Syntheses and Characterization of New Nickel Coordination Polymers with 4,4’-Dipyridylsulfide. Dynamic Rearrangements of One-Dimensional Chains Responding to External Stimuli: Temperature Variation and Guest Releases/Re-Inclusions
Abstract
:1. Introduction
2. Results and Discussion
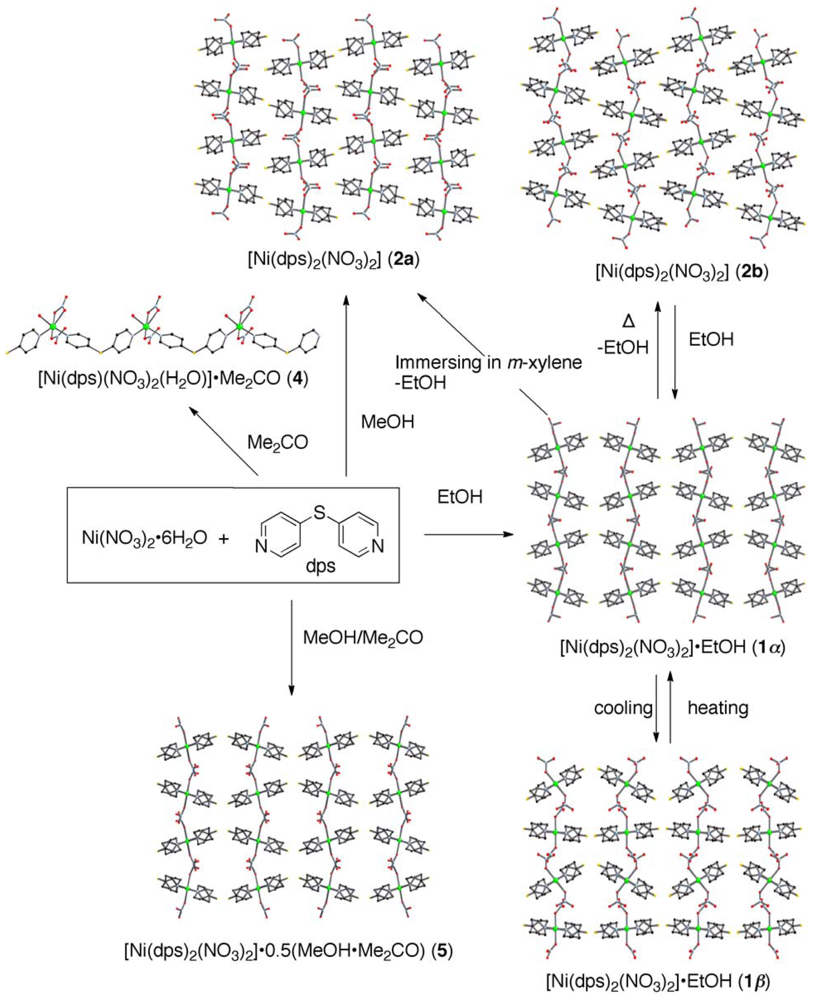
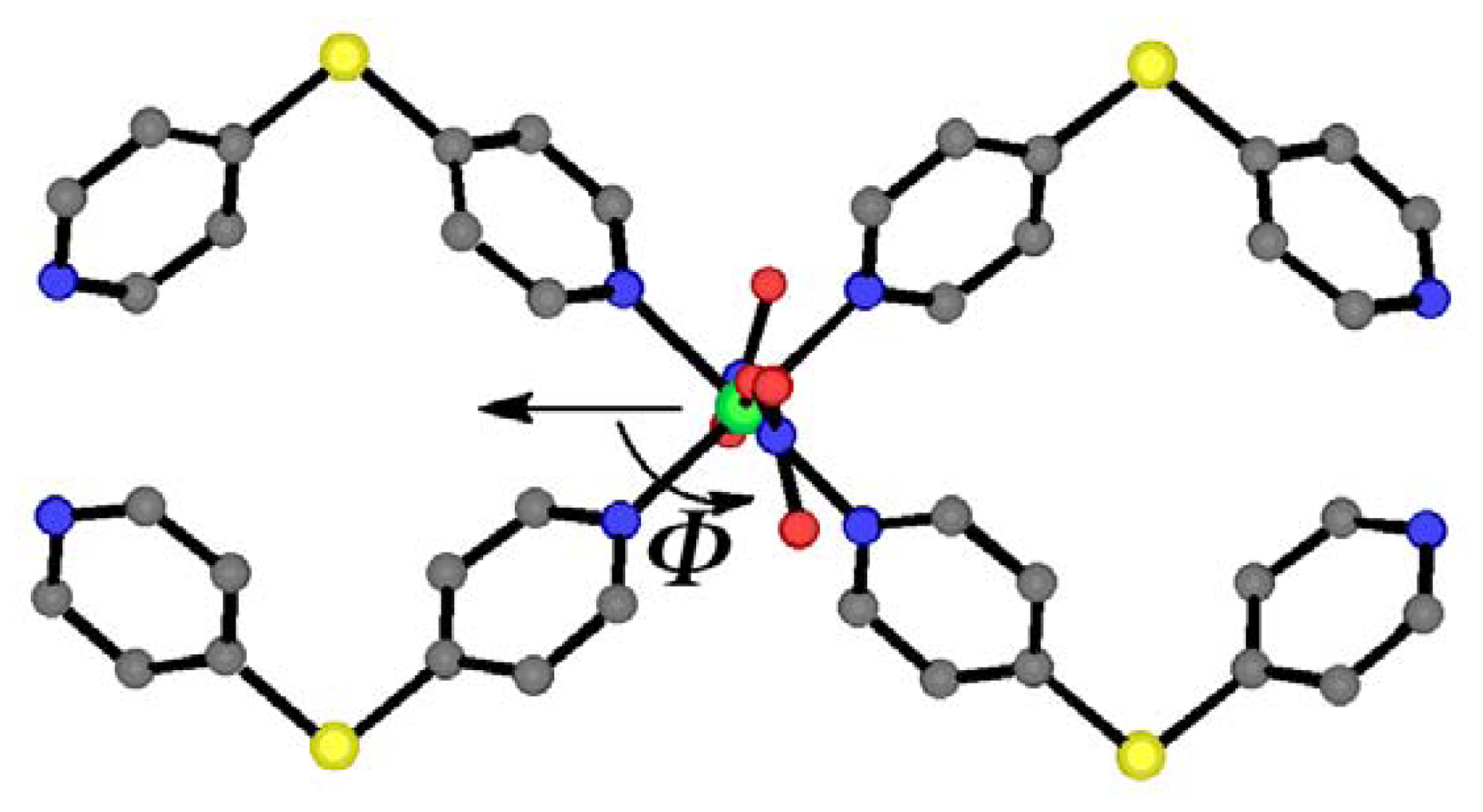
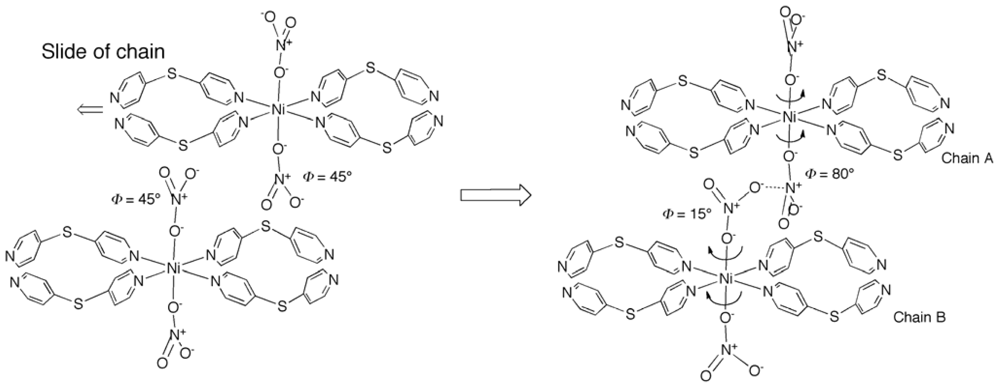
2.1. Overview of the Structural Rearrangement of the Ni-dps System
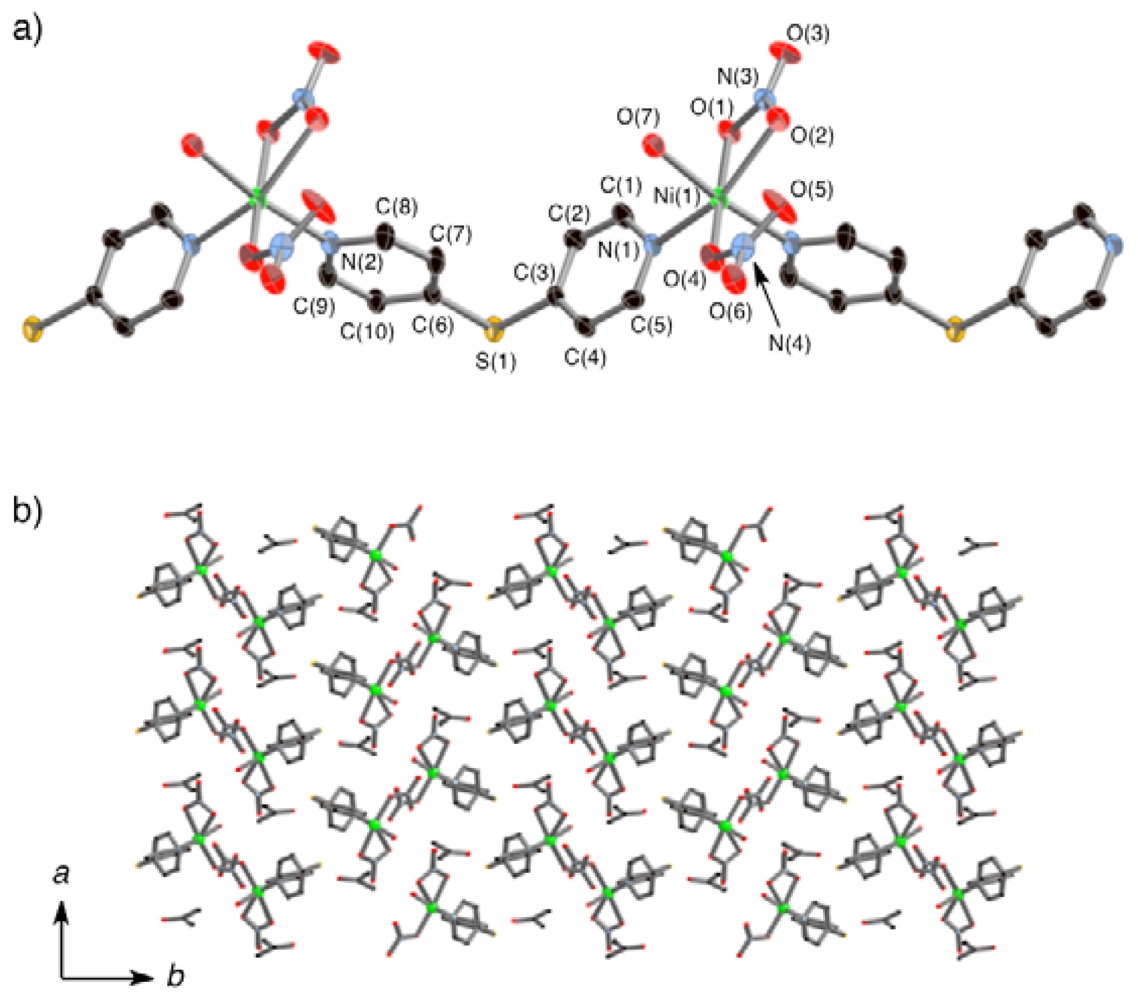
2.2. Crystal Structures of 1α and 1β
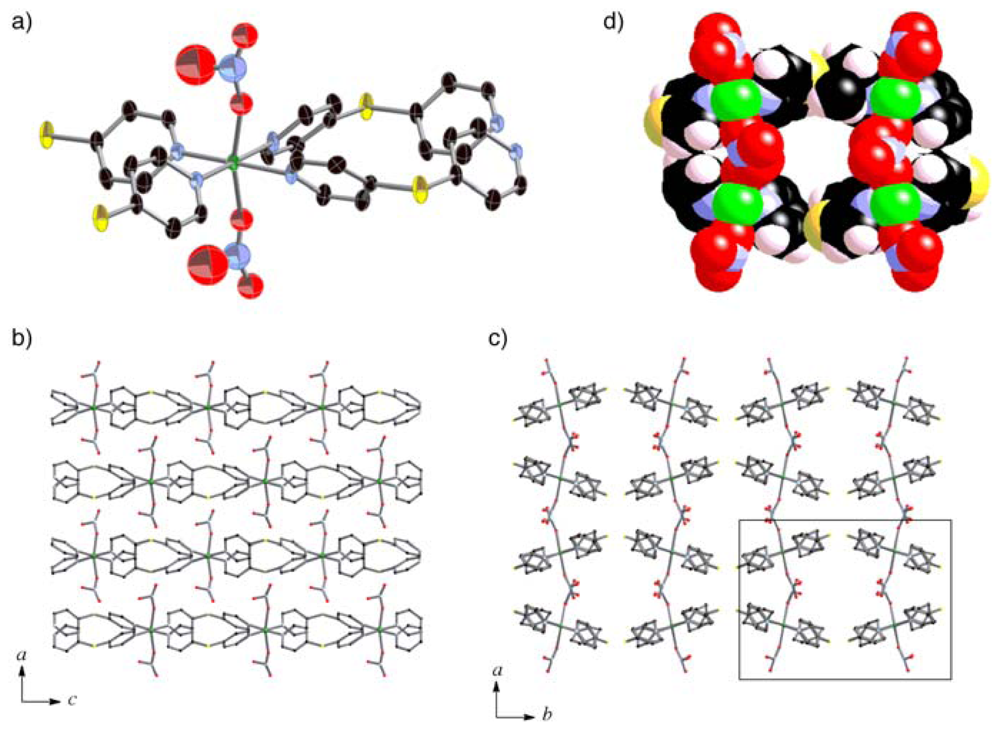
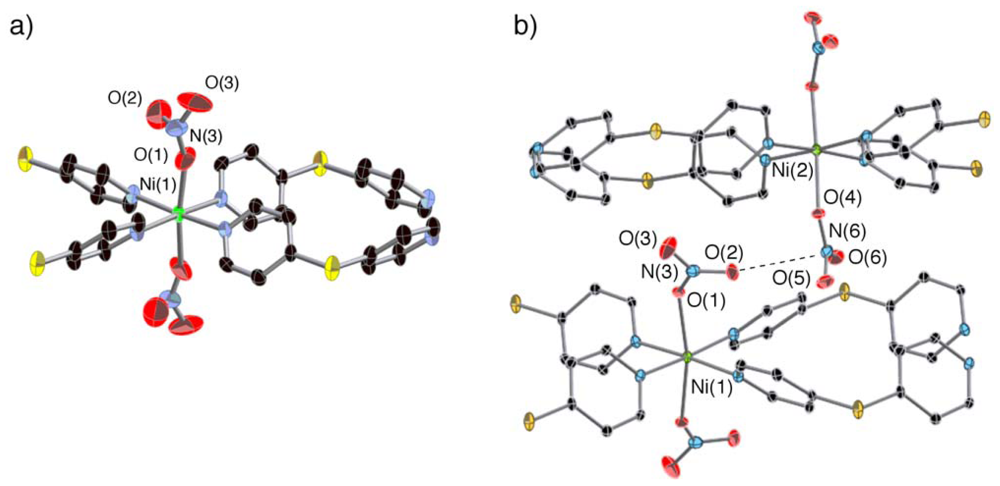
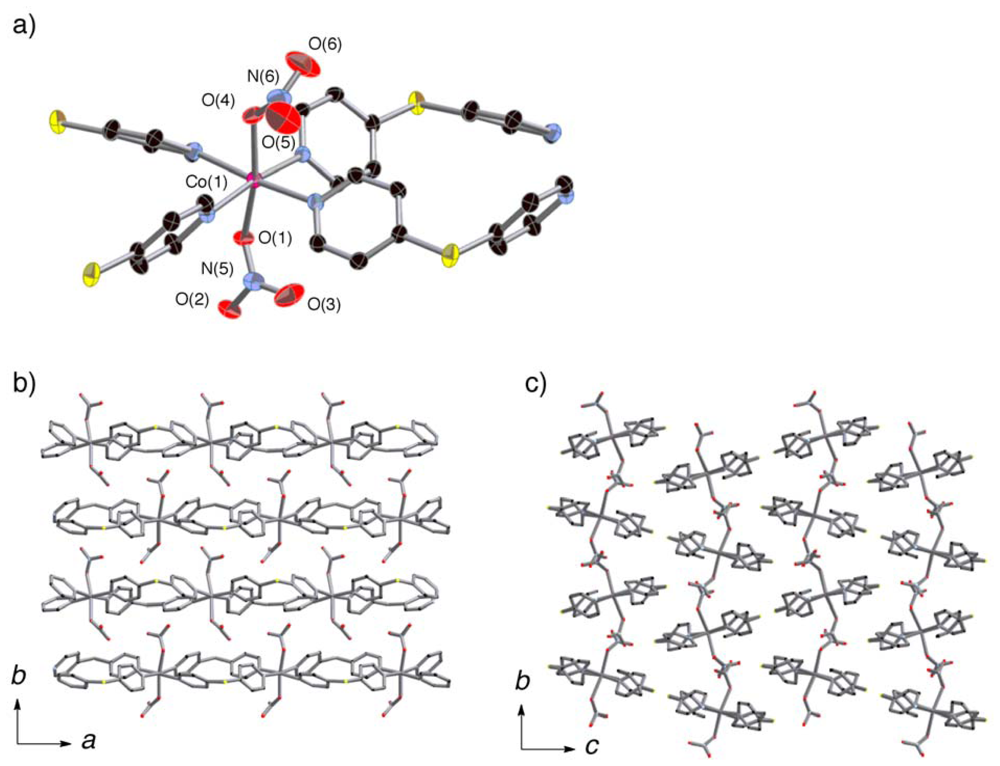
2.3. Crystal Structures of 2a and 3
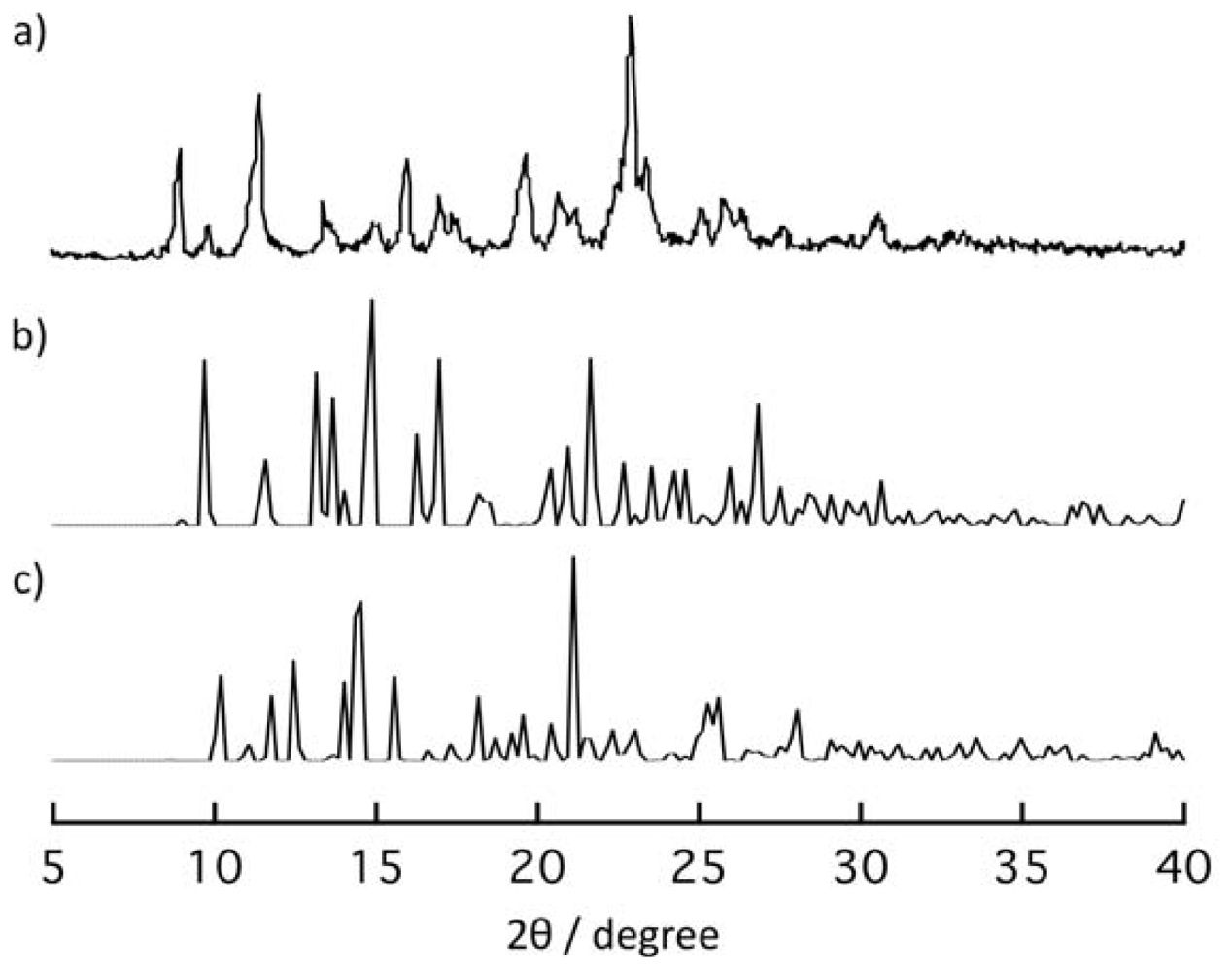
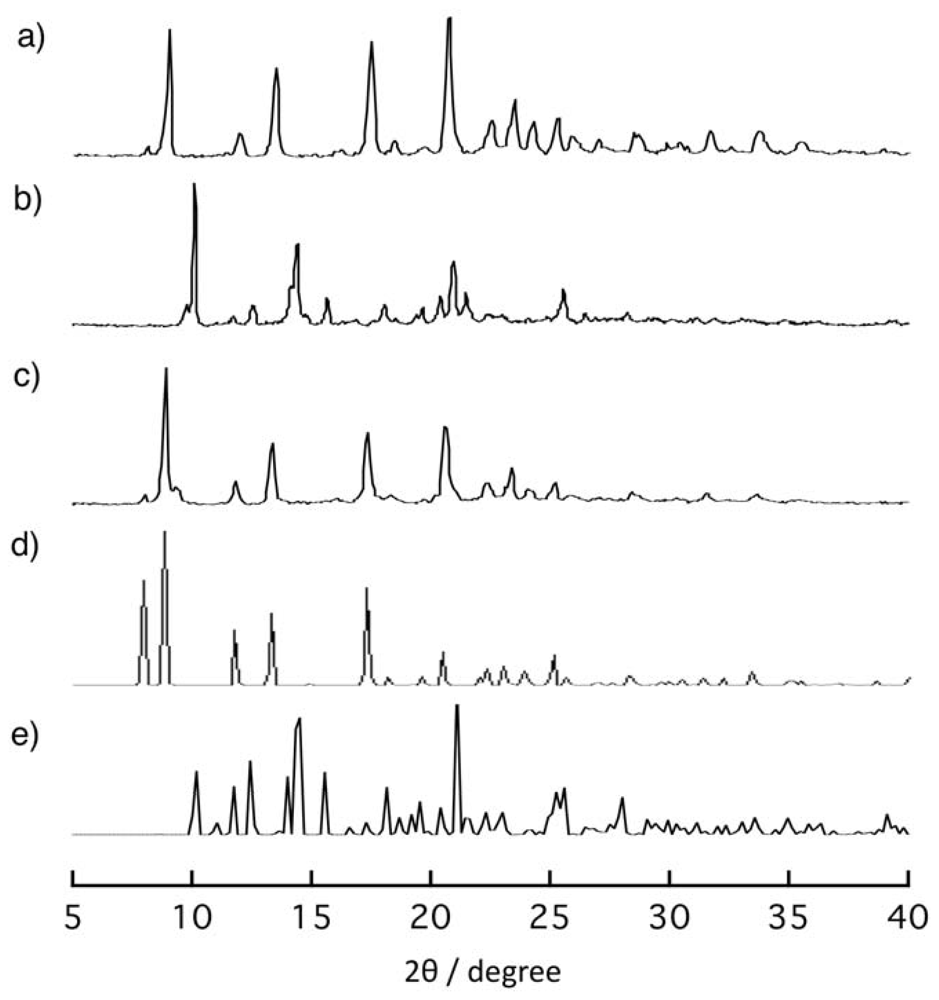
2.4. Rearrangement of (Ni-dps2) Chains by Guest Releases and Re-Inclusions
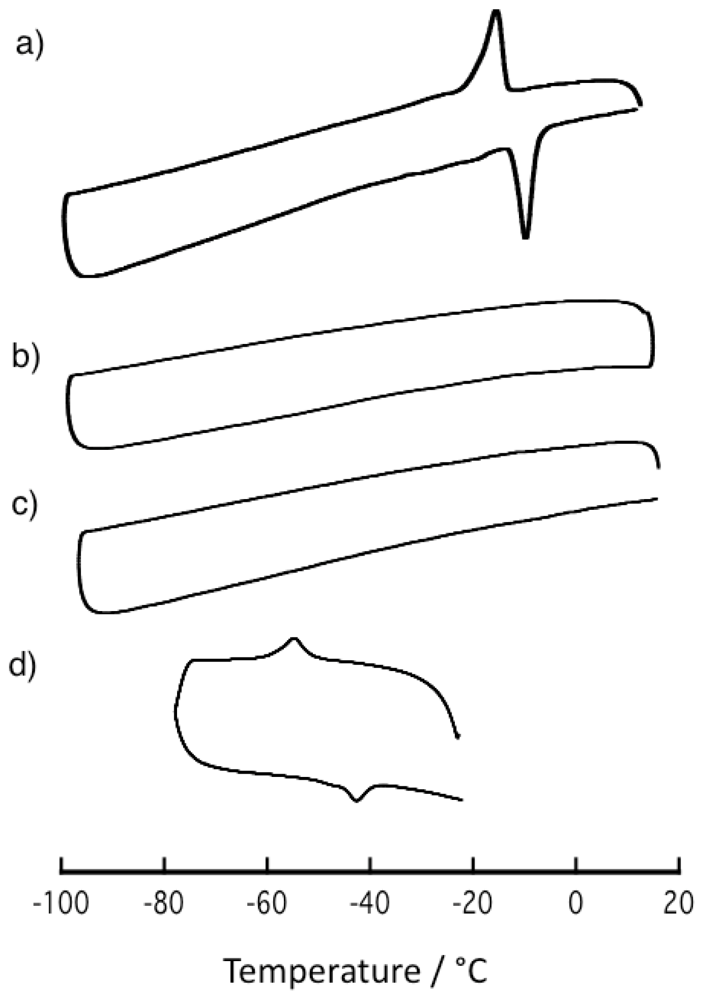
2.5. Thermal Property of Ni-dps Compounds
3. Experimental Section
3.1. Reagents and Materials
3.2. Crystal Structure Determinations
4. Conclusions
Supporting Information

Acknowledgements
References
- Férey, G. Hybrid porous solids: Past, present, future. Chem. Soc. Rev 2008, 37, 191–214. [Google Scholar]
- Maji, T; Kitagawa, S. Chemistry of porous coordination polymers. Pure Appl. Chem 2007, 79, 2155–2177. [Google Scholar] [Green Version]
- Zhang, J-P; Lin, Y-Y; Zhang, W-X; Chen, X-M. Temperature- or guest-induced drastic single-crystal-to-single-crystal transformations of a nanoporous coordination polymer. J. Am. Chem. Soc 2005, 127, 14162–14163. [Google Scholar]
