Abstract
The new compounds 2-[4-(6-cyanobenzothiazol-2-yl)phenyl]-5-(6-cyano- benzothiazol-2-yl)furan (6a) and 2-[4-(6-Cyanobenzothiazol-2-yl)phenyl]-5-(6-cyano- benzothiazol-2-yl)thiophene (6b) were synthesized by multi-step reactions from the corresponding 2-furan and 2-thiophene carboxaldehydes (route A), as well as from 2- furan and 2- thiophene carboxylic acids (route B). Route B involves one less step than route A, but the overall yields of the reactions are considerably lower.
Introduction
Benzothiazoles are heterocyclic compounds with multiple applications and, although they have been known from long ago to be biologically active, [1,2,3], their varied biological features are still of great scientific interest nowadays. They show, for example, very intensive antitumor activity, especially the phenyl-substituted benzothiazoles [4,5,6], while condensed pyrimido[2,1-b]benzothiazoles and benzothiazolo[2,3-b]-quinazolines exert antiviral activity [7]. Recently, Racanè at al. [8] have described the synthesis of bis-substituted amidinobenzothiazoles as potential anti-HIV agents. Substituted benzamido- and phenylacetamido-substituted 2-phenylbenzothiazoles [9,10,11], 2- substituted 6-nitro- and 6-aminobenzothiazoles [12], fluorobenzothiazoles [13] and Schiff bases derived from benzothiazoles [14] show microbiological activity.
On the other hand, bis-benzothiazoles and substituted bis-benzothiazoles are frequently fluorescent compounds and therefore convenient for fluorimetric measurements, which could serve as a potential method for detection of binding the biologically active compounds on DNA [15]. However, there is little data describing compounds containing two benzothiazole rings attached via a heterocyclic system, such as 2,5-benzothiazolylfuran and thiophene and its derivatives [16,17,18], as well as their vinylogues [19,20].
Results and Discussion
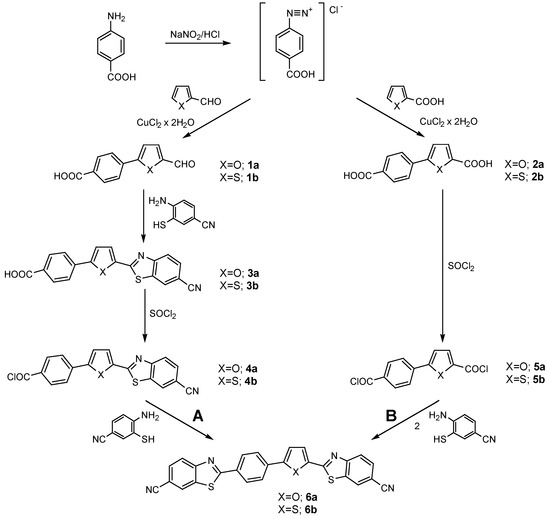
Cyano substituted bis-benzothiazolyl compounds 6a and 6b were synthesized starting from 2- furan- and 2-thiophenecarboxaldehydes or from 2-furan- and 2-thiophenecarboxylic acids via multistep reactions by the two routes designated A and B, respectively (Scheme 1).
In the first step of the reaction, 2-furan- or 2-thiophenecarboxaldehydes, and 2-furan- or 2- thiophenecarboxylic acids were arylated by the diazonium salts of p-aminobenzoic acid by the well known Meerwein arylation procedure [20,21]. In agreement with the greater aromaticity of thiophenes with respect to furans, it is evident from the arylation yields that the furan derivatives are more reactive than the corresponding thiophene derivatives. Thus, 2-furancarboxaldehyde yields 60% of 1a [20] upon arylation; 2-furancarboxylic acid yields 58% of 2a [20], 2-thiophenealdehyde yields 17% of 1b [21] and 2-thiophenecarboxylic acid yields 19% of 2b, respectively.
Benzothiazolyl compounds 3a and 3b, synthesized from the appropriate aldehydes and 4-amino-3- mercaptobenzonitrile [22], according to a modified condensation method [18,23], have been converted to the corresponding chlorocarbonyl derivatives 4a and 4b. In the last step (route A), these chlorocarbonyl compounds were condensed with 4-amino-3mercaptobenzonitrile to obtain the bis- cyanobenzothiazolyl compounds 6a and 6b in good yields of about 75%.
The syntheses of compounds 6a and 6b (route B) have been carried out with 5-(4-carboxyphenyl)- 2-furylcarboxylic acid (2a) [20] and 5-(4-carboxyphenyl)-2-thienylcarboxylic acid (2b). Dicarboxylic compounds 2a and 2b have been converted to the dichlorocarbonyl derivatives 5a and 5b and then these were condensed with 4-amino-3-mercaptobenzonitrile in yields of about 35%. Route B has one step less then route A, but the overall yield of the reactions is considearbly lower.
