Dual Substituent Parameter Modeling of Theoretical, NMR and IR Spectral Data of 5-Substituted Indole-2,3-diones
Abstract
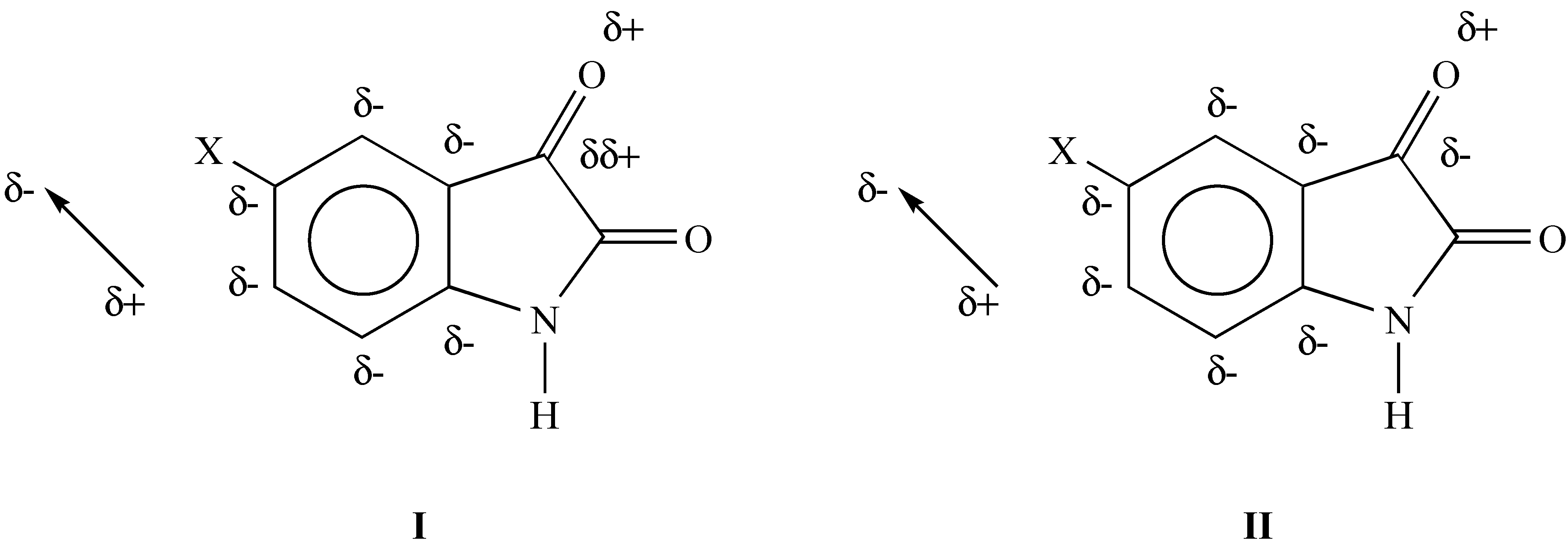
:Introduction
Results and Discussion
| Reynolds’ model: | ρF = 3.22, ρR = 3.37 | Taft’s model: | ρF = 3.80, ρF = 3.09 |
| R = 0.965 | R = 0.982 | ||
| F = 58.9, f = 0.289 | F = 68.4, f = 0.221 |
R = 0.990
F = 80.2, f = 0.087
R = -0.950
F = 38.8, f = 0.211
R = 0.982
F = 88.4, f = 0.221
R = 0.970
F = 55.3, f = 0.117
Conclusions
- 1)
- Application of the Taft’s model provides always better correlation results for both Coulson charge densities and Mulliken charges than the use of Reynolds model.
- 2)
- For the C7 site a reverse substituent effects was observed and is believed to be connected with the extended π-polarization.
- 3)
- The DSP correlation analysis for the C3 atom of the investigated molecules shows a normal substituent effect.
- 4)
- The Taft’s model DSP correlations for C3=O bond vibrational wave numbers and bond orders show that the contribution of the field effect to this bond is roughly twice the contribution of the resonance effect.
- 5)
- The previously reported vibrational-coupling model proposed on the basis of MSP correlations was confirmed using the results of DSP correlations studied in this work.