- Chen, C-L; Goforth, AM; Smith, MD; Su, C-Y; zur Loye, H-C. [Co2(ppca)2(H2O)(V4O12)0.5]: A framework material exhibiting reversible shrinkage and expansion through a single-crystal-to-single-crystal transformation involving a change in the cobalt coordination environment. Angew. Chem. Int. Ed 2005, 44, 6673–6677. [Google Scholar]
- Shimomura, S; Horike, S; Matsuda, R; Kitagawa, S. Guest-specific function of a flexible undulating channel in a 7,7,8,8-tetracyano-p-quinodimethane dimer-based porous coordination polymer. J. Am. Chem. Soc 2007, 129, 10990–10991. [Google Scholar]
- Kondo, M; Shimizu, Y; Miyazawa, M; Irie, Y; Nakamura, A; Naito, T; Maeda, K; Uchida, F; Nakamoto, T; Inaba, A. A new nickel coordination polymer with dynamic channels that mechanically capture and release including guest molecules responding to a temperature variation. Chem. Lett 2004, 514–515. [Google Scholar]
- Kaneko, W; Ohba, M; Kitagawa, S. A flexible coordination polymer crystal providing reversible structural and magnetic conversions. J. Am. Chem. Soc 2007, 129, 13706–13712. [Google Scholar]
- Byrne, P; Lloyd, GO; Anderson, KM; Clarke, N; Steed, JW. Anion hydrogen bond efects in the formation of planar or quintuple helical coordination polymers. Chem. Commun 2008, 3720–3722. [Google Scholar]
- Takamizawa, S; Nakata, E-I; Yokoyama, H; Mochizuki, K; Mori, W. Carbon dioxide inclusion phases of a transformable 1D coordination polymer host [Rh2(O2CPh)4(pyz)]n. Angew. Chem. Int. Ed 2003, 115, 4467–4470. [Google Scholar]
