Microwave-Accelerated or Conventionally Heated Iodination Reactions of Some Aromatic Amines, Using ortho-Periodic Acid as the Oxidant †
Abstract
:Introduction
Results and Discussion
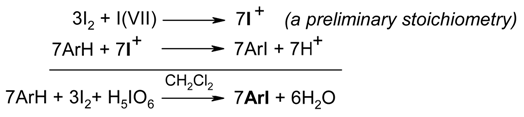
- a)
- by conventional heating: the vigorously stirred reaction mixtures were gently boiled under a reflux condenser for 10-210 minutes (see Table 1);
- b)
- by focused monomode microwave irradiation, under an externally attached reflux condenser and with stirring: the reaction mixtures were placed into the microwave cavity and were then irradiated for 2-20 minutes (Table 1); an appropriate power output was used to secure a mild, uninterrupted reflux of the solvent.
| Iodinated product | M.p. (˚C) | Lit. [7] m.p. (˚C) | Conventional method | MW method | ||
| time [min] | yield [%] | time [min] | yield [%] | |||
| 4-I-C6H4NH2 | 62-63 | 62-63 | 30 | 62 | 5 | 68 |
| 4-I-C6H4NHMe | 28-29 | 29-31 | 60 | 59 | 20 | 47.5 |
| 4-I-C6H4NMe2 | 79-80 | 81-82 | 30 | 79 | 2 | 77 |
| 4-I-C6H4NHEt | 53.5-54.3 | not found b) | 60 | 66 | 10 | 61 |
| 4-I-C6H4NEt2 | 26-27 | 26-29 | 60 | 79 | 2 | 79 |
| 2-Cl-4-I-C6H3NH2 | 63-64 | 62-63 | 30 | 12 | 5 | 11 |
| 3-Cl-4-I-C6H3NH2c) | 67-69 | 64-68 | 210 | 46 | 10 | 19 |
| 4-I-2-Me-C6H3NH2 | 86-87 | 86-88 | 10 | 71 | 5 | 58 |
| 4-I-3-Me-C6H3NH2 | 43-44 | 40-42 | 30 | 57.5 | 2 | 71.5 |
| 2-Br-4-I-C6H3NH2 | 70-71 | 71-72 | 30 | 8 | 20 | 14 |
| 4-Br-2-I-C6H3NH2 | 76-77 | 77-78 | 60 | 11 | 10 | 11 |
- a)
- Satisfactory microanalyses obtained for the purified products: I ±0.3%.
- b)
- Probably a new compound. Calcd. for C8H10IN: C, 39.02; H, 4.07; I, 50.96%; Found: C, 39.0; H, 4.2; I, 50.9%.
- c)
- Admixed with ca. 10% of its ortho isomer (as established from its 1H- and 13C-NMR spectra).
Experimental
General
Microwave-Accelerated Iodination Reactions
References and Notes
- a) Microwave-Enhanced Chemistry Fundamentals, Sample Preparation and Applications; Kingston, H. M.; Haswell, S. J. (Eds.) American Chemical Society: Washington, D. C., 1997. b) Microwaves in Organic Synthesis; Loupy, A. (Ed.) Wiley-VCH: Weinheim, 2002. c) Microwave Synthesis; Chemistry at the Speed of Light; Hayes, B. L. (Ed.) CEM Publishing: Matthews, NC, 2002.
- Perreux, L.; Loupy, A. A tentative realization of microwave effects in organic synthesis according to the reaction medium, and mechanistic consideration. Tetrahedron 2001, 57, 9199–9223, A review. [Google Scholar]
- Lindstroem, P.; Tierney, J.; Whathey, B.; Westman, J. Microwave assisted organic synthesis – a review. Tetrahedron 2001, 57, 9225–9283. [Google Scholar]
- For a review see: Fini, A.; Breccia, A. Chemistry by Microwaves. Pure Appl. Chem. 1999, 71, 573–579. [Google Scholar]
- Lulinski, P.; Kryska, A.; Sosnowski, M.; Skulski, L. Eco-friendly Oxidative Iodination of Various Arenes with a Urea-Hydrogen Peroxide Adduct (UHP) as the Oxidant. Synthesis 2004, 441–445. [Google Scholar]
- Sosnowski, M.; Skulski, L. Microwave-Accelerated Iodination of Some Aromatic Amines, Using Urea-Hydrogen Peroxide Addition Compound (UHP) as the Oxidant. Molecules 2002, 7, 867–880, Avail. at URL: http://www.mdpi.org/molecules/papers/71200867.pdf. [Google Scholar]
- Dictionary of Organic Compounds, 6th ed.; Chapman & Hall: London, 1996.
- Sample Availability: Available from the authors.
© 2004 by MDPI (http:www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Sosnowski, M.; Skulski, L.; Wolowik, K. Microwave-Accelerated or Conventionally Heated Iodination Reactions of Some Aromatic Amines, Using ortho-Periodic Acid as the Oxidant †. Molecules 2004, 9, 617-621. https://doi.org/10.3390/90700617
Sosnowski M, Skulski L, Wolowik K. Microwave-Accelerated or Conventionally Heated Iodination Reactions of Some Aromatic Amines, Using ortho-Periodic Acid as the Oxidant †. Molecules. 2004; 9(7):617-621. https://doi.org/10.3390/90700617
Chicago/Turabian StyleSosnowski, Maciej, Lech Skulski, and Katarzyna Wolowik. 2004. "Microwave-Accelerated or Conventionally Heated Iodination Reactions of Some Aromatic Amines, Using ortho-Periodic Acid as the Oxidant †" Molecules 9, no. 7: 617-621. https://doi.org/10.3390/90700617
APA StyleSosnowski, M., Skulski, L., & Wolowik, K. (2004). Microwave-Accelerated or Conventionally Heated Iodination Reactions of Some Aromatic Amines, Using ortho-Periodic Acid as the Oxidant †. Molecules, 9(7), 617-621. https://doi.org/10.3390/90700617