Scheme 1.
Scheme 1.
Experimental
General
Melting points were determined on a Koffler block apparatus and are uncorrected. IR spectra were determined with a Nicolet Magna 760 infrared spectrophotometer using KBr pellets. 1H-NMR spectral data were determined with a Brucker Avance DPX 300 MHz NMR spectrometer with tetramethylsilane as an internal standard. Elemental analyses were carried out in the Microanalytical Laboratory at the Rugjer Boskovic Institute.
General Procedure for the Arylation of Compounds 1a, 1b, 2a and 2b.
A solution of p-aminobenzoic acid (75 mmol) in water (120 mL) and concentrated HCl (40 mL) was cooled to 5 °C and diazotized with a solution of NaNO2 (91 mmol) in H2O (35 mL). After 20 min a solution of 75 mmol of 2-furanaldehyde for 1a [20], 2-thiophenealdehyde for 1b [21], 2-furan- carboxylic acid for 2a [20] or 2-thiophenecarboxylic acid for 2b, respectively, in acetone (50 mL) and a solution of CuCl2·2H2O (23 mmol) in water (25 mL) were added to the stirred reaction mixture. The reaction mixture was left to stand at room temperature for two days with occasional shaking. After dilution with water (500 mL) the precipitated crystals were filtered off and washed with abundant hot water.
5-(4-carboxyphenyl)-2-thiophenecarboxylicacid (2b). Yield: 3.9 g (21%); m.p.: >300 °C (from DMF - water); IR (KBr): 1674 (COOH) cm-1; 1H-NMR (300 MHz; DMSO-d6) δ: 13.22 (s, 2H, H-COOH), 8.01 (d, J=8.4 Hz, 2H, H-arom.), 7.88 (d, J=8.4 Hz, 2H, H-arom.), 7.76 (d, J=3.9, 1H, H-thioph.), 7.72 (d, J=3.9 Hz, 1H, H-thioph.); Anal. Calcd. for C12H8O4S: C 58.06, H 3.25, S 12.92. Found: C 58.32, H 3.13, S 12.78.
General Procedure for the Synthesis of Benzothiazolyl Compounds 3a and 3b.
4-Amino-3-mercaptobenzonitrile (21 mmol) was added to a solution of the appropiate aldehyde 1a or 1b (21 mmol) in pyridine (60 mL) and the stirred reaction mixture was refluxed 4 h. The mixture was then poured into 2 M hydrochloric acid (400 mL), and after cooling overnight, the crystalline product obtained was oxidized with an ethanolic solution of FeCl3 to obtain the benzothiazole compounds 3a and 3b, respectively.
2-(4-Carboxyphenyl)-5-(6-cyanobenzothiazol-2-yl)furan (3a). Yield: 5.0 g (70%); m.p.>300°C (DMF); IR (KBr): 2210 (CN), 1670 (COOH) cm-1; 1H-NMR (300 MHz; DMSO-d6) δ: 13.18 (s, 1H, H-COOH), 8.78 (s, 1H, H-arom.), 8.50 (d, J=8.6 Hz, 1H, H-arom.), 8.07 (d, J=8.5 Hz, 2H, H-arom.), 8.01 (d, =8.5 Hz, 2H, H-arom.), 7.97 (d, J=8.9 Hz, 1H, H-arom.), 7.68 (d, J=3.8 Hz, 1H, H-furan), 7.52 (d, J=3.8 Hz, 1H, H-furan); Anal. Calcd. for C19H10N2O3S: C 65.89, H 2.91, N 8.09 S 9.26. Found: C 65.52, H 2.82, N 8.31 S 9.13.
2-(4-Carboxyphenyl)-5-(6-cyanobenzothiazol-2-yl)thiophene (3b). Yield: 4.5 g (62%); m.p.>300°C (from DMF - ethanol); IR (KBr): 2224 (CN), 1687 (COOH) cm-1; 1H-NMR (300 MHz; TFA) δ: 8.33 (s, 1H, H-arom.), 8.11 (d, J=4.2 Hz, 1H, H-thioph.), 8.06 (d, J=8.1 Hz, 2H, H-arom.), 7.99 (d, J=8.7 Hz, 1H, H-arom.), 7.91 (d, J=8.4 Hz, 1H, H-arom.), 7.68 (d, J=8.1 Hz, 2H, H-arom.), 7.56 (d, J=4.2 Hz, 1H, H-thioph.); Anal. Calcd. for C19H10N2O2S2: C 62.97, H 2.78, N 7.73 S 17.70. Found: C 62.95, H 2.82, N 8.01 S 17.58.
