Klebsiella pneumoniae yggG Gene Product: A Zinc-Dependent Metalloprotease
Abstract
:1. Introduction
2. Results
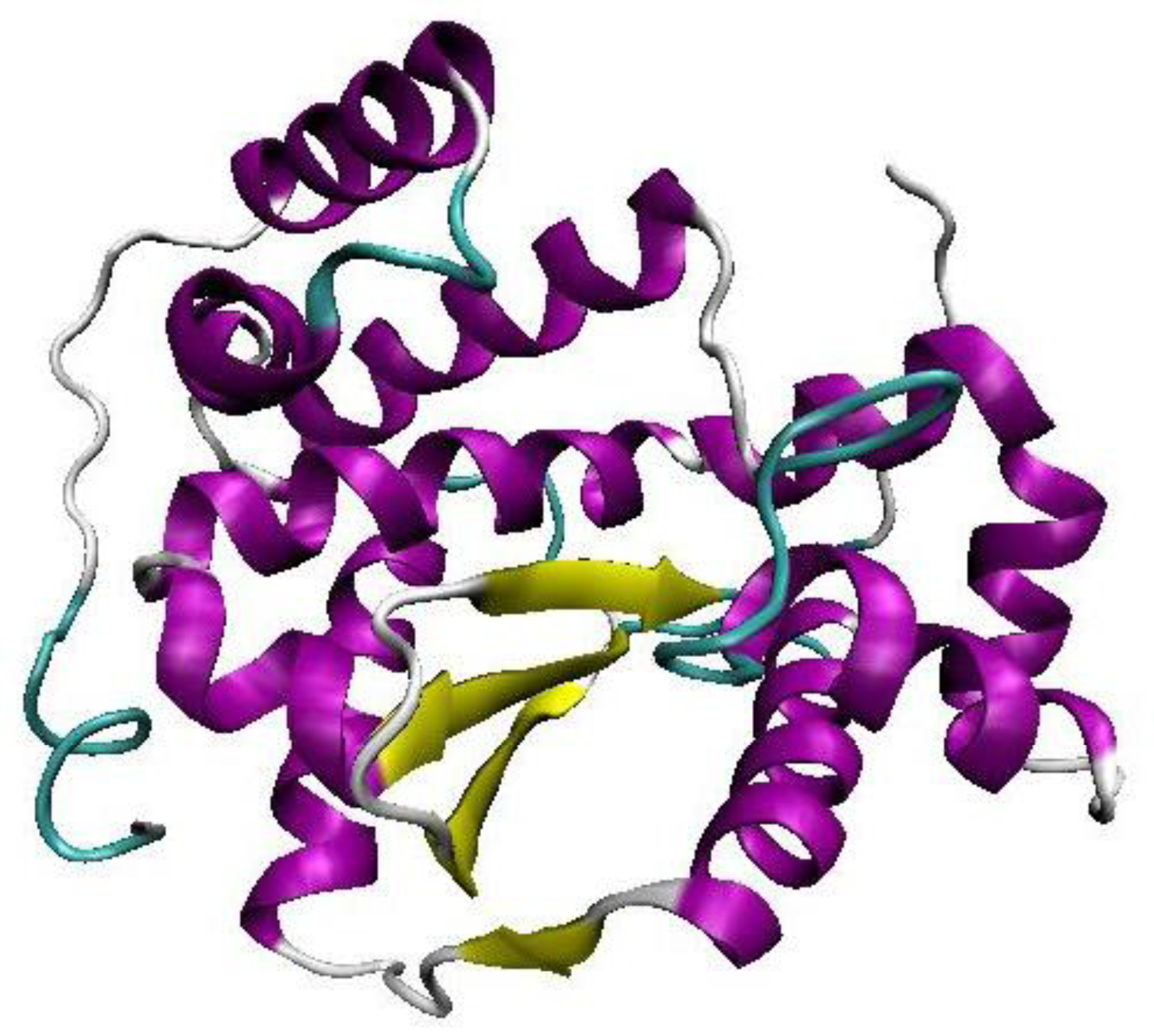
2.1. Homology Modeling of YggG Protein and Model Assessment
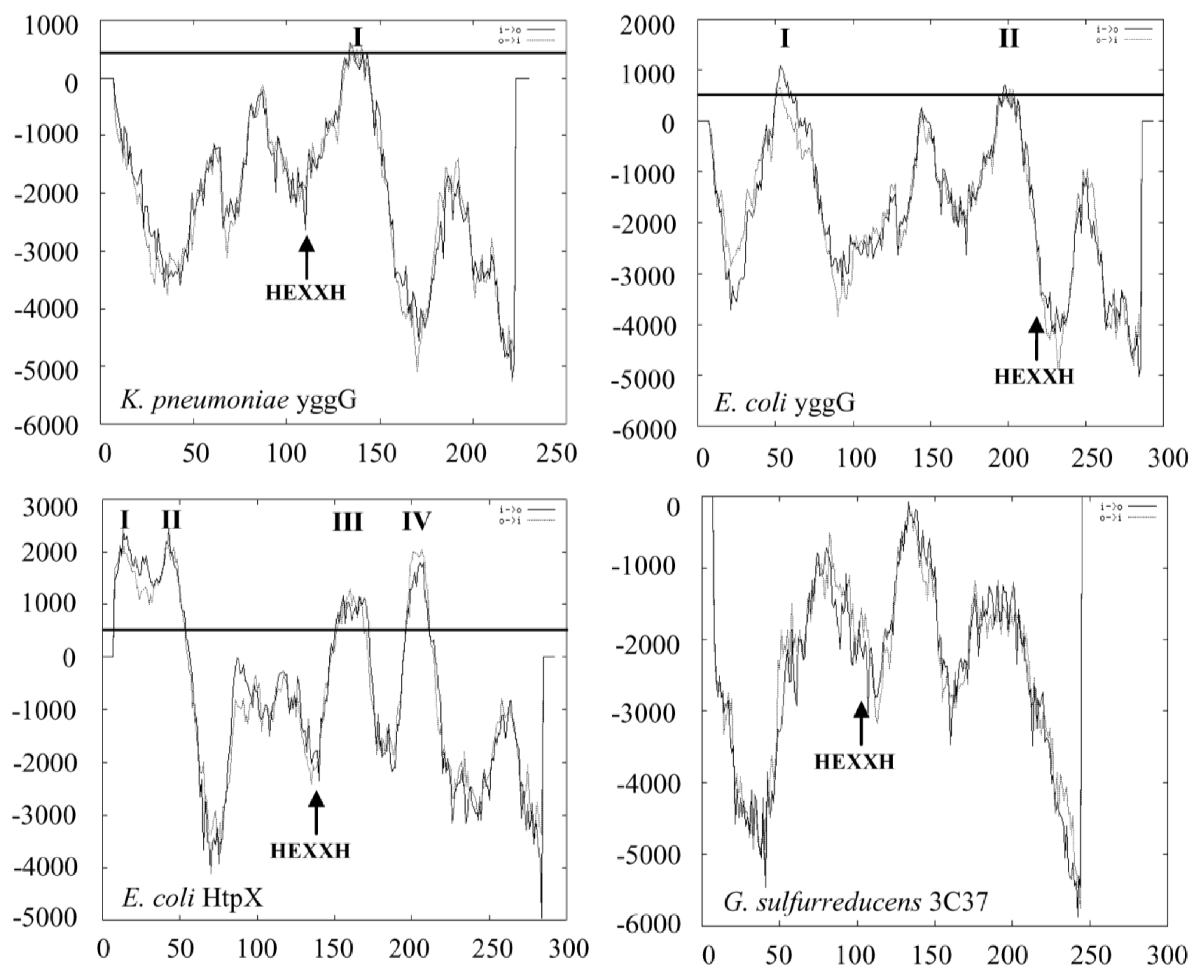
2.2. Domain Family, Amino Acid and Membrane Topology Analyses of YggG Protein
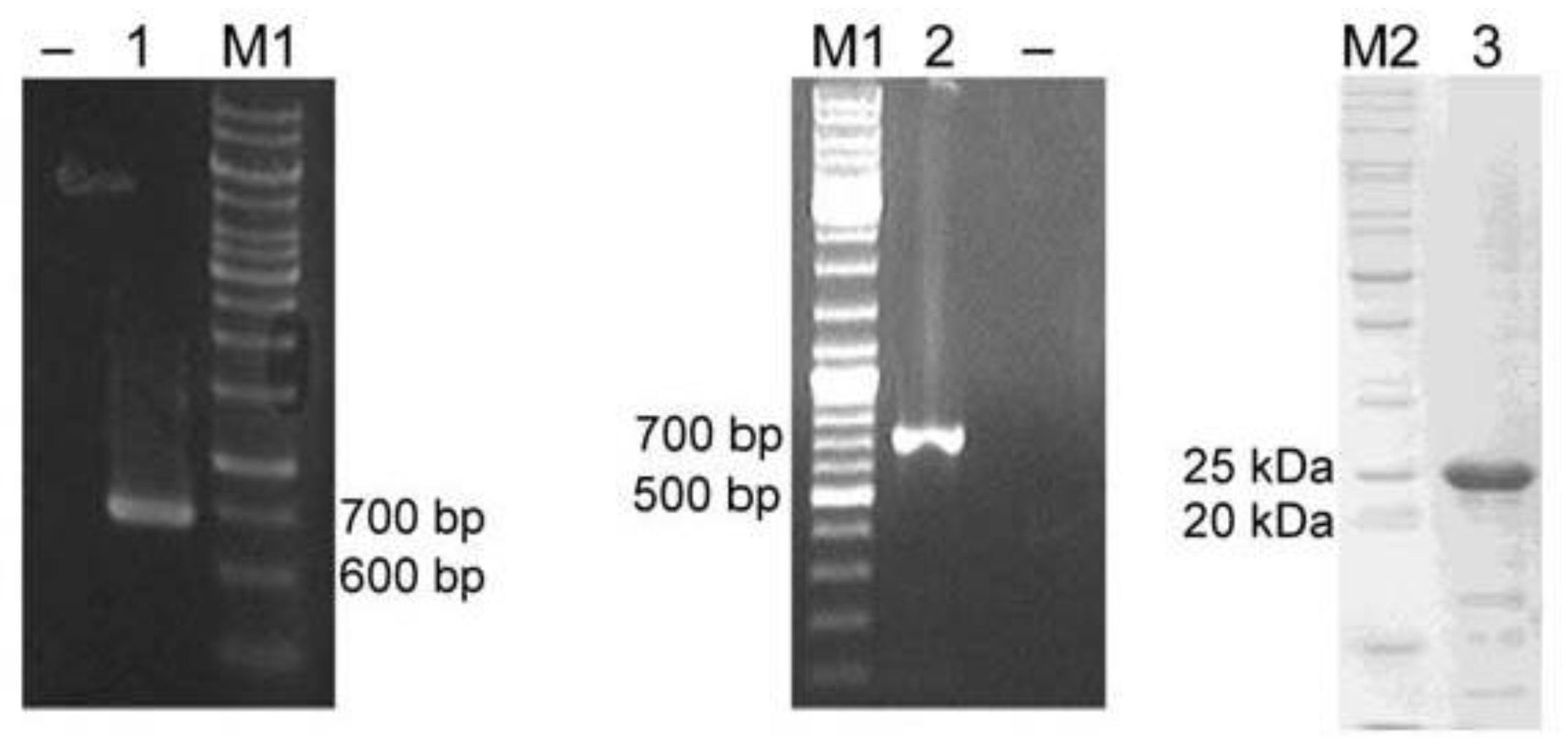
2.3. Cloning and Heterologous Expression of YggG in E. coli
2.4. Detection of yggG mRNA Expression in K. pneumoniae
2.5. Proteolytic Activity of Purified K. pneumoniae YggG
3. Discussion
4. Experimental Section
4.1. Computational Methodology for Homology Modelling
4.2. Bacterial Strain, Growth and Culture Conditions
4.3. Total Genomic DNA and RNA Extractions from K. pneumoniae MGH 78578
4.4. PCR Cloning of K. pneumoniae yggG Open Reading Frame
4.5. Expression and Purification of YggG Protein
4.6. Reverse Transcription PCR of yggG Gene
4.7. Casein Hydrolysis Assay
4.8. Determination of Temperature and pH Optima of the Activity of Purified YggG
5. Conclusions
Acknowledgements
References
- Alcantar-Curiel, MD; Garcia-Latorre, E; Santos, JI. Klebsiella pneumoniae 35 and 36 kDa porins are common antigens in different serotypes and induce opsonizing antibodies. Arch. Med. Res 2000, 31, 28–36. [Google Scholar]
- Ishida, T; Hashimoto, T; Arita, M; Ito, I; Osawa, M. Etiology of community-acquired pneumonia in hospitalized patients: A 3-year prospective study in Japan. Chest 1998, 114, 1588–1593. [Google Scholar]
- Lee, KH; Hui, KP; Tan, WC; Lim, TK. Klebsiella bacteraemia: A report of 101 cases from National University Hospital, Singapore. J. Hosp. Infect 1994, 27, 299–305. [Google Scholar]
- Tang, LM; Chen, ST; Hsu, WC; Chen, CM. Klebsiella meningitis in Taiwan: An overview. Epidemiol. Infect 1997, 119, 135–142. [Google Scholar]