- Ohara, K; Martí-Rujas, J; Haneda, T; Kawano, M; Hashizume, D; Izumi, F; Fujita, M. Formation of a thermally stable, porous coordination network via a crystalline-to-amorphous-to-crystalline phase transition. J. Am. Chem. Soc 2009, 131, 2860–3861. [Google Scholar]
- Biradha, K; Hongo, Y; Fujita, M. Crystal-to-crystal sliding of 2D coordination layers triggered by guest exchange. Angew. Chem. Int. Ed 2002, 41, 3395–3398. [Google Scholar]
- Biradha, K; Fujita, M. A sprinlike 3D-coordination network that shrink or swells in a crystal-to-crystal manner upon guest removal or readsorption. Angew. Chem. Int. Ed 2002, 41, 3392–3395. [Google Scholar]
- Noguchi, H; Kondoh, A; Hattori, Y; Kanoh, H; Kajiro, H; Kaneko, K. Clathrate-formation mediated adsorption of methane on Cu-complex crystals. J. Phys. Chem. B 2005, 109, 13851–13853. [Google Scholar]
- Hanzawab, Y; Kanoha, H; Kanekoa, K. Hydrogen-bond change-associated gas adsorption in inorganic–organic hybrid microporous crystals. Appl. Surf. Sci 2002, 196, 81–88. [Google Scholar]
- Llewellyn, PL; Horcajada, P; Maurin, G; Devic, T; Rosenbach, N; Bourrelly, S; Serre, C; Vincent, D; Loera-Serna, S; Filinchuk, Y; Frey, G. Complex adsorption of short linear alkanes in the flexible metal-organic-framework MIL-53(Fe). J. Am. Chem. Soc 2009, 131, 13002–13008. [Google Scholar]
- Kasai, K; Aoyagi, M; Fujita, M. Flexible coordination networks with fluorinated backbones. Remarkable ability for induced-fit enclathration of organic molecules. J. Am. Chem. Soc 2000, 122, 2140–2141. [Google Scholar]
