Determining the Antimicrobial Actions of Tea Tree Oil
Abstract
:Introduction
Results and Discussion
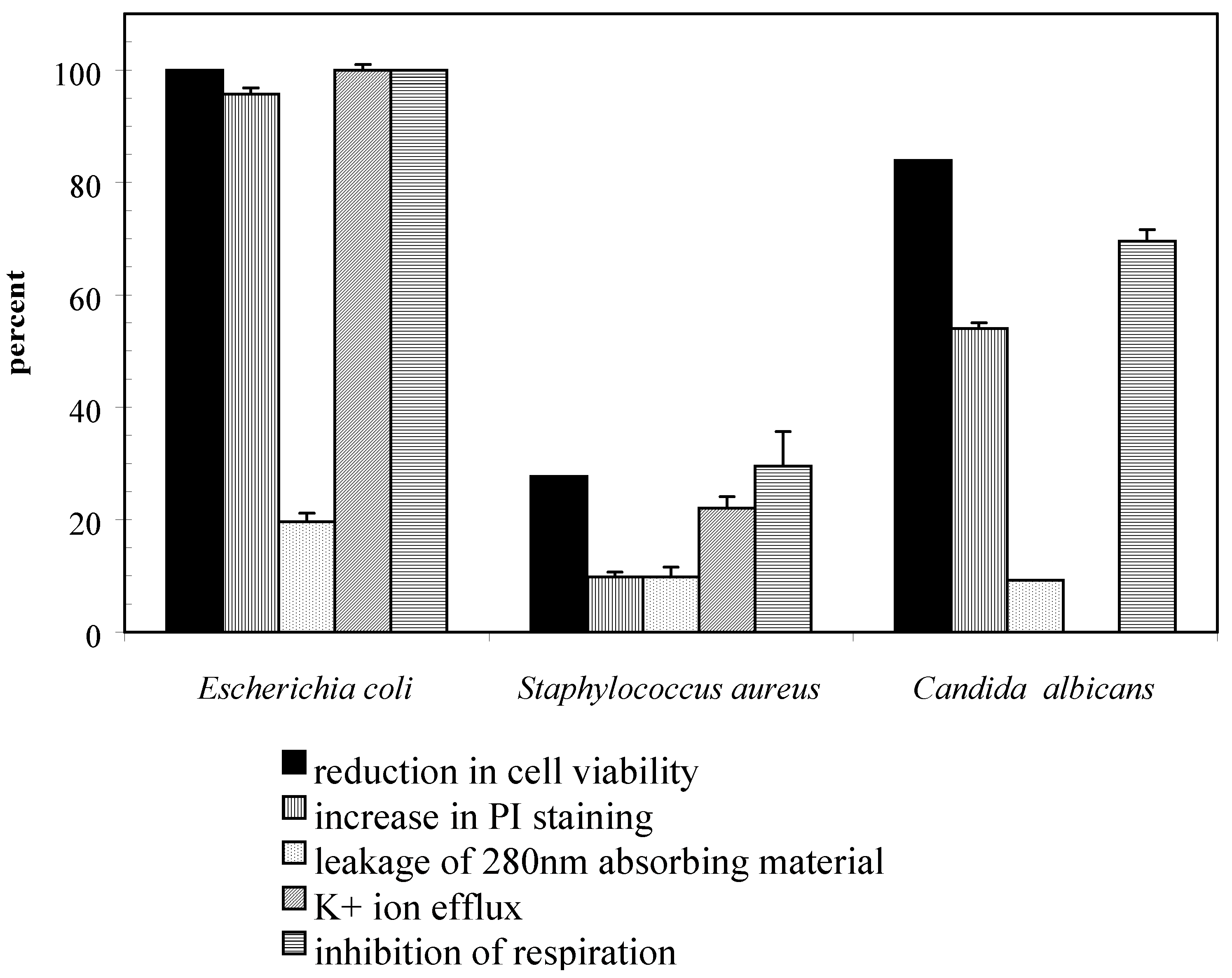
Conclusions
Experimental
References
- Janssen, A.M.; Scheffer, J.J.C.; Baerheim Svendsen, A. Antimicrobial activity of essential oils. A 1976-1986 literature review. Aspects of test methods. Planta Medica 1986, 53, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Beylier, M.F. Bacteriostatic activity of some Australian essential oils. Perfumer and Flavorist 1979, 4, 23–25. [Google Scholar]
- Andrews, R.E.; Parks, L.W.; Spence, K.D. Some effects of Douglas fir terpenes on certain microorganisms. Appl. Environ. Microbiol. 1980, 40, 301–304. [Google Scholar] [PubMed]
- Uribe, S.; Ramirez, J.; Peña, A. Effects of β-pinene on yeast membrane functions. J. Bacteriol. 1985, 161, 1195–1200. [Google Scholar] [PubMed]
- Sikkema, J.; de Bont, J.A.M.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev. 1995, 59, 201–222. [Google Scholar] [PubMed]
- Cox, S.D.; Gustafson, J.E.; Mann, C.M.; Markham, J.L.; Liew, Y.C.; Hartland, R.P.; Bell, H.C.; Warmington, J.R.; Wyllie, S.G. Tea tree oil causes K+ leakage and inhibits respiration in Escherichia coli. Lett. Appl. Microbiol. 1998, 26, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Southwell, I.A.; Hayes, A.J.; Markham, J.L.; Leach, D.N. The search for optimally bioactive Australian tea tree oil. Acta Hort. 1993, 334, 265–275. [Google Scholar] [CrossRef]
- Mason, D.J.; Dybowski, R.; Larrick, J.W.; Gant, V.A. Antimicrobial action of rabbit leukocyte CAP18106-137. Antimicrob. Agents Chemother. 1997, 41, 624–629. [Google Scholar] [PubMed]
- Trumpower, B.L.; Gennis, R.B. Energy transduction by cytochrome complexes in mitochondrial and bacterial respiration: the enzymology of coupling electron transfer reactions to transmembrane proton translocation. Annu. Rev. Biochem. 1994, 63, 675–716. [Google Scholar] [CrossRef] [PubMed]
