Synthesis and Characterization of Benzo-15-Crown-5 Ethers with Appended N2O Schiff Bases
Abstract
:Introduction
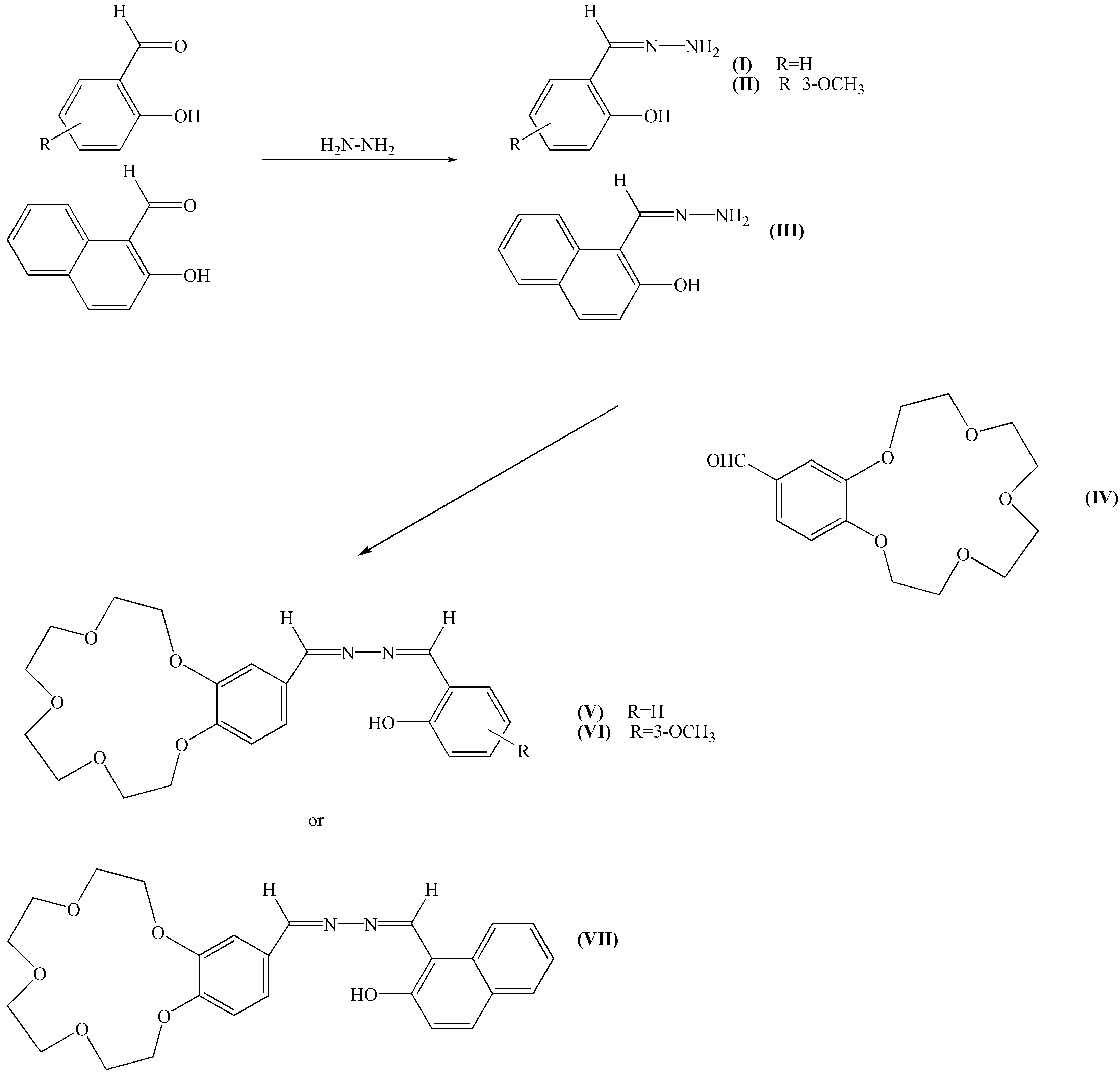
Results and Discussion
Conclusions
Acknowledgments
Experimental
General
Syntheses
N-(aminoformimidoyl)phenol (Hsh, I).
