Anti-Fatigue Properties of Tartary Buckwheat Extracts in Mice
Abstract
:1. Introduction
2. Results and Discussion
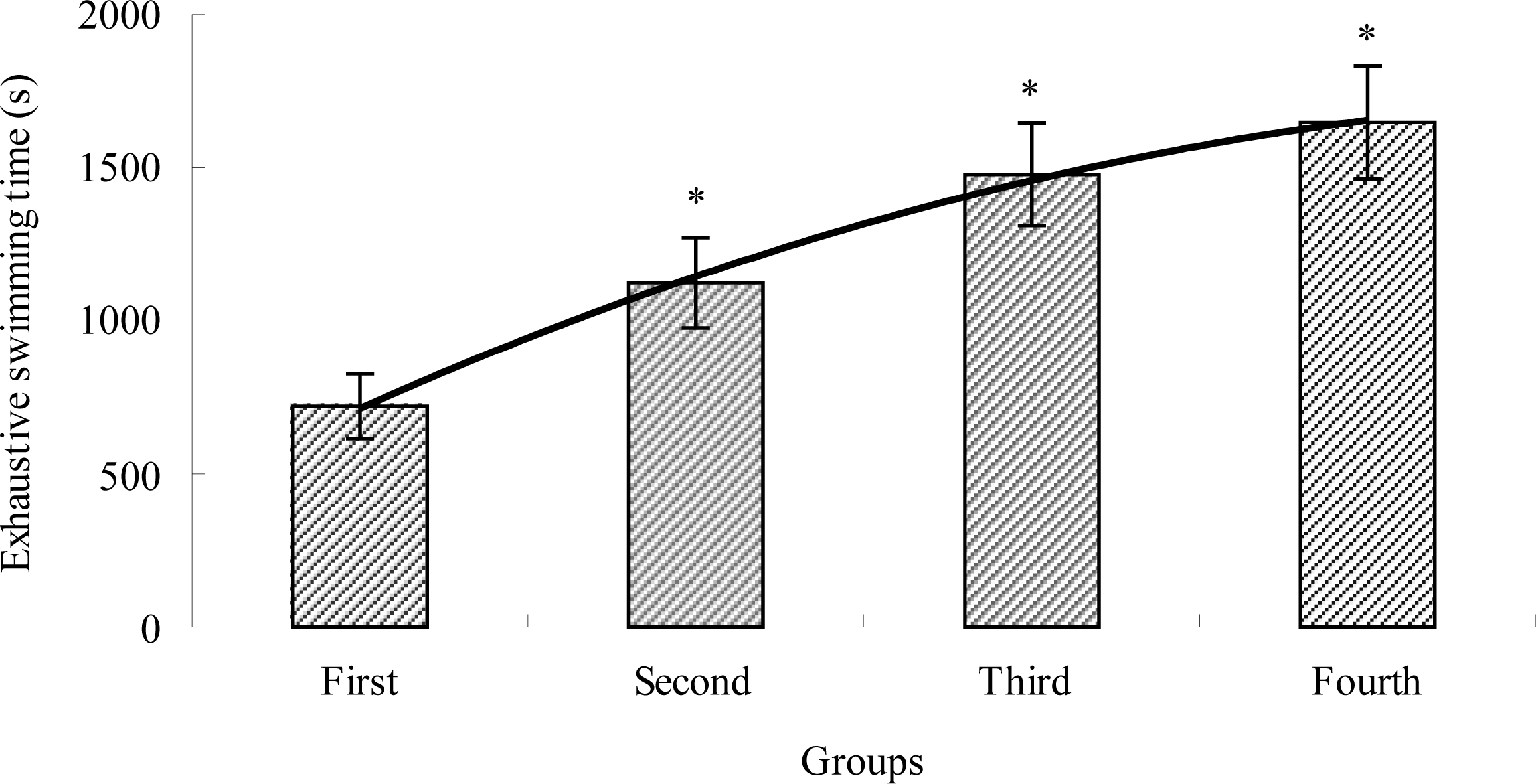
2.1. The Effects of TBE on Exhaustive Swimming Time of Mice
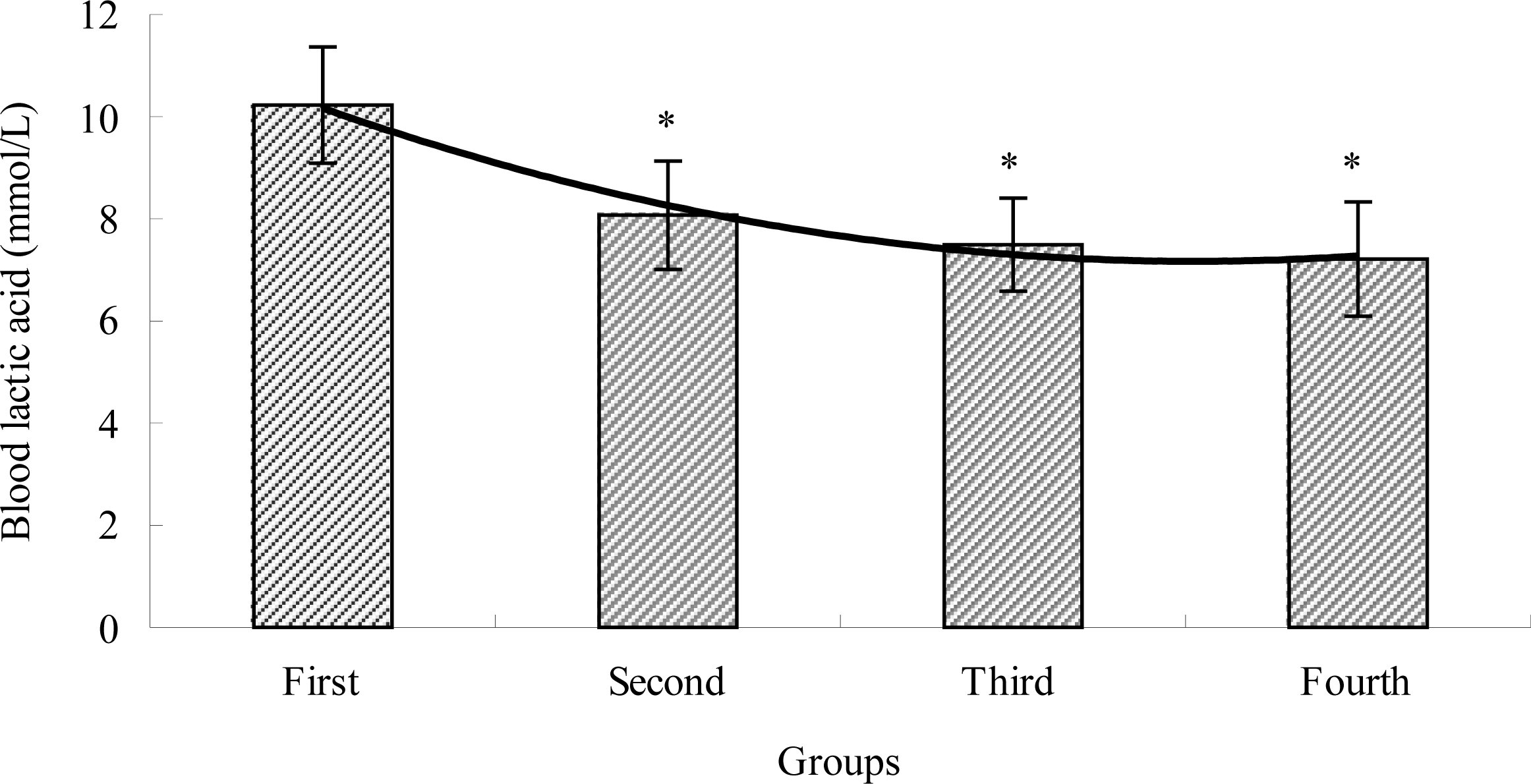
2.2. The Effects of TBE on BLA of Mice
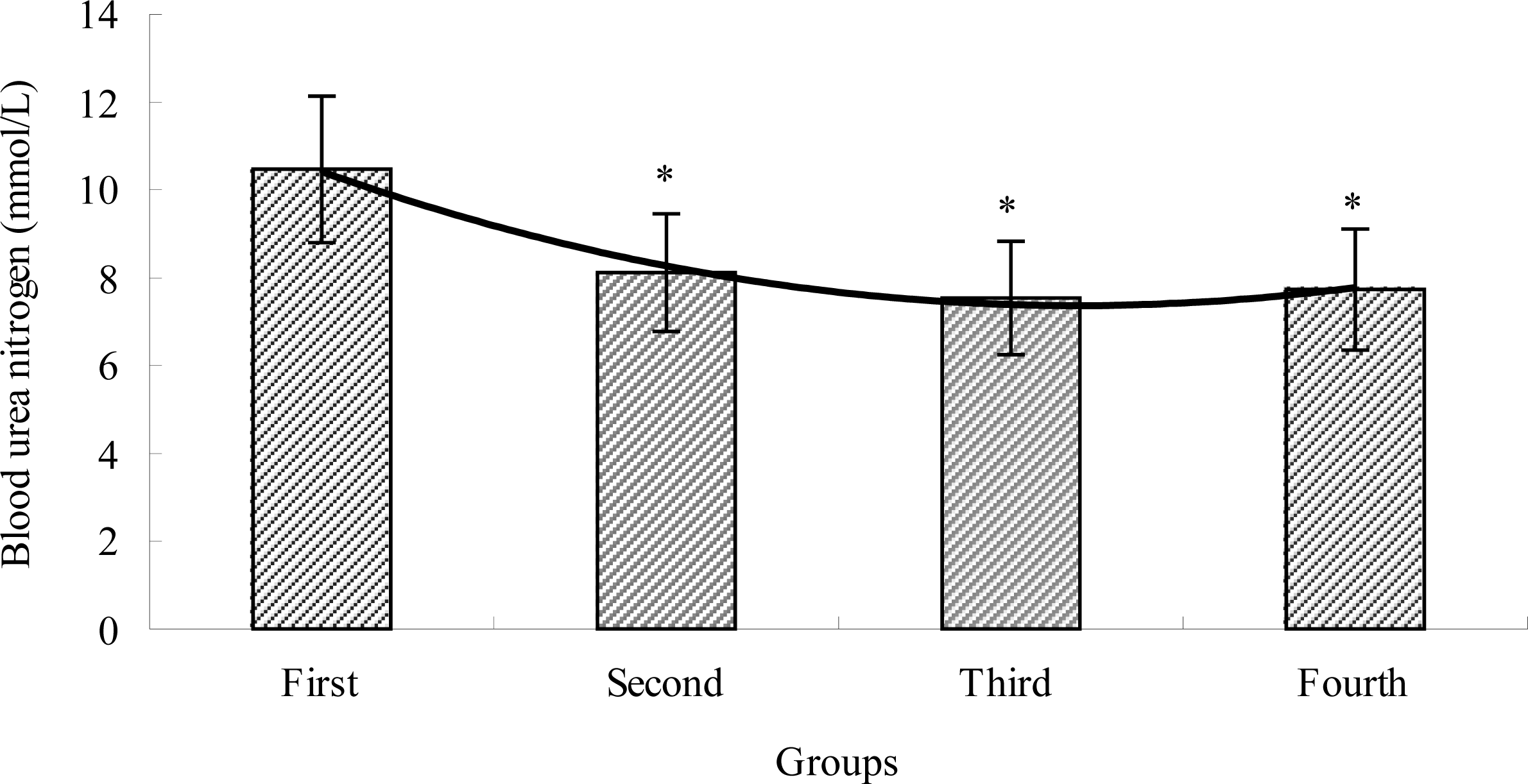
2.3. The Effects of TBE on BUN of Mice
2.4. The Effects of TBE on Tissue Glycogen of Mice
2.5. The Effects of TBE on GPx and SOD of Mice
3. Experimental Section
3.1. Plant Material
3.2. Chemicals and Reagents
3.3. Preparation of Tartary Buckwheat Extracts
3.4. Measuring the Contents of Flavonoids
3.5. Animals and Care
3.6. Grouping of Animal
3.7. Exhaustive Swimming Test
3.8. Analysis of Biochemical Parameters
3.9. Statistical Analysis
4. Conclusions
References
- Huang, LZ; Huang, BK; Ye, Q; Qin, LP. Bioactivity-guided fractionation for anti-fatigue property of Acanthopanax senticosus. J. Ethnopharmacol 2011, 133, 213–219. [Google Scholar]
- Akazawa, KH; Cui, Y; Tanaka, M; Kataoka, Y; Yoneda, Y; Watanabe, Y. Mapping of regional brain activation in response to fatigue-load and recovery in rats with c-Fos immunohistochemistry. Neurosci. Res 2010, 66, 372–379. [Google Scholar]
- Chen, JR; Wang, TJ; Huang, HY; Chen, LJ; Huang, YS; Wang, YJ; Tseng, GF. Fatigue reversibly reduced cortical and hippocampal dendritic spines concurrent with compromise of motor endurance and spatial memory. Neuroscience 2009, 161, 1104–1113. [Google Scholar]
- Liu, W; Liu, WL; Liu, CM; Liu, JH; Yang, SB; Zheng, HJ; Lei, HW; Ruan, R; Li, T; Tu, ZC; et al. Medium-chain fatty acid nanoliposomes for easy energy supply. Nutrition 2011, 27, 700–706. [Google Scholar]
- Tanaka, M; Baba, Y; Kataoka, Y; Kinbara, N; Sagesaka, YM; Kakuda, T; Watanabe, Y. Effects of (-)-epigallocatechin gallate in liver of an animal model of combined (physical and mental) fatigue. Nutrition 2008, 24, 599–603. [Google Scholar]