Experimental
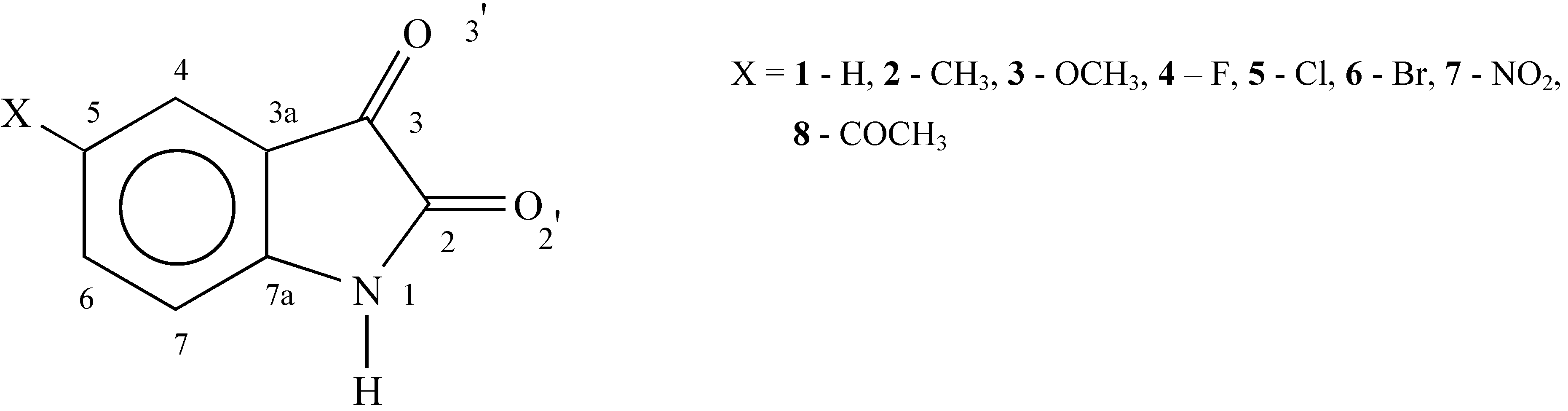
| Compound | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| 13C SCS C3)b ppm | 184.33 | 184.56 | -d | 184.92 | 183.29 | 183.12 | 182.31 | -d |
| ν(C3=O)c cm-1 | 1744.0 | 1740.1 | 1744.0 | 1744.8 | 1750.8 | 1750.4 | 1749.0 | 1753.6 |
| Comp. | AM1 | PM3 | ||||||||
| qC(C3) | qM(C3) | qC(C7) | qM(C7) | qC(C7a) | qM(C7a) | qO(C3’) | qM(C3’) | νc(C3=O)a | p(C3=O) | |
| 1 | 0.2710 | 0.3065 | -0.1857 | -0.2398 | 0.1110 | 0.1261 | -0.2674 | -0.2963 | 2134 | 1.9538 |
| 2 | 0.2686 | 0.3036 | -0.1488 | -0.1991 | 0.0727 | 0.0883 | -0.2714 | -0.2999 | 2135 | 1.9578 |
| 3 | 0.2708 | 0.3059 | -0.1811 | -0.2342 | 0.1045 | 0.1195 | -0.2690 | -0.2978 | 2134 | 1.9535 |
| 4 | 0.2692 | 0.050 | -0.1611 | -0.2139 | 0.0976 | 0.1128 | -0.2619 | -0.2906 | 2135 | 1.9611 |
| 5 | 0.2781 | 0.3080 | -0.1880 | -0.2416 | 0.1245 | 0.1392 | -0.2591 | -0.2881 | 2135 | 1.9589 |
| 6 | 0.2709 | 0.3069 | -0.1785 | -0.2321 | 0.1126 | 0.1276 | -0.2612 | -0.2901 | 2135 | 1.9589 |
| 7 | 0.2731 | 0.3111 | -0.2016 | -0.2510 | 0.1558 | 0.1703 | -0.2420 | -0.2712 | 2137 | 1.9678 |
| 8 | 0.2732 | 0.3099 | -0.2042 | -0.2579 | 0.1389 | 0.1535 | -0.2572 | -0.2863 | 2136 | 1.9598 |
| ρF | ρR | R | Fa | fb | ρF | ρR | R | Fa | fb | |
| qC(C3)c | 0.0012 | 0.0070 | 0.950 | 78.7 | 0.3302 | 0.0010 | 0.0051 | 0.965 | 87.1 | 0.2186 |
| qM(C3)c | 0.0041 | 0.0111 | 0.982 | 69.3 | 0.2119 | 0.0046 | 0.0084 | 0.966 | 95.3 | 0.2097 |
| qC(C7)c | -0.0058 | -0.0866 | 0.960 | 66.0 | 0.2521 | -0.0104 | -0.0665 | 0.979 | 67.0 | 0.2122 |
| qM(C7)c | -0.0071 | -0.0890 | 0.964 | 69.3 | 0.2481 | -0.0107 | -0.0686 | 0.982 | 70.5 | 0.1117 |
| qC(C7a)c | -0.0425 | 0.1070 | 0.985 | 85.3 | 0.1083 | 0.0461 | 0.0815 | 0.990 | 98.4 | 0.1002 |
| qM(C7a)c | 0.0415 | 0.1065 | 0.985 | 86.7 | 0.1073 | 0.0452 | 0.0802 | 0.991 | 113.7 | 0.1013 |
| qC(O3’)d | 0.0249 | 0.0166 | 0.975 | 68.0 | 0.2613 | 0.0241 | 0.0153 | 0.984 | 83.5 | 0.2411 |
| qM(O3’)d | 0.0250 | 0.0175 | 0.976 | 67.9 | 0.2791 | 0.0253 | 0.0160 | 0.983 | 82.3 | 0.2489 |
Acknowledgements
References and Notes
- Stojceva-Radovanovic, B.C.; Andjekovic, S.S. J. Serb. Chem. 1998, 63, 397.
