Convenient Replacement of the Hydroxy by an Amino Group in 4 Hydroxycoumarin and 4-Hydroxy-6-methyl-2-pyrone under Microwave Irradiation
Abstract
:Introduction
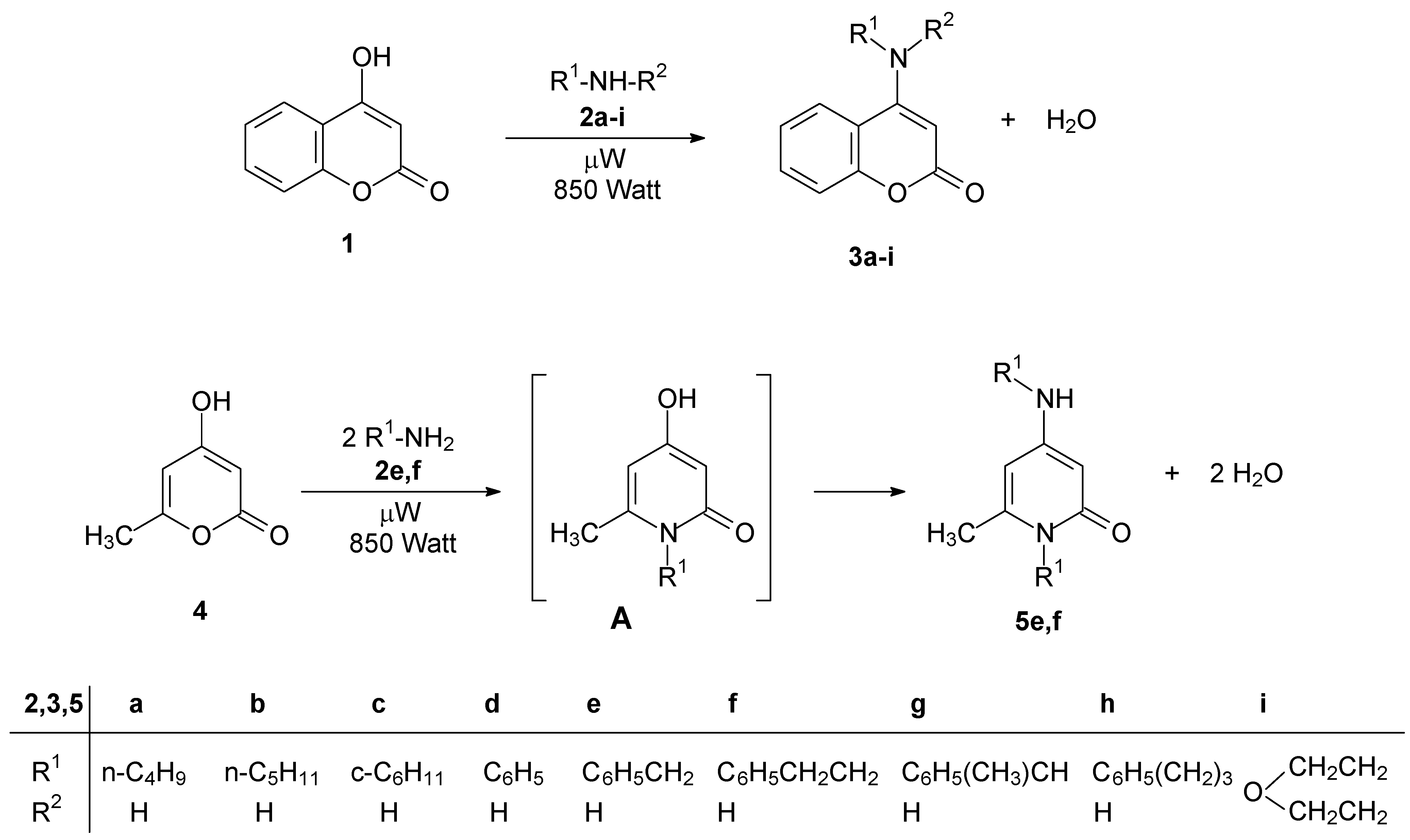
Results and Discussion
| Starting compounds | Reaction time a (min) | Product | M.p., °C | Yield, % |
| 1 + butylamine (2a) | 2.0 | 3a | 117-119 [3] | 90 |
| 1 + pentylamine (2b) | 2.0 | 3b | 162-164 [3] | 83 |
| 1 + cyclohexylamine (2c) | 2.0 | 3c | 193-195 [3] | 40 |
| 1 + aniline (2d) | 2.0 | 3d | 267-268 [4] | 97 |
| 1 + benzylamine (2e) | 2.0 | 3e | 240-242 [4] | 98 |
| 1 + phenethylamine (2f) | 2.0 | 3f | 169-171 [4] | 97 |
| 1 + 1-phenylethylamine (2g) | 2.0 | 3g | 186-187 | 95 |
| 1 + 3-phenylpropylamine (2h) | 2.0 | 3h | 167-169 | 94 |
| 1 + morpholine (2i) | 1.5 | 3i | 140-141 [5] | 73 |
| 4 + benzylamine (2e) | 10 | 5e | 185-187 [7,8] | 90 |
| 4 + phenethylamine (2f) | 10 | 5f | 166-168 [7,8] | 93 |
Conclusions
Experimental
General procedure
Spectral Data
References and Notes
- Hepworth, J. D. Comprehensive Heterocyclic Chemistry; Katritzky, A. R., Rees, C. W., Boulton, A. J., McKillop, A., Eds.; Pergamon Press: Oxford, 1984; Vol. 3, Chapter 2.24; pp. 799–810. [Google Scholar]
- Hishmat, O. H.; Gohar, A. M.; Wassef, M. E.; Shalash, M. R.; Ismail, I. Synthesis and biological activity of some coumarin and furocoumarin derivatives. Pharm. Acta Helv. 1977, 52, 252–255, and references cited therein. [Google Scholar]
- Ivanov, I. C.; Karagiosov, S. K.; Manolov, I. Synthesis of 4-(monoalkylamino)coumarins. Arch. Pharm. (Weinheim). 1991, 324, 61–62, and references cited therein. [Google Scholar]
- Tabakovic, K.; Tabacovic, I.; Ajdini, N.; Leci, O. A novel transformation of 4-arylaminocoumarins to 6H-1-benzopyrano[4,3-b]quinolin-6-ones under Vilsmeier-Haack conditions. Synthesis 1987, 308–310. [Google Scholar]