- Jin, G; Kataoka, Y; Tanaka, M; Mizuma, H; Nozaki, S; Tahara, T; Mizuno, K; Yamato, M; Watanabe, Y. Changes in plasma and tissue amino acid levels in an animal model of complex fatigue. Nutrition 2009, 25, 597–607. [Google Scholar]
- You, LJ; Zhao, MM; Regenstein, JM; Ren, JY. In vitro antioxidant activity and in vivo anti-fatigue effect of loach (Misgurnus anguillicaudatus) peptides prepared by papain digestion. Food Chem 2011, 124, 188–194. [Google Scholar]
- Wang, L; Zhang, HL; Lu, R; Zhou, YJ; Ma, R; Lv, JQ; Li, XL; Chen, LJ; Yao, Z. The decapeptide CMS001 enhances swimming endurance in mice. Peptides 2008, 29, 1176–1182. [Google Scholar]
- Huang, SC; Lee, FT; Kuo, TY; Yang, JH; Chien, CT. Attenuation of long-term Rhodiola rosea supplementation on exhaustive swimming-evoked oxidative stress in the rat tissues. Chin. J. Physiol 2009, 52, 316–324. [Google Scholar]
- Powers, SK; DeRuisseau, KC; Quindry, J; Hamilton, KL. Dietary antioxidants and exercise. J. Sports Sci 2004, 22, 81–94. [Google Scholar]
- Abbey, EL; Rankin, JW. Effect of quercetin supplementation on repeated-sprint performance, xanthine oxidase activity, and inflammation. Int. J. Sport Nutr. Exerc. Metab 2011, 21, 91–96. [Google Scholar]
- König, D; Wagner, KH; Elmadfa, I; Berg, A. Exercise and oxidative stress: Significance of antioxidants with reference to inflammatory, muscular, and systemic stress. Exerc. Immunol. Rev 2001, 7, 108–133. [Google Scholar]
- Kim, KM; Yu, KW; Kang, DH; Koh, JH; Hong, BS; Suh, HJ. Anti-stress and anti-fatigue effects of fermented rice bran. Biosci. Biotechnol. Biochem 2001, 65, 2294–2296. [Google Scholar]
- Ikeda, K. Buckwheat: Composition, chemistry, and processing. Adv. Food Nutr. Res 2002, 44, 395–434. [Google Scholar]
- Cao, W; Chen, WJ; Suo, ZR; Yao, YP. Protective effects of ethanolic extracts of buckwheat groats on DNA damage caused by hydroxyl radicals. Food Res. Inter 2008, 41, 924–929. [Google Scholar]