- Uemura, K; Kitagawa, S; Kondo, M; Fukui, K; Kitaura, R; Chang, H-C; Mizutani, T. Novel felxible frameworks of porous cobalt(II) coordination polymers that show selective guest adsorption based on the switching of hydrogen-bond pairs of amide groups. Chem. Eur. J 2002, 8, 3587–3600. [Google Scholar]
- Zeng, M-H; Wang, Q-X; Tan, Y-X; Hu, S; Zhao, H-X; Long, L-S; Kurmoo, M. Rigid pillars and double walls in a porous metal-organic framework: Single-crystal to single-crystal, controlled uptake and release of iodine and electrical conductivity. J. Am. Chem. Soc 2010, 132, 2561–2563. [Google Scholar]
- Wheatley, PS; Butler, AR; Crane, MS; Fox, S; Xiao, B; Rossi, AG; Megson, IL; Morris, RE. NO-releasing zeolites and their antithrombotic properties. J. Am. Chem. Soc 2006, 128, 502–509. [Google Scholar]
- Yaghi, OM; Li, H; Davis, C; Richardson, D; Groy, TL. Synthetic strategies, structure properties in the chemistry of modular porous solids. Acc. Chem. Res 1998, 31, 474–484. [Google Scholar]
- Ohmori, O; Fujita, M. Heterogeneous catalysis of a coordination network: cyanosilylation of imines catalyzed by a Cd(II)-(4,4′-bipyridine) square grid complex. Chem. Commun 2004, 1586–1587. [Google Scholar]
- Ma, L; Abney, C; Lin, W. Enantioselective catalysis with homochiral metal–organic frameworks. Chem. Soc. Rev 2009, 38, 1248–1256. [Google Scholar]
- Lee, JY; Farha, OK; Roberts, J; Scheidt, KA; Nguyen, ST; Hupp, JT. Metal–organic framework materials as catalysts. Chem. Soc. Rev 2009, 38, 1450–1459. [Google Scholar]