- Zagorevskii, V. A.; Savel’ev, V. L.; Meshcheryakova, L. M. Study in pyrane series and relative compounds. 42. On synthesis of 4-aminocoumarins and on mechanism of halogen substitution in 4-chlorocoumarin. Khim. Geterotsikl. Soed. 1970, 8, 1019–1023, [Chem. Abstr. 1971, 74, 76269c] and references cited therein. [Google Scholar]
- Ivanov, I. C.; Stoyanov, E. V.; Denkova, P. S.; Dimitrov, V. S. Synthesis of substituted 1,2-dihydro-2-imino-7-methyl-1,6(6H)-naphthyridin-5-ones. Liebigs Ann./Recueil 1997, 1777–1781. [Google Scholar]
- Ivanov, I. C.; Stoyanov, E. V.; Alexandrova, S. V. On the preparation of 4-hydroxy- and 4-amino-6-methyl-2(1H)-pyridones from 4-hydroxy-6-methyl-2-pyrone. Farmatsiya (Sofia). 1997, 44, 3–6, [Chem. Abstr. 1998, 128, 243925w]. [Google Scholar]
- Heber, D.; Stoyanov, E. V. The microwave assisted nucleophilic substitution of 4-hydroxy-6-methyl-2(1H)-pyridones. Synlett 1999, 1747–1748. [Google Scholar]
- Samples Availability: Available from the authors.
© 2004 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Stoyanov, E.V.; Ivanov, I.C. Convenient Replacement of the Hydroxy by an Amino Group in 4 Hydroxycoumarin and 4-Hydroxy-6-methyl-2-pyrone under Microwave Irradiation. Molecules 2004, 9, 627-631. https://doi.org/10.3390/90800627
Stoyanov EV, Ivanov IC. Convenient Replacement of the Hydroxy by an Amino Group in 4 Hydroxycoumarin and 4-Hydroxy-6-methyl-2-pyrone under Microwave Irradiation. Molecules. 2004; 9(8):627-631. https://doi.org/10.3390/90800627
Chicago/Turabian StyleStoyanov, Edmont V., and Ivo C. Ivanov. 2004. "Convenient Replacement of the Hydroxy by an Amino Group in 4 Hydroxycoumarin and 4-Hydroxy-6-methyl-2-pyrone under Microwave Irradiation" Molecules 9, no. 8: 627-631. https://doi.org/10.3390/90800627
APA StyleStoyanov, E. V., & Ivanov, I. C. (2004). Convenient Replacement of the Hydroxy by an Amino Group in 4 Hydroxycoumarin and 4-Hydroxy-6-methyl-2-pyrone under Microwave Irradiation. Molecules, 9(8), 627-631. https://doi.org/10.3390/90800627