- Bonafaccia, G; Gambelli, L; Fabjan, N; Kreft, I. Trace elements in flour and bran from common and tartary buck wheat. Food Chem 2003, 83, 1–5. [Google Scholar]
- Guo, XN; Yao, HY; Chen, Z. Effect of heat, rutin and disulfide bond reduction on in vitro pepsin digestibility of Chinese tartary buckwheat protein fractions. Food Chem 2007, 102, 118–122. [Google Scholar]
- Liu, CL; Chen, YS; Yang, JH; Chiang, BH. Antioxidant activity of tartary (Fagopyrum tataricum (L.) Gaertn.) and common (Fagopyrum esculentum moench) buckwheat sprouts. J. Agric. Food Chem 2008, 56, 173–178. [Google Scholar]
- Gu, Y. Processing technology for non-staple cereals-buckwheat processing. Cereal Feed Ind 1999, 7, 19–26. [Google Scholar]
- Li, D; Xiao, G; Ding, X. Study on antioxidant effect of tartary buckwheat flavonoids. J. Wuxi Univ. Light Ind 2001, 20, 44–47. [Google Scholar]
- Tomotake, H; Yamamoto, N; Yanaka, N; Ohinata, H; Yamazaki, R; Kayashita, J. High protein buckwheat flour suppresses hypercholesterolemia in rats and gallstone formation in mice by hypercholesterolemic diet and body fat in rats because of its low protein digestibility. Nutrition 2006, 22, 166–173. [Google Scholar]
- Yao, Y; Shan, F; Bian, J; Chen, F; Wang, M; Ren, G. D-chiro-inositol-enriched tartary buckwheat bran extract lowers the blood glucose level in KK-Ay mice. J. Agric. Food Chem 2008, 56, 10027–10031. [Google Scholar]
- Wang, M; Liu, JR; Gao, JM; Parry, JW; Wei, YM. Antioxidant activity of Tartary buckwheat bran extract and its effect on the lipid profile of hyperlipidemic rats. J. Agric. Food Chem 2009, 57, 5106–5112. [Google Scholar]
- Ushida, Y; Matsui, T; Tanaka, M; Matsumoto, K; Hosoyama, H; Mitomi, A; Sagesaka, Y; Kakuda, T. Endothelium-dependent vasorelaxation effect of rutin-free tartary buckwheat extract in isolated rat thoracic aorta. J. Nutr. Biochem 2008, 19, 700–707. [Google Scholar]