- Wu, C-D; Lin, W. Heterogeneous asymmetric catalysis with homochiral metal–organic frameworks: Network-structure-dependent catalytic activity. Angew. Chem. Int. Ed 2007, 46, 1075–1078. [Google Scholar]
- Latroche, M; Surble’, S; Serre, C; Mellot-Draznieks, C; Llewellyn, PL; Lee, J-H; Chang, J-S; Jhun, SH; Férey, G. Hydrogen storage in the giant-pore metal–organic frameworks MIL-100 and MIL-101. Angew. Chem. Int. Ed 2006, 45, 8227–8231. [Google Scholar]
- Liu, Y; Eubank, JF; Cairns, AJ; Eckert, J; Kravtsov, VC; Luebke, R; Eddaoudi, M. Assembly of metal–organic frameworks (MOFs) based on indium-trimer building blocks: A porous MOF with soc topology and high hydrogen storage. Angew. Chem. Int. Ed 2007, 46, 3278–3283. [Google Scholar]
- Li, H; Eddaoudi, M; O’Keeffe, M; Yaghi, OM. Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature 1999, 402, 276–279. [Google Scholar]
- Rosi, N; Eddaoudi, M; Vodak, D; Eckert, J; O’Keeffe, M; Yaghi, OM. Hydrogen storage in microporous metal-orgabnic frameworks. Science 2003, 300, 1127–1129. [Google Scholar]
- Rowsell, JLC; Yaghi, OM. Metal-organic frameworks: A new class of porous materials. Micropor. Mesopor. Mater 2004, 73, 3–14. [Google Scholar]
- Alaerts, L; Kirshhock, CEA; Maes, M; van der Veen, MA; Finsy, V; Depla, A; Martens, JA; Baron, GV; Jacobs, PA; Denayer, JFM; De Vos, DE. Selective adsorption and separation of xylene isomers and ethylbenzene with the microporous vanadium(IV) terephthalate MIL-47. Angew. Chem. Int. Ed 2007, 46, 4293–4297. [Google Scholar]
