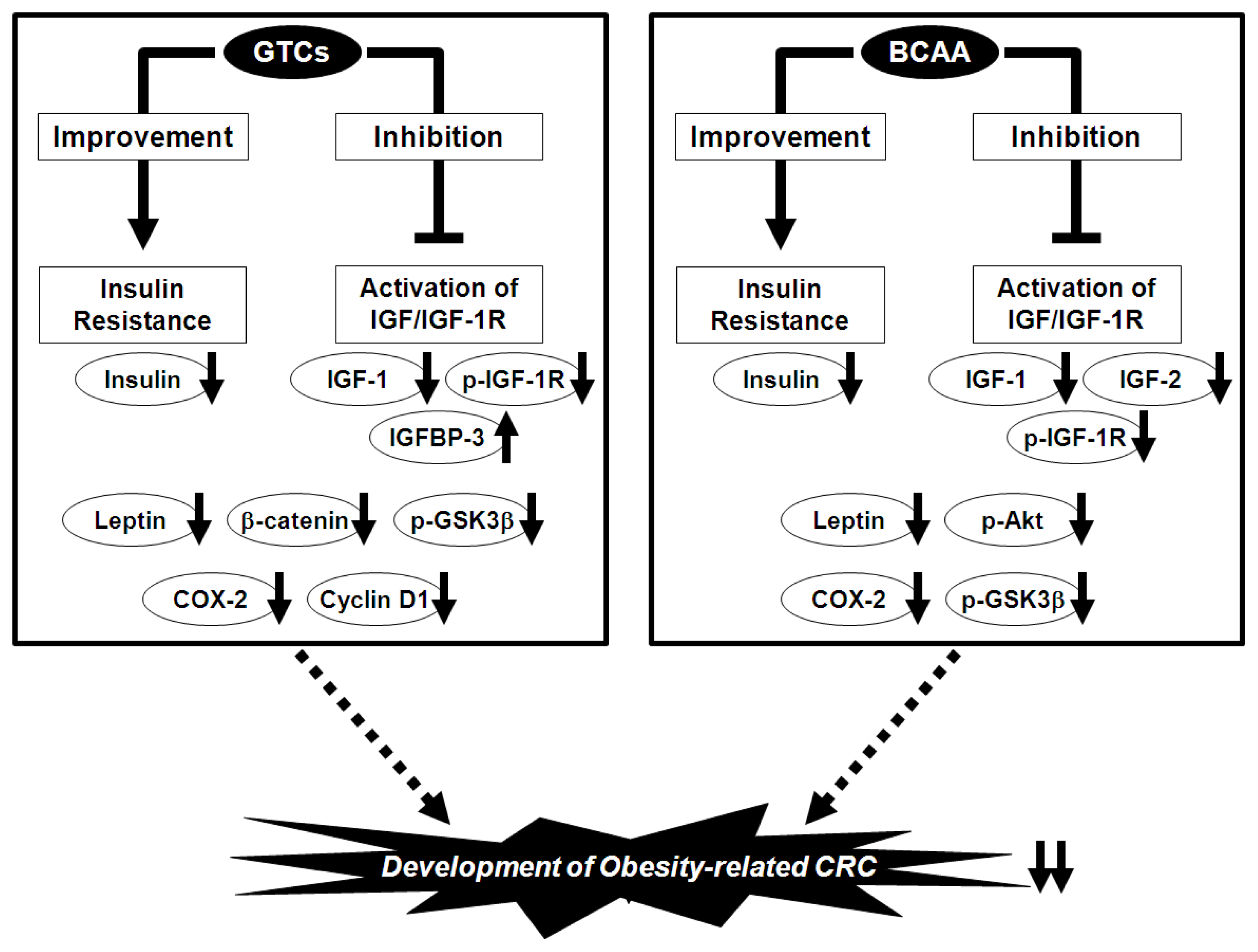
We used this experimental model to investigate in detail the effects of EGCG and BCAA on the prevention of obesity-related colorectal carcinogenesis. We found that drinking water with EGCG significantly decreased the number of ACF and BCAC, which accumulate the IGF-1R protein, and this decrease was associated with inhibited expression of IGF-1R, p-IGF-1R, the phosphorylated form of glycogen synthase kinase-3β (GSK-3β), β-catenin, COX-2, and cyclin D1 on the colonic mucosa [95]. EGCG also increased the serum level of IGFBP-3 while decreasing the serum levels of IGF-1, insulin, triglycerides, total cholesterol, and leptin [95]. In accordance with this study, supplementation with BCAA also caused a significant reduction in the number of ACF and BCAC compared with the control diet-fed groups by inhibiting the phosphorylation of IGF-1R, GSK-3β, and Akt on the colonic mucosa [99]. The serum levels of insulin, IGF-1, IGF-2, triglycerides, total cholesterol, and leptin were also decreased [99]. These findings suggest that both EGCG and BCAA effectively suppress the development of premalignant CRC lesions by suppressing the IGF/IGF-1R axis; improving hyperlipidemia, hyperinsulinemia, and hyperleptinemia; and inhibiting the expression of COX-2, which is involved in CRC development because it mediates inflammatory signaling pathways and can therefore be an important target for chemoprevention (Figure 2) [100].