- Fujital, K; Inoue, N; Yang, ZF; Hagiwara, SJ; Hagiwara, M. Varietal differences of antioxidant activity in tartary buckwheat flour as evaluated by chemiluminescence. Fagopyrum 2003, 20, 47–52. [Google Scholar]
- Fabjan, N; Rode, J; Kosir, IJ; Wang, Z; Zhang, Z; Kreft, I. Tartary buckwheat (Fagopyrum tataricum Gaertn.) as a source of dietary rutin and quercitrin. J. Agric. Food Chem 2003, 51, 6452–6455. [Google Scholar]
- Belluardo, N; Westerblad, H; Mudo, G; Casabona, A; Bruton, J; Caniglia, G; Pastoris, O; Grassi, F; Ibanez, CF. Neuromuscular junction disassembly and muscle fatigue in mice lacking neurotrophin-4. Mol. Cell. Neurosci 2001, 18, 56–67. [Google Scholar]
- Tanaka, M; Nakamura, F; Mizokawa, S; Matsumura, A; Nozaki, S; Watanabe, Y. Establishment and assessment of a rat model of fatigue. Neurosci. Lett 2003, 352, 159–162. [Google Scholar]
- Wei, W; Zheng, LY; Yu, MY; Jiang, N; Yang, ZR; Luo, X. Anti-fatigue activity of extract form the submerged fermentation of Ganoderma Lucidum using Radix astragali as substrate. J. Anim. Plant Sci 2010, 3, 677–684. [Google Scholar]
- Cairns, SP. Lactic acid and exercise performance: Culprit or friend? Sports Med 2006, 36, 279–291. [Google Scholar]
- Yao, LQ; Li, FL. Lycium barbarum polysaccharides ameliorates physical fatigue. Afr. J. Agric. Res 2010, 5, 2153–2157. [Google Scholar]
- Wang, L; Zhang, HL; Lu, R; Zhou, YJ; Ma, R; Lv, JQ; Li, XL; Chen, LJ; Yao, Z. The decapeptide CMS001 enhances swimming endurance in mice. Peptides 2008, 29, 1176–1182. [Google Scholar]
- Zhang, Y; Yao, X; Bao, B; Zhang, Y. Anti-fatigue activity of a triterpenoid-rich extract from Chinese bamboo shavings (Caulis bamfusae in Taeniam). Phytother Res 2006, 20, 872–876. [Google Scholar]
- Ding, JF; Li, YY; Xu, JJ; Su, XR; Gao, X; Yue, FP. Study on effect of jellyfish collagen hydrolysate on anti-fatigue and anti-oxidation. Food Hydrocol 2011, 25, 1350–1353. [Google Scholar]
- Favier, RJ; Koubi, HE. Metabolic and structural adaptations to exercise in chronic intermittent fasted rats. Am. J. Physiol 1988, 254, 877–884. [Google Scholar]