- Ko, WC; Paterson, DL; Sagnimeni, AJ; Hansen, DS; Von Gottberg, A; Mohapatra, S; Casellas, JM; Goossens, H; Mulazimoglu, L; Trenholme, G; et al. Community-acquired Klebsiella pneumoniae bacteremia: global differences in clinical patterns. Emerg. Infect. Dis 2002, 8, 160–166. [Google Scholar]
- Ogawa, W; Li, DW; Yu, P; Begum, A; Mizushima, T; Kuroda, T; Tsuchiya, T. Multidrug resistance in Klebsiella pneumoniae MGH78578 and cloning of genes responsible for the resistance. Biol. Pharm. Bull 2005, 28, 1505–1508. [Google Scholar]
- Jain, E; Bairoch, A; Duvaud, S; Phan, I; Redaschi, N; Suzek, BE; Martin, MJ; McGarvey, P; Gasteiger, E. Infrastructure for the life sciences: design and implementation of the UniProt website. BMC Bioinform 2009, 10, 136. [Google Scholar]
- Kanehisa, M; Goto, S; Furumichi, M; Tanabe, M; Hirakawa, M. KEGG for representation and analysis of molecular networks involving diseases and drugs. Nucleic Acids Res 2010, 38, D355–D360. [Google Scholar]
- Miyoshi, S; Shinoda, S. Microbial metalloproteases and pathogenesis. Microbes. Infect 2000, 2, 91–98. [Google Scholar]
- Dreyfus, LA; Iglewski, BH. Purification and characterization of an extracellular protease of Legionella pneumophila. Infect. Immun 1986, 51, 736–743. [Google Scholar]
- Hase, CC; Finkelstein, RA. Cloning and nucleotide sequence of the Vibrio cholerae hemagglutinin/protease (HA/protease) gene and construction of an HA/protease-negative strain. J. Bacteriol 1991, 173, 3311–3317. [Google Scholar]
- Fakruddin, JM; Biswas, S; Sharma, YD. Metalloprotease activity in a small heat shock protein of the human malaria parasite Plasmodium vivax. Infect. Immun 2000, 68, 1202–1206. [Google Scholar]