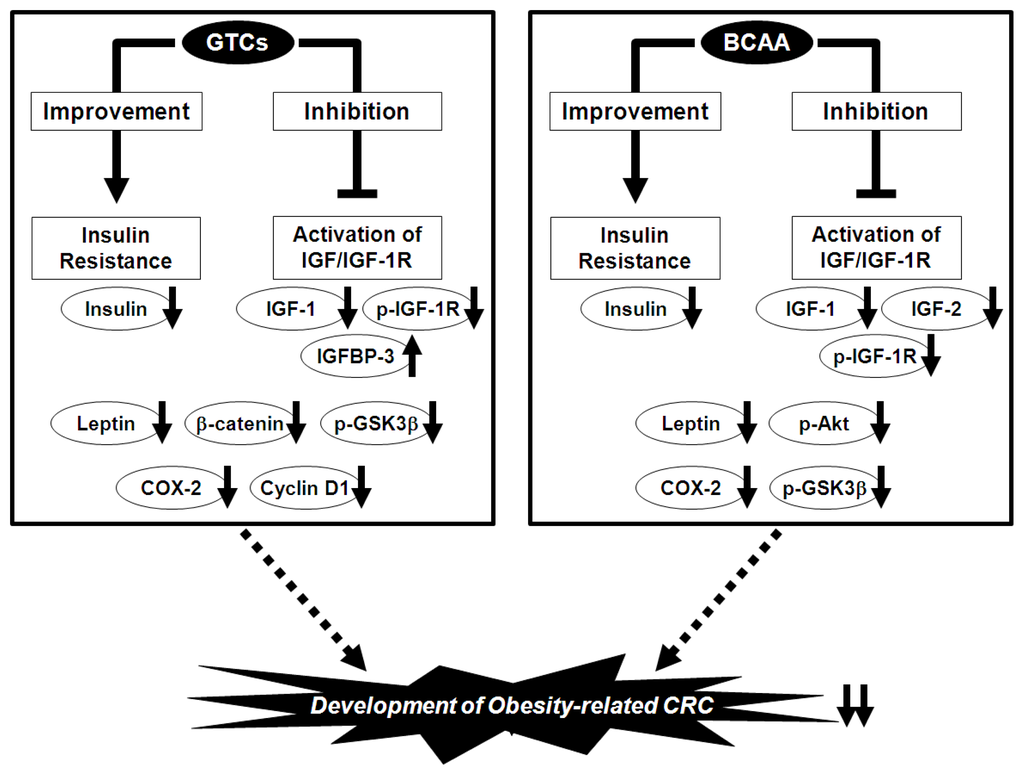
Figure 2.
Mechanisms of action of green tea catechins (GTCs) and branched-chain amino acids (BCAA) in the inhibition of obesity-related colorectal carcinogenesis.
7. Prevention of Obesity-Related HCC via the Nutraceutical Approach—BCAA and GTCs Effectively Prevent Obesity-Related Liver Carcinogenesis
In addition to established risk factors such as hepatitis and alcohol consumption, obesity and its related metabolic abnormalities increase the risk of HCC [6–8,11]. NASH is also an important pathological condition when considering the prevention of obesity-related HCC because it progresses to cirrhosis and finally develops into HCC [53,54]. In order to elucidate the pathogenesis of obesity-and NASH-associated HCC and evaluate the mechanisms of how chemopreventive agents suppress these diseases, we developed a useful preclinical model using db/db mice and a liver carcinogen N-diethylnitrosamine (DEN) [101]. We found that db/db mice, which have severe steatosis, are more susceptible to DEN-induced liver tumorigenesis than the genetic control mice, and this oncogenic sensitivity is associated with the activation of the IGF/IGF-1R axis and induction of chronic inflammation in the liver [13,101–103].
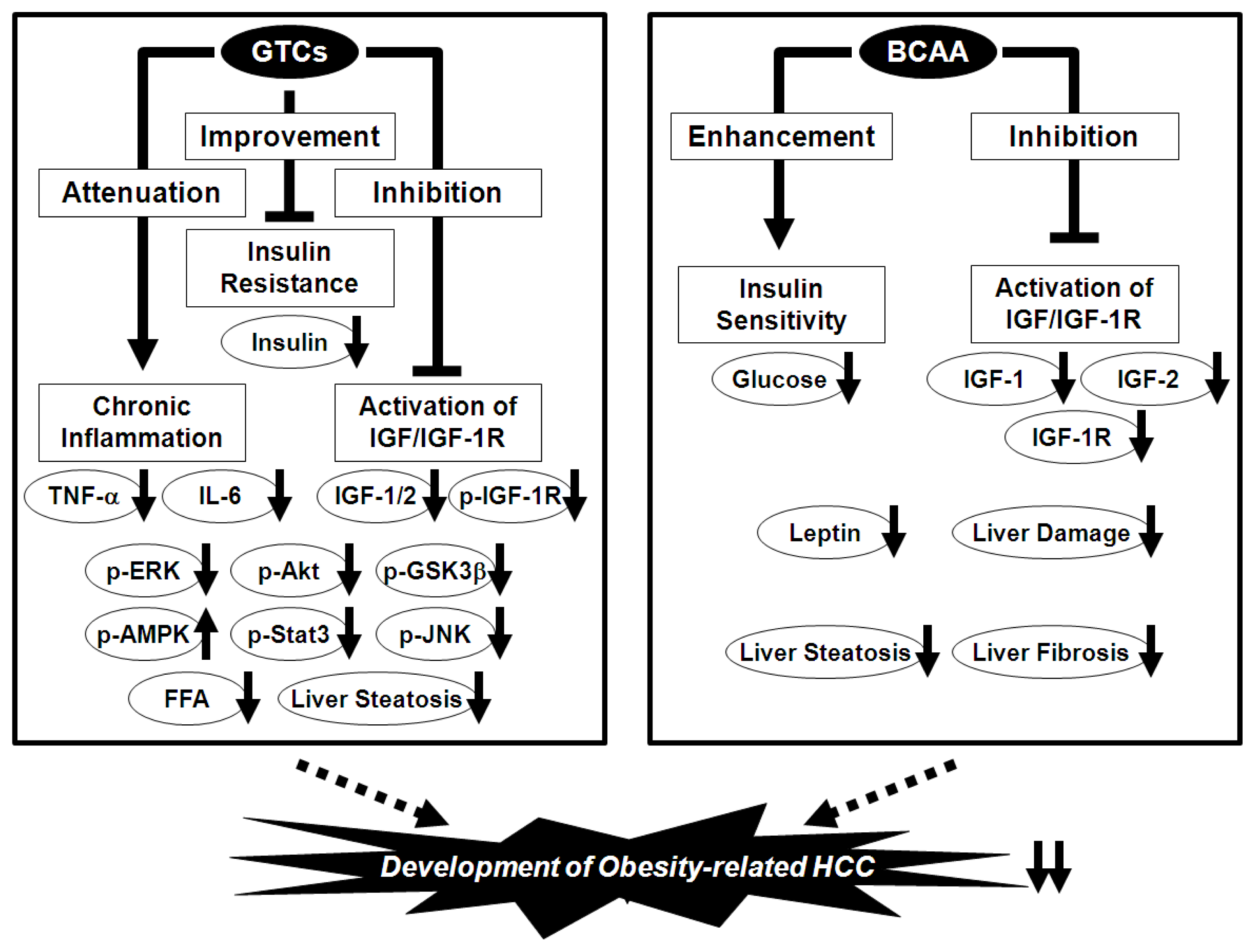
Using this experimental model, we also investigated the possible inhibitory effects of BCAA and EGCG on obesity-related liver tumorigenesis. We found that BCAA supplementation significantly suppressed the development of hepatic preneoplastic lesions, known as foci of cellular alteration (FCA), in obese and diabetic db/db mice by inhibiting the expression of IGF-1, IGF-2, and IGF-1R in the liver [101]. The development of liver neoplasms, including hepatic adenoma and HCC, was also reduced by BCAA supplementation and this was associated with improvement of insulin resistance, reduction of serum levels of leptin, and attenuation of hepatic steatosis and fibrosis [101]. Yoshiji et al. [104] also reported that the chemopreventive effect exerted by BCAA supplementation against HCC in obese and diabetic rats was associated with the suppression of vascular endothelial growth factor expression and hepatic neovascularization. In addition, drinking water containing EGCG significantly inhibited the development of FCA and hepatic adenoma, and improved hepatic steatosis [103]. The serum levels of insulin, IGF-1, and IGF-2 and the phosphorylation of the IGF-1R, ERK, Akt, and GSK-3β proteins in the liver were reduced by EGCG consumption [103]. EGCG also decreased the levels of free fatty acids and TNF-α in the serum and the expression of TNF-α, IL-6, IL-1β, and IL-18 mRNAs in the liver, indicating that it prevents obesity-related liver tumorigenesis by inhibiting the IGF/IGF-1R axis, improving hyperinsulinemia, and attenuating chronic inflammation [103]. Thus, both BCAA and GTCs may be useful for the chemoprevention of liver carcinogenesis in obese individuals (Figure 3).