N-(aminoformimidoyl)-6-methoxyphenol (Hmsh, II).
N-(aminoformimidoyl)naphthol (Hnh, III).
4’-Formylbenzo-15-crown-5 (IV).
N-(formimidoyl-benzo-15-crown-5)-N´-(formimidoyl)phenol (Hsh-b-15-c-5, V).
N-(formimidoyl-benzo-15-crown-5)-N´-(formimidoyl)-6-methoxyphenol (Hmsh-b-15-c-5 VI).
N-(formimidoyl-benzo-15-crown-5)-N´-(formimidoyl)-naphthol (Hnh-b-15-c-5 VII).
References
- van Veggel, F. C. J. M.; Verboom, W.; Reinhoudt, D. N. Chem. Rev. 1994, 94, 279.
- Beer, P. D. Adv. Inorg. Chem. 1992, 39, 79.
- Beer, P. D. Endeavour 1992, 16, 182.
- Beer, P. D.; Gale, P. A.; Chen, G. Z. Coord. Chem.Rev. 1992, 39, 79.
- Santos, I. C.; Vilas-Boas, M.; Piedade, F.; Freire, C.; Duarte, M. T.; de Castro, B. Polyhedron 2000, 19, 655.
- Freire, C.; de Castro, B. Polyhedron 1998, 17, 4227.
- Freire, C.; de Castro, B. J. Chem. Soc. Dalton Trans. 1998, 1491.
- Azevedo, F.; Carrondo, M. A. A. de C. T.; de Castro, B.; Convery, M.; Domingues, D.; Freire, C.; Duarte, M. T.; Nielson, K.; Santos, I. Inorg. Chim. Acta 1994, 219, 43.
- Carrondo, M. A. A. de C. T.; de Castro, B.; Coelho, A. M.; Domingues, D.; Freire, C.; Morais, J. Inorg. Chim. Acta 1993, 205, 157.
- Freire, C.; de Castro, B. Inorg. Chem. 1990, 29, 5113.
- Pedersen, C. J.; Frensdorff, H. K. Angew. Chem. Int. Ed. Engl. 1972, 11, 16.
- Pedersen, C. J. J. Am. Chem. Soc. 1967, 89, 7917.
- Gokel, G. W.; Durst, H. D. Synthesis 1976, 168.
- Alexander, V. Chem. Rev. 1999, 95, 271.
- Sousa, C.; Borges, J. E.; Freire, C.; Romão, C.; de Castro, B. Het. Comm. 2000, 6, 323.
- Sousa, C.; Freire, C.; Romão, C.; de Castro, B. unpublished results.
- Elder, R. C. Aust. J. Chem. 1978, 31, 35.
- Sousa, C.; Freire, C.; Romão, C.; de Castro, B. J. Coord. Chem. 2001, 54, 1.
- Gul, A.; Okur, A. I.; Tan, N.; Bekaroglu, O. Synth. React. Inorg. Met.-Org. Chem. 1986, 16, 871.
- Ungaro, R.; El Haj, B.; Smid, J. J. Am. Chem. Soc. 1976, 98, 5198.
- Sample Availability: Available from the authors.
© 2003 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Sousa, C.; Freire, C.; De Castro, B. Synthesis and Characterization of Benzo-15-Crown-5 Ethers with Appended N2O Schiff Bases. Molecules 2003, 8, 894-900. https://doi.org/10.3390/81200894
Sousa C, Freire C, De Castro B. Synthesis and Characterization of Benzo-15-Crown-5 Ethers with Appended N2O Schiff Bases. Molecules. 2003; 8(12):894-900. https://doi.org/10.3390/81200894
Chicago/Turabian StyleSousa, Carla, Cristina Freire, and Baltazar De Castro. 2003. "Synthesis and Characterization of Benzo-15-Crown-5 Ethers with Appended N2O Schiff Bases" Molecules 8, no. 12: 894-900. https://doi.org/10.3390/81200894
APA StyleSousa, C., Freire, C., & De Castro, B. (2003). Synthesis and Characterization of Benzo-15-Crown-5 Ethers with Appended N2O Schiff Bases. Molecules, 8(12), 894-900. https://doi.org/10.3390/81200894