- Rehman, A.-U.; Subhan, A.S; Iqbal, C.M.; Azeen, A.; Rehman, A.-U. J. Chem. Soc. Pak. 1997, 19, 239.
- Singh, G.S.; Singh, T.; Lakhan, R. Indian J. Chem., Sect. B: Org. Incl. Med Chem. 1997, B36, 951.
- Pandeya, S.N; Srivam, D.; Nath, G.; DeClercq, E. Sci. Pharm. 1999, 67, 103.
- Falsone, G.; Cateni, F.; El-Alali, A.; Papaiannu, A.; Ravalico, L.; Furlani, A. Pharmacol. Let. 1992, 2, 104.
- Radhy, H.A.; Fadhil, G.F.; Perjéssy, A.; Kolehmainen, E.; Fabian, W.M.F.; Šamalíková, M.; Laihia, K.; Šusteková, Z. Heterocycl. Commun. 2001, 7, 387.
- Fadhil, G.F. Collect. Czech. Chem. Commun. 1993, 58, 385.
- Craik, D.J.; Brownlee, R.T.C. Progr. Physic. Org. Chem. 1983, 14, 1.
- Craik, D.J. Substituent Effects on Nuclear Shielding, Annual Reports on NMR Spectroscopy; Academic Press: London, 1983. [Google Scholar]
- Al-Shawi, S.A.O. Ph.D. Thesis, University of Basrah, Basrah, 1998.
- Al-Amood, K.H. M.Sc. Thesis, University of Basrah, Basrah, 1999.
- Saleh, B.A. M.Sc. Thesis, University of Basrah, Basrah, 1999.
- Dewar, M.J.S.; Zoebisch, E.G.; Healy, E.F.; Stewart, J.J.P. J. Am. Chem. Soc. 1985, 107, 3902.
- AMPAC 6.55, 1994, Semichem, 7128 Summit, KS 66216, USA.
- Ehrenson, S.; Brownlee, R.T.C.; Taft, R. W. Progr. Phys. Org. Chem. 1973, 10, 1.
- Exner, O. Advances in Linear Free Energy Relationships; Chapman, N. B., Shorter, J., Eds.; Plenum Press: London, 1972. [Google Scholar]
- Reynolds, W.F.A.; Gomes, A.; Maron, A.; McIntyre, D.W.; Tanin, A.; Hamer, G.K.; Peat, I.R. Can. J. Chem. 1983, 61, 2376.
- Levitt, L.S.; Widing, H. F. Progr. Phys. Org. Chem. 1976, 12, 119.
- Sample Availability: Available from the authors.
© 2002 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Fadhil, G.F.; Radhy, H.A.; Perjéssy, A.; Šamalíková, M.; Kolehmainen, E.; Fabian, W.M.F.; Laihia, K.; Šusteková, Z. Dual Substituent Parameter Modeling of Theoretical, NMR and IR Spectral Data of 5-Substituted Indole-2,3-diones. Molecules 2002, 7, 833-839. https://doi.org/10.3390/71100833
Fadhil GF, Radhy HA, Perjéssy A, Šamalíková M, Kolehmainen E, Fabian WMF, Laihia K, Šusteková Z. Dual Substituent Parameter Modeling of Theoretical, NMR and IR Spectral Data of 5-Substituted Indole-2,3-diones. Molecules. 2002; 7(11):833-839. https://doi.org/10.3390/71100833
Chicago/Turabian StyleFadhil, Ghazwan F., Hanan A. Radhy, Alexander Perjéssy, Mária Šamalíková, Erkki Kolehmainen, Walter M.F. Fabian, Katri Laihia, and Zora Šusteková. 2002. "Dual Substituent Parameter Modeling of Theoretical, NMR and IR Spectral Data of 5-Substituted Indole-2,3-diones" Molecules 7, no. 11: 833-839. https://doi.org/10.3390/71100833
APA StyleFadhil, G. F., Radhy, H. A., Perjéssy, A., Šamalíková, M., Kolehmainen, E., Fabian, W. M. F., Laihia, K., & Šusteková, Z. (2002). Dual Substituent Parameter Modeling of Theoretical, NMR and IR Spectral Data of 5-Substituted Indole-2,3-diones. Molecules, 7(11), 833-839. https://doi.org/10.3390/71100833