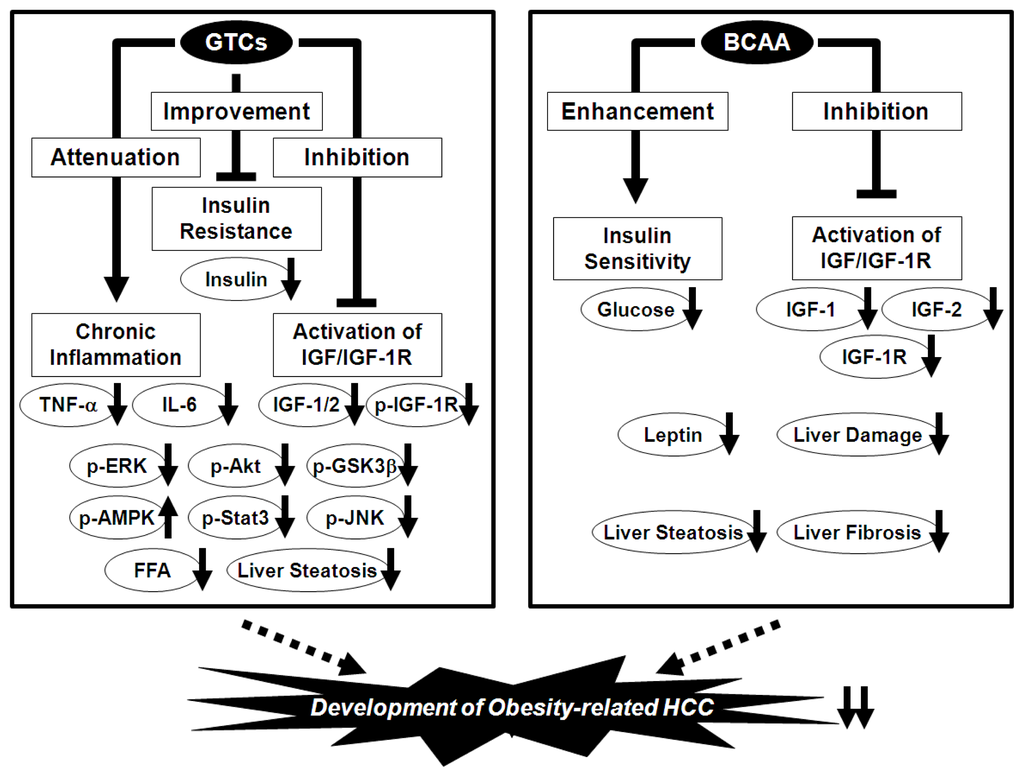
Figure 3.
Mechanisms of action of GTCs and BCAA in the inhibition of obesity-related liver carcinogenesis.
8. Conclusions
In the present social and medical circumstances, the consequences of obesity and its related metabolic abnormalities, including cancer, are critical issues that need to be resolved. Among human cancers, CRC and HCC are the most representative malignancies affected by obesity. In this review, we indicate the possibility that the nutraceutical approach for targeting and restoring metabolic homeostasis may be a promising strategy to prevent the development of obesity-related CRC and HCC. Tea catechins, especially GTCs, are considered one of the most practical agents for the prevention of obesity-related carcinogenesis because the safety and efficacy of GTCs as chemopreventive agents have been demonstrated in recent interventional trials [69,71]. BCAA is also a feasible agent because its preparations are widely used in clinical practice for patients with chronic liver diseases, and a randomized controlled trial has shown that BCAA supplementation can prevent HCC in such patients who are obese [8,20]. Thus, active intervention using GTCs and BCAA might be an effective approach for the chemoprevention of obesity-related CRC and HCC.
Acknowledgments
This review was based on studies supported in part by Grants-in-Aid from the Ministry of Education, Science, Sports and Culture of Japan (No. 22790638 to M.S. and No. 21590838 to H.M.) and by Grant-in-Aid for the 3rd Term Comprehensive 10-Year Strategy for Cancer Control from the Ministry of Health, Labour and Welfare of Japan.
- Conflict of Interest The authors declare no conflict of interest.
References
- WHO: World Health Organization. Fact Sheet for World Wide Prevalence of Obesity. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/indexhtml (accessed on 27 December 2011).