- Choi, HJ; Dincâ, M; Long, JR. Broadly hysteretic H2 adsorption in the microporous metal-organic framework Co(1,4-benzenedipyrazolate). J. Am. Chem. Soc 2008, 130, 7848–7850. [Google Scholar]
- Li, J-R; Kuppler, RJ; Zhou, H-C. Selective gas adsorption and separation in metal–organic frameworks. Chem. Soc. Rev 2009, 38, 1477–1504. [Google Scholar]
- Kondo, M; Irie, Y; Shimizu, Y; Miyazawa, M; Kawaguchi, H; Nakamura, A; Naito, T; Maeda, K; Uchida, F. Dynamic coordination polymers with 4,4’-oxybis(benzoate): Reversible transformations of nano- and nonporous coordination frameworks responding to present solvents. Inorg. Chem 2004, 43, 6139–6141. [Google Scholar]
- Kondo, M; Shimizu, E; Horiba, T; Tanaka, H; Fuwa, Y; Nabari, K; Unoura, K; Naito, T; Maeda, K; Uchida, F. New copper(II) complexes connected by NH···O=C and NH···S=C intermolecular hydrogen bonds. Chem. Lett 2003, 944–945. [Google Scholar]



| Compound | 1α | 1β | 2a | 2b | 3 | 4 | 5 |
| Formula | C22H22N6O7NiS2 | C22H22N6O7NiS2 | C20H22N6O7NiS2 | C20H16N6O6NiS2 | C20H16CoN6O6S2 | C13H16N4O8NiS | C22H21N12NiO7S2 |
| Formula weight | 605.27 | 605.27 | 559.20 | 559.20 | 559.44 | 447.05 | 604.26 |
| Lattice | orthorhombic | orthorhombic | monoclinic | monoclinic | monoclinic | monoclinic | orthorhombic |
| a, Å | 13.27(1) | 13.1233(8) | 10.016(4) | 10.11(1) | 10.200(1) | 9.499(1) | 13.20(2) |
| b, Å | 19.88(2) | 19.468(1) | 12.554(5) | 13.11(2) | 12.9500(9) | 21.841(2) | 19.727(9) |
| c, Å | 10.100(9) | 10.1241(5) | 18.960(9) | 17.57(2) | 17.4300(7) | 10.293(2) | 10.082(4) |