- Jung, K; Kim, IH; Han, D. Effect of medicinal plant extracts on forced swimming capacity in mice. J. Ethnopharmacol 2004, 93, 75–81. [Google Scholar]
- Shang, H; Cao, S; Wang, J; Zheng, H; Putheti, R. Glabridin from Chinese herb licorice inhibits fatigue in mice. Afr. J. Tradit. Complement. Altern. Med 2009, 7, 17–23. [Google Scholar]
- Powers, SK; DeRuisseau, KC; Quindry, J; Hamilton, KL. Dietary antioxidants and exercise. J. Sports Sci 2004, 22, 81–94. [Google Scholar]
- Chen, CY; Holtzman, GI; Bakhit, RM. High-genistin isoflavone supplementation modulated erythrocyte antioxidant enzymes and increased running endurance in rats undergoing one session of exhausting exercise—A pilot study. Pak. J. Nutr 2004, 1, 1–7. [Google Scholar]
- Powers, S; Lennon, SL. Analysis of cellular responses to free radicals: Focus on exercise and skeletal muscle. Proc. Nutr. Soc 1999, 58, 1025–1033. [Google Scholar]
- Zhang, M; Chen, HX; Li, JL; Pei, Y; Liang, Y. Antioxidant properties of tartary buckwheat extracts as affected by different thermal processing methods. LWT Food Sci. Technol 2010, 43, 181–185. [Google Scholar]
- Zhu, HB; Wang, YZ; Liu, YX; Xia, YL. Analysis of flavonoids in Portulaca oleracea L. by UV-Vis spectrophotometry with comparative study on different extraction technologies. Food Anal. Method 2010, 3, 90–97. [Google Scholar]
- Xu, YQ; Zhang, R; Fu, H. Optimal process to extract flavonoids from red-raspberry studies on the optimal process to extract flavonoids from red-raspberry fruits. Nat. Sci 2005, 3, 43–45. [Google Scholar]
- Chen, Y; Kong, LD; Xia, X; Kung, HF; Zhang, L. Behavioral and biochemical studies of total furocoumarins from seeds of Psoralea corylifolia in the forced swimming test in mice. J. Ethnopharmacol 2005, 96, 451–459. [Google Scholar]