- Calle, E.E.; Kaaks, R. Overweight, obesity and cancer: Epidemiological evidence and proposed mechanisms. Nat. Rev. Cancer 2004, 4, 579–591. [Google Scholar]
- Anderson, A.S.; Caswell, S. Obesity management—An opportunity for cancer prevention. Surgeon 2009, 7, 282–285. [Google Scholar]
- Giovannucci, E.; Michaud, D. The role of obesity and related metabolic disturbances in cancers of the colon, prostate, and pancreas. Gastroenterology 2007, 132, 2208–2225. [Google Scholar]
- Frezza, E.E.; Wachtel, M.S.; Chiriva-Internati, M. Influence of obesity on the risk of developing colon cancer. Gut 2006, 55, 285–291. [Google Scholar]
- El-Serag, H.B.; Rudolph, K.L. Hepatocellular carcinoma: Epidemiology and molecular carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar]
- El-Serag, H.B.; Tran, T.; Everhart, J.E. Diabetes increases the risk of chronic liver disease and hepatocellular carcinoma. Gastroenterology 2004, 126, 460–468. [Google Scholar]
- Muto, Y.; Sato, S.; Watanabe, A.; Moriwaki, H.; Suzuki, K.; Kato, A.; Kato, M.; Nakamura, T.; Higuchi, K.; Nishiguchi, S.; et al. Overweight and obesity increase the risk for liver cancer in patients with liver cirrhosis and long-term oral supplementation with branched-chain amino acid granules inhibits liver carcinogenesis in heavier patients with liver cirrhosis. Hepatol. Res 2006, 35, 204–214. [Google Scholar]
- World Cancer Research Fund/American Institute for Cancer Research, Food, Nutrition, Physical activity, and the Prevention of Cancer: A Global Perspective 2007; AICR: Washington, DC, USA, 2007.
- Renehan, A.G.; Tyson, M.; Egger, M.; Heller, R.F.; Zwahlen, M. Body-mass index and incidence of cancer: A systematic review and meta-analysis of prospective observational studies. Lancet 2008, 371, 569–578. [Google Scholar]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N. Engl. J. Med 2003, 348, 1625–1638. [Google Scholar]
- Yasuda, Y.; Shimizu, M.; Shirakami, Y.; Sakai, H.; Kubota, M.; Hata, K.; Hirose, Y.; Tsurumi, H.; Tanaka, T.; Moriwaki, H. Pitavastatin inhibits azoxymethane-induced colonic preneoplastic lesions in C57BL/KsJ-db/db obese mice. Cancer Sci 2010, 101, 1701–1707. [Google Scholar]
- Shimizu, M.; Yasuda, Y.; Sakai, H.; Kubota, M.; Terakura, D.; Baba, A.; Ohno, T.; Kochi, T.; Tsurumi, H.; Tanaka, T.; et al. Pitavastatin suppresses diethylnitrosamine-induced liver preneoplasms in male C57BL/KsJ-db/db obese mice. BMC Cancer 2011, 11. [Google Scholar] [CrossRef]
- Kubota, M.; Shimizu, M.; Sakai, H.; Yasuda, Y.; Ohno, T.; Kochi, T.; Tsurumi, H.; Tanaka, T.; Moriwaki, H. Renin-angiotensin system inhibitors suppress azoxymethane-induced colonic preneoplastic lesions in C57BL/KsJ-db/db obese mice. Biochem. Biophys. Res. Commun 2011, 410, 108–113. [Google Scholar]
- Kao, Y.H.; Chang, H.H.; Lee, M.J.; Chen, C.L. Tea, obesity, and diabetes. Mol. Nutr. Food Res 2006, 50, 188–210. [Google Scholar]
- Yang, C.S.; Wang, X.; Lu, G.; Picinich, S.C. Cancer prevention by tea: Animal studies, molecular mechanisms and human relevance. Nat. Rev. Cancer 2009, 9, 429–439. [Google Scholar]
- Shimizu, M.; Shirakami, Y.; Moriwaki, H. Targeting receptor tyrosine kinases for chemoprevention by green tea catechin, EGCG. Int. J. Mol. Sci 2008, 9, 1034–1049. [Google Scholar]
- Shimizu, M.; Adachi, S.; Masuda, M.; Kozawa, O.; Moriwaki, H. Cancer chemoprevention with green tea catechins by targeting receptor tyrosine kinases. Mol. Nutr. Food Res 2011, 55, 832–843. [Google Scholar]
- Shimizu, M.; Weinstein, I.B. Modulation of signal transduction by tea catechins and related phytochemicals. Mutat. Res 2005, 591, 147–160. [Google Scholar]
- Muto, Y.; Sato, S.; Watanabe, A.; Moriwaki, H.; Suzuki, K.; Kato, A.; Kato, M.; Nakamura, T.; Higuchi, K.; Nishiguchi, S.; et al. Effects of oral branched-chain amino acid granules on event-free survival in patients with liver cirrhosis. Clin. Gastroenterol. Hepatol 2005, 3, 705–713. [Google Scholar]
- Marchesini, G.; Bianchi, G.; Merli, M.; Amodio, P.; Panella, C.; Loguercio, C.; Fanelli, F.R.; Abbiati, R. Italian BCAA Study Group. Nutritional supplementation with branched-chain amino acids in advanced cirrhosis: A double-blind randomized trial. Gastroenterology 2003, 124, 1792–1801. [Google Scholar]
- Kawaguchi, T.; Nagao, Y.; Matsuoka, H.; Ide, T.; Sata, M. Branched-chain amino acid-enriched supplementation improves insulin resistance in patients with chronic liver disease. Int. J. Mol. Med 2008, 22, 105–112. [Google Scholar]
- Chang, C.K.; Ulrich, C.M. Hyperinsulinaemia and hyperglycaemia: Possible risk factors of colorectal cancer among diabetic patients. Diabetologia 2003, 46, 595–607. [Google Scholar]
- Pollak, M. Insulin and insulin-like growth factor signalling in neoplasia. Nat. Rev. Cancer 2008, 8, 915–928. [Google Scholar]