| β, ° | 107.921(6) | 96.31(2) | 95.790(2) | 117.987(5) | |||
| V, Å3 | 2665(3) | 2586.6(2) | 2268(1) | 2314(4) | 2290.6(3) | 1885.8(4) | 2611(7) |
| Space group | Ccc2 (No. 37) | Pnc2 (No. 30) | P21/n (No. 14) | P21/c (No. 14) | P21/c (No. 14) | P21/n (No. 14) | Ccc2 (No. 37) |
| Z | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
| ρ (calcd) g cm−3 | 1.508 | 1.554 | 1.637 | 1.605 | 1.622 | 1.574 | 1.529 |
| μ (MoKa), mm−1 | 0.937 | 0.966 | 1.091 | 1.069 | 0.983 | 1.188 | 0.952 |
| Radiation (λ, Å) | 0.7107 | 0.7107 | 0.7107 | 0.7107 | 0.7107 | 0.7107 | 0.7107 |
| Temperature (K) | 298 | 233 | 298 | 298 | 298 | 298 | 298 |
| Reflns collected | 1969 | 20712 | 20623 | 6854 | 17163 | 14993 | 9423 |
| Unique reflections | 1641 | 5334 | 3171 | 2317 | 4294 | 3243 | 3237 |
| Param refined | 158 | 344 | 316 | 316 | 3116 | 244 | 131 |
| R [I > 2σ (I)] | 0.0774 | 0.0380 | 0.0591 | 0.243 | 0.0422 | 0.0443 | 0.0789 |
| Rw [I > 2σ (I)] | 0.1120 | 0.0557 | 0.0600 | 0.2878 | 0.0706 | 0.0529 | 0.1165 |
| Goodness-of-fit | 1.346 | 1.221 | 1.097 | 3.522 | 1.246 | 1.023 | 0.933 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kondo, M.; Takahashi, H.; Watanabe, H.; Shimizu, Y.; Yamanishi, K.; Miyazawa, M.; Nishina, N.; Ishida, Y.; Kawaguchi, H.; Uchida, F. Syntheses and Characterization of New Nickel Coordination Polymers with 4,4’-Dipyridylsulfide. Dynamic Rearrangements of One-Dimensional Chains Responding to External Stimuli: Temperature Variation and Guest Releases/Re-Inclusions. Int. J. Mol. Sci. 2010, 11, 2821-2838. https://doi.org/10.3390/ijms11082821