General Procedure for the Synthesis of Chlorocarbonyl Compounds 4a, 4b, 5a and 5b.
Heating of a mixture of compounds 3a or 3b (11.5 mmol) with thionyl chloride (230 mmol) or of compounds 2a or 2b (18 mmol) with thionyl chloride (344 mmol) for 4 h on an oil bath at 85 °C afforded the corresponding monochlorocarbonyl compound 4a and 4b or the dichlorocarbonyl compound 5a and 5b.
2-(4-Chlorocarbonylphenyl)-5-(6-cyanobenzothiazol-2-yl)furan (4a). Yield: 3.1 g (74%); m.p.>300°C (CHCl3); IR (KBr): 2210 (CN), 1760 (COCl) 1720 (COCl) cm-1; 1H-NMR (300 MHz; DMSO-d6) δ: 8.78 (s, 1H, H-arom.), 8.20 (d, J=8.5 Hz, 1H, H-arom.), 8.07 (d, J=8.2 Hz, 2H, H-arom.), 8.00 (d, J=8.5 Hz, 2H, H-arom.), 7.96 (d, J=9.0 Hz, 1H, H-arom.), 7.68 (d, J=3.8 Hz, 1H, H-furan), 7.51 (d, J=3.8 Hz, 1H, H-furan).
2-(4-Chlorocarbonylphenyl)-5-(6-cyanobenzothiazol-2-yl)thiophene (4b). Yield: 2.7 g (62%); m.p.>300°C (CHCl3);. IR (KBr): 2222 (CN), 1769 (COCl) 1720 (COCl) cm-1; 1H-NMR (300 MHz; DMSO-d6) δ: 8.75 (s, 1H, H-arom.), 8.17 (d, J=8.7 Hz, 1H, H-arom.), 8.07 (d, J=3.9 Hz, 1H, H-thioph.), 8.03 (d, J=8.1 Hz, 2H, H-arom.), 7.95 (m, 3H, H-arom.), 7.86 (d, J=3.9 Hz, 1H, H-thioph.).
2-(4-Chlorocarbonylphenyl)-5-chlorocarbonylfuran (5a). Yield: 2.2 g (46%); m.p.102-106 °C (from benzene - cyclohexane); IR (KBr): 1740 (COCl) cm-1; 1H-NMR (300 MHz; CDCl3) δ: 8.21 (d, J=8.7 Hz, 2H, H-arom.), 7.95 (d, J=8.7 Hz, 2H, H-arom.), 7.60 (d, J=3.8 Hz, 1H, H-furan), 7.05 (d, J=3.8 Hz, 1H, H-furan).
2-(4-Chlorocarbonylphenyl)-5-chlorocarbonylthiophene (5b). Yield: 3.7 g (81%); m.p.88-92 °C (from benzene - cyclohexane); IR (KBr): 1771 (COCl) 1736 (COCl) cm-1; 1H-NMR (300 MHz; CDCl3) δ: 8.20 (d, J=8.5 Hz, 2H, H-arom.), 7.99 (d, J=4.1 Hz, 1H, H-thioph.), 7.80 (d, J=8.5 Hz, 2H, H-arom.), 7.52 (d, J=4.1 Hz, 1H, H-thioph.).
General Procedure for the Synthesis of bis-Benzothiazolyl Compounds 6a and 6b.
A solution of monochlorocarbonyl compounds 4a or 4b (5.0 mmol, route A), or of dichloro- carbonyl compounds 5a or 5b (2.5 mmol, route B) in dry chlorobenzene (100 mL) was stirred under a stream of nitrogen. To these solutions 4-amino-3-mercaptobenzonitrile (5.1 mmol) was added. The reaction mixture was heated under reflux under the stream of nitrogen for 70 h. After cooling, a crystalline product was obtained.
2-[4-(6-Cyanobenzothiazol-2-yl)phenyl]-5-(6-cyanobenzothiazol-2-yl)furan (6a). Yields: 1.73 g (75%, method A), 0.38 g (33%, method B); m.p.>300°C (DMF); IR (KBr): 2226 (CN) cm-1; 1H-NMR (300 MHz; TFA) δ: 9.35 (s, 1H, H-arom.), 9.27 (s, 1H, H-arom.), 9.03 (d, J=8.1 Hz, 2H, H-arom.), 8.96-8.80 (m, 6H, H-arom.), 8.74 (d, J=3.9 Hz, 1H, H-furan), 8.16 (d, J=3.9 Hz, 1H, H-furan). Anal. Calcd. for C26H12N4OS2: C 67.81, H 2.63, N 12.17 S 13.92. Found: C 67.86, H 2.54, N 12.10, S 14.02.