- Clayton, P.E.; Banerjee, I.; Murray, P.G.; Renehan, A.G. Growth hormone, the insulin-like growth factor axis, insulin and cancer risk. Nat. Rev. Endocrinol 2011, 7, 11–24. [Google Scholar]
- Bjork, J.; Nilsson, J.; Hultcrantz, R.; Johansson, C. Growth-regulatory effects of sensory neuropeptides, epidermal growth factor, insulin, and somatostatin on the non-transformed intestinal epithelial cell line IEC-6 and the colon cancer cell line HT 29. Scand. J. Gastroenterol 1993, 28, 879–884. [Google Scholar]
- Tran, T.T.; Medline, A.; Bruce, W.R. Insulin promotion of colon tumors in rats. Cancer Epidemiol. Biomark. Prev 1996, 5, 1013–1015. [Google Scholar]
- Singh, P.; Rubin, N. Insulinlike growth factors and binding proteins in colon cancer. Gastroenterology 1993, 105, 1218–1237. [Google Scholar]
- Kajantie, E.; Fall, C.H.; Seppala, M.; Koistinen, R.; Dunkel, L.; Yliharsila, H.; Osmond, C.; Andersson, S.; Barker, D.J.; Forsen, T.; et al. Serum insulin-like growth factor (IGF)-I and IGF-binding protein-1 in elderly people: Relationships with cardiovascular risk factors, body composition, size at birth, and childhood growth. J. Clin. Endocrinol. Metab 2003, 88, 1059–1065. [Google Scholar]
- Esposito, K.; Nappo, F.; Marfella, R.; Giugliano, G.; Giugliano, F.; Ciotola, M.; Quagliaro, L.; Ceriello, A.; Giugliano, D. Inflammatory cytokine concentrations are acutely increased by hyperglycemia in humans: Role of oxidative stress. Circulation 2002, 106, 2067–2072. [Google Scholar]
- Hotamisligil, G.S.; Shargill, N.S.; Spiegelman, B.M. Adipose expression of tumor necrosis factor-alpha: Direct role in obesity-linked insulin resistance. Science 1993, 259, 87–91. [Google Scholar]
- Hotamisligil, G.S.; Peraldi, P.; Budavari, A.; Ellis, R.; White, M.F.; Spiegelman, B.M. IRS-1-mediated inhibition of insulin receptor tyrosine kinase activity in TNF-alpha- and obesity-induced insulin resistance. Science 1996, 271, 665–668. [Google Scholar]
- Kern, P.A.; Ranganathan, S.; Li, C.; Wood, L.; Ranganathan, G. Adipose tissue tumor necrosis factor and interleukin-6 expression in human obesity and insulin resistance. Am. J. Physiol. Endocrinol. Metab 2001, 280, E745–E751. [Google Scholar]
- Szlosarek, P.; Charles, K.A.; Balkwill, F.R. Tumour necrosis factor-alpha as a tumour promoter. Eur. J. Cancer 2006, 42, 745–750. [Google Scholar]
- Valko, M.; Izakovic, M.; Mazur, M.; Rhodes, C.J.; Telser, J. Role of oxygen radicals in DNA damage and cancer incidence. Mol. Cell. Biochem 2004, 266, 37–56. [Google Scholar]
- Leslie, N.R. The redox regulation of PI 3-kinase-dependent signaling. Antioxid. Redox Signal 2006, 8, 1765–1774. [Google Scholar]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L.; et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med 1996, 334, 292–295. [Google Scholar]
- Barb, D.; Williams, C.J.; Neuwirth, A.K.; Mantzoros, C.S. Adiponectin in relation to malignancies: A review of existing basic research and clinical evidence. Am. J. Clin. Nutr 2007, 86, s858–s866. [Google Scholar]
- Amemori, S.; Ootani, A.; Aoki, S.; Fujise, T.; Shimoda, R.; Kakimoto, T.; Shiraishi, R.; Sakata, Y.; Tsunada, S.; Iwakiri, R.; et al. Adipocytes and preadipocytes promote the proliferation of colon cancer cells in vitro. Am. J. Physiol. Gastrointest. Liver Physiol 2007, 292, G923–G929. [Google Scholar]
- Stattin, P.; Lukanova, A.; Biessy, C.; Soderberg, S.; Palmqvist, R.; Kaaks, R.; Olsson, T.; Jellum, E. Obesity and colon cancer: Does leptin provide a link? Int. J. Cancer 2004, 109, 149–152. [Google Scholar]
- Faggioni, R.; Feingold, K.R.; Grunfeld, C. Leptin regulation of the immune response and the immunodeficiency of malnutrition. FASEB J 2001, 15, 2565–2571. [Google Scholar]
- Molina, A.; Vendrell, J.; Gutierrez, C.; Simon, I.; Masdevall, C.; Soler, J.; Gomez, J.M. Insulin resistance, leptin and TNF-alpha system in morbidly obese women after gastric bypass. Obes. Surg 2003, 13, 615–621. [Google Scholar]
- Fenton, J.I.; Hursting, S.D.; Perkins, S.N.; Hord, N.G. Interleukin-6 production induced by leptin treatment promotes cell proliferation in an ApcMin/+ colon epithelial cell line. Carcinogenesis 2006, 27, 1507–1515. [Google Scholar]
- Kang, S.; Song, J.; Kang, H.; Kim, S.; Lee, Y.; Park, D. Insulin can block apoptosis by decreasing oxidative stress via phosphatidylinositol 3-kinase- and extracellular signal-regulated protein kinase-dependent signaling pathways in HepG2 cells. Eur. J. Endocrinol 2003, 148, 147–155. [Google Scholar]
- Tornkvist, A.; Parpal, S.; Gustavsson, J.; Stralfors, P. Inhibition of Raf-1 kinase expression abolishes insulin stimulation of DNA synthesis in H4IIE hepatoma cells. J. Biol. Chem 1994, 269, 13919–13921. [Google Scholar]
- Imai, K.; Takai, K.; Nishigaki, Y.; Shimizu, S.; Naiki, T.; Hayashi, H.; Uematsu, T.; Sugihara, J.; Tomita, E.; Shimizu, M.; et al. Insulin resistance raises the risk for recurrence of stage I hepatocellular carcinoma after curative radiofrequency ablation in hepatitis C virus-positive patients: A prospective, case series study. Hepatol. Res 2010, 40, 376–382. [Google Scholar]