Kondo M, Takahashi H, Watanabe H, Shimizu Y, Yamanishi K, Miyazawa M, Nishina N, Ishida Y, Kawaguchi H, Uchida F. Syntheses and Characterization of New Nickel Coordination Polymers with 4,4’-Dipyridylsulfide. Dynamic Rearrangements of One-Dimensional Chains Responding to External Stimuli: Temperature Variation and Guest Releases/Re-Inclusions. International Journal of Molecular Sciences. 2010; 11(8):2821-2838. https://doi.org/10.3390/ijms11082821
Chicago/Turabian StyleKondo, Mitsuru, Hideaki Takahashi, Hirotaka Watanabe, Yusuke Shimizu, Katsunori Yamanishi, Makoto Miyazawa, Naoko Nishina, Yutaka Ishida, Hiroyuki Kawaguchi, and Fumio Uchida. 2010. "Syntheses and Characterization of New Nickel Coordination Polymers with 4,4’-Dipyridylsulfide. Dynamic Rearrangements of One-Dimensional Chains Responding to External Stimuli: Temperature Variation and Guest Releases/Re-Inclusions" International Journal of Molecular Sciences 11, no. 8: 2821-2838. https://doi.org/10.3390/ijms11082821
APA StyleKondo, M., Takahashi, H., Watanabe, H., Shimizu, Y., Yamanishi, K., Miyazawa, M., Nishina, N., Ishida, Y., Kawaguchi, H., & Uchida, F. (2010). Syntheses and Characterization of New Nickel Coordination Polymers with 4,4’-Dipyridylsulfide. Dynamic Rearrangements of One-Dimensional Chains Responding to External Stimuli: Temperature Variation and Guest Releases/Re-Inclusions. International Journal of Molecular Sciences, 11(8), 2821-2838. https://doi.org/10.3390/ijms11082821