- Szumanski, MB; Boyle, SM. Influence of cyclic AMP, agmatine, and a novel protein encoded by a flanking gene on speB (agmatine ureohydrolase) in Escherichia coli. J. Bacteriol 1992, 174, 758–764. [Google Scholar]
- Huang, Y; Zhang, B; Dong, K; Zhang, X; Hou, L; Wang, T; Chen, N; Chen, S. Up-regulation of yggG promotes the survival of Escherichia coli cells containing era-1 mutant protein. FEMS Microbiol. Lett 2007, 275, 8–15. [Google Scholar]
- Huang, Y; Dong, K; Zhang, X; Zhang, B; Hou, L; Chen, N; Chen, S. Expression and regulation of the yggG gene of Escherichia coli. Curr. Microbiol 2008, 56, 14–20. [Google Scholar]
- Ojima, Y; Komaki, M; Nishioka, M; Iwatani, S; Tsujimoto, N; Taya, M. Introduction of a stress-responsive gene, yggG, enhances the yield of l-phenylalanine with decreased acetic acid production in a recombinant Escherichia coli. Biotechnol. Lett 2009, 31, 525–530. [Google Scholar]
- Rawlings, ND; Barrett, AJ. Evolutionary families of metallopeptidases. Methods Enzymol 1995, 248, 183–228. [Google Scholar]
- Halbhuber, Z; Petrmichlova, Z; Alexciev, K; Thulin, E; Stys, D. Overexpression and purification of recombinant membrane PsbH protein in Escherichia coli. Protein Expr. Purif 2003, 32, 18–27. [Google Scholar]
- Siddiqui, AA; Jalah, R; Sharma, YD. Expression and purification of HtpX-like small heat shock integral membrane protease of an unknown organism related to Methylobacillus flagellatus. J. Biochem. Biophys. Methods 2007, 70, 539–546. [Google Scholar]
- Laskowski, RA; Macarthur, MW; Moss, DS; Thornton, JM. Procheck—a Program to Check the Stereochemical Quality of Protein Structures. J. Appl. Cryst 1993, 26, 283–291. [Google Scholar]
- Humphrey, W; Dalke, A; Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph 1996, 14, 33–38. [Google Scholar]
- Kornitzer, D; Teff, D; Altuvia, S; Oppenheim, AB. Isolation, characterization, and sequence of an Escherichia coli heat shock gene, htpX. J. Bacteriol 1991, 173, 2944–2953. [Google Scholar]