2-[4-(6-Cyanobenzothiazol-2-yl)phenyl]-5-(6-cyanobenzothiazol-2-yl)thiophene (6b). Yields: 1.95 g (82%, method A), 0.43 g (36%, method B); m.p.>300°C (DMF); IR (KBr): 2224 (CN) cm-1; 1H-NMR (300 MHz; TFA) δ: 8.49 (s, 1H, H-arom.), 8.38 (s, 1H, H-arom.), 8.18-8.05 (m, 4H, H-arom.), 8.01 (d, J=3.9 Hz, 1H, H-thioph.), 7.97-7.92 (m, 4H, H-arom.), 7.70 (d, J=4.0 Hz, 1H, H-thioph.); Anal. Calcd. for C26H12N4S3: C 65.52, H 2.54, N 11.76, S 20.18. Found: C 65.38, H 2.51, N 11.66, S 20.32.
Acknowledgments
This study was financially supported by the Ministry of Science of the Republic of Croatia within project No. 125005, and US-Croatian Scientific and Technogical Cooperation Award No. JF 146.
References
- Lácová, M.; Chovancová, J.; Hýblová, O.; Varkonda, S. Synthesis and Pesticidal Activity of Acyl Derivatives of 4-chloro-2-aminobenzothiazole and the Products of their Reduction. Chem. Pap. 1991, 45, 411–418. [Google Scholar]
- Chulák, I.; Sutorius, V.; Sekerka, V. Benzothiazole compound XXXV. Synthesis of 3-substituted 2-benzylbenzothiazolium Salts and their Growth-regulating Effect on Triticum Aestivum L. Chem. Pap. 1990, 44, 131–138. [Google Scholar]
- Papenfuhs, T. Preparation of Benzothiazoles as Intermediates for Dyes, Plant Protectants and Pharmaceuticals. Ger. Offen. DE 3,528,032, 1987. [Google Scholar]
- Bradshaw, T.D.; Bibby, M.C.; Double, J.A.; Fichtner, I.; Cooper, P.A.; Alley, M.C.; Donohue, S.; Stinson, S.F.; Tomaszewjski, J. E.; Sausville, E.A.; Stevens, M.F.G. Preclinical Evaluation of Amino Acid Prodrugs of Novel Antitumor 2-(4-amino-3-methylphenyl)benzothiazoles. Mol. Cancer Therapeutics. 2002, 1, 239–246. [Google Scholar]
- Bradshaw, T.D.; Chua, M.S.; Browne, H.L.; Trapani, V.; Sausville, E.A.; Stevens, M.F.G. In Vitro Evaluation of Amino Acid Prodrugs of Novel Antitumor 2-(4-amino-3- methylphenyl)benzothiazoles. Br. J. Cancer. 2002, 86, 1348–1354. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, I.; Jennings, S.A.; Vishnuvajjala, B.R.; Westwell, A.D.; Stevens, M.F.G. Antitumor Benzothiazoles. 16. Synthesis and Pharmacutical Properties of Antitumor 2-(4- aminophenyl)benzothiazole Amino Acid Prodrugs. J. Med. Chem. 2002, 45, 744–747. [Google Scholar] [CrossRef] [PubMed]
- El-Sherbeny, M.A. Synthesis of Certain Pyrimido[2,1-b]benzothiazole and Benzothiazolo[2,3- b]quinazoline Derivatives for in Vitro Antitumor and Antiviral Activities. Arzeneim. Forsch. 2000, 50, 848–853. [Google Scholar] [CrossRef]
- Racanè, L.; Tralić-Kulenović, V.; Fišer-Jakić, L.; Boykin, D.W.; Karminski-Zamola, G. Synthesis of bis-Substituted Amidinobenzothiazoles as Potential Anti-HIV Agents. Heterocycles. 2001, 55, 2085–2098. [Google Scholar]
- Sener, E.A.; Arpaci, O.T.; Yalcin, I.; Altanlar, N. Synthesis and Microbiological Activity of Some Novel 5-benzamido- and 5-phenylacetamido- Substituted 2-phenylbenzoxazole Derivatives. Farmaco. 2000, 55, 397–405. [Google Scholar] [CrossRef]
- Mruthyunjayaswamy, B.H.M.; Shanthaveerappa, B.K. Synthesis and Pharmacological Evaluation of 3,5-disubstituted indole-2-[N beta-(substituted benzopyran-2’-one-3’-carboxyl)]carboxy hydrazides and 2H-3-(various substituted indol-3’-yl)methyl-1,3-benzothiazoles. Indian J. Chem. Sect. B. 2000, 39, 433–439. [Google Scholar]