- Powell, E.E.; Jonsson, J.R.; Clouston, A.D. Steatosis: Co-factor in other liver diseases. Hepatology 2005, 42, 5–13. [Google Scholar]
- Alexia, C.; Fallot, G.; Lasfer, M.; Schweizer-Groyer, G.; Groyer, A. An evaluation of the role of insulin-like growth factors (IGF) and of type-I IGF receptor signalling in hepatocarcinogenesis and in the resistance of hepatocarcinoma cells against drug-induced apoptosis. Biochem. Pharmacol 2004, 68, 1003–1015. [Google Scholar]
- Shimizu, M.; Shirakami, Y.; Sakai, H.; Tatebe, H.; Nakagawa, T.; Hara, Y.; Weinstein, I.B.; Moriwaki, H. EGCG inhibits activation of the insulin-like growth factor (IGF)/IGF-1 receptor axis in human hepatocellular carcinoma cells. Cancer Lett 2008, 262, 10–18. [Google Scholar]
- Chen, C.; Chang, Y.C.; Liu, C.L.; Liu, T.P.; Chang, K.J.; Guo, I.C. Leptin induces proliferation and anti-apoptosis in human hepatocarcinoma cells by up-regulating cyclin D1 and down-regulating Bax via a Janus kinase 2-linked pathway. Endocr. Relat. Cancer 2007, 14, 513–529. [Google Scholar]
- Watanabe, N.; Takai, K.; Imai, K.; Shimizu, M.; Naiki, T.; Nagaki, M.; Moriwaki, H. Increased levels of serum leptin are a risk factor for the recurrence of stage I/II hepatocellular carcinoma after curative treatment. J. Clin. Biochem. Nutr 2011, 49, 153–158. [Google Scholar]
- Angulo, P. Nonalcoholic fatty liver disease. N. Engl. J. Med 2002, 346, 1221–1231. [Google Scholar]
- Siegel, A.B.; Zhu, A.X. Metabolic syndrome and hepatocellular carcinoma: Two growing epidemics with a potential link. Cancer 2009, 115, 5651–5661. [Google Scholar]
- Ratziu, V.; Bonyhay, L.; Di Martino, V.; Charlotte, F.; Cavallaro, L.; Sayegh-Tainturier, M.H.; Giral, P.; Grimaldi, A.; Opolon, P.; Poynard, T. Survival, liver failure, and hepatocellular carcinoma in obesity-related cryptogenic cirrhosis. Hepatology 2002, 35, 1485–1493. [Google Scholar]
- Day, C.P.; James, O.F. Steatohepatitis: A tale of two “hits”? Gastroenterology 1998, 114, 842–845. [Google Scholar]
- Chitturi, S.; Abeygunasekera, S.; Farrell, G.C.; Holmes-Walker, J.; Hui, J.M.; Fung, C.; Karim, R.; Lin, R.; Samarasinghe, D.; Liddle, C.; et al. NASH and insulin resistance: Insulin hypersecretion and specific association with the insulin resistance syndrome. Hepatology 2002, 35, 373–379. [Google Scholar]
- Feldstein, A.E.; Werneburg, N.W.; Canbay, A.; Guicciardi, M.E.; Bronk, S.F.; Rydzewski, R.; Burgart, L.J.; Gores, G.J. Free fatty acids promote hepatic lipotoxicity by stimulating TNF-alpha expression via a lysosomal pathway. Hepatology 2004, 40, 185–194. [Google Scholar]
- Crespo, J.; Cayon, A.; Fernandez-Gil, P.; Hernandez-Guerra, M.; Mayorga, M.; Dominguez-Diez, A.; Fernandez-Escalante, J.C.; Pons-Romero, F. Gene expression of tumor necrosis factor alpha and TNF-receptors, p55 and p75, in nonalcoholic steatohepatitis patients. Hepatology 2001, 34, 1158–1163. [Google Scholar]
- Chitturi, S.; Farrell, G.; Frost, L.; Kriketos, A.; Lin, R.; Fung, C.; Liddle, C.; Samarasinghe, D.; George, J. Serum leptin in NASH correlates with hepatic steatosis but not fibrosis: A manifestation of lipotoxicity? Hepatology 2002, 36, 403–409. [Google Scholar]
- Wong, V.W.; Wong, G.L.; Tsang, S.W.; Fan, T.; Chu, W.C.; Woo, J.; Chan, A.W.; Choi, P.C.; Chim, A.M.; Lau, J.Y.; et al. High prevalence of colorectal neoplasm in patients with non-alcoholic steatohepatitis. Gut 2011, 60, 829–836. [Google Scholar]
- Hursel, R.; Viechtbauer, W.; Westerterp-Plantenga, M.S. The effects of green tea on weight loss and weight maintenance: A meta-analysis. Int. J. Obes 2009, 33, 956–961. [Google Scholar]
- Rains, T.M.; Agarwal, S.; Maki, K.C. Antiobesity effects of green tea catechins: A mechanistic review. J. Nutr. Biochem 2011, 22, 1–7. [Google Scholar]
- Grove, K.A.; Lambert, J.D. Laboratory, epidemiological, and human intervention studies show that tea (Camellia sinensis) may be useful in the prevention of obesity. J. Nutr 2010, 140, 446–453. [Google Scholar]
- Thielecke, F.; Boschmann, M. The potential role of green tea catechins in the prevention of the metabolic syndrome—A review. Phytochemistry 2009, 70, 11–24. [Google Scholar]
- Ramadan, G.; El-Beih, N.M.; El-Ghffar, E.A.A. Modulatory effects of black v. green tea aqueous extract on hyperglycaemia, hyperlipidaemia and liver dysfunction in diabetic and obese rat models. Br. J. Nutr 2009, 102, 1611–1619. [Google Scholar]
- Murase, T.; Nagasawa, A.; Suzuki, J.; Hase, T.; Tokimitsu, I. Beneficial effects of tea catechins on diet-induced obesity: Stimulation of lipid catabolism in the liver. Int. J. Obes 2002, 26, 1459–1464. [Google Scholar]