- Sakoh, M; Ito, K; Akiyama, Y. Proteolytic activity of HtpX, a membrane-bound and stress-controlled protease from Escherichia coli. J. Biol. Chem 2005, 280, 33305–33310. [Google Scholar]
- Hirokawa, T; Boon-Chieng, S; Mitaku, S. SOSUI: Classification and secondary structure prediction system for membrane proteins. Bioinformatics 1998, 14, 378–379. [Google Scholar]
- Tusnady, GE; Simon, I. The HMMTOP transmembrane topology prediction server. Bioinformatics 2001, 17, 849–850. [Google Scholar]
- Tusnady, GE; Simon, I. Principles governing amino acid composition of integral membrane proteins: application to topology prediction. J. Mol. Biol 1998, 283, 489–506. [Google Scholar]
- Krogh, A; Larsson, B; von Heijne, G; Sonnhammer, EL. Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J. Mol. Biol 2001, 305, 567–580. [Google Scholar]
- Combet, C; Blanchet, C; Geourjon, C; Deleage, G. NPS@: Network protein sequence analysis. Trends Biochem. Sci 2000, 25, 147–150. [Google Scholar]
- Fujimura-Kamada, K; Nouvet, FJ; Michaelis, S. A novel membrane-associated metalloprotease, Ste24p, is required for the first step of NH2-terminal processing of the yeast a-factor precursor. J. Cell Biol 1997, 136, 271–285. [Google Scholar]
- Shimohata, N; Chiba, S; Saikawa, N; Ito, K; Akiyama, Y. The Cpx stress response system of Escherichia coli senses plasma membrane proteins and controls HtpX, a membrane protease with a cytosolic active site. Genes Cells 2002, 7, 653–662. [Google Scholar]
- da Silveira, RB; Wille, AC; Chaim, OM; Appel, MH; Silva, DT; Franco, CR; Toma, L; Mangili, OC; Gremski, W; Dietrich, CP; et al. Identification, cloning, expression and functional characterization of an astacin-like metalloprotease toxin from Loxosceles intermedia (brown spider) venom. Biochem. J 2007, 406, 355–363. [Google Scholar]