- Temiz-Arpaci, O.; Aki-Sener, E.; Yalcin, I; Altanlar, N. Synthesis and Antimicrobial Activity of Some 2-[p-substituted-phenyl]benzoxazol-5-yl-arylcarboxyamides. Arch. Pharm. Weinheim, Ger. 2002, 335, 283–288. [Google Scholar] [CrossRef]
- Delmas, F.; Di Giorgio, C.; Robin, M.; Azas, N.; Gasquet, M.; Detang, C.; Costa, M.; Timon- David, P.; Galy, J.P. In Vitro Activities of Position 2 Substitution–bearing 6-nitro and 6-amino- benzothiazoles and their Corresponding Anthranilic Acid Derivatives Against Leishmania Infantum and Trichomonas Vaginalis. Antimicrob. Ag. Chemother. 2002, 46, 2588–2594. [Google Scholar] [CrossRef]
- Pattan, S.R.; Babu, S.N.N.; Angadi, J. Synthesis and Biological Activity of 2-amino [5’-(4’- sulphonylbenzylidene)-2,4-thiazolidine dione]-7-(substituted)-6-fluoro benzothiazoles. Indian J. Heterocyclic Chem. 2002, 11, 333–334. [Google Scholar]
- Mahmood-ul-Hasan; Chohan, Z.H.; Supuran, C.T. Antibacterial Zn(II) Compounds of Schiff Bases Derived from Some Benzothiazoles. Main Group Met. Chem. 2002, 25, 291–296. [Google Scholar] [CrossRef]
- Akyama, S.; Ochiai, T.; Nakatsuji, S.; Nakashima, K.; Ohkura, Y. Preparation and Evaluation of Fatty Acid Esters of Fluorescent p-Substituted Phenols as Substrates for Measurement of Lipase Activity. Chem. Pharm. Bull. 1987, 35, 3029–3032. [Google Scholar] [CrossRef]
- Farcasan, V.; Balazs, I. Derivati ai Furanului (VII). Stud. Univ. Babes-Bolyai-Ser. Chem. 1968, 13, 123–127. [Google Scholar]
- Dotrong, M.; Mehta, R.; Balchin, G.A.; Tomlinson, R.C.; Sinsky, M.; Lee, C.Y.-C.; Evers, R.C. Synthesis, Processing, and Third-Order Nonlinear Optical Properties of Benzobisthiazole Polymers Containing Thiophene Moieties. J. Pol. Sci. Part A: Polym. Chem. 1993, 31, 723–729. [Google Scholar] [CrossRef]
- Racanè, L.; Tralić-Kulenović, V.; Karminski-Zamola, G.; Fišer-Jakić, L. Synthesis and Fluorescent Properties of Some New Unsymmetric bis-Benzothiazolyl Furans and Thiophenes. Monatsh. Chem. 1995, 126, 1375–1381. [Google Scholar] [CrossRef]
- Kornilov, M.Y.; Ruban, E.M.; Fredchuk, V.N.; Starinskaya, E.V.; Buznik, M.V. Reaction of Chromophores in Asymetric 2,5-disubstituted Derivatives of Furan and Thiophene. Zh. Org. Khim. 1973, 9, 2577–2582. [Google Scholar]
- Malinowski, S. Reaction of Diazo Compounds with Unsaturated Compounds II. Arylation of furfural. Polish J. Chem. 27, 54–61.
- Tralić-Kulenović, V.; Fišer-Jakić, L.; Lazarević, Z. Synthesis and Absorption Spectral Properties of Substituted Phenylfurylbenzothiazoles and their Vinylogues. Monatsh. Chem. 1994, 125, 209–215. [Google Scholar] [CrossRef]
- Tralić-Kulenović, V.; Karminski-Zamola, G.; Racanè, L.; Fišer-Jakić, L. Synthesis of Symmetric and Unsymmetric bis-Cyanosubstituted Heterocycles. Heterocyclic Comm. 1998, 4, 423–428. [Google Scholar]
- Bogert, M.T.; Naiman, B. Research on Thiazoles XX. J. Am. Chem. Soc. 1935, 57, 1529–1533. [Google Scholar] [CrossRef]
- Sample availability: Samples of compounds 2b, 3b, 6a and 6b are available from MDPI.
© 2003 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