- Booth, I.R. Regulation of cytoplasmic pH in bacteria. Microbiol. Rev. 1985, 49, 359–378. [Google Scholar] [PubMed]
- Poolman, B.; Driessen, A.J.M.; Konings, W.N. Regulation of solute transport in Streptococci by external and internal pH values. Microbiol. Rev. 1987, 51, 498–508. [Google Scholar] [PubMed]
- Anon. Oil of Melaleuca, terpinen-4-ol type (Tea Tree Oil). In ISO 4730 1996 (E); 1996; International Standards Organization: Geneva. [Google Scholar]
- Cox, S.D.; Mann, C.M.; Markham; Bell, H.C.; Gustafson, J.E.; Warmington, J.R.; Wyllie, S.G. The mode of antimicrobial action of the essential oil of Melaleuca alternifolia (tea tree oil). J. Appl. Microbiol. 2000, 88, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples are available from the authors.
© 2001 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Cox, S.D.; Mann, C.M.; Markham, J.L.; Gustafson, J.E.; Warmington, J.R.; Wyllie, S.G. Determining the Antimicrobial Actions of Tea Tree Oil. Molecules 2001, 6, 87-91. https://doi.org/10.3390/60100087
Cox SD, Mann CM, Markham JL, Gustafson JE, Warmington JR, Wyllie SG. Determining the Antimicrobial Actions of Tea Tree Oil. Molecules. 2001; 6(2):87-91. https://doi.org/10.3390/60100087
Chicago/Turabian StyleCox, Sean D., Cindy M. Mann, Julie L. Markham, John E. Gustafson, John R. Warmington, and S. Grant Wyllie. 2001. "Determining the Antimicrobial Actions of Tea Tree Oil" Molecules 6, no. 2: 87-91. https://doi.org/10.3390/60100087
APA StyleCox, S. D., Mann, C. M., Markham, J. L., Gustafson, J. E., Warmington, J. R., & Wyllie, S. G. (2001). Determining the Antimicrobial Actions of Tea Tree Oil. Molecules, 6(2), 87-91. https://doi.org/10.3390/60100087