- Jing, Y; Toubarro, D; Hao, Y; Simoes, N. Cloning, characterisation and heterologous expression of an astacin metalloprotease, Sc-AST, from the entomoparasitic nematode Steinernema carpocapsae. Mol. Biochem. Parasitol 2010, 174, 101–108. [Google Scholar]
- Mansfeld, J. Metalloproteases. In Industrial Enzymes: Structure, Function and Applications; Polaina, J, MacCabe, AP, Eds.; Springer: Amsterdam, The Netherlands, 2007; pp. 221–242. [Google Scholar]
- Fouts, DE; Tyler, HL; DeBoy, RT; Daugherty, S; Ren, Q; Badger, JH; Durkin, AS; Huot, H; Shrivastava, S; Kothari, S; et al. Complete genome sequence of the N2-fixing broad host range endophyte Klebsiella pneumoniae 342 and virulence predictions verified in mice. PLoS Genet 2008, 4, e1000141. [Google Scholar]
- Wu, KM; Li, LH; Yan, JJ; Tsao, N; Liao, TL; Tsai, HC; Fung, CP; Chen, HJ; Liu, YM; Wang, JT; et al. Genome sequencing and comparative analysis of Klebsiella pneumoniae NTUH-K2044, a strain causing liver abscess and meningitis. J. Bacteriol 2009, 191, 4492–4501. [Google Scholar]
- Sali, A; Blundell, TL. Comparative protein modelling by satisfaction of spatial restraints. J. Mol. Biol 1993, 234, 779–815. [Google Scholar]
- Finn, RD; Mistry, J; Tate, J; Coggill, P; Heger, A; Pollington, JE; Gavin, OL; Gunasekaran, P; Ceric, G; Forslund, K; et al. The Pfam protein families database. Nucleic Acids Res 2010, 38, D211–D222. [Google Scholar]
- Larkin, MA; Blackshields, G; Brown, NP; Chenna, R; McGettigan, PA; McWilliam, H; Valentin, F; Wallace, IM; Wilm, A; Lopez, R; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar]
- Hofmann, K; Stoel, W. Tmbase—A database of membrane spanning protein segments. Biol. Chem. Hoppe-Seyler 1993, 374, 166. [Google Scholar]
