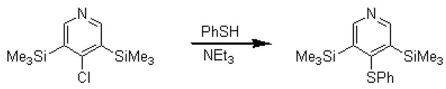
3,5-Bis(trimethylsilyl)-4-thiophenoxypyridine
Supplementary Material
Reference
- Kay, A. J.; Woolhouse, A. D. Molecules 2001, 6, M241. [CrossRef]
Sample Availability: Available from the authors and from MDPI. |
© 2001 MDPI, Basel, Switzerland. All rights reserved. Molecules website www.mdpi.org/molecules/.
Share and Cite
Kay, A.J.; Woolhouse, A.D. 3,5-Bis(trimethylsilyl)-4-thiophenoxypyridine. Molecules 2001, 6, M266. https://doi.org/10.3390/M266
Kay AJ, Woolhouse AD. 3,5-Bis(trimethylsilyl)-4-thiophenoxypyridine. Molecules. 2001; 6(12):M266. https://doi.org/10.3390/M266
Chicago/Turabian StyleKay, A.J., and A.D. Woolhouse. 2001. "3,5-Bis(trimethylsilyl)-4-thiophenoxypyridine" Molecules 6, no. 12: M266. https://doi.org/10.3390/M266
APA StyleKay, A. J., & Woolhouse, A. D. (2001). 3,5-Bis(trimethylsilyl)-4-thiophenoxypyridine. Molecules, 6(12), M266. https://doi.org/10.3390/M266