- Bose, M.; Lambert, J.D.; Ju, J.; Reuhl, K.R.; Shapses, S.A.; Yang, C.S. The major green tea polyphenol, (−)-epigallocatechin-3-gallate, inhibits obesity, metabolic syndrome, and fatty liver disease in high-fat-fed mice. J. Nutr 2008, 138, 1677–1683. [Google Scholar]
- Qin, B.; Polansky, M.M.; Harry, D.; Anderson, R.A. Green tea polyphenols improve cardiac muscle mRNA and protein levels of signal pathways related to insulin and lipid metabolism and inflammation in insulin-resistant rats. Mol. Nutr. Food Res 2010, 54, S14–S23. [Google Scholar]
- Bettuzzi, S.; Brausi, M.; Rizzi, F.; Castagnetti, G.; Peracchia, G.; Corti, A. Chemoprevention of human prostate cancer by oral administration of green tea catechins in volunteers with high-grade prostate intraepithelial neoplasia: A preliminary report from a one-year proof-of-principle study. Cancer Res 2006, 66, 1234–1240. [Google Scholar]
- Li, N.; Sun, Z.; Han, C.; Chen, J. The chemopreventive effects of tea on human oral precancerous mucosa lesions. Proc. Soc. Exp. Biol. Med 1999, 220, 218–224. [Google Scholar]
- Shimizu, M.; Fukutomi, Y.; Ninomiya, M.; Nagura, K.; Kato, T.; Araki, H.; Suganuma, M.; Fujiki, H.; Moriwaki, H. Green tea extracts for the prevention of metachronous colorectal adenomas: A pilot study. Cancer Epidemiol. Biomark. Prev 2008, 17, 3020–3025. [Google Scholar]
- Shirakami, Y.; Shimizu, M.; Tsurumi, H.; Hara, Y.; Tanaka, T.; Moriwaki, H. EGCG and Polyphenon E attenuate inflammation-related mouse colon carcinogenesis induced by AOM plus DDS. Mol. Med. Rep 2008, 1, 355–361. [Google Scholar]
- Adachi, S.; Nagao, T.; Ingolfsson, H.I.; Maxfield, F.R.; Andersen, O.S.; Kopelovich, L.; Weinstein, I.B. The inhibitory effect of (−)-epigallocatechin gallate on activation of the epidermal growth factor receptor is associated with altered lipid order in HT29 colon cancer cells. Cancer Res 2007, 67, 6493–6501. [Google Scholar]
- Adachi, S.; Nagao, T.; To, S.; Joe, A.K.; Shimizu, M.; Matsushima-Nishiwaki, R.; Kozawa, O.; Moriwaki, H.; Maxfield, F.R.; Weinstein, I.B. (−)-Epigallocatechin gallate causes internalization of the epidermal growth factor receptor in human colon cancer cells. Carcinogenesis 2008, 29, 1986–1993. [Google Scholar]
- Shimizu, M.; Deguchi, A.; Hara, Y.; Moriwaki, H.; Weinstein, I.B. EGCG inhibits activation of the insulin-like growth factor-1 receptor in human colon cancer cells. Biochem. Biophys. Res. Commun 2005, 334, 947–953. [Google Scholar]
- Yasuda, Y.; Shimizu, M.; Sakai, H.; Iwasa, J.; Kubota, M.; Adachi, S.; Osawa, Y.; Tsurumi, H.; Hara, Y.; Moriwaki, H. (−)-Epigallocatechin gallate prevents carbon tetrachloride-induced rat hepatic fibrosis by inhibiting the expression of the PDGFRbeta and IGF-1R. Chem. Biol. Interact 2009, 182, 159–164. [Google Scholar]
- Moriwaki, H.; Miwa, Y.; Tajika, M.; Kato, M.; Fukushima, H.; Shiraki, M. Branched-chain amino acids as a protein- and energy-source in liver cirrhosis. Biochem. Biophys. Res. Commun 2004, 313, 405–409. [Google Scholar]
- Kawaguchi, T.; Izumi, N.; Charlton, M.R.; Sata, M. Branched-chain amino acids as pharmacological nutrients in chronic liver disease. Hepatology 2011, 54, 1063–1070. [Google Scholar]
- Petrides, A.S.; Vogt, C.; Schulze-Berge, D.; Matthews, D.; Strohmeyer, G. Pathogenesis of glucose intolerance and diabetes mellitus in cirrhosis. Hepatology 1994, 19, 616–627. [Google Scholar]
- Mehta, S.H.; Brancati, F.L.; Sulkowski, M.S.; Strathdee, S.A.; Szklo, M.; Thomas, D.L. Prevalence of type 2 diabetes mellitus among persons with hepatitis C virus infection in the United States. Ann. Intern. Med 2000, 133, 592–599. [Google Scholar]
- She, P.; Reid, T.M.; Bronson, S.K.; Vary, T.C.; Hajnal, A.; Lynch, C.J.; Hutson, S.M. Disruption of BCATm in mice leads to increased energy expenditure associated with the activation of a futile protein turnover cycle. Cell Metab 2007, 6, 181–194. [Google Scholar]
- Zhang, Y.; Guo, K.; LeBlanc, R.E.; Loh, D.; Schwartz, G.J.; Yu, Y.H. Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes 2007, 56, 1647–1654. [Google Scholar]
- Ikehara, O.; Kawasaki, N.; Maezono, K.; Komatsu, M.; Konishi, A. Acute and chronic treatment of l-isoleucine ameliorates glucose metabolism in glucose-intolerant and diabetic mice. Biol. Pharm. Bull 2008, 31, 469–472. [Google Scholar]
- Higuchi, N.; Kato, M.; Miyazaki, M.; Tanaka, M.; Kohjima, M.; Ito, T.; Nakamuta, M.; Enjoji, M.; Kotoh, K.; Takayanagi, R. Potential role of branched-chain amino acids in glucose metabolism through the accelerated induction of the glucose-sensing apparatus in the liver. J. Cell. Biochem 2011, 112, 30–38. [Google Scholar]
- Hinault, C.; Mothe-Satney, I.; Gautier, N.; Lawrence, J.C., Jr; van Obberghen, E. Amino acids and leucine allow insulin activation of the PKB/mTOR pathway in normal adipocytes treated with wortmannin and in adipocytes from db/dbmice. FASEB J 2004, 18, 1894–1896. [Google Scholar]