- Enoksson, M; Zhu, W; Salvesen, G. Identification of Protease Substracts by Mass Spectrometry Approaches-1. In The Cancer Degradome; Edwards, D, Hoyer-Hansen, G, Blasi, F, Sloane, BF, Eds.; Springer Science + Business Media, LLC: New York, NY, USA, 2008; pp. 67–82. [Google Scholar]
- Walters, S; Belanger, M; Rodrigues, P; Whitlock, J; Progulske-Fos, A. A member of the peptidase M48 superfamily of Porphyromonas gingivalis is associated with virulence in vitro andin vivo. J Oral Microbiol 2009, 1. [Google Scholar] [CrossRef]


© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kuan, C.S.; Wong, M.T.; Choi, S.B.; Chang, C.C.; Yee, Y.H.; Wahab, H.A.; Normi, Y.M.; Too, W.C.S.; Few, L.L. Klebsiella pneumoniae yggG Gene Product: A Zinc-Dependent Metalloprotease. Int. J. Mol. Sci. 2011, 12, 4441-4455. https://doi.org/10.3390/ijms12074441
Kuan CS, Wong MT, Choi SB, Chang CC, Yee YH, Wahab HA, Normi YM, Too WCS, Few LL. Klebsiella pneumoniae yggG Gene Product: A Zinc-Dependent Metalloprotease. International Journal of Molecular Sciences. 2011; 12(7):4441-4455. https://doi.org/10.3390/ijms12074441
Chicago/Turabian StyleKuan, Chee Sian, Mun Teng Wong, Sy Bing Choi, Ching Ching Chang, Yoke Hiang Yee, Habibah A. Wahab, Yahaya Mohd Normi, Wei Cun See Too, and Ling Ling Few. 2011. "Klebsiella pneumoniae yggG Gene Product: A Zinc-Dependent Metalloprotease" International Journal of Molecular Sciences 12, no. 7: 4441-4455. https://doi.org/10.3390/ijms12074441
APA StyleKuan, C. S., Wong, M. T., Choi, S. B., Chang, C. C., Yee, Y. H., Wahab, H. A., Normi, Y. M., Too, W. C. S., & Few, L. L. (2011). Klebsiella pneumoniae yggG Gene Product: A Zinc-Dependent Metalloprotease. International Journal of Molecular Sciences, 12(7), 4441-4455. https://doi.org/10.3390/ijms12074441