- Wu, JL; Wu, QP; Huang, JM; Chen, R; Cai, M; Tan, JB. Effects of L-malate on physical stamina and activities of enzymes related to the malate-aspartate shuttle in liver of mice. Physiol. Res 2007, 56, 213–220. [Google Scholar]
- Wang, ZB; Yan, B. Gastrodia elata Blume extract ameliorates exercise induced fatigue. Afr. J. Biotechnol 2010, 9, 5978–5982. [Google Scholar]
- Jung, KA; Han, D; Kwon, EK; Lee, CH; Kim, YE. Antifatigue effect of Rubus coreanus Miquel extract in mice. J. Med. Food 2007, 10, 689–693. [Google Scholar]
- Tang, W; Zhang, Y; Gao, J; Ding, X; Gao, S. The anti-fatigue effect of 20(R)-ginsenoside Rg3 in mice by intranasally administration. Biol. Pharm. Bull 2008, 31, 2024–2027. [Google Scholar]
| Groups | Liver glycogen (mg/g) | Muscle glycogen (mg/g) |
|---|---|---|
| First | 7.25 ± 1.19 | 1.36 ± 0.21 |
| Second | 14.86 ± 2.34 * | 2.04 ± 0.34 * |
| Third | 18.73 ± 2.06 * | 2.48 ± 0.27 * |
| Fourth | 19.16 ± 3.28 * | 2.97 ± 0.38 * |
| Groups | GPx (u/mg protein) | SOD (NU/mg protein) |
|---|---|---|
| First | 47.81 ± 4.16 | 76.38 ± 5.69 |
| Second | 58.29 ± 4.87 * | 91.25 ± 6.84 * |
| Third | 67.84 ± 5.23 * | 113.68 ± 7.25 * |
| Fourth | 78.23 ± 4.28 * | 148.26 ± 6.81 * |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jin, H.-M.; Wei, P. Anti-Fatigue Properties of Tartary Buckwheat Extracts in Mice. Int. J. Mol. Sci. 2011, 12, 4770-4780. https://doi.org/10.3390/ijms12084770
Jin H-M, Wei P. Anti-Fatigue Properties of Tartary Buckwheat Extracts in Mice. International Journal of Molecular Sciences. 2011; 12(8):4770-4780. https://doi.org/10.3390/ijms12084770
Chicago/Turabian StyleJin, Hong-Mei, and Ping Wei. 2011. "Anti-Fatigue Properties of Tartary Buckwheat Extracts in Mice" International Journal of Molecular Sciences 12, no. 8: 4770-4780. https://doi.org/10.3390/ijms12084770
APA StyleJin, H.-M., & Wei, P. (2011). Anti-Fatigue Properties of Tartary Buckwheat Extracts in Mice. International Journal of Molecular Sciences, 12(8), 4770-4780. https://doi.org/10.3390/ijms12084770