- Nishitani, S.; Takehana, K.; Fujitani, S.; Sonaka, I. Branched-chain amino acids improve glucose metabolism in rats with liver cirrhosis. Am. J. Physiol. Gastrointest. Liver Physiol 2005, 288, G1292–G1300. [Google Scholar]
- Arakawa, M.; Masaki, T.; Nishimura, J.; Seike, M.; Yoshimatsu, H. The effects of branched-chain amino acid granules on the accumulation of tissue triglycerides and uncoupling proteins in diet-induced obese mice. Endocr. J 2011, 58, 161–170. [Google Scholar]
- Nishimura, J.; Masaki, T.; Arakawa, M.; Seike, M.; Yoshimatsu, H. Isoleucine prevents the accumulation of tissue triglycerides and upregulates the expression of PPARalpha and uncoupling protein in diet-induced obese mice. J. Nutr 2010, 140, 496–500. [Google Scholar]
- Sakaida, I.; Tsuchiya, M.; Okamoto, M.; Okita, K. Late evening snack and the change of blood glucose level in patients with liver cirrhosis. Hepatol. Res 2004, 30, 67–72. [Google Scholar]
- Urata, Y.; Okita, K.; Korenaga, K.; Uchida, K.; Yamasaki, T.; Sakaida, I. The effect of supplementation with branched-chain amino acids in patients with liver cirrhosis. Hepatol. Res 2007, 37, 510–516. [Google Scholar]
- Hagiwara, A.; Nishiyama, M.; Ishizaki, S. Branched-chain amino acids prevent insulin-induced hepatic tumor cell proliferation by inducing apoptosis through mTORC1 and mTORC2-dependent mechanisms. J. Cell. Physiol 2011. [Google Scholar] [CrossRef]
- Forte, A.; de Sanctis, R.; Leonetti, G.; Manfredelli, S.; Urbano, V.; Bezzi, M. Dietary chemoprevention of colorectal cancer. Ann. Ital. Chir 2008, 79, 261–267. [Google Scholar]
- Hirose, Y.; Hata, K.; Kuno, T.; Yoshida, K.; Sakata, K.; Yamada, Y.; Tanaka, T.; Reddy, B.S.; Mori, H. Enhancement of development of azoxymethane-induced colonic premalignant lesions in C57BL/KsJ-db/db mice. Carcinogenesis 2004, 25, 821–825. [Google Scholar]
- Lee, G.H.; Proenca, R.; Montez, J.M.; Carroll, K.M.; Darvishzadeh, J.G.; Lee, J.I.; Friedman, J.M. Abnormal splicing of the leptin receptor in diabetic mice. Nature 1996, 379, 632–635. [Google Scholar]
- Shimizu, M.; Shirakami, Y.; Sakai, H.; Adachi, S.; Hata, K.; Hirose, Y.; Tsurumi, H.; Tanaka, T.; Moriwaki, H. (−)-Epigallocatechin gallate suppresses azoxymethane-induced colonic premalignant lesions in male C57BL/KsJ-db/db mice. Cancer Prev. Res 2008, 1, 298–304. [Google Scholar]
- Suzuki, R.; Kohno, H.; Yasui, Y.; Hata, K.; Sugie, S.; Miyamoto, S.; Sugawara, K.; Sumida, T.; Hirose, Y.; Tanaka, T. Diet supplemented with citrus unshiu segment membrane suppresses chemically induced colonic preneoplastic lesions and fatty liver in male db/db mice. Int. J. Cancer 2007, 120, 252–258. [Google Scholar]
- Hayashi, K.; Suzuki, R.; Miyamoto, S.; Shin-Ichiroh, Y.; Kohno, H.; Sugie, S.; Takashima, S.; Tanaka, T. Citrus auraptene suppresses azoxymethane-induced colonic preneoplastic lesions in C57BL/KsJ-db/db mice. Nutr. Cancer 2007, 58, 75–84. [Google Scholar]
- Miyamoto, S.; Yasui, Y.; Ohigashi, H.; Tanaka, T.; Murakami, A. Dietary flavonoids suppress azoxymethane-induced colonic preneoplastic lesions in male C57BL/KsJ-db/db mice. Chem. Biol. Interact 2010, 183, 276–283. [Google Scholar]
- Shimizu, M.; Shirakami, Y.; Iwasa, J.; Shiraki, M.; Yasuda, Y.; Hata, K.; Hirose, Y.; Tsurumi, H.; Tanaka, T.; Moriwaki, H. Supplementation with branched-chain amino acids inhibits azoxymethane-induced colonic preneoplastic lesions in male C57BL/KsJ-db/db mice. Clin. Cancer Res 2009, 15, 3068–3075. [Google Scholar]
- Gupta, R.A.; Dubois, R.N. Colorectal cancer prevention and treatment by inhibition of cyclooxygenase-2. Nat. Rev. Cancer 2001, 1, 11–21. [Google Scholar]
- Iwasa, J.; Shimizu, M.; Shiraki, M.; Shirakami, Y.; Sakai, H.; Terakura, Y.; Takai, K.; Tsurumi, H.; Tanaka, T.; Moriwaki, H. Dietary supplementation with branched-chain amino acids suppresses diethylnitrosamine-induced liver tumorigenesis in obese and diabetic C57BL/KsJ-db/db mice. Cancer Sci 2010, 101, 460–467. [Google Scholar]
- Shimizu, M.; Sakai, H.; Shirakami, Y.; Iwasa, J.; Yasuda, Y.; Kubota, M.; Takai, K.; Tsurumi, H.; Tanaka, T.; Moriwaki, H. Acyclic retinoid inhibits diethylnitrosamine-induced liver tumorigenesis in obese and diabetic C57BLKS/J-+Leprdb/+Leprdb mice. Cancer Prev. Res 2011, 4, 128–136. [Google Scholar]
- Shimizu, M.; Sakai, H.; Shirakami, Y.; Yasuda, Y.; Kubota, M.; Terakura, D.; Baba, A.; Ohno, T.; Hara, Y.; Tanaka, T.; et al. Preventive Effects of (−)-Epigallocatechin gallate on diethylnitrosamine-induced liver tumorigenesis in obese and diabetic C57BL/KsJ-db/db mice. Cancer Prev. Res 2011, 4, 396–403. [Google Scholar]
- Yoshiji, H.; Noguchi, R.; Kitade, M.; Kaji, K.; Ikenaka, Y.; Namisaki, T.; Yoshii, J.; Yanase, K.; Yamazaki, M.; Tsujimoto, T.; et al. Branched-chain amino acids suppress insulin-resistance-based hepatocarcinogenesis in obese diabetic rats. J. Gastroenterol 2009, 44, 483–491. [Google Scholar]
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).